TPPS4—Sensitized Photooxidation of Micropollutants—Singlet Molecular Oxygen Kinetic Study
Abstract
:1. Introduction
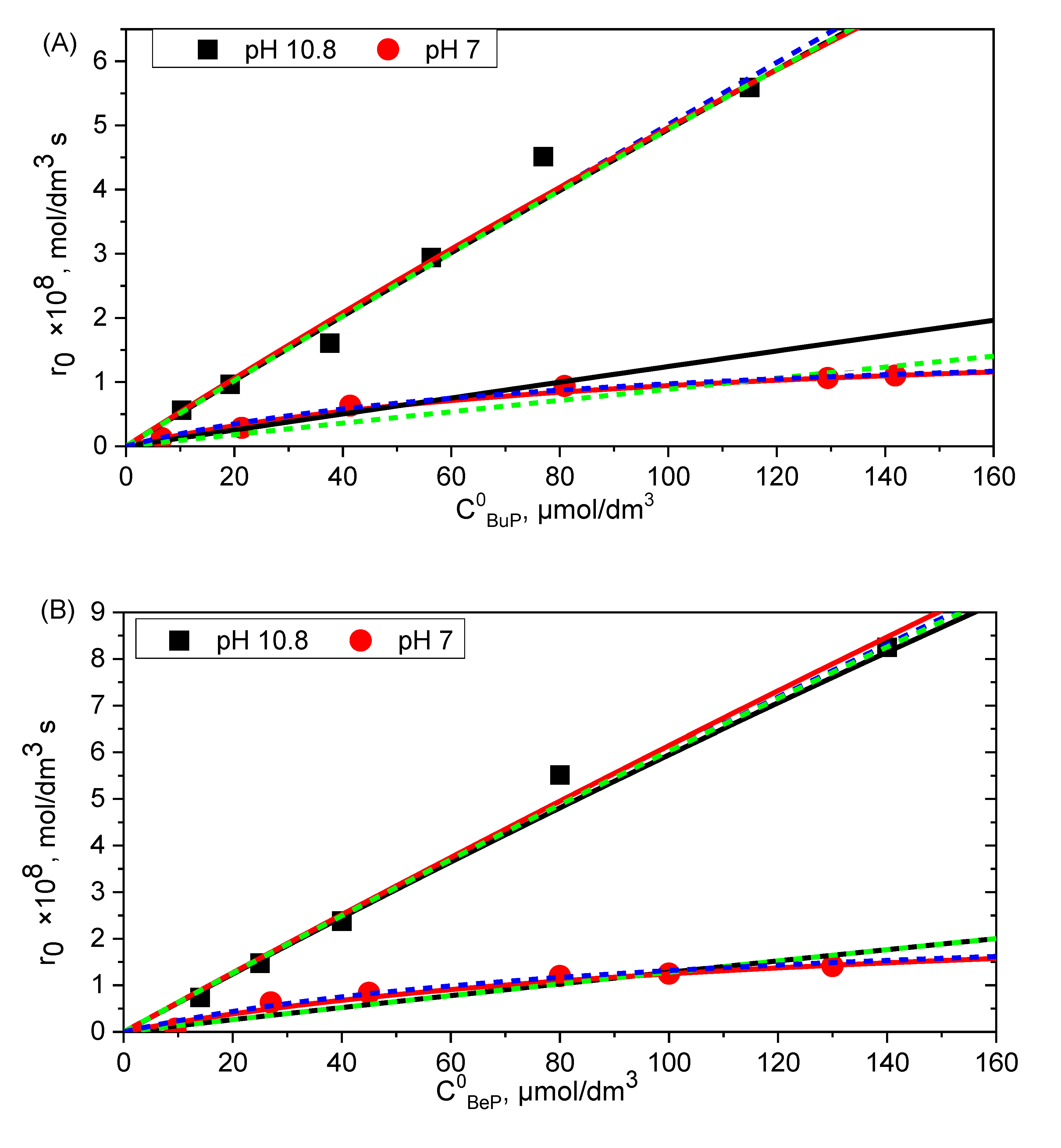
2. Results
2.1. Time Resolve Spectroscopy Measurements
2.2. Kinetic Models for Photooxidation
3. Discussion
4. Materials and Methods
5. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
Nomenclature/Abbreviations
| 1O2 | singlet oxygen |
| BuP | butylparaben |
| BeP | benzylparaben |
| 2CP | 2-chlorophenol |
| MPs | micropollutants |
| Ea | flux of absorbed photons per unit of the reaction volume |
| k | rate constant |
| kd | rate constant of singlet oxygen decay in water |
| kq | rate constant for physical quenching of singlet oxygen |
| kr | rate constant for the chemical reaction of singlet oxygen |
| rate constant for chemical reaction of singlet oxygen with TPPS4 | |
| rate constant for physical and chemical decay of singlet oxygen with MP | |
| kD | rate constant for triplet TPPS4 decay |
| rate constant for triplet TPPS4 reaction with oxygen which gives 1O2 | |
| rate constant for triplet TPPS4 quenching by oxygen | |
| rate constant for triplet TPPS4 quenching by ground state TPPS4 | |
| rate constant for triplet TPPS4 quenching by MPs | |
| O2 | molecular oxygen (triplet 3O2) |
| r | reaction rate |
| TPPS4 | meso-tetrakis(4-sulfonatophenyl)porphyrin |
| t | time |
| α | dissociation degree |
| φ | quantum yield |
| φT | quantum yield of triplet formation |
| φΔ | quantum yield of singlet oxygen formation |
| λ | wavelength |
| * | excited state |
| 0 | initial conditions |
References
- Ogilby, P.R. Solvent effects on the radiative transitions of singlet oxygen. Acc. Chem. Res. 1999, 32, 512–519. [Google Scholar] [CrossRef]
- Min, D.B.; Boff, J.M. Chemistry and Reaction of Singlet Oxygen in Foods. Compr. Rev. Food Sci. Food Saf. 2002, 1, 58–72. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, R.; Merkel, P.B.; Kearns, D.R. Unambiguous Evidence for the Participation of Singlet Oxygen (1δ) in Photodynamic Oxidation of Amino Acids. Photochem. Photobiol. 1972, 16, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Lion, Y.; Delmelle, M.; Van De Vorst, A. New method of detecting singlet oxygen production. Nature 1976, 263, 442–443. [Google Scholar] [CrossRef]
