Potential Therapeutic Mechanism of Traditional Chinese Medicine on Diabetes in Rodents: A Review from an NMR-Based Metabolomics Perspective
Abstract
:1. Introduction
2. Metabolomics as a Powerful Tool for the Modernization of TCM
3. Potential Metabolic Mechanisms of TCM on Diabetes Care
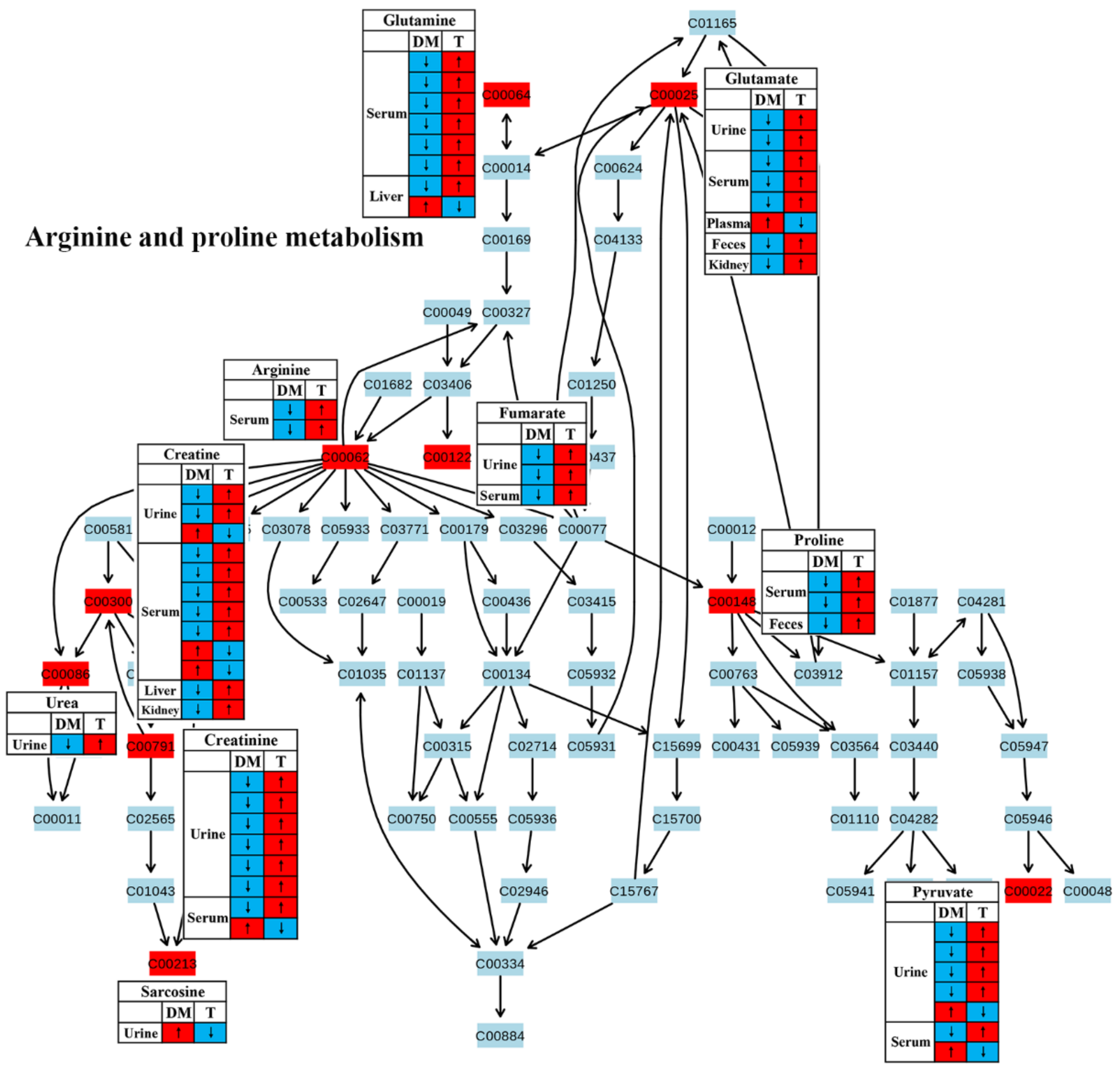
3.1. Amino Acid Metabolism
3.2. Energy Metabolism
3.3. Synthesis and Degradation of Ketone Bodies
3.4. Taurine and Hypotaurine Metabolism
4. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- International Diabetes Federation. IDF Diabetes Atlas, 10th ed.; International Diabetes Federation: Brussels, Belgium, 2021; Available online: https://www.diabetesatlas.org (accessed on 11 March 2022).
