A Two-Photon Fluorescent Probe for the Visual Detection of Peroxynitrite in Living Cells and Zebrafish
Abstract
:1. Introduction
2. Experimental Section
2.1. General Comments
2.2. Synthesis of HDBT-ONOO−
2.3. Statistical Methods
3. Results and Discussion
3.1. Design of the Probe HDBT-ONOO−
3.2. Spectral Response of HDBT-ONOO− to ONOO−
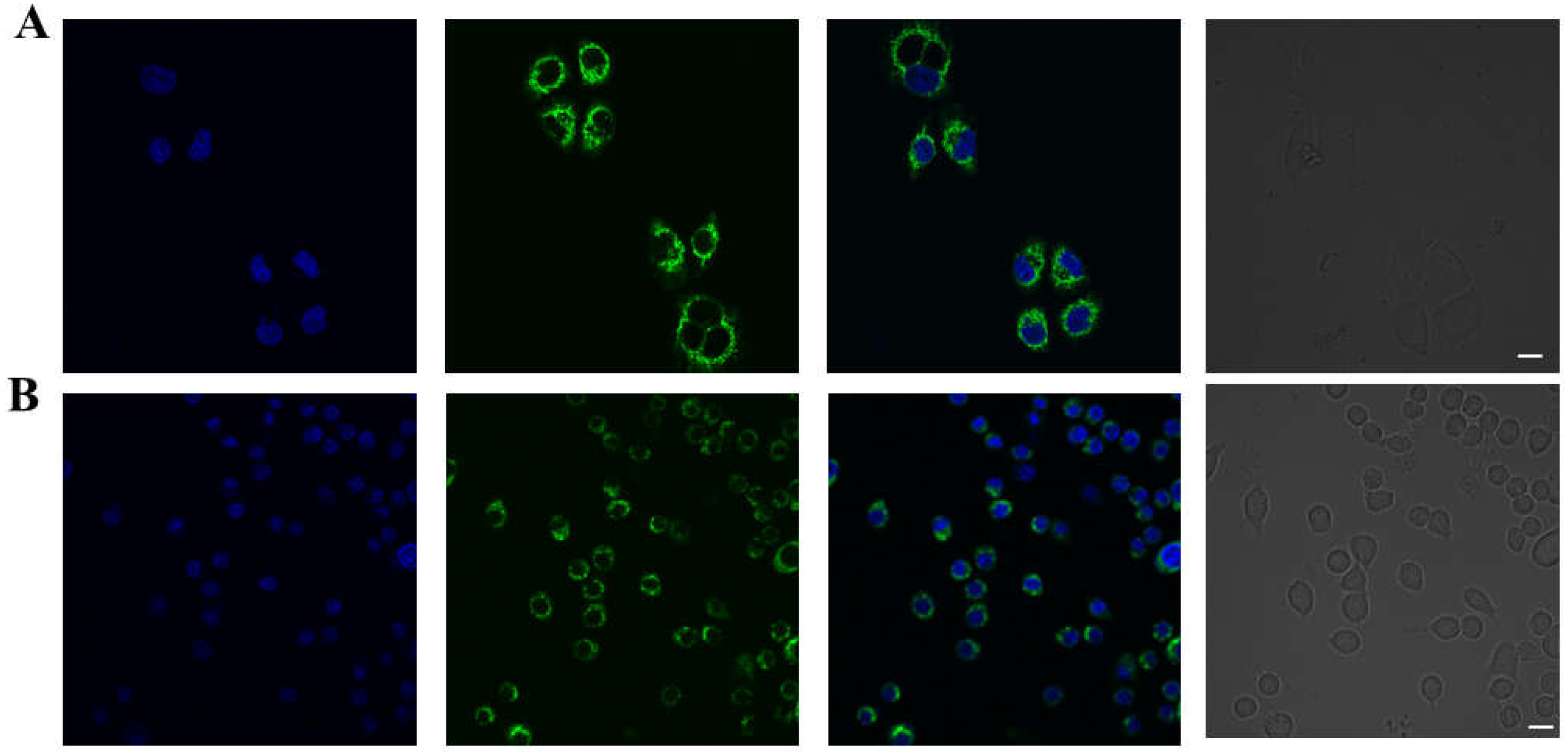
3.3. Fluorescence Imaging in Living Cells
3.4. Imaging of ONOO− in Zebrafish
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Serrano-Luginbuehl, S.; Kissner, R.; Koppenol, W.H. Reaction of CO2 with ONOO−: One Molecule of CO2 Is Not Enough. Chem. Res. Toxicol. 2018, 31, 721–730. [Google Scholar] [CrossRef] [PubMed]
- Beckman, J.S.; Beckman, T.W.; Chen, J.; Marshall, P.A.; Freeman, B.A. Apparent hydroxyl radical production by peroxynitrite: Implications for endothelial injury from nitric oxide and superoxide. Proc. Natl. Acad. Sci. USA 1990, 87, 1620–1624. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ischiropoulos, H.; Zhu, L.; Chen, J.; Tsai, M.; Martin, J.C.; Smith, C.D.; Beckman, J.S. Peroxynitrite-mediated tyrosine nitration catalyzed by superoxide dismutase. Arch. Biochem. Biophys. 1992, 298, 431–437. [Google Scholar] [CrossRef]
- Ferrer-Sueta, G.; Radi, R. Chemical biology of peroxynitrite: Kinetics, diffusion, and radicals. ACS Chem. Biol. 2009, 4, 161–177. [Google Scholar] [CrossRef]
- Koppenol, W.H.; Moreno, J.J.; Pryor, W.A.; Ischiropoulos, H.; Beckman, J.S. Peroxynitrite, a cloaked oxidant formed by nitric oxide and superoxide. Chem. Res. Toxicol. 1992, 5, 834–842. [Google Scholar] [CrossRef]
- Nathan, C.; Cunningham-Bussel, A. Beyond oxidative stress: An immunologist’s guide to reactive oxygen species. Nat. Rev. Immunol. 2013, 13, 349–361. [Google Scholar] [CrossRef] [Green Version]
- Radi, R. Peroxynitrite, a stealthy biological oxidant. J. Biol. Chem. 2013, 288, 26464–26472. [Google Scholar] [CrossRef] [Green Version]
- Szabo, C. Multiple pathways of peroxynitrite cytotoxicity. Toxicol. Lett. 2003, 140–141, 105–112. [Google Scholar] [CrossRef]
- Alvarez, B.; Radi, R. Peroxynitrite reactivity with amino acids and proteins. Amino Acids 2003, 25, 295–311. [Google Scholar] [CrossRef]
- Ferrer-Sueta, G.; Campolo, N.; Trujillo, M.; Bartesaghi, S.; Carballal, S.; Romero, N.; Alvarez, B.; Radi, R. Biochemistry of Peroxynitrite and Protein Tyrosine Nitration. Chem. Rev. 2018, 118, 1338–1408. [Google Scholar] [CrossRef]
- Szabo, C.; Ischiropoulos, H.; Radi, R. Peroxynitrite: Biochemistry, pathophysiology and development of therapeutics. Nat. Rev. Drug Discov. 2007, 6, 662–680. [Google Scholar] [CrossRef]
