Classification of Polish Natural Bee Honeys Based on Their Chemical Composition
Abstract
:1. Introduction
2. Results and Discussion
2.1. Sugar Profile of Polish Honeys
2.2. Amino Acids Profile and HMF Content in Polish Honeys
2.3. One-Way ANOVA
2.4. Multivariate Data Analysis
3. Materials and Methods
3.1. Honey Samples
3.2. Sample Preparation
3.3. NMR Experiments
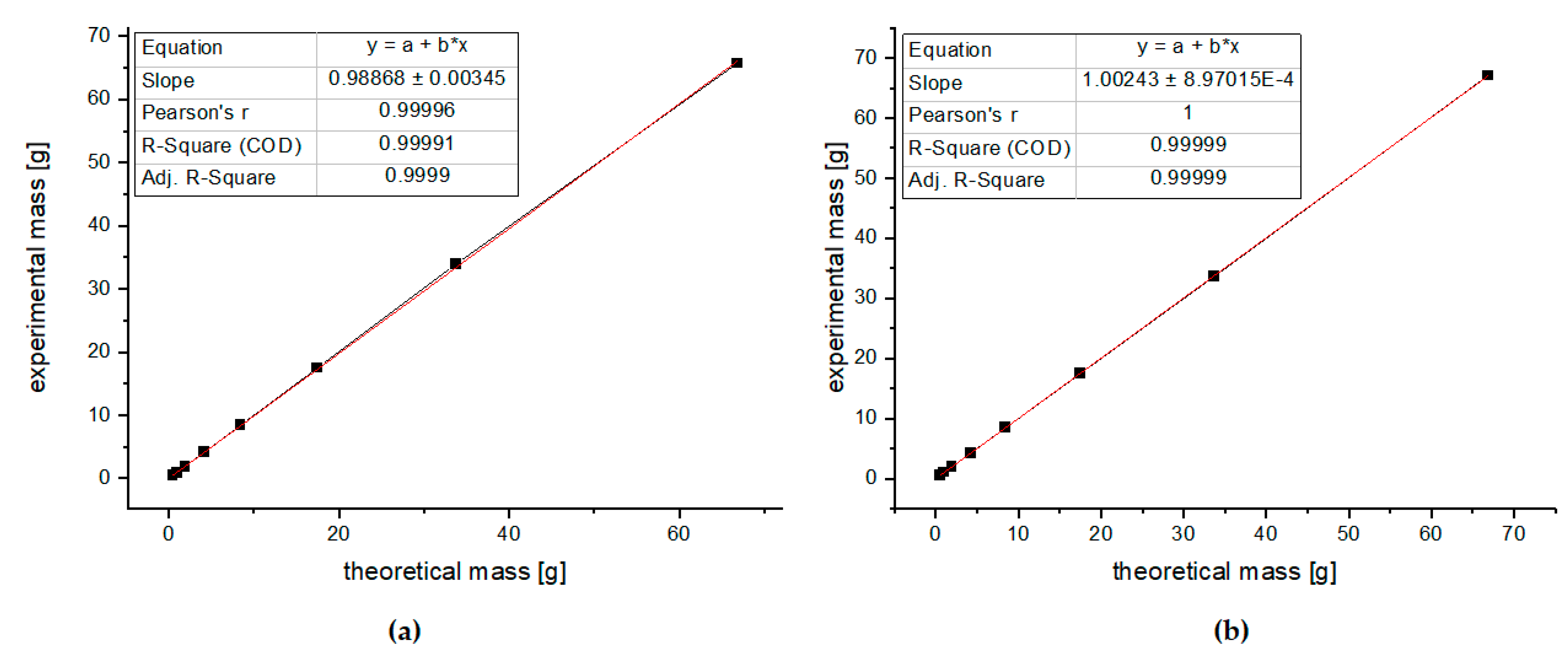
3.3.1. Calibration Curve
3.3.2. Inter-Day and Intra-Day Repeatability
3.4. Quantification of Sugars and Amino Acids
3.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Honey Market Size, Share, Industry Report, 2022–2030. Available online: https://grandviewresearch.com (accessed on 13 June 2022).
