1. Introduction
One of the most commonly detected toxins in the environment is Cr(VI), which is extremely hazardous to living things and may cause cancer in people. There are two types of chromium: Cr(VI) and (III). As a nutrient, Cr(III) is vital for animals, due to its ability to conserve active glucose, protein, and lipid metabolism [
1]. In contrast, because it can permeate across cell membranes as CrO
42− or HCrO
4−, Cr(VI) is extremely harmful. The consumption of water contaminated with Cr(VI) poses health problems due to its toxicity [
2]. The permissible limit concentration for Cr(VI) ions in drinking water is less than 50 µg/L, as recommended by the World Health Organization (WHO) and the United States Environmental Protection Agency (USEPA) [
3,
4]. In industrial operations such as the tanning of leather, electroplating, preservation of wood, dye manufacture, and the production of paint and paper, as well as petroleum refining activities, chromium is produced in significant quantities. Concentrations of Cr in states of (VI) and (III) in the effluents from these businesses range from tens to hundreds of milligrams per litre, depending on the process.
Skin irritation, lung tumours, and kidney, liver, and stomach damage are all possible side effects of Cr(VI), which is 500 times more dangerous in its hexavalent form than its trivalent counterpart [
5].
The molecular systems that regulate lipid, glucose, and protein metabolism depend on Cr(III) and Cr(VI) regularly. Due to the unique chemical characteristics of Cr (III) and Cr (VI), the separation of the two Cr species has become extremely significant [
6,
7]. Metals can be identified with high precision and accuracy using flame atomic absorption spectrometry (FAAS). However, it lacks sufficient sensitivity to detect traces of components. An effective method of pre-concentration is therefore typically necessary. FAAS is unable to distinguish between distinct chemical species of analyses and can only detect the overall quantity of analyte. Chromate ion-selective sorption has been effectively achieved using flow injection (FI) through line column pre-concentration procedures that utilize solid phase extraction (SPE) [
8]. Spectrophotometers are also one of the most essential tools for determining chromium with great precision and accuracy.
Chemical precipitation, ion exchange redox, and adsorption have all been used in the past to remove Cr(VI). When it comes to removing Cr(VI) from industrial effluent, the most successful and straightforward procedure is adsorption [
9,
10]. The adsorption technique requires an inexpensive adsorbent, has a high adsorption capacity, and is readily available. Other management approaches, for example, adsorption membrane, reduction, bioremediation, and photocatalytic, have been advanced to counteract the disadvantages of the precipitation process [
11,
12,
13]. Adsorption has become more popular due to its wide variety of applications, high efficiency, low cost, and remarkable reproducibility [
14,
15,
16,
17]. The contact between the active groups (for example, carboxyl, ammonium groups, and hydroxyl) in the adsorbents and the pore volumes of those materials during the physical adsorption phase of the process is critical to the elimination of Cr(VI) [
18,
19]. Carbon materials have become a good choice for adsorbents [
20,
21,
22] because they have a lot of pores, a lot of active groups, strong properties, and good thermal stability.
Chemical interaction between any amine of primary type and a ketone or an aldehyde under mild conditions produces Schiff bases, commonly known as imines, which have an imine or azomethine (C=N) group. They can be used in a variety of industries, including the production of thermally resistant, semi-conductive, and anticorrosive materials. Due to their antimicrobial, antimalarial, and antibacterial characteristics, the synthesis of Schiff bases has been highlighted in biological simulations, the design of molecular magnets, and the production of pharmaceuticals [
23,
24,
25]. Schiff bases are very important in coordination chemistry due to their ability to act as donors.
To date, Schiff bases have been one of the most popular and thoroughly studied molecular chemo-sensors for choosy sensing of metal ions due to their simplicity of synthesis as well as their good biological activity and strong photophysical characteristics [
26,
27,
28]. Schiff bases with something like a C=N bridge can easily isomerize in the excitation state and have extremely weak fluorescence because of the C=N isomerization [
29,
30].