- Pacher, P.; Beckman, J.S.; Liaudet, L. Nitric oxide and peroxynitrite in health and disease. Physiol. Rev. 2007, 87, 315–424. [Google Scholar] [CrossRef] [Green Version]
- Torreilles, F.; Salman-Tabcheh, S.; Guerin, M.; Torreilles, J. Neurodegenerative disorders: The role of peroxynitrite. Brain Res. Rev. 1999, 30, 153–163. [Google Scholar] [CrossRef]
- Szabo, C. Role of nitrosative stress in the pathogenesis of diabetic vascular dysfunction. Br. J. Pharmacol. 2009, 156, 713–727. [Google Scholar] [CrossRef] [Green Version]
- Okun, Z.; Kupershmidt, L.; Amit, T.; Mandel, S.; Bar-Am, O.; Youdim, M.B.; Gross, Z. Manganese corroles prevent intracellular nitration and subsequent death of insulin-producing cells. ACS Chem. Biol. 2009, 4, 910–914. [Google Scholar] [CrossRef]
- Daiber, A.; Oelze, M.; August, M.; Wendt, M.; Sydow, K.; Wieboldt, H.; Kleschyov, A.L.; Munzel, T. Detection of superoxide and peroxynitrite in model systems and mitochondria by the luminol analogue L-012. Free Radic Res. 2004, 38, 259–269. [Google Scholar] [CrossRef]
- Dikalov, S.; Grigor’ev, I.A.; Voinov, M.; Bassenge, E. Detection of superoxide radicals and peroxynitrite by 1-hydroxy-4-phosphonooxy-2,2,6,6-tetramethylpiperidine: Quantification of extracellular superoxide radicals formation. Biochem. Biophys. Res. Commun. 1998, 248, 211–215. [Google Scholar] [CrossRef]
- Xue, J.; Ying, X.; Chen, J.; Xian, Y.; Jin, L. Amperometric ultramicrosensors for peroxynitrite detection and its application toward single myocardial cells. Anal. Chem. 2000, 72, 5313–5321. [Google Scholar] [CrossRef]
- Yamada, K.; Mito, F.; Matsuoka, Y.; Ide, S.; Shikimachi, K.; Fujiki, A.; Kusakabe, D.; Ishida, Y.; Enoki, M.; Tada, A.; et al. Fluorescence probes to detect lipid-derived radicals. Nat. Chem. Biol. 2016, 12, 608–613. [Google Scholar] [CrossRef]
- Kawatani, M.; Yamamoto, K.; Yamada, D.; Kamiya, M.; Miyakawa, J.; Miyama, Y.; Kojima, R.; Morikawa, T.; Kume, H.; Urano, Y. Fluorescence Detection of Prostate Cancer by an Activatable Fluorescence Probe for PSMA Carboxypeptidase Activity. J. Am. Chem. Soc. 2019, 141, 10409–10416. [Google Scholar] [CrossRef]
- Ning, J.; Tian, Z.; Wang, J.; Wang, B.; Tian, X.; Yu, Z.; Huo, X.; Feng, L.; Cui, J.; James, T.D.; et al. Rational Design of a Two-Photon Fluorescent Probe for Human Cytochrome P450 3A and the Visualization of Mechanism-Based Inactivation. Angew. Chem. Int. Ed. Engl. 2022, 61, e202113191. [Google Scholar] [CrossRef]