- National Apiculture Programmes 2020–2022. (europa.eu). Available online: https://honey-apiculture-programmes-overview-2020-2022.pdf (accessed on 13 June 2022).
- Semkiw, P. Sektor Pszczelarski w Polsce w 2020 Roku. Puławy: Zakład Pszczelnictwa w Puławach (inhort.pl). Available online: http://www.inhort.pl/files/program_wieloletni/PW_2015_2020_IO/spr_2020/4.3_Semkiw_Sektor_pszczelarski_2020.pdf (accessed on 12 June 2022).
- Jaafar, M.B.; Othman, M.B.; Yaacob, M.; Talip, B.A.; Ilyas, M.A.; Ngajikin, N.H.; Fauzi, N.A.M. A review on honey adulteration and the available detection approaches. Int. J. Struct. Integr. 2020, 12, 125–131. [Google Scholar] [CrossRef]
- Codex Alimentarius Commission. Draft Revised Standard for Honey (at 10 Step of the Codex Procedure); Codex Alimentarius Commission: Rome, Italy, 2001; Volume 25, pp. 19–26. [Google Scholar]
- Council Directive 2001/110/EC of 20 December 2001 relating to honey. Off. J. Eur. Communities 2002, L.10, 47–52.
- Janiszewska, K.; Aniołowska, M.; Nowakowski, P. Free amino acids content of honeys from Poland. Pol. J. Food Nutr. Sci. 2012, 62, 85–89. [Google Scholar] [CrossRef] [Green Version]
- El Sohaimy, S.A.; Masry, S.H.D.; Shehata, M.G. Physicochemical characteristics of honey from different origins. Ann. Agric. Sci. 2015, 60, 279–287. [Google Scholar] [CrossRef] [Green Version]
- Da Silva, P.M.; Gauche, C.; Gonzaga, L.V.; Costa, A.C.O.; Fett, R. Honey: Chemical composition, stability and authenticity. Food Chem. 2016, 196, 309–323. [Google Scholar] [CrossRef]
- Molan, P.C. The limitations of the methods of identifying the floral source of honeys. Bee World 1998, 79, 59–68. [Google Scholar] [CrossRef]
- Escuredo, O.; Míguez, M.; Fernández-González, M.; Seijo, M.C. Nutritional value and antioxidant activity of honeys produced in a European Atlantic area. Food Chem. 2013, 138, 851–856. [Google Scholar] [CrossRef]
- Beretta, G.; Artali, R.; Caneva, E.; Orlandini, S.; Centini, M.; Facino, R.M. Quinoline alkaloids in honey: Further analytical (HPLC-DAD-ESI-MS, multidimensional diffusion-ordered NMR spectroscopy), theoretical and chemometric studies. J. Pharm. Biomed. 2009, 50, 432–439. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Matute, A.I.; Brokl, M.; Soria, A.C.; Sanz, M.L.; Martínez-Castro, I. Gas chromatographic-mass spectrometric characterisation of tri- and tetrasaccharides in honey. Food Chem. 2010, 120, 637–642. [Google Scholar] [CrossRef]
- Kowalski, S.; Kopuncova, M.; Ciesarova, Z.; Kukurova, K. Free amino acids profile of Polish and Slovak honeys based on LC–MS/MS method without the prior derivatization. J. Food Sci. Technol. 2017, 54, 3716–3723. [Google Scholar] [CrossRef] [PubMed]
- Kek, S.P.; Chin, N.L.; Tan, S.W.; Yusof, Y.A.; Chua, L.S. Classification of honey from its Bee origin via chemical profiles and mineral content. Food Anal. Methods 2017, 10, 19–30. [Google Scholar] [CrossRef]