However, when this efficient group can coordinate and form a complex with metal ions, the C=N isomerization is prevented, resulting in considerably brighter fluorescence signals. Due to their ease of synthesis, electrical characteristics, and high solubility, Schiff base ligands have been widely explored in coordination chemistry. The stability of these imine ligands under a wide range of oxidative and reductive conditions shows the border between hard and soft Lewis bases, which provides a borderline. Biochemical, analytical, and antibacterial reagents are becoming increasingly significant [
31,
32].
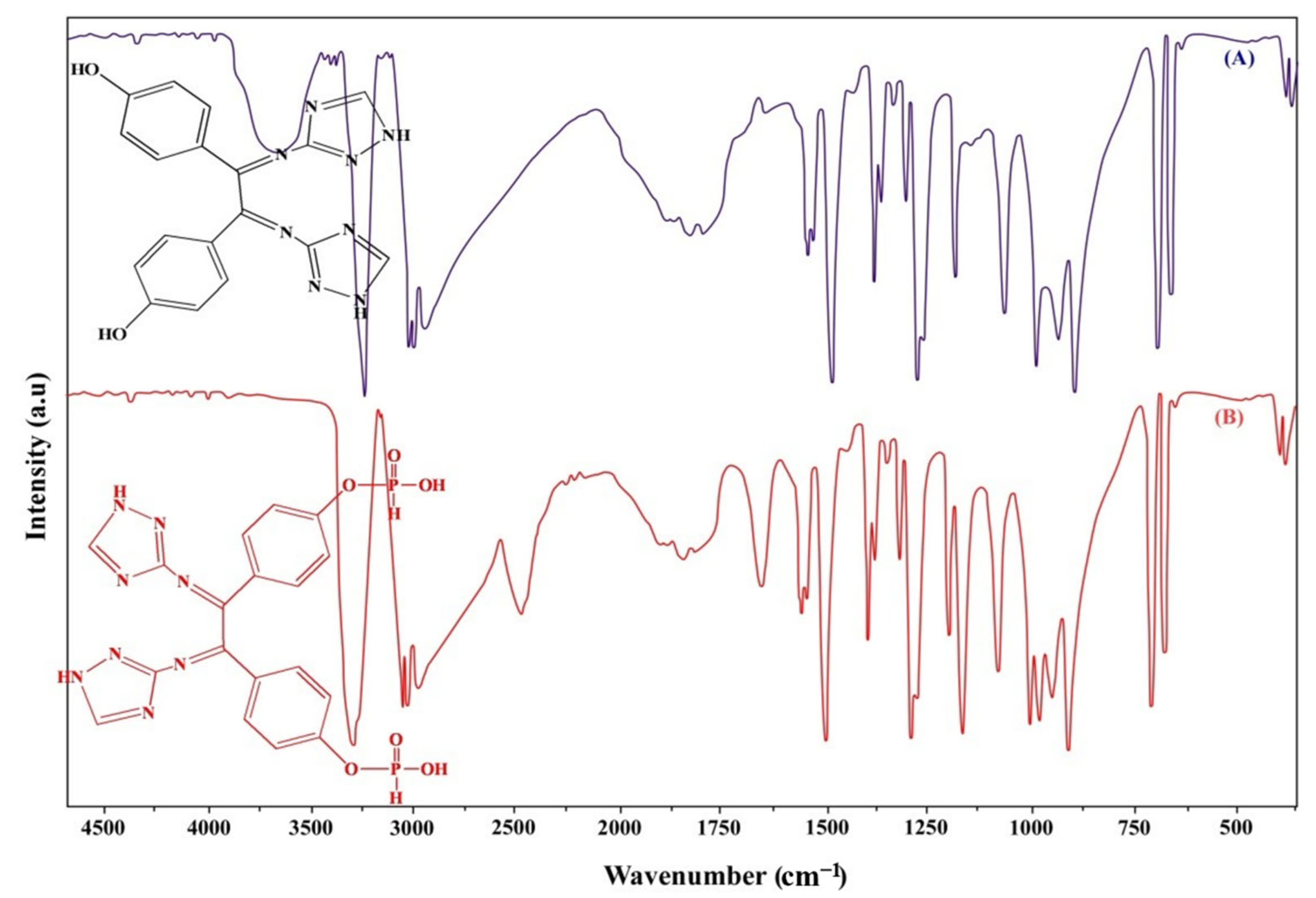
In this paper, the 3-Amino-1H-1,2,4-triazole and 4,4’-Dihydroxybenzil were used as the starting material to synthesise a new Schiff base structure, TIHP. The new Schiff base structure was then subjected to a phosphorylation reaction to increase the adsorption capacity. Scanning electron microscopy (SEM), Fourier transform infrared spectrometer (FT-IR), Brunauer-Emmett-Teller specific surface area analyzer (BET), solid-state 13C nuclear magnetic resonance (13CNMR), and X-rays were used to figure out the physical and chemical properties of the new adsorbent materials, TIHP. The adsorption performance of the TIHP materials was tested by static adsorption experiments.
