The High-Temperature Soft Ferromagnetic Molecular Materials Based on [W(CN)6(bpy)]2−/− System
Abstract
:1. Introduction
2. Results
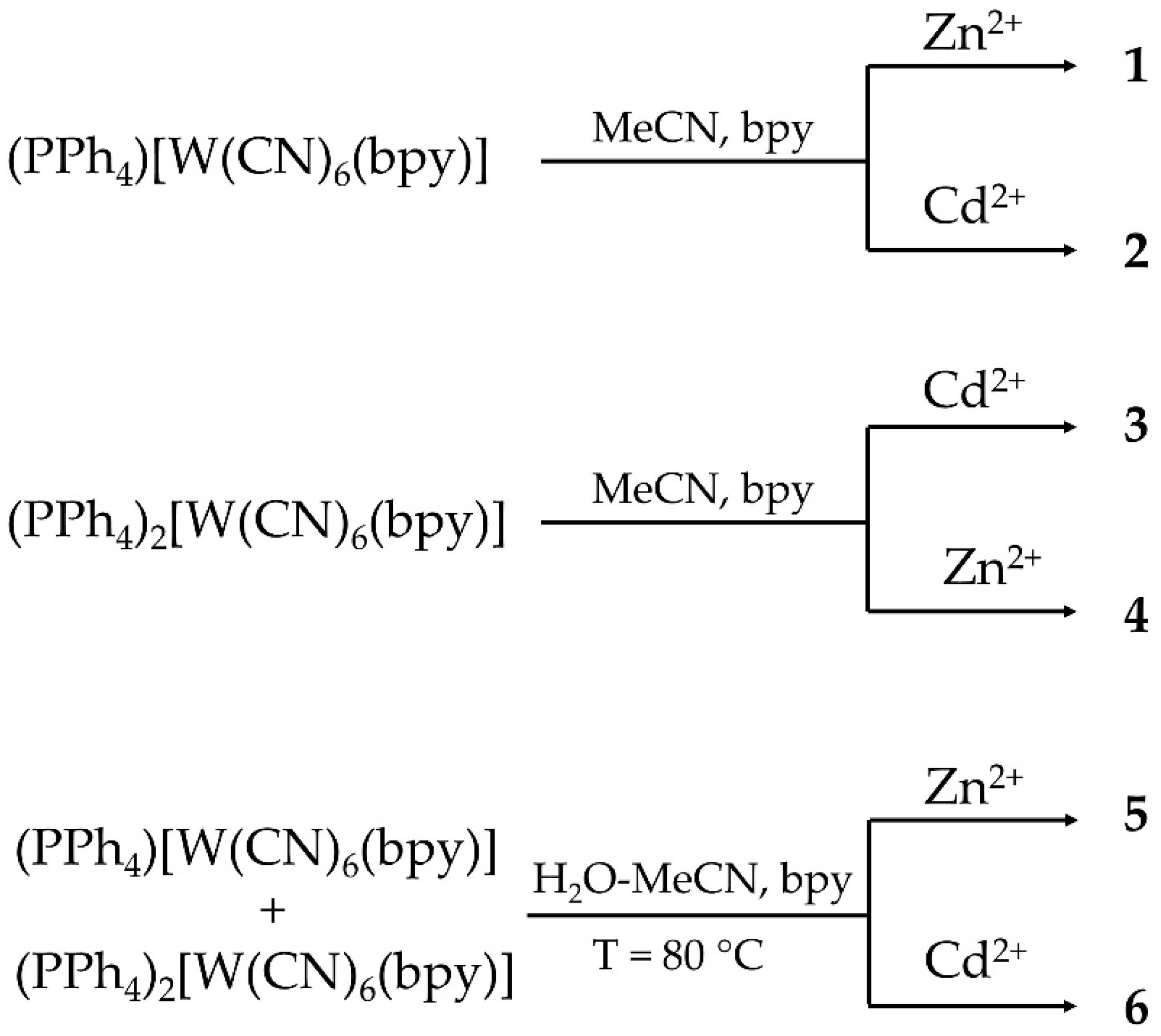
2.1. Design and Characterization
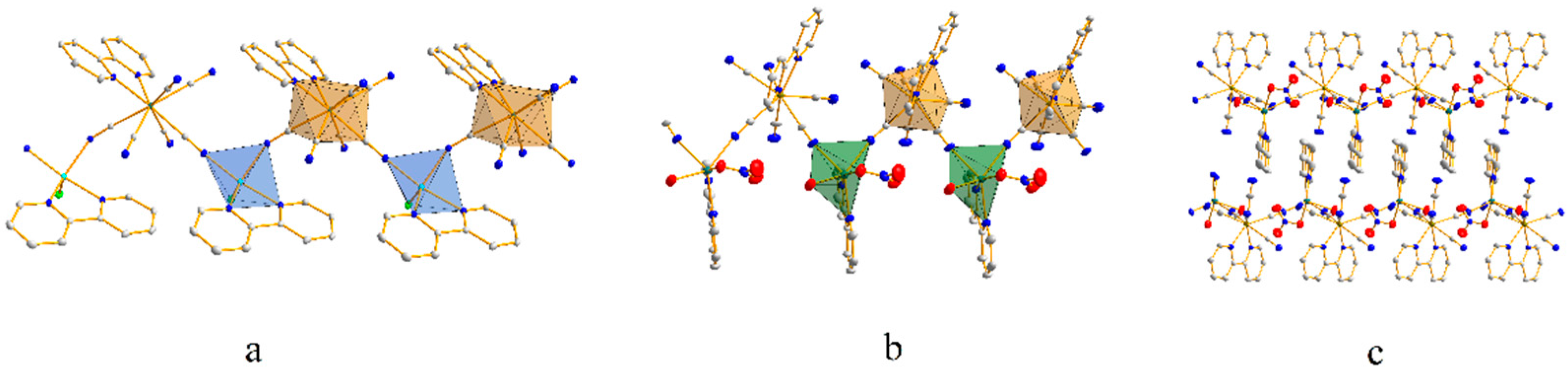
2.2. The structures of Precursors
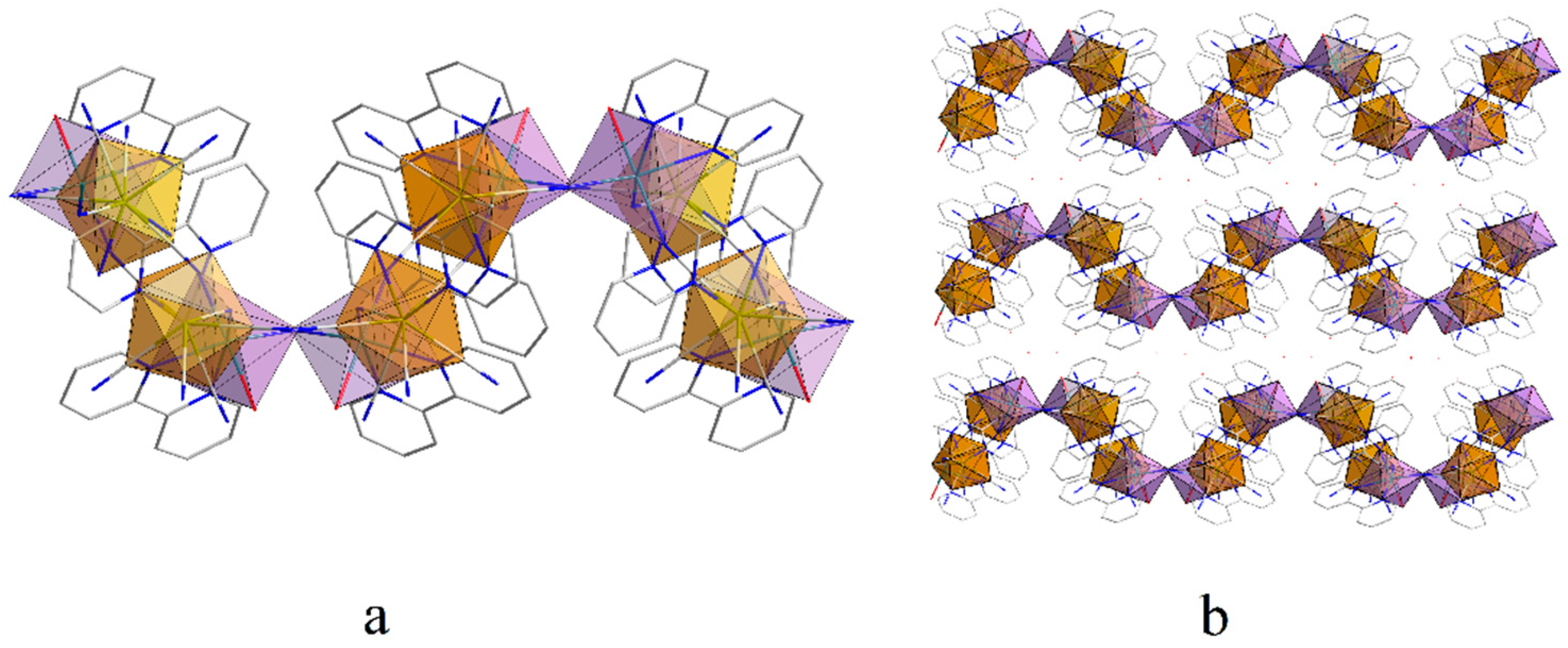
2.3. The Ferromagnetic Transformation
3. Discussion
4. Materials and Methods
4.1. Chemicals and Materials
4.2. Synthesis
4.2.1. Synthesis of [Zn(bpy)Cl][W(CN)6(bpy)] (1)
4.2.2. Synthesis of [Cd(bpy)(H2O)(NO3)][W(CN)6(bpy)] (2)
4.2.3. Synthesis of [Cd(bpy)(H2O)][W(CN)6(bpy)]·2H2O (3)
4.2.4. Synthesis of {[Zn(bpy)(NO3)]}2[W(CN)6(bpy)]·2H2O (4)
4.2.5. Synthesis of Ferromagnetic Material 5 and 6
4.3. Physicochemical Measurements
4.4. Single-Crystal Structure Determination
4.5. XRD Measurements of Powder Samples
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Kahn, O. Molecular Magnetism; VCV Publishers: New York, NY, USA, 1993. [Google Scholar]
- Hodorowicz, M.; Jurowska, A.; Szklarzewicz, J. X-ray crystal structures of K+ and Rb+ salts of [W(CN)6(bpy)]2− ion. The unusual cation–Anion interactions and structure changes going from Li+ to Cs+ salts. CrystEngComm 2021, 23, 1207–1217. [Google Scholar] [CrossRef]
- Hodorowicz, M.; Jurowska, A.; Szklarzewicz, J. Structures of alkali metal salts with [W(CN)6(bpy)]− ion. Comparative studies to W(IV) analogues. Polyhedron 2021, 207, 115369. [Google Scholar] [CrossRef]