- Dai, F.; Jin, F.; Long, Y.; Jin, X.L.; Zhou, B. A 1,8-naphthalimide-based turn-on fluorescent probe for imaging mitochondrial hydrogen peroxide in living cells. Free Radic Res. 2018, 52, 1288–1295. [Google Scholar] [CrossRef]
- Li, M.; Fan, J.; Li, H.; Du, J.; Long, S.; Peng, X. A ratiometric fluorescence probe for lysosomal polarity. Biomaterials 2018, 164, 98–105. [Google Scholar] [CrossRef]
- Lee, D.; Lim, C.S.; Ko, G.; Kim, D.; Cho, M.K.; Nam, S.J.; Kim, H.M.; Yoon, J. A Two-Photon Fluorescent Probe for Imaging Endogenous ONOO− near NMDA Receptors in Neuronal Cells and Hippocampal Tissues. Anal. Chem. 2018, 90, 9347–9352. [Google Scholar] [CrossRef]
- Miao, J.; Huo, Y.; Liu, Q.; Li, Z.; Shi, H.; Shi, Y.; Guo, W. A new class of fast-response and highly selective fluorescent probes for visualizing peroxynitrite in live cells, subcellular organelles, and kidney tissue of diabetic rats. Biomaterials 2016, 107, 33–43. [Google Scholar] [CrossRef]
- Palanisamy, S.; Wu, P.Y.; Wu, S.C.; Chen, Y.J.; Tzou, S.C.; Wang, C.H.; Chen, C.Y.; Wang, Y.M. In vitro and in vivo imaging of peroxynitrite by a ratiometric boronate-based fluorescent probe. Biosens. Bioelectron. 2017, 91, 849–856. [Google Scholar] [CrossRef]
- Qu, W.; Niu, C.; Zhang, X.; Chen, W.; Yu, F.; Liu, H.; Zhang, X.; Wang, S. Construction of a novel far-red fluorescence light-up probe for visualizing intracellular peroxynitrite. Talanta 2019, 197, 431–435. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Wang, H.L.; Sun, Z.N.; Chung, N.W.; Shen, J.G. A highly selective fluorescent probe for the detection and imaging of peroxynitrite in living cells. J. Am. Chem. Soc. 2006, 128, 6004–6005. [Google Scholar] [CrossRef]
- Wang, C.; Dong, B.; Kong, X.; Zhang, N.; Song, W.; Lin, W. Dual site-controlled two-photon fluorescent probe for the imaging of lysosomal pH in living cells. Luminescence 2018, 33, 1275–1280. [Google Scholar] [CrossRef]
- Ren, T.B.; Xu, W.; Zhang, Q.L.; Zhang, X.X.; Wen, S.Y.; Yi, H.B.; Yuan, L.; Zhang, X.B. Enhancing the Anti-Solvatochromic Two-Photon Fluorescence for Cirrhosis Imaging by Forming a Hydrogen-Bond Network. Angew. Chem. Int. Ed. Engl. 2018, 57, 7473–7477. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Dong, B.; Kong, X.; Zhang, N.; Song, W.; Lin, W. A new xanthene-based two-photon fluorescent probe for the imaging of 1,4-dithiothreitol (DTT) in living cells. Luminescence 2018, 33, 1048–1053. [Google Scholar] [CrossRef] [PubMed]