2. Materials and Methods
2.1. Chemicals Utilized and Used Reagent
The aluminium chloride (AlCl3) and sodium hydroxide were supplied by Scharlau Chemie Company S.A. (Barcelona, Spain), 3-Amino-1H-1,2,4-triazole was purchased from Thermo Fisher Scientific-Acros Organics Inc. ((Morris Plains, NJ, USA), 4,4’-Dihydroxybenzil and DMF were obtained from Fluka (Charlotte, NC, England). Ethanol, isopropanol and phosphoryl trichloride were obtained from Sigma-Aldrich (St. Louis, MO, USA). Prior to usage, the solvents were purified and decontaminated using fixed scientific techniques and then newly distilled to ensure extreme effectiveness. All procedures were carried out in flame-dried glassware. The progress of the reaction was detected using thin paper chromatography (PC). The mixture of (ethyl acetate + ethanol 50:50 v/v) as an eluent has been used as spots on the thin paper chromatography plates which have been identified with a simple UV lamp at 250 nm.
2.2. Equipment
FTIR and SEM were applied to examine the new adsorbent (a Hitachi S-4160 scanning electron microscope). The concentration of metal ions was determined via inductively coupled plasma (ICP-AES) (Varian liberty 150 XL).
The Perkin-Elmer Frontier was applied to take FT-IR spectra. Dimethyl sulfoxide (DMSO) or chloroform (CDCl3) were applied as solvent materials for routine spectra of NMR at ambient temp. on an Avance TM 400 spectrometer. It is important to note that all chemical modifications are stated in ppm δ relative to the trace resonance of protonated chloroform, CDCl3 dimethyl sulfoxide, DMSO, and external 85% aqueous H3PO4, as (δ 7.25 ppm), (δ 77.0 ppm), or (δ 2.50 ppm), (δ 39.51 ppm) and (δ 0.0 ppm), respectively.
A mass spectrum was examined by using a GC Finnigan MAT SSQ-7000 type of mass spectrometer. The progress of the reactions and examination of the compound’s purity was completed. The latter was done via thin Layer Chromatography (TLC) on silica gel-precoated aluminium sheets (Type 60, F 254, Merck, Darmstadt, Germany) with an eluent of petroleum ether (60–80 °C)/ethyl acetate, and the spots were identified by exposure to UV light at a lamp at λ254 nm for several seconds. The chemical names of the synthesized chemicals are designated using the IUPAC nomenclature. Conventional drying and purification processes were utilized.
2.3. Batch Adsorption
The standard solution of 1000 mg/L of chromium was made via dissolving 2.82 g of K2Cr2O7 in 1 L of distilled water and then diluting it. The dilution principle was used to sequentially dilute this stock.