- Szklarzewicz, J. New cyano complex of W(V), (AsPh4)[W(bpy)(CN)6]: Reversible redox system W(bpy)(CN)6−/W(bpy)(CN)62−. Inorg. Chim. Acta 1993, 205, 85–89. [Google Scholar] [CrossRef]
- Coronado, E. Molecular magnetism: From chemical design to spin control in molecules, materials and devices. Nat. Rev. Mater. 2020, 5, 87–104. [Google Scholar] [CrossRef]
- McAdams, S.G.; Ariciu, A.-M.; Kostopoulos, A.K.; Walsh, J.P.S.; Tuna, F. Molecular single-ion magnets based on lanthanides and actinides: Design considerations and new advances in the context of quantum technologies. Coord. Chem. Rev. 2017, 346, 216–239. [Google Scholar] [CrossRef] [Green Version]
- Verdaguer, M.; Gleizes, A.N. Magnetism: Molecules to Build Solids. Eur. J. Inorg. Chem. 2020, 2020, 723–731. [Google Scholar] [CrossRef] [Green Version]
- Liu, F.; Velkos, G.; Krylov, D.S.; Spree, L.; Zalibera, M.; Ray, R.; Samoylova, N.A.; Chen, C.-H.; Rosenkranz, M.; Schiemenz, S.; et al. Air-stable redox-active nanomagnets with lanthanide spins radical-bridged by a metal-metal bond. Nat. Commun. 2019, 10, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Serrano, G.; Poggini, L.; Briganti, M.; Sorrentino, A.L.; Cucinotta, G.; Malavolti, L.; Cortigiani, B.; Otero, E.; Sainctavit, P.; Loth, S.; et al. Quantum dynamics of a single molecule magnet on superconducting Pb(III). Nat. Mater. 