- Tong, X.L.; Dong, L.; Chen, L.; Zhen, Z. Treatment of diabetes using traditional Chinese medicine: Past, present and future. Am. J. Chin. Med. 2012, 40, 877–886. [Google Scholar] [CrossRef] [PubMed]
- Covington, M.B. Traditional Chinese medicine in the treatment of diabetes. Diabetes Spectr. 2001, 14, 154–159. [Google Scholar] [CrossRef]
- Bai, L.; Li, X.; He, L.; Zheng, Y.; Lu, H.; Li, J.; Zhong, L.; Tong, R.; Jiang, Z.; Shi, J.; et al. Antidiabetic potential of flavonoids from traditional Chinese medicine: A review. Am. J. Chin. Med. 2019, 47, 933–957. [Google Scholar] [CrossRef] [PubMed]
- Li, W.F.; Jiang, J.G.; Chen, J. Chinese medicine and its modernization demands. Arch. Med. Res. 2008, 39, 246–251. [Google Scholar] [CrossRef]
- Qiu, J. China plans to modernize traditional medicine. Nature 2007, 446, 590–592. [Google Scholar] [CrossRef]
- Cai, F.F.; Zhou, W.J.; Wu, R.; Su, S.B. Systems biology approaches in the study of Chinese herbal formulae. Chin. Med. 2018, 13, 65. [Google Scholar] [CrossRef]
- Patti, G.J.; Yanes, O.; Siuzdak, G. Innovation: Metabolomics: The apogee of the omics trilogy. Nat. Rev. Mol. Cell Biol. 2012, 13, 263–269. [Google Scholar] [CrossRef]
- Jiang, W.Y. Therapeutic wisdom in traditional Chinese medicine: A perspective from modern science. Trends Pharmacol. Sci. 2005, 26, 558–563. [Google Scholar] [CrossRef]
- Wang, M.; Lamers, R.-J.A.N.; Korthout, H.A.A.J.; Van Nesselrooij, J.H.J.; Witkamp, R.F.; Van Der Heijden, R.; Voshol, P.J.; Havekes, L.M.; Verpoorte, R.; Van Der Greef, J. Metabolomics in the context of systems biology: Bridging traditional Chinese medicine and molecular pharmacology. Phytother. Res. 2005, 19, 173–182. [Google Scholar] [CrossRef]
- Zheng, H.; Clausen, M.; Dalsgaard, T.; Bertram, H. Metabolomics to explore impact of dairy intake. Nutrients 2015, 7, 4875–4896. [Google Scholar] [CrossRef]
- Zheng, H.; Ji, H.; Fan, K.; Xu, H.; Huang, Y.; Zheng, Y.; Xu, Q.; Li, C.; Zhao, L.; Li, Y.; et al. Targeting Gut microbiota and host metabolism with Dendrobium officinale dietary fiber to prevent obesity and improve glucose homeostasis in diet-induced obese mice. Mol. Nutr. Food Res. 2022, 66, 2100772. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Gao, H.; Zhao, Y.; Lin, D. Metabonomic analysis of the therapeutic effect of Zhibai Dihuang Pill in treatment of streptozotocin-induced diabetic nephropathy. J. Ethnopharmacol. 2012, 142, 647–656. [Google Scholar] [CrossRef] [PubMed]
- Tian, N.; Wang, J.; Wang, P.; Song, X.; Yang, M.; Kong, L. NMR-based metabonomic study of Chinese medicine Gegen Qinlian Decoction as an effective treatment for type 2 diabetes in rats. Metabolomics 2013, 9, 1228–1242. [Google Scholar] [CrossRef]
- Perumal, V.; Murugesu, S.; Lajis, N.H.; Khatib, A.; Saari, K.; Abdul-Hamid, A.; Khoo, W.C.; Mushtaq, M.Y.; Abas, F.; Ismail, I.S.; et al. Evaluation of antidiabetic properties of Momordica charantia in streptozotocin induced diabetic rats using metabolomics approach. Int. Food Res. J. 2015, 22, 1298–1306. [Google Scholar]
- Mediani, A.; Abas, F.; Maulidiani, M.; Khatib, A.; Tan, C.P.; Ismail, I.S.; Shaari, K.; Ismail, A.; Lajis, N.H. Metabolic and biochemical changes in streptozotocin induced obese-diabetic rats treated with Phyllanthus niruri extract. J. Pharm. Biomed. Anal. 2016, 128, 302–312. [Google Scholar] [CrossRef]
- Akhtar, M.T.; Sarib, M.S.B.M.; Ismail, I.S.; Abas, F.; Ismail, A.; Lajis, N.H.; Shaari, K. Anti-diabetic activity and metabolic changes induced by Andrographis Paniculata plant extract in obese diabetic rats. Molecules 2016, 21, 1026. [Google Scholar] [CrossRef]
- Maulidiani; Abas, F.; Khatib, A.; Perumal, V.; Suppaiah, V.; Ismail, A.; Hamid, M.; Shaari, K.; Lajis, N. Metabolic alteration in obese diabetes rats upon treatment with Centella asiatica extract. J. Ethnopharmacol. 2016, 180, 60–69. [Google Scholar] [CrossRef]
- Shen, X.L.; Liu, H.; Xiang, H.; Qin, X.M.; Du, G.H.; Tian, J.S. Combining biochemical with 1H NMR-based metabolomics approach unravels the antidiabetic activity of genipin and its possible mechanism. J. Pharm. Biomed. Anal. 2016, 129, 80–89. [Google Scholar] [CrossRef]