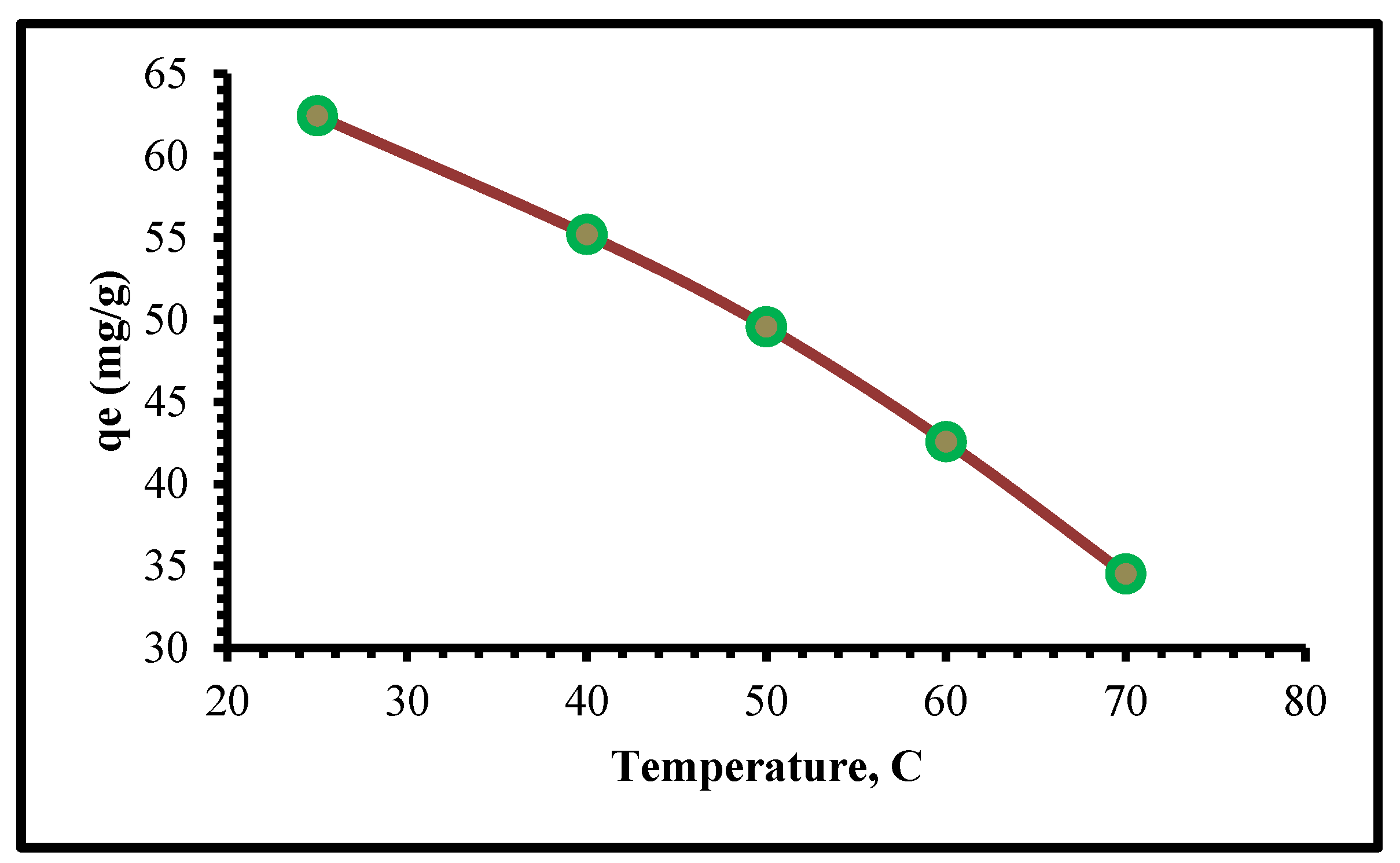
The aqueous solution was reserved in interaction with a suitable dosage of TIHP in an Erlenmeyer (100 mL) in a shaker-incubator instrument type (FTSH-301 MINI (SP)) at 150 rpm for the batch-mode adsorption tests. Before we start the experimental adsorption studies, we start studying the best-controlling factors affecting the adsorption of Cr(VI), each separately, a single experiment for adsorption of Cr(VI) using 0.1 g/L TIHP, in the pH range of 2–7 with an initial Cr(VI) concentration that is 250 mg/L, shaking for 30 min at 25 °C temperature.