2020, 19, 546–551. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, P.; Riso, C.; Amir, U.; Rojas-Dotti, C.; Martinez-Lillo, J. Exploring room-temperature transport of single-molecule magnet-based molecular spintronics devices using the magnetic tunnel junction as a device platform. RSC Adv. 2020, 10, 13006–13015. [Google Scholar] [CrossRef] [PubMed]
- Szklarzewicz, J.; Hodorowicz, M. Synthesising a Molecular Magnetic Material. US Patent 17/067,777, 12 October 2020. [Google Scholar]
- Szklarzewicz, J.; Hodorowicz, M. A Molecular Magnetic Material and a Method for Preparation Thereof. EP19183528.9, 1 July 2019. [Google Scholar]
- Szklarzewicz, J.; Hodorowicz, M. Sposób Syntezy Molekularnego Materiału Magnetycznego Oraz Molekularny Materiał Magnetyczny Otrzymany Tym Sposobem. EP 20199624.6 (P.431440), 1 October 2020. [Google Scholar]
- Szklarzewicz, J.; Samotus, A. A novel cyano complex of tungsten(IV) with 2,2′-bipyridyl. Transit. Met. Chem. 1988, 13, 69–71. [Google Scholar] [CrossRef]
- Hodorowicz, M.; Szklarzewicz, J.; Jurowska, A. The versatility of lithium cation coordination modes in salts with [W(CN)6(bpy)]2− anions. CrystEngComm 2020, 22, 3991–3998. [Google Scholar] [CrossRef]
- Rigaku Oxford Diffraction, CrysAlisPro; Version. 1.171.36.20; Rigaku Oxford Diffraction: Oxfordshire, UK, 2015.