- Dos Santos, J.S.; dos Santos, N.S.; dos Santos, M.L.P.; dos Santos, S.N.; Lacerda, J.J. Honey classification from semi-arid, Atlantic and transitional forest zones in Bahia, Brazil. J. Braz. Chem. Soc. 2008, 19, 502–508. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Kliks, M.M.; Qu, W.; Jun, S.; Shi, G.; Li, Q.X. Rapid determination of the geographical origin of honey based on protein fingerprinting and barcoding using MALDI TOF MS. J. Agric. Food Chem. 2009, 57, 10081–10088. [Google Scholar] [CrossRef]
- Pierna, J.A.F.; Abbas, O.; Dardenne, P.; Baeten, V. Discrimination of Corsican honey by FT-Raman spectroscopy and chemometrics. Biotechnol. Agron. Soc. Environ. 2011, 15, 75–84. [Google Scholar]
- Zhu, X.; Li, S.; Shan, Y.; Zhang, Z.; Li, G.; Su, D.; Liu, F. Detection of adulterants such as sweeteners materials in honey using near-infrared spectroscopy and chemometrics. J. Food Eng. 2010, 101, 92–97. [Google Scholar] [CrossRef]
- Cozzolino, D.; Corbella, E.; Smyth, H. Quality control of honey using infrared spectroscopy: A review. Appl. Spectrosc. Rev. 2011, 46, 523–538. [Google Scholar] [CrossRef]
- Labsvards, K.D.; Rudovica, V.; Kluga, R.; Rusko, J.; Busa, L.; Bertins, M.; Eglite, I.; Naumenko, J.; Salajeva, M.; Viksna, A. Determination of Floral Origin Markers of Latvian Honey by Using IRMS, UHPLC-HRMS, and 1H-NMR. Foods 2022, 11, 42. [Google Scholar] [CrossRef]
- Ohmenhaeuser, M.; Monakhova, Y.B.; Kuballa, T.; Lachenmeier, D.W. Qualitative and Quantitative Control of Honeys Using NMR Spectroscopy and Chemometrics. IRSN Anal. Chem. 2013, 2013, 825318. [Google Scholar] [CrossRef]
- Consonni, R.; Cagliani, L.R.; Cogliati, C. Geographical discrimination of honeys by saccharides analysis. Food Control 2013, 32, 543–548. [Google Scholar] [CrossRef]
- Kazalaki, A.; Misiak, M.; Spyros, A.; Dais, P. Identification and quantitative determination of carbohydrate molecules in Greek honey by employing 13C NMR spectroscopy. Anal. Methods 2015, 7, 5962–5972. [Google Scholar] [CrossRef]
- Spiteri, C.; Lia, F.; Farrugia, C. Determination of the geographical origin of maltese honey using 1H NMR fingerprinting. Foods 2020, 9, 1455. [Google Scholar] [CrossRef]
- Zieliński, Ł.; Deja, S.; Jasicka-Misiak, I.; Kafarski, P. Chemometrics as a tool of origin determination of Polish monofloral and multifloral honeys. J. Agric. Food Chem. 2014, 62, 2973–2981. [Google Scholar] [CrossRef] [PubMed]
- Yong, C.H.; Muhammed, S.A.; Aziz, F.A.; Nasir, F.I.; Mustafa, M.Z.; Ibrahim, B.; Kelly, S.D.; Cannavan, A.; Seow, E.K. Detecting adulteration of stingless bee honey using untargeted 1H NMR metabolomics with chemometrics. Food Chem. 2022, 368, 130808. [Google Scholar] [CrossRef]