- Lindig, B.A.; Rodgers, M.A.J.; Schaaplc, A.P. Determination of the Lifetime of Singlet Oxygen in D20 Using 9,10-Anthracenedipropionic Acid, a Water-Soluble Probe. J. Am. Chem. Soc. 1980, 102, 5590–5593. [Google Scholar] [CrossRef]
- Kraljić, I.; Mohsni, S. El A new method for the detection of singlet oxygen in aqueous solutions. Photochem. Photobiol. 1978, 28, 577–581. [Google Scholar] [CrossRef]
- Jockusch, S.; Turro, N.J.; Thompson, E.K.; Gouterman, M.; Callis, J.B.; Khalil, G.E. Singlet molecular oxygen by direct excitation. Photochem. Photobiol. Sci. 2008, 7, 235–239. [Google Scholar] [CrossRef]
- Turro, N.J. Modern Molecular Photochemistry; University Science Books: Mill-Valley, CA, USA, 1991. [Google Scholar]
- Di Mascio, P.; Kaiser, S.; Sies, H. Lycopene as the most efficient biological carotenoid singlet oxygen quencher. Arch. Biochem. Biophys. 1989, 274, 532–538. [Google Scholar] [CrossRef]
- Darijani, M.; Shahraki, M.; Mostafa Habibi-Khorassani, S. Theoretical study on the mechanism and kinetics of the formation β-carotene epoxides from the oxidative degradation of β-carotene. Food Chem. 2022, 389, 133082. [Google Scholar] [CrossRef]
- Tratnyek, P.G.; Holgné, J. Oxidation of Substituted Phenols in the Environment: A QSAR Analysis of Rate Constants for Reaction with Singlet Oxygen. Environ. Sci. Technol. 1991, 25, 1596–1604. [Google Scholar] [CrossRef]
- Miller, J.S. Rose bengal-sensitized photooxidation of 2-chlorophenol in water using solar simulated light. Water Res. 2005, 39, 412–422. [Google Scholar] [CrossRef]
- Barrios, B.; Mohrhardt, B.; Doskey, P.V.; Minakata, D. Mechanistic insight into the reactivities of aqueous-phase singlet oxygen with organic compounds. Environ. Sci. Technol. 2021, 55, 8054–8067. [Google Scholar] [CrossRef]
- Gmurek, M.; Miller, J.S.; Ledakowicz, S. Kinetics of the photosensitized degradation of benzyl 4-hydroxybenzoate in homogeneous aqueous solution under visible-light irradiation. Chem. Eng. J. 2012, 210, 417–424. [Google Scholar] [CrossRef]
- Wilkinson, F.; Brummer, J.G. Rate constants for the decay and reactions of the lowest electronically excited singlet state of molecular oxygen in solution. J. Phys. Chem. Ref. Data 1981, 10, 809–999. [Google Scholar] [CrossRef]
- Dántola, M.L.; Neyra Recky, J.R.; Lorente, C.; Thomas, A.H. Photosensitized Dimerization of Tyrosine: The Oxygen Paradox. Photochem. Photobiol. 2022, 98, 687–695. [Google Scholar] [CrossRef]