- Azam, A.A.; Pariyani, R.; Ismail, I.S.; Ismail, A.; Khatib, A.; Abas, F.; Shaari, K. Urinary metabolomics study on the protective role of Orthosiphon stamineus in Streptozotocin induced diabetes mellitus in rats via 1H NMR spectroscopy. BMC Complement. Altern. Med. 2017, 17, 278. [Google Scholar] [CrossRef]
- Zheng, H.; Pan, L.; Xu, P.; Zhu, J.; Wang, R.; Zhu, W.; Hu, Y.; Gao, H. An NMR-based metabolomic approach to unravel the preventive effect of water-soluble extract from Dendrobium officinale Kimura & Migo on streptozotocin-induced diabetes in mice. Molecules 2017, 22, 1543. [Google Scholar]
- AL-Zuaidy, M.H.; Mumtaz, M.W.; Hamid, A.A.; Ismail, A.; Mohamed, S.; Razis, A.F.A. Biochemical characterization and 1H NMR based metabolomics revealed Melicopelunu-ankenda leaf extract a potent anti-diabetic agent in rats. BMC Complement. Altern. Med. 2017, 17, 359. [Google Scholar] [CrossRef] [PubMed]
- Sajak, A.A.B.; Mediani, A.; Maulidiani; Dom, N.S.M.; Machap, C.; Hamid, M.; Ismail, A.; Khatib, A.; Abas, F. Effect of Ipomoea aquatica ethanolic extract in streptozotocin (STZ) induced diabetic rats via 1H NMR-based metabolomics approach. Phytomedicine 2017, 36, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.S.; Zhao, L.; Shen, X.L.; Liu, H.; Qin, X.M. 1H NMR-based metabolomics approach to investigating the renal protective effects of Genipin in diabetic rats. Chin. J. Nat. Med. 2018, 16, 261–270. [Google Scholar] [CrossRef]
- Li, H.; Chu, S.; Zhao, H.; Liu, D.; Liu, X.; Qu, X.; Chen, J.; Li, Z.; Li, J. Effect of Zishen Jiangtang pill, a Chinese herbal product, on rats with diabetic osteoporosis. Evid. Based Complement. Altern. Med. 2018, 2018, 7201914. [Google Scholar] [CrossRef]
- Gao, K.; Yang, R.; Zhang, J.; Wang, Z.; Jia, C.; Zhang, F.; Li, S.; Wang, J.; Murtaza, G.; Xie, H.; et al. Effects of Qijian mixture on type 2 diabetes assessed by metabonomics, gut microbiota and network pharmacology. Pharmacol. Res. 2018, 130, 93–109. [Google Scholar] [CrossRef]
- Singh, A.K.; Raj, V.; Keshari, A.K.; Rai, A.; Kumar, P.; Rawat, A.; Maity, B.; Kumar, D.; Prakash, A.; De, A.; et al. Isolated mangiferin and naringenin exert antidiabetic effect via PPAR γ/GLUT4 dual agonistic action with strong metabolic regulation. Chem. Biol. Interact. 2018, 280, 33–44. [Google Scholar] [CrossRef]
- Chen, M.; Xiao, D.; Liu, W.; Song, Y.; Zou, B.; Li, L.; Li, P.; Cai, Y.; Liu, D.; Liao, Q.; et al. Intake of Ganoderma lucidum polysaccharides reverses the disturbed gut microbiota and metabolism in type 2 diabetic rats. Int. J. Biol. Macromol. 2020, 155, 890–902. [Google Scholar] [CrossRef]
- Su, Z.; Ling, X.; Ji, K.; Huang, H.; Liu, X.; Yin, C.; Zhu, H.; Guo, Y.; Mo, Y.; Lu, Y.; et al. 1H NMR-based urinary metabonomic study of the antidiabetic effects of Rubus Suavissimus S. Lee in STZ-induced T1DM rats. J. Chromatogr. B 2020, 1158, 122347. [Google Scholar] [CrossRef]
- Niu, W.; Miao, J.; Li, X.; Guo, Q.; Deng, Z.; Wu, L. Metabolomics combined with systematic pharmacology reveals the therapeutic effects of Salvia miltiorrhiza and Radix Pueraria lobata herb pair on type 2 diabetes rats. J. Func. Foods 2022, 89, 104950. [Google Scholar] [CrossRef]
- Chen, K.; Wei, X.; Zhang, J.; Pariyani, R.; Jokioja, J.; Kortesniemi, M.; Linderborg, K.M.; Heinonen, J.; Sainio, T.; Zhang, Y.; et al. Effects of anthocyanin extracts from bilberry (Vaccinium myrtillus L.) and purple potato (Solanum tuberosum L. var. ‘SynkeäSakari’) on the plasma metabolomic profile of Zucker diabetic fatty rats. J. Agric. Food Chem. 2020, 68, 9436–9450. [Google Scholar] [CrossRef]