- Escuredo, O.; Dobre, I.; Fernández-González, M.; Seijo, M.C. Contribution of botanical origin and sugar composition of honeys on the crystallization phenomenon. Food Chem. 2014, 149, 84–90. [Google Scholar] [CrossRef]
- Chua, L.S.; Adnan, N.A. Biochemical and nutritional components of selected honey samples. Acta Sci. Pol. Technol. Aliment. 2014, 13, 169–179. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borawska, M.; Arciuch, L.; Puścion-Jakubik, A.; Lewoc, D. Content of sugars (fructose, glucose, sucrose) and proline in different varieties of natural bee honey. Probl. Hig. Epidemiol. 2015, 96, 816–820. [Google Scholar]
- Hao, S.; Yuan, J.; Cui, J.; Yuan, W.; Zhang, H.; Xuan, H. The rapid detection of acacia honey adulteration by alternating current impedance spectroscopy combined with 1H NMR profile. LWT 2022, 161, 113377. [Google Scholar] [CrossRef]
- Krauze, A.; Zalewski, R.I. Classification of honeys by principal component analysis on the basis of chemical and physical parameters. Z. Lebensm. Unters. Forsch. 1991, 192, 19–23. [Google Scholar] [CrossRef]
- Rodriguez, G.O.; Ferrer, B.S.; Ferrer, A.; Rodriguez, B. Characterization of honey produced in Venezuela. Food Chem. 2004, 84, 499–502. [Google Scholar] [CrossRef]
- Hermosın, I.; Chicon, R.M.; Cabezudo, M.D. Free amino acid composition and botanical origin of honey. Food Chem. 2003, 83, 263–268. [Google Scholar] [CrossRef]
- Iglesias, M.T.; Lorenzo, C.; Polo, M.C.; Martın-Alvarez, P.J.; Pueyo, E.N. Usefulness of amino acids composition to discriminate between honeydew and floral honeys. Application to honeys from a small geographic area. J. Agric. Food Chem. 2004, 52, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Paramas, A.M.; Gomez-Barez, J.A.; Cordon-Marcos, C.; García-Villanova, R.J.; Sanchez-Sanchez, J. HPLC-fluorimetric method for analysis of amino acids in products of hive (honey and bee-pollen). Food Chem. 2006, 95, 148–156. [Google Scholar] [CrossRef]
- Czipa, N.; Borbély, M.; Győri, Z. Proline content of different honey types. Acta Aliment. 2012, 41, 26–32. [Google Scholar] [CrossRef]
- Kirvak, S. Analysis of Amino Acid and Phenolic Content in Honey by UPLC-ESI-MS/MS. In Honey Analysis; de Alencar, V., de Toledo, A., Eds.; BoD-Books on Demand: Rijeka, Croatia, 2017; Chapter 04. [Google Scholar] [CrossRef] [Green Version]
- Truzzi, C.; Annibaldi, A.; Illuminati, S.; Finale, C.; Scarponi, G. Determination of proline in honey: Comparison between official methods, optimization and validation of the analytical methodology. Food Chem. 2014, 150, 477–481. [Google Scholar] [CrossRef] [PubMed]
- Iglesias, M.T.; Martın-Alvarez, P.J.; Polo, M.C.; Lorenzo, C.; Gonzalez, M.; Pueyo, E.N. Changes in free amino acid contents of honeys during storage at ambient temperature. J. Agric. Food Chem. 2006, 54, 9099–9194. [Google Scholar] [CrossRef]