- Sheldrick, G.M. Crystal Structure Refinement with SHELXL. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. SHELX2017; Programs for Crystal Structure Determination; Universität Göttingen: Göttingen, Germany, 2017. [Google Scholar]
- Putz, H.; Brandenburg, K. Diamond-Crystal and Molecular Structure Visualization Crystal Impact; GbR: Bonn, Germany, 2022. [Google Scholar]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szklarzewicz, J.; Hodorowicz, M.; Jurowska, A.; Baran, S. The High-Temperature Soft Ferromagnetic Molecular Materials Based on [W(CN)6(bpy)]2−/− System. Molecules 2022, 27, 4525. https://doi.org/10.3390/molecules27144525
Szklarzewicz J, Hodorowicz M, Jurowska A, Baran S. The High-Temperature Soft Ferromagnetic Molecular Materials Based on [W(CN)6(bpy)]2−/− System. Molecules. 2022; 27(14):4525. https://doi.org/10.3390/molecules27144525
Chicago/Turabian StyleSzklarzewicz, Janusz, Maciej Hodorowicz, Anna Jurowska, and Stanisław Baran. 2022. "The High-Temperature Soft Ferromagnetic Molecular Materials Based on [W(CN)6(bpy)]2−/− System" Molecules 27, no. 14: 4525. https://doi.org/10.3390/molecules27144525
APA StyleSzklarzewicz, J., Hodorowicz, M., Jurowska, A., & Baran, S. (2022). The High-Temperature Soft Ferromagnetic Molecular Materials Based on [W(CN)6(bpy)]2−/− System. Molecules, 27(14), 4525. https://doi.org/10.3390/molecules27144525