- Du, H.; Li, Q.; Yi, H.; Xu, T.; Xu, X.M.; Kuang, T.T.; Zhang, J.; Huang, A.Q.; Fan, G. Anti-diabetic effects of Berberis kansuensis extract on type 2 diabetic rats revealed by 1H-NMR-based metabolomics and biochemistry analysis. Chem. Biodiver. 2020, 17, e2000413. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Niu, W.; Li, X.; Guo, H.; Zhang, N.; Wang, X.; Wu, L. Study on hypoglycemic effect of the drug pair of Astragalus radix and Dioscoreaerhizoma in T2DM rats by network pharmacology and metabonomics. Molecules 2019, 24, 4050. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Lu, B.; Gong, J.; Li, L.; Chen, G.; Zhang, J.; Chen, Y.; Tian, X.; Han, B.; Guo, Y.; et al. Chickpea extract ameliorates metabolic syndrome symptoms via restoring intestinal ecology and metabolic profile in type 2 diabetic rats. Mol. Nutr. Food Res. 2021, 65, 2100007. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.-R.; Yoon, D.; Kim, H.-G.; Oh, S.; Yoo, Y.; Lee, Y.-S.; Kim, K.-W.; Yi, T.-H.; Lee, D. NMR-based metabolomics approach to investigate the effects of fruits of Acanthopanax sessiliflorus in a high-fat diet induced mouse model. Metabolites 2021, 11, 505. [Google Scholar] [CrossRef] [PubMed]
- Guo, F.; Han, M.; Lin, S.; Ye, H.; Chen, J.; Zhu, H.; Lin, W. Enteromorpha prolifera polysaccharide prevents high-fat diet-induced obesity in hamsters: A NMR-based metabolomic evaluation. J. Food Sci. 2021, 86, 3672–3685. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Zhao, C.; Zhang, Y.; Du, H.; Xu, T.; Xu, X.; Zhang, J.; Kuang, T.; Lai, X.; Fan, G.; et al. 1H NMR-based metabolomics coupled with molecular docking reveal the anti-diabetic effects and potential active components of Berberis vernae on type 2 diabetic rats. Front. Pharmacol. 2020, 11, 932. [Google Scholar] [CrossRef]
- Pang, Z.; Chong, J.; Zhou, G.; de Lima Morais, D.A.; Chang, L.; Barrette, M.; Gauthier, C.; Jacques, P.-É.; Li, S.; Xia, J. MetaboAnalyst 5.0: Narrowing the gap between raw spectra and functional insights. Nucleic Acids Res. 2021, 49, W388–W396. [Google Scholar] [CrossRef]
- Newsholme, P.; Bender, K.; Kiely, A.; Brennan, L. Amino acid metabolism, insulin secretion and diabetes. Biochem. Soc. Trans. 2007, 35, 1180–1186. [Google Scholar] [CrossRef]
- Wang-Sattler, R.; Yu, Z.H.; Herder, C.; Messias, A.C.; Floegel, A.; He, Y.; Heim, K.; Campillos, M.; Holzapfel, C.; Thorand, B.; et al. Novel biomarkers for pre-diabetes identified by metabolomics. Mol. Syst. Biol. 2012, 8, 615. [Google Scholar] [CrossRef]
- El-Hafidi, M.; Franco, M.; Ramírez, A.R.; Sosa, J.S.; Flores, J.A.P.; Acosta, O.L.; Salgado, M.C.; Cardoso-Saldaña, G. Glycine increases insulin sensitivity and glutathione biosynthesis and protects against oxidative stress in a model of sucrose-induced insulin resistance. Oxid. Med. Cell. Longev. 2018, 2018, 2101562. [Google Scholar] [CrossRef]
- Tuttle, R.L.; Gill, N.S.; Pugh, W.; Lee, J.P.; Koeberlein, B.; Furth, E.E.; Polonsky, K.S.; Naji, A.; Birnbaum, M.J. Regulation of pancreatic β-cell growth and survival by the serine/threonine protein kinase Akt1/PKBα. Nat. Med. 2001, 7, 1133–1137. [Google Scholar] [CrossRef] [PubMed]
- Copps, K.D.; White, M.F. Regulation of insulin sensitivity by serine/threonine phosphorylation of insulin receptor substrate proteins IRS1 and IRS2. Diabetologia 2012, 55, 2565–2582. [Google Scholar] [CrossRef] [PubMed]
- Brennan, L.; Shine, A.; Hewage, C.; Malthouse, J.P.; Brindle, K.M.; McClenaghan, N.; Flatt, P.R.; Newsholme, P. A nuclear magnetic resonance-based demonstration of substantial oxidative l-alanine metabolism and l-alanine-enhanced glucose metabolism in a clonal pancreatic β-cell line: Metabolism of l-alanine is important to the regulation of insulin secretion. Diabetes 2002, 51, 1714–1721. [Google Scholar] [CrossRef]
- Maechler, P.; Wollheim, C.B. Mitochondrial glutamate acts as a messenger in glucose-induced insulin exocytosis. Nature 1999, 402, 685–689. [Google Scholar] [CrossRef] [PubMed]
- Di Sebastiano, K.M.; Bell, K.E.; Barnes, T.; Weeraratne, A.; Premji, T.; Mourtzakis, M. Glutamate supplementation is associated with improved glucose metabolism following carbohydrate ingestion in healthy males. Br. J. Nutr. 2013, 110, 2165–2172. [Google Scholar] [CrossRef] [PubMed]