- Fridovich, I. Fundamental aspects of reactive oxygen species, or what’s the matter with oxygen? Ann. N. Y. Acad. Sci. 1999, 893, 13–18. [Google Scholar] [CrossRef]
- Mosinger, J.; Deumié, M.; Lang, K.; Kubát, P.; Wagnerová, D.M. Supramolecular sensitizer: Complexation of meso-tetrakis(4-sulfonatophenyl)porphyrin with 2-hydroxypropyl-cyclodextrins. J. Photochem. Photobiol. A Chem. 2000, 130, 13–20. [Google Scholar] [CrossRef]
- Dědic, R.; Kořínek, M.; Molnár, A.; Svoboda, A.; Hála, J. Singlet oxygen quenching by oxygen in tetraphenyl-porphyrin solutions. J. Lumin. 2006, 119–120, 209–213. [Google Scholar] [CrossRef]
- Parra, G.G.; Correa, D.S.; Silveira-Alves, E.; Almeida, L.M.; Souza, M.A.R.; De Boni, L.; Misoguti, L.; Mendonça, C.R.; Zílio, S.C.; Barbosa Neto, N.M.; et al. Effects of meso-tetrakis (4-sulfonatophenyl) porphyrin (TPPS4) aggregation on its spectral and kinetic characteristics and singlet oxygen production. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021, 261, 120063. [Google Scholar] [CrossRef]
- Schmidt, R. Influence of Heavy Atoms on the Deactivation of Singlet Oxygen (1Δg) in Solution. J. Am. Chem. Soc. 1989, 111, 6983–6987. [Google Scholar] [CrossRef]
- Gonçalves, P.J.; Aggarwal, L.P.F.; Marquezin, C.A.; Ito, A.S.; De Boni, L.; Neto, N.M.B.; Rodrigues, J.J.; Zílio, S.C.; Borissevitch, I.E. Effects of interaction with CTAB micelles on photophysical characteristics of meso-tetrakis(sulfonatophenyl) porphyrin. J. Photochem. Photobiol. A Chem. 2006, 181, 378–384. [Google Scholar] [CrossRef]
- Gmurek, M.; Kubat, P.; Mosinger, J.; Miller, J.S. Comparison of two photosensitizers Al(III) phthalocyanine chloride tetrasulfonic acid and meso-tetrakis(4-sulfonatophenyl)porphyrin in the photooxidation of n-butylparaben. J. Photochem. Photobiol. A Chem. 2011, 223, 50–56. [Google Scholar] [CrossRef]
- Scully, F.E.; Hoigné, J. Rate constants for reactions of singlet oxygen with phenols and other compounds in water. Chemosphere 1987, 16, 681–694. [Google Scholar] [CrossRef]
- Palumbo, M.C.; García, N.A.; Gutiérrez, M.I.; Luiz, M. Singlet molecular oxygen—mediated photooxidation of monochloro and mononitrophenols. A kinetic study. Toxicol. Environ. Chem 2008, 29, 85–94. [Google Scholar] [CrossRef]
- Fukahori, M.; Takatsuji, Y.; Yamakita, T.; Takahashi, H.; Sato, H.; Yotsuyanagi, T. Aggregate Formation of p-Hydroxybenzoic Acid Esters in Aqueous Solution. Chem. Pharm. Bull. 1996, 44, 245–248. [Google Scholar] [CrossRef]