- Czernel, G.; Matwijczuk, A.; Karcz, D.; Gorecki, A.; Niemczynowicz, A.; Szczes, A.; Gladyszewski, G.; Matwijczuk, A.; Gladyszewska, B.; Niewiadomy, A. Spectroscopic Studies of Dual Fluorescence in 2-(4-Fluorophenylamino)-5-(2,4-dihydroxybenzeno)-1,3,4-thiadiazole: Effect of Molecular Aggregation in a Micellar System. Molecules 2018, 23, 2861. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murphy, M.P.; Smith, R.A. Targeting antioxidants to mitochondria by conjugation to lipophilic cations. Annu. Rev. Pharmacol. Toxicol. 2007, 47, 629–656. [Google Scholar] [CrossRef] [PubMed]
- Porteous, C.M.; Logan, A.; Evans, C.; Ledgerwood, E.C.; Menon, D.K.; Aigbirhio, F.; Smith, R.A.; Murphy, M.P. Rapid uptake of lipophilic triphenylphosphonium cations by mitochondria in vivo following intravenous injection: Implications for mitochondria-specific therapies and probes. Biochim. Biophys. Acta 2010, 1800, 1009–1017. [Google Scholar] [CrossRef]
- Kaur, A.; Brigden, K.W.; Cashman, T.F.; Fraser, S.T.; New, E.J. Mitochondrially targeted redox probe reveals the variations in oxidative capacity of the haematopoietic cells. Org. Biomol. Chem. 2015, 13, 6686–6689. [Google Scholar] [CrossRef]
- Saeki, M.; Kamisaki, Y.; Maeda, S. Potentiation of carbachol-induced Ca2+ release by peroxynitrite in human neuroblastoma SH-SY5Y cells. Neurochem. Res. 2000, 25, 909–914. [Google Scholar] [CrossRef]
- Pipicz, M.; Kocsis, G.F.; Sarvary-Arantes, L.; Bencsik, P.; Varga, Z.V.; Ferdinandy, P.; Csont, T. Low-Dose Endotoxin Induces Late Preconditioning, Increases Peroxynitrite Formation, and Activates STAT3 in the Rat Heart. Molecules 2017, 22, 433. [Google Scholar] [CrossRef] [Green Version]
- Yoo, B.K.; Choi, J.W.; Shin, C.Y.; Jeon, S.J.; Park, S.J.; Cheong, J.H.; Han, S.Y.; Ryu, J.R.; Song, M.R.; Ko, K.H. Activation of p38 MAPK induced peroxynitrite generation in LPS plus IFN-gamma-stimulated rat primary astrocytes via activation of iNOS and NADPH oxidase. Neurochem. Int. 2008, 52, 1188–1197. [Google Scholar] [CrossRef]
- Asagiri, K.; Nakatsuka, M.; Konishi, H.; Noguchi, S.; Takata, M.; Habara, T.; Kudo, T. Involvement of peroxynitrite in LPS-induced apoptosis of trophoblasts. J. Obstet. Gynaecol. Res. 2003, 29, 49–55. [Google Scholar] [CrossRef]
- Takumida, M.; Popa, R.; Anniko, M. Lipopolysaccharide-induced expression of reactive oxygen species and peroxynitrite in the guinea pig vestibular organ. ORL J. Otorhinolaryngol. Relat. Spec. 1998, 60, 254–262. [Google Scholar] [CrossRef]
- Srikun, D.; Miller, E.W.; Domaille, D.W.; Chang, C.J. An ICT-Based Approach to Ratiometric Fluorescence Imaging of Hydrogen Peroxide Produced in Living Cells. J. Am. Chem. Soc. 2008, 130, 4596–4597. [Google Scholar] [CrossRef]