- Gheni, G.; Ogura, M.; Iwasaki, M.; Yokoi, N.; Minami, K.; Nakayama, Y.; Harada, K.; Hastoy, B.; Wu, X.; Takahashi, H.; et al. Glutamate acts as a key signal linking glucose metabolism to incretin/cAMP action to amplify insulin secretion. Cell Rep. 2014, 9, 661–673. [Google Scholar] [CrossRef] [PubMed]
- Monti, L.D.; Setola, E.; Lucotti, P.C.G.; Marrocco-Trischitta, M.M.; Comola, M.; Galluccio, E.; Poggi, A.; Mammì, S.; Catapano, A.L.; Comi, G.; et al. Effect of a long-term oral l-arginine supplementation on glucose metabolism: A randomized, double-blind, placebo-controlled trial. Diabetes Obes. Metab. 2012, 14, 893–900. [Google Scholar] [CrossRef] [PubMed]
- Krause, M.S.; McClenaghan, N.H.; Flatt, P.R.; de Bittencourt, P.I.H.; Murphy, C.; Newsholme, P. L-arginine is essential for pancreatic β-cell functional integrity, metabolism and defense from inflammatory challenge. J. Endocrinol. 2011, 211, 87–97. [Google Scholar] [CrossRef]
- Clemmensen, C.; Madsen, A.N.; Smajilovic, S.; Holst, B.; Bräuner-Osborne, H. L-arginine improves multiple physiological parameters in mice exposed to diet-induced metabolic disturbances. Amino Acids 2012, 43, 1265–1275. [Google Scholar] [CrossRef]
- Lu, H.; Koshkin, V.; Allister, E.M.; Gyulkhandanyan, A.V.; Wheeler, M.B. Molecular and metabolic evidence for mitochondrial defects associated with beta-cell dysfunction in a mouse model of type 2 diabetes. Diabetes 2010, 59, 448–459. [Google Scholar] [CrossRef]
- Kwak, S.H.; Park, K.S.; Lee, K.U.; Lee, H.K. Mitochondrial metabolism and diabetes. J. Diabetes Investig. 2010, 1, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Szendroedi, J.; Phielix, E.; Roden, M. The role of mitochondria in insulin resistance and type 2 diabetes mellitus. Nat. Rev. Endocrinol. 2011, 8, 92–103. [Google Scholar] [CrossRef] [PubMed]
- Rutter, G.A.; Georgiadou, E.; Martinez-Sanchez, A.; Pullen, T.J. Metabolic and functional specialisations of the pancreatic beta cell: Gene disallowance, mitochondrial metabolism and intercellular connectivity. Diabetologia 2020, 63, 1990–1998. [Google Scholar] [CrossRef] [PubMed]
- Newman, J.C.; Verdin, E. Ketone bodies as signaling metabolites. Trends Endocrinol. Metab. 2014, 25, 42–52. [Google Scholar] [CrossRef]
- Fukao, T.; Lopaschuk, G.D.; Mitchell, G.A. Pathways and control of ketone body metabolism: On the fringe of lipid biochemistry. Prostag. Leukotr. Essen. Fatty Acids 2004, 70, 243–251. [Google Scholar] [CrossRef]
- Keller, U.; Lustenberger, M.; Stauffacher, W. Effect of insulin on ketone body clearance studied by a ketone body “clamp” technique in normal man. Diabetologia 1988, 31, 24–29. [Google Scholar] [CrossRef]
- Mahendran, Y.; Vangipurapu, J.; Cederberg, H.; Stančáková, A.; Pihlajamäki, J.; Soininen, P.; Kangas, A.J.; Paananen, J.; Civelek, M.; Saleem, N.K.; et al. Association of ketone body levels with hyperglycemia and type 2 diabetes in 9,398 Finnish men. Diabetes 2013, 62, 3618–3626. [Google Scholar] [CrossRef]
- Batista, T.M.; Ribeiro, R.A.; Amaral, A.G.; de Oliveira, C.A.; Boschero, A.C.; Carneiro, E.M. Taurine supplementation restores glucose and carbachol-induced insulin secretion in islets from low-protein diet rats: Involvement of Ach-M3R, Synt 1 and SNAP-25 proteins. J. Nutr. Biochem. 2012, 23, 306–312. [Google Scholar] [CrossRef]
- Cappelli, A.P.; Zoppi, C.C.; Barbosa-Sampaio, H.C.; Costa, J.M., Jr.; Protzek, A.O.; Morato, P.N.; Boschero, A.C.; Carneiro, E.M. Taurine-induced insulin signalling improvement of obese malnourished mice is associated with redox balance and protein phosphatases activity modulation. Liver Int. 2014, 34, 771–783. [Google Scholar] [CrossRef]
- Chen, W.; Guo, J.; Zhang, Y.; Zhang, J. The beneficial effects of taurine in preventing metabolic syndrome. Food Funct. 2016, 7, 1849–1863. [Google Scholar] [CrossRef]