- Shapla, U.M.; Solayman, M.; Alam, N.; Khalil, I.; Hua, S. 5-Hydroxymethylfurfural (HMF) levels in honey and other food products: Efects on bees and human health. Chem. Cent. J. 2018, 12, 35. [Google Scholar] [CrossRef]
- Turkut, G.M.; Degirmenci, A.; Yildiz, O.; Can, Z.; Cavrar, S.; Karahalil, F.; Kolaylı, S. Investigating 5-hydroxymethylfurfural formation kinetic and antioxidant activity in heat treated honey from different floral sources. J. Food Meas. Charact. 2018, 12, 2358–2365. [Google Scholar] [CrossRef]
- Anklam, E. A review of the analytical methods to determine the geographical and botanical origin of honey. Food Chem. 1998, 63, 549–562. [Google Scholar] [CrossRef]
- Harun, K.; Edita, S.; Nevzeta, A.; Nađa, M. Determination of Hydroxymethylfurfural Content (HMF) in Fresh Bee Honey Produced in Bosnia and Herzegovina (B&H) by HPLC DAD Method. Int. J. Environ. Sci. Nat. Res. 2021, 26, 556199. [Google Scholar] [CrossRef]
- Conte, L.S.; Miorini, M.; Giomo, A.; Bertacco, G.; Zironi, R. Evaluation of some fixed components for unifloral honey characterization. J. Agric. Food Chem. 1998, 46, 1844–1849. [Google Scholar] [CrossRef]
- Kortesniemi, M.; Slupsky, C.M.; Ollikka, T.; Kauko, L.; Spevacek, A.R.; Sjövall, O.; Yang, B.; Kallio, H. NMR profiling clarifies the characterization of Finnish honeys of different botanical origins. Food Res. Int. 2016, 86, 83–92. [Google Scholar] [CrossRef] [Green Version]
- Lolli, M.; Bertelli, D.; Plessi, M.; Sabatini, A.G.; Restani, C. Classification of Italian honeys by 2D HR-NMR. J. Agric. Food Chem. 2009, 56, 1298–1304. [Google Scholar] [CrossRef] [PubMed]
- Ralli, E.; Amargianitaki, M.; Manolopoulou, E.; Misiak, M.; Markakis, G.; Tachtalidou, S.; Kolesnikova, A.; Dais, P.; Spyros, A. NMR Spectroscopy Protocols for Food Metabolomics Applications. In Metabolic Profiling. Methods in Molecular Biology; Theodoridis, G., Gika, H., Wilson, I., Eds.; Humana Press: New York, NY, USA, 2018; Volume 1738. [Google Scholar] [CrossRef]
- Bharti, S.K.; Roy, R. Quantitative 1H NMR spectroscopy. Trends Analyt. Chem. 2012, 35, 5–26. [Google Scholar] [CrossRef]





| Variety of Honey | Mean ± SD [g/100g] | |||
|---|---|---|---|---|
| Glc (Min–Max) | Fru (Min–Max) | Suc (Min–Max) | Fru/Glc Ratio | |
| Lime | 33.11 ± 1.09 (31.41–33.71) | 37.02 ± 1.30 (34.86–38.44) | 0.48 ± 0.06 (0.42–0.61) | 1.11 ± 0.06 (1.01–1.22) |
| Rape | 38.74 ± 3.25 (37.51–45.28) | 38.81 ± 0.87 (37.92–40.03) | 0.18 ± 0.004 (0.18–0.19) | 1.01 ± 0.09 (0.84–1.14) |
| Acacia | 27.62 ± 1.73 (25.56–30.34) | 42.26 ± 0.76 (41.38–43.09) | 1.03 ± 0.06 (0.98–1.13) | 1.52 ± 0.10 (1.39–1.62) |
| Multiflorous | 35.63 ± 1.13 (34.07–37.74) | 43.51 ± 1.22 (41.99–45.24) | 1.19 ± 0.05 (1.12–1.27) | 1.23 ± 0.07 (1.11–1.32) |