- Zhang, J.; Zhen, X.; Zeng, J.; Pu, K. A Dual-Modal Molecular Probe for Near-infrared Fluorescence and Photoacoustic Imaging of Peroxynitrite. Anal. Chem. 2018, 90, 9301–9307. [Google Scholar] [CrossRef]
- Hou, J.-T.; Yang, J.; Li, K.; Liao, Y.-X.; Yu, K.-K.; Yu, X.-Q. A Highly Selective Water-Soluble Optical Probe for Endogenous Peroxynitrite. Chem. Comm. 2014, 50, 9947–9950. [Google Scholar] [CrossRef]
- Kim, J.; Park, J.; Lee, H.; Choi, Y.; Ki, Y. A Boronate-Based Fluorescent Probe for the Selective Detection of Cellular Peroxynitrite. Chem. Comm. 2014, 50, 9353–9356. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, F.; Xiong, J.; Mao, Z.; Liu, Z. Investigations of drug-induced liver injury by a peroxynitrite activatable two-photon fluorescence probe. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2020, 246, 118960. [Google Scholar] [CrossRef]
- Xia, L.L.; Tong, Y.; Li, L.S.; Cui, M.Y.; Gu, Y.Q.; Wang, P. A Selective Fluorescent Turn-On Probe for Imaging Peroxynitrite in Living Cells and Drug Damaged Liver Tissues. Talanta 2019, 9140, 431–437. [Google Scholar] [CrossRef]
- Chen, L.; Cui, M.; Chen, J.; Xia, L.; Deng, D.; Gu, Y.; Wang, P. A Novel Highly Selective Fluorescent Probe with New Chalcone Fluorophore for Monitoring and Imaging Endogenous Peroxynitrite in Living Cells and Drug-Damaged Liver Tissue. Talanta 2020, 1, 120934. [Google Scholar] [CrossRef]
- Cui, J.; Zang, S.; Nie, H.; Shen, T.; Jing, J.; Zhang, X. An Ict-Based Fluorescent Probe for Ratiometric Monitoring the Fluctuations of Peroxynitrite in Mitochondria. Sens. Actuators B Chem. 2021, 1, 129069. [Google Scholar] [CrossRef]







Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, Z.; Qian, J.; Ge, Y.; Wang, Y.; Chen, H. A Two-Photon Fluorescent Probe for the Visual Detection of Peroxynitrite in Living Cells and Zebrafish. Molecules 2022, 27, 4858. https://doi.org/10.3390/molecules27154858
Xu Z, Qian J, Ge Y, Wang Y, Chen H. A Two-Photon Fluorescent Probe for the Visual Detection of Peroxynitrite in Living Cells and Zebrafish. Molecules. 2022; 27(15):4858. https://doi.org/10.3390/molecules27154858
Chicago/Turabian StyleXu, Zhencai, Jin Qian, Yufeng Ge, Yalan Wang, and Hongwei Chen. 2022. "A Two-Photon Fluorescent Probe for the Visual Detection of Peroxynitrite in Living Cells and Zebrafish" Molecules 27, no. 15: 4858. https://doi.org/10.3390/molecules27154858
APA StyleXu, Z., Qian, J., Ge, Y., Wang, Y., & Chen, H. (2022). A Two-Photon Fluorescent Probe for the Visual Detection of Peroxynitrite in Living Cells and Zebrafish. Molecules, 27(15), 4858. https://doi.org/10.3390/molecules27154858