- Wu, N.; Lu, Y.; He, B.; Zhang, Y.Y.; Lin, J.F.; Zhao, S.; Zhang, W.; Li, Y.; Han, P. Taurine prevents free fatty acid-induced hepatic insulin resistance in association with inhibiting JNK1 activation and improving insulin signaling in vivo. Diabetes Res. Clin. Prac. 2010, 90, 288–296. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, E.M.; Latorraca, M.Q.; Araujo, E.; Beltrá, M.; Oliveras, M.J.; Navarro, M.; Berná, G.; Bedoya, F.J.; Velloso, L.A.; Soria, B.; et al. Taurine supplementation modulates glucose homeostasis and islet function. J. Nutr. Biochem. 2009, 20, 503–511. [Google Scholar] [CrossRef] [PubMed]
- L’Amoreaux, W.J.; Cuttitta, C.; Santora, A.; Blaize, J.F.; Tachjadi, J.; El Idrissi, A. Taurine regulates insulin release from pancreatic beta cell lines. J. Biomed. Sci. 2010, 17, S11. [Google Scholar] [CrossRef] [PubMed]
- Zierer, J.; Jackson, M.A.; Kastenmüller, G.; Mangino, M.; Long, T.; Telenti, A.; Mohney, R.P.; Small, K.S.; Bell, J.T.; Steves, C.J.; et al. The fecal metabolome as a functional readout of the gut microbiome. Nat. Genet. 2018, 50, 790–795. [Google Scholar] [CrossRef]











| NMR | MS | |
|---|---|---|
| Advantage | High reproducibility | High sensitivity |
| Minimal sample preparation | More metabolite detection | |
| Non-destructive | Flexible technique | |
| Good quantitative analysis | Minimal sample size | |
| No separation and fast analysis | ||
| Good software/database for identification | ||
| Disadvantage | Relatively low sensitivity | Low reproducibility |
| Larger sample size | Sample derivatization for GC-MS | |
| Cannot detect non-protonated metabolites | Sample not recoverable | |
| Relatively poor quantitative analysis | ||
| Difficult identification |
| No. | Metabolite | Chemical Shift (ppm) a | Metabolic Pathway |
|---|---|---|---|
| 1 | 3-Hydroxybutyrate | 1.18(d) | Ketone body metabolism |
| 2 | AMP b | 6.15(d), 8.26(s), 8.58(s) | Energy metabolism |
| 3 | NAG c | 2.05(m), 3.75(m) | - e |
| 4 | α-Glucose | 5.21(d) | Energy metabolism |
| 5 | β-Glucose | 4.65(d) | Energy metabolism |
| 6 | Phenylalanine | 7.37(t), 7.45(t) | Amino acid metabolism |
| 7 | Alanine | 1.48(d) | Amino acid metabolism |
| 8 | Acetone | 2.37(s) | Ketone body metabolism |
| 9 | Pyruvate | 2.40(s) | Energy metabolism |
| 10 | Choline | 3.20(s) | Choline metabolism |
| 11 | LDL/VLDL d | 0.85(m), 1.25(m) | - |
| 12 | Butyrate | 0.89(t), 1.55(m) | Fatty acid metabolism |
| 13 | Glycine | 3.55(s) | Amino acid metabolism |
| 14 | Glycerol | 3.67(q) | Glycerolipid metabolism |
| 15 | Glutamate | 2.15(m), 3.75(m) | Amino acid metabolism |
| 16 | Glutamine | 2.45(m), 3.78(t) | Amino acid metabolism |
| 17 | Glutathione | 2.15(m) | Amino acid metabolism |
| 18 | Succinate | 2.39(s) | Energy metabolism |
| 19 | Creatine | 3.03(s), 3.93(s) | Energy metabolism |
| 20 | Methanol | 3.35(s) | - |
| 21 | Methylhistidine | 7.05(s) | Amino acid metabolism |
| 22 | Formate | 8.44(s) | Fatty acid metabolism |
| 23 | Lysine | 1.71(m) | Amino acid metabolism |
| 24 | Tyrosine | 6.89(d), 7.20(d) | Amino acid metabolism |
| 25 | Leucine | 0.95(t) | Amino acid metabolism |
| 26 | Uracil | 5.80(d) | Nucleotide metabolism |
| 27 | Citrate | 2.55(d) | Energy metabolism |
| 28 | Taurine | 3.25(t), 3.41(t) | Amino acid metabolism |
| 29 | Glucose/amino acid region | 3.35–3.92(m) | - |
| 30 | Lactate | 1.32(d), 4.11(q) | Energy metabolism |
| 31 | Aspartate | 2.80(d), 3.15(d) | Amino acid metabolism |
| 32 | Valine | 0.98(d), 1.05(d) | Amino acid metabolism |
| 33 | Fumarate | 7.11(s) | Energy metabolism |
| 34 | Acetate | 1.91(s) | Fatty acid metabolism |
| 35 | Isoleucine | 0.99(d) | Amino acid metabolism |
| 36 | Histidine | 7.79(s) | Amino acid metabolism |
| 37 | Tryptophan | 7.34(d) | Amino acid metabolism |
| Treatment | Dose/Time | Model | Type | Glucose Lowering | Sample | Metabolic Change a | Reference |
|---|---|---|---|---|---|---|---|
| Zhibai Dihuang pill | 4 g/kg; 30 days | STZ-induced diabetic nephropathy rats | T1D | Yes, but no significant difference | Urine Serum Kidney | Urine: (↓)3-hydroxybutyrate, lactate Serum: (↑) creatine, methionine, lactate, pyruvate; (↓) VLDL/LDL, 3-hydroxybutyrate Kidney: (↑) betaine, choline, glutamate; (↓)glucose, lactate | [13] |