| Foreign commercial multiflorous | 37.32 ± 10.98 (31.41–59.67) | 49.82 ± 17.74 (38.44–85.47) | 3.76 ± 1.38 (2.96–6.51) | 1.32 ± 0.09 (1.18–1.43) |
| Variety of Honey | Mean ± SD [mg/kg] | Mean ± SD [mg/100 g] | |||
|---|---|---|---|---|---|
| 5-HMF (min-max) | Ala (min-max) | Pro (min-max) | Tyr (min-max) | Phe (min-max) | |
| Lime | 1.96 ± 0.32 (1.59–2.37) | 2.33 ± 0.33 (1.94–2.83) | 64.77 ± 9.99 (52.73–77.17) | 2.21 ± 0.15 (2.00–2.38) | 4.46 ± 0.25 (4.24–4.82) |
| Rape | 16.18 ± 1.39 (14.71–18.53) | 2.11 ± 0.13 (1.91–2.28) | 26.83 ± 3.06 (22.03–31.02) | 1.44 ± 0.25 (1.16–1-81) | 3.78 ± 0.23 (3.30–3.97) |
| Acacia | 0.83 ± 0.08 (0.73–0.95) | 2.59 ± 0.39 (1.81–2.97) | 11.34 ± 2.94 (8.27–16.72) | 7.21 ± 0.66 (6.48–8.06) | 1.81 ± 0.14 (1.66–2.04) |
| Multiflorous | 7.57 ± 0.59 (6.91–8.42) | 2.30 ± 0.13 (2.11–2.44) | 41.04 ± 4.75 (31.21–44.31) | 3.51 ± 0.39 (3.01–4.03) | 8.09 ± 0.99 (6.81–9.28) |
| Foreign multiflorous | 12.17 ± 0.85 (11.13–13.32) | 2.35 ± 0.10 (2.27–2.54) | 67.55 ± 4.71 (62.66–74.17) | 5.03 ± 0.62 (4.27–5.98) | 7.56 ± 0.71 (6.74–8.44) |
| Honey Samples | Number of Samples | Origin | Symbol 1 |
|---|---|---|---|
| Lime | 7 | Beekeeper |  |
| Rape | 7 | Beekeeper |  |
| Acacia | 7 | Beekeeper |  |
| Multiflorous | 7 | Beekeeper |  |
| Foreign multiflorous | 6 | Commercial |  |
| Experiment Number | Intra-Day Repeatability | Inter-Day Repeatability |
|---|---|---|
| 1 | 1.9198 | 1.9176 |
| 2 | 1.9224 | 1.9188 |
| 3 | 1.9211 | 1.9184 |
| 4 | 1.9207 | 1.9214 |
| 5 | 1.9178 | 1.9192 |
| 6 | 1.9216 | 1.9204 |
| 7 | 1.9179 | 1.9231 |
| 8 | 1.9194 | 1.9174 |
| Average | 1.9201 | 1.9195 |
| SD | 0.0017 | 0.0020 |
| CV% | 0.0872 | 0.1026 |
| Experiment Number | Intra-Day Repeatability | Inter-Day Repeatability |
|---|---|---|
| 1 | 37.5147 | 37.5289 |
| 2 | 37.5157 | 37.5282 |
| 3 | 37.5161 | 37.5167 |
| 4 | 37.5149 | 37.5187 |
| 5 | 37.5152 | 37.5193 |
| 6 | 37.5168 | 37.5235 |
| 7 | 37.5156 | 37.5288 |
| 8 | 37.5164 | 37.5282 |
| Average | 37.5157 | 37.5240 |
| SD | 0.0007 | 0.0052 |
| CV% | 0.0019 | 0.014 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pacholczyk-Sienicka, B.; Ciepielowski, G.; Modranka, J.; Bartosik, T.; Albrecht, Ł. Classification of Polish Natural Bee Honeys Based on Their Chemical Composition. Molecules 2022, 27, 4844. https://doi.org/10.3390/molecules27154844
Pacholczyk-Sienicka B, Ciepielowski G, Modranka J, Bartosik T, Albrecht Ł. Classification of Polish Natural Bee Honeys Based on Their Chemical Composition. Molecules. 2022; 27(15):4844. https://doi.org/10.3390/molecules27154844
Chicago/Turabian StylePacholczyk-Sienicka, Barbara, Grzegorz Ciepielowski, Jakub Modranka, Tomasz Bartosik, and Łukasz Albrecht. 2022. "Classification of Polish Natural Bee Honeys Based on Their Chemical Composition" Molecules 27, no. 15: 4844. https://doi.org/10.3390/molecules27154844