When adjusting the pH, NaOH and HCl solutions were used. The second series of adsorption studies were done in the dosage variety of 0.01–0.25 g/L at the best pH to find the optimum adsorbent dose. There were no new operating settings in this series of tests. Temperatures of 25 °C, initial concentration of 250 mg/L, and time intervals of 5 min to 90 min were used in the kinetic tests. The starting concentration of Cr(VI) was ranging from 250 to 1500 mg/L during the equilibrium trials. The adsorption kinetics were used to determine the equilibrium time. Adsorbents were tested for their ability to adsorb Cr(VI) and another interfering ion in different binary adsorption studies. The final Cr(VI) concentrations were determined by atomic absorption spectroscopy (Varian AA240) and UV-VIS spectroscopy type (Rigol-Ultra-3660 spectrophotometer at the extreme wavelength (λ = 590 nm)) in the multicomponent adsorption studies. Every experiment was attended twice and the average values were provided. Here is how the adsorption capacity and efficiency of Cr(VI) ions were considered using equations (1) and (2), which were used to mathematically calculate the adsorption capability (q
t (mg/g)), as follows:
For a given pollutant concentration, Co (mg/L) is the starting pollutant concentration, Ct (mg/L) is the pollutant concentration at a specified time, Ce (mg/L) is the equilibrium pollution concentration, V is the volume of solution (L), and m denotes the mass of adsorbent (g).
2.4. Determination of Cr(VI) Concentration
The content of Cr(VI) was determined using the 1,5-diphenylcarbazide method [
33], which involved determining the absorbance of each sample at (540 nm) with a Hitachi U-2900 UV-Vis spectrophotometer equipped (Tokyo, Japan).
To determine the presence of chromium Cr(VI), the certified analytical methods of the Society of Leather Technologists and Chemists [
34,
35] were followed. The material (25 mL) was placed in an Erlenmeyer flask (250 mL) and treated with HNO
3 (15 mL), followed by an HClO
4/H
2SO
4 mixture (15 mL). After that, the mixture was slightly heated until it turned a bright orange-red color, after which it was left to boil for another minute. This was followed by eliminating the flask from the heat source and fast cooling it in a cold water bath while it was whirling around. Following that, 75 mL of distilled water was wisely added along with a few glass beads (anti-bumping granules) and the mixture was brought to a boil for 10 min to eliminate any remaining free chlorine. Slowly, 10 mL of 30 percent H
2SO
4 was added to the liquid, which was then cooled to 20 °C. The working solution was titrated with newly made ferrous ammonium sulphate in which N-phenyl anthranilic acid acted as an indicator. It was signified by a color shifted from a violet color to a green one, and then the Cr(VI) concentration was mathematically calculated.
2.5. Preparation of TIHP
At first, 3-Amino-1H-1,2,4-triazole (0.1 mol/L) was measured in a 250 mL three-neck flask. Then, 60 mL of ethanol was added and stirred until 3-Amino-1H-1,2,4-triazole was completely dissolved. A 30 mL ethanolic solution of 4,4’-Dihydroxybenzil (1 mmol, 1.02 g) was added to the ethanolic solution of 3-Amino-1H-1,2,4-triazole. The mixture was stirred to reflux at 150 ℃ for 5 h. The precipitate was filtered off and recrystallized from the mixed solution of ethanol and isopropanol to obtain a pale yellowish precipitate formed and 4,4’-((1E)-1,2-bis((1H-1,2,4-triazol-3-yl)imino)ethane-1,2-diyl)diphenol was obtained.
The produced Schiff base was then subjected to phosphorylation reaction via interaction with 0.15 g AlCl
3 (anhydrous) with 20 mL phosphorus oxychloride for 30 h at the temperature of 115 °C. A tail gas absorber to absorb HCl in the reaction process was installed. Then, in an ice bath, 200 mL of distilled water was gently added to the resulting mixture slowly drop by drop. The working mixture was filtered and drained with distilled water several times and dried in a vacuum at 80 °C for 30 h. Finally, a white precipitate of 4-((1
E)-1,2-bis((1
H-1,2,4-triazol-3-yl)imino)-2-(4-((hydroxyhydrophosphoryl) oxy) phenyl) ethyl) phenyl hydrogen phosphonate TIHP was obtained. The synthesis process is shown in
Scheme 1.
FTIR (Nicolet 6700 spectrometer), mass spectra, 13C analysis, and 31P NMR studies were used to characterize the produced product. A Bruker instrument, AVANCE III HD 400, was used for the NMR analysis.