| Gegen Qinlian decoction | 8 g/kg; 5 weeks | High-fat diet/STZ-induced diabetic rats | T2D | Yes | Plasma | (↑)lipoprotein, valine, TMAO, dimethylamine, arginine; (↓) choline, glucose, glycerol, taurine, creatine, creatinine, tyrosine | [14] |
| Momordica charantia ethanol extract | 200 mg/kg; 1 week | STZ-induced diabetic rats | T1D | Yes | Urine | (↑) succinate, creatine, creatinine, urea, phenylacetylglycine; (↓) lactate, glucose | [15] |
| Phyllanthus niruri ethanol extract | 500 mg/kg; 4 weeks | High-fat diet/STZ-induced diabetic rats | T2D | Yes | Urine Serum | Urine: (↑) hippurate, formate, fumarate, methylnicotinamide, pyruvate, acetone, phenylacetylglycine, allantoin, alanine, succinate, lactate; (↓) glucose, choline, taurine, creatine Serum: (↓) glucose, triglyceride, cholesterol, LDL, HDL | [16] |
| Andrographis paniculata water extract | 200 mg/kg; 4 weeks | High-fat diet/STZ-induced diabetic rats | T2D | Yes | Urine | (↑) lactate, formate, pyruvate, citrate, 2-oxoglutarate, succinate, acetoacetate, 3-hydroxybutyrate, acetate, dimethylglycine, dimethylamine, alanine, allantoin; (↓) glucose, taurine | [17] |
| Centella asiatica ethanol extract | 300 mg/kg; 4 weeks | High-fat diet/STZ-induced diabetic rats | T2D | Yes | Urine Serum | Urine: (↑) pyruvate, lactate, citrate, fumarate, succinate, 2-oxoglutarate, 3-hydoxybutyrate, acetoacetate, acetone, acetate, alanine, hippurate, dimethylamine, creatinine, trimethylamine, allantoin; (↓) glucose Serum: (↑) lactate, choline, succinate; (↓) glucose | [18] |
| Genipin, derived from the fruit of Gardenia jasminoides | 100 mg/kg; 2 weeks | Alloxan-induced diabetic rats | T1D | Yes | Serum | (↑) citrate, succinate, 3-hydroxybutyrate, acetone | [19] |
| Orthosiphon stamineus aqueous extract | 500 mg/kg; 2 weeks | STZ-induced diabetic rats | T1D | Yes | Urine | (↑) hippurate, allantoin, creatinine, glutamate, 3-hydroxybutyrate, pyruvate, citrate; (↓) glucose, taurine, betaine, leucine, acetoacetate | [20] |
| Dendrobium officinale water extract | 700 mg/kg; 2 weeks | STZ-induced diabetic mice | T1D | Yes | Serum Liver | Serum: (↑) citrate, glutamine; (↓) glucose, creatine Liver: (↑) creatine, alanine, leucine, isoleucine, valine, glutamine, glutathione, taurine, 3-hydroxybutyrate | [21] |
| Melicopelunu-ankenda leaf ethanol extract | 400 mg/kg; 8 weeks | High-fat diet/STZ-induced diabetic rats | T2D | Yes | Serum | (↑) lactate, formate, 2-oxoglutarate, succinate, leucine, isoleucine, hippurate; (↓) glucose, acetoacetate, 3-hydroxybutyrate, choline, creatine | [22] |
| Ipomoea aquatic ethanolic extract | 250 mg/kg; 4 weeks | High-fat diet/STZ-induced diabetic rats | T2D | Yes, but no significant difference | Urine | (↑)creatine, creatinine, hippurate, leucine, 1-methylnicotinamice, taurine, 3-hydroxybutyrate, lysine, trigonelline, allantoin, formate; (↓) glucose, citrate, carnitine, 2-oxoglutarate, succinate, tryptophan, acetoacetate, dimethylamine | [23] |
| Genipin, derived from the fruit of Gardenia jasminoides | 100 mg/kg; 2 weeks | Alloxan-induced diabetic rats | T1D | Not mentioned | Urine Kidney | Urine: (↑) isoleucine, glutamate, acetoacetate, hippurate, N-acetyl-glycoprotein, creatinine, methylamine, dimethylglycine; (↓) 2-oxoglutarate, betaine, sarcosine Kidney: (↑) creatine; (↓) glycine, betaine | [24] |
| Zishen Jiangtang pill | 3.0 g/kg; 8 weeks | STZ-induced rats with diabetic osteoporosis | T1D | Yes | Blood Urine | Blood: (↑) tryptophan, malate, propylene glycol, xanthosine, fumarate Urine: (↓) butyrate | [25] |
| Qijian mixture | 5.385 g/kg; 8 weeks | Male KKay mice | T2D | Yes | Liver Kidney | Liver: (↑) glucose, taurine, glycerol; (↓) isoleucine, valine, lactate, alanine, acetate, homoserine, glutarate, 3-hydroxybutyrate, glutamine, glutathione, choline, anserine, niacinamide, xanthine, inosine Kidney: (↑) phosphocholine, TMAO, myo-inositol, xanthine; (↓) citrate | [26] |
| Mangiferin (SA1) and naringenin (SA2) from the leaves of Salacia oblonga | 100 mg/kg; 15 days | STZ-induced diabetic rats | T2D | Yes | Serum | SA1: (↑) isoleucine, leucine, valine, lactate, alanine, acetate, proline, N-acetyl-glycoprotein, O-acetyl-glycoprotein, acetone, glutamate, glutamine, lipid, creatine, creatinine, malonate, choline, methanol, myo-inositol, serine, gluconate, threonine, allantoin, tyrosine, phenylalanine, histidine; (↓) glucose SA2: (↑) HDL/LDL, LDL/VLDL, isoleucine, leucine, valine, lactate, alanine, acetate, proline, N-acetyl-glycoprotein, O-acetyl-glycoprotein, acetone, glutamate, glutamine, lipid, creatine, malonate, choline, methanol, myo-inositol, glycerol, serine, gluconate, threonine, allantoin, tyrosine, phenylalanine, histidine; (↓) glucose | [27] |
| Ganoderma lucidum polysaccharides | 400 mg/kg; 4 weeks | STZ-induced T2D rats | T2D | Yes | Feces | (↓) xanthine, deoxycholic acid, imidazole, n-Heptanoate, Urocanate, valine; (↑) methanol | [28] |
| Rubus suavissimus S. Lee | 3 g/kg; 6 weeks | STZ-induced T1D rats | T1D | Yes | Urine | (↑) creatinine, allantoin, hippurate; (↓) lactate, pyruvate, succinate, 2-oxoglutarate, citrate | [29] |
| Salvia miltiorrhiza and Radix Pueraria lobata herb pair | 3.15 g/kg; 4 weeks | STZ-induced T2D rats | T2D | Yes | Feces | (↑) alanine, succinate, lactate, proline, valine, leucine, glutamate, glucose, isoleucine, α-ketoisovalerate, hypoxanthine; (↓) butyrate | [30] |
| Anthocyanin Extracts from Bilberry and Purple Potato | 25 and 50 mg/kg; 8 weeks | Zucker diabetic rats | T2D | Yes | Plasma | (↓) lactate, lipid, valine, leucine, isoleucine, glutamate | [31] |
| Berberis kansuensis extract | 0.84 g/kg; 30 days | High-fat diet/STZ-induced diabetic rats | T2D | Yes | Serum | (↑) LDL/VLDL, isoleucine, valine, NAG, acetoacetate, glutamate; (↓) betaine, glucose | [32] |
| Astragalus radix and Dioscoreae rhizoma | 6.3 g/kg; 4 weeks | High-fat diet/STZ-induced diabetic rats | T2D | Yes | Serum | (↑) taurine, glycine, glutamine; (↓) lipid, pyruvate, TMAO, glycerol, isoleucine, leucine, valine, glucose, tyrosine, 3-hydroxybutyrate, acetoacetate, succinate, xanthine | [33] |
| Chickpea extract | 3 g/kg; 4 weeks | High-fat diet/STZ-induced diabetic rats | T2D | Yes | Cecum | (↑) acetate, propionate, butyrate | [34] |
| Acanthopanax sessiliflorus fruits | 3 mg/kg; 4 weeks | High-fat diet-induced mouse model | T2D | Not mentioned | Liver | (↑) formate, inosine, pyroglutamate, taurine; (↓) alanine, tyrosine | [35] |
| Enteromorpha prolifera polysaccharide | 450 mg/kg; 12 weeks | High-fat diet-fed hamsters | T2D | Not mentioned | Serum | (↑) arginine; (↓) 2-hydroxyisovalerate, 2-oxoglutarate, 3-hydroxybutyrate, 3-hydroxyisobutyrate, betaine, citrate, glucose, lactate | [36] |
| Berberis vernae extract | 0.84 g/kg; 30 days | High-fat diet/STZ-induced diabetic rats | T2D | Yes | Serum | (↑) LDL/VLDL, isoleucine, valine, lipid, NAG, acetoacetate; (↓) TMAO, betaine, glucose | [37] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, Y.; Lu, J.; Zhao, Q.; Chen, J.; Dong, W.; Lin, M.; Zheng, H. Potential Therapeutic Mechanism of Traditional Chinese Medicine on Diabetes in Rodents: A Review from an NMR-Based Metabolomics Perspective. Molecules 2022, 27, 5109. https://doi.org/10.3390/molecules27165109
Huang Y, Lu J, Zhao Q, Chen J, Dong W, Lin M, Zheng H. Potential Therapeutic Mechanism of Traditional Chinese Medicine on Diabetes in Rodents: A Review from an NMR-Based Metabolomics Perspective. Molecules. 2022; 27(16):5109. https://doi.org/10.3390/molecules27165109
Chicago/Turabian StyleHuang, Yinli, Jiahui Lu, Qihui Zhao, Junli Chen, Wei Dong, Minjie Lin, and Hong Zheng. 2022. "Potential Therapeutic Mechanism of Traditional Chinese Medicine on Diabetes in Rodents: A Review from an NMR-Based Metabolomics Perspective" Molecules 27, no. 16: 5109. https://doi.org/10.3390/molecules27165109
APA StyleHuang, Y., Lu, J., Zhao, Q., Chen, J., Dong, W., Lin, M., & Zheng, H. (2022). Potential Therapeutic Mechanism of Traditional Chinese Medicine on Diabetes in Rodents: A Review from an NMR-Based Metabolomics Perspective. Molecules, 27(16), 5109. https://doi.org/10.3390/molecules27165109