- Mártire, D.O.; Braslavsky, S.E.; García, N.A. Sensitized photo-oxidation of dihydroxybenzenes and chlorinated derivatives. A kinetic study. J. Photochem. Photobiol. A Chem. 1991, 61, 113–124. [Google Scholar] [CrossRef]
- Michaeli, A.; Feitelson, J. Reactivity of singlet oxygen toward amino acids and peptides. Photochem. Photobiol. 1994, 59, 284–289. [Google Scholar] [CrossRef]
- Wilkinson, F.; Helman, W.P.; Ross, A.B.; Wilkinson, F.; Helman, W.P.; Ross, A.B. Rate Constants for the Decay and Reactions of the Lowest Electronically Excited Singlet State of Molecular Oxygen in Solution. An Expanded and Revised Compilation. JPCRD 1995, 24, 663–677. [Google Scholar] [CrossRef]
- Li, M.Y.; Cline, C.S.; Koker, E.B.; Carmichael, H.H.; Chignell, C.F.; Bilski, P. Quenching of Singlet Molecular Oxygen (1O2) by Azide Anion in Solvent Mixtures. Photochem. Photobiol. 2001, 74, 760–764. [Google Scholar] [CrossRef]
- Ogryzlo, E.A.; Tang, C.W. Quenching of Oxygen (1Δg by Amines. J. Am. Chem. Soc. 1970, 92, 5034–5036. [Google Scholar] [CrossRef]
- Egorov, S.Y.; Kurella, E.G.; Boldyrev, A.A.; Krasnovsky, A.A. Quenching of singlet molecular oxygen by carnosine and related antioxidants. Monitoring 1270-nm phosphorescence in aqueous media. IUBMB Life 1997, 41, 687–694. [Google Scholar] [CrossRef] [PubMed]





| pH 7 | |||
| kt, dm3/mol s | BuP | BEP | 2-CP |
| (5.06 ± 0.2) × 107 | (8.86 ± 0.3) × 107 | (2.92 ± 0.3) × 107 | |
| pH 9 | |||
| kt, dm3/mol s | BuP | BEP | 2-CP |
| (8.45 ± 0.5) × 107 | (1.02 ± 0.04) × 108 | (1.66 ± 0.03) × 108 | |
| pH 10.8 | |||
| kt, dm3/mol s | BuP | BEP | 2-CP |
| (1.15 ± 0.04) × 108 | (1.21 ± 0.06) × 108 | (2.03 ± 0.07) × 108 | |
| Constant | φT | φΔ | kD 1/s | kd 1/s | krTPPS4 dm3/mol s | ||
|---|---|---|---|---|---|---|---|
| Value | 0.78 [23] | 0.62 [18] | 2.8 × 103 [22] | 1.77 × 109 [this study] | 1.46 × 109 a | 2.4 × 105 [21] | <108 [14] |
| Rate constant, dm3/mol s |  |  | ||
| pH = 7 | pH = 10.8 | |||
| Equation (26) | ||||
| Method 1 | Method 2 | Method 1 | Method 2 | |
| (1.25 ± 0.5) × 106 | (1.9 ± 0.1) × 106 | (5.1 ± 0.4) × 106 | (5.3 ± 0.6) × 106 | |
| (5.1 ± 0.2) × 107 | (2.5 ± 0.5) × 109 | (1.1 ± 0.04) × 108 | (2.0 ± 0.2) × 108 | |
| (1.2 ± 0.5) × 106 | (2.12 ± 0.9) × 106 | no quenching | (1.01 ± 0.7) × 106 | |
| Rate constant, dm3/mol s 1 | Equation (27) | |||
| pH = 7 | pH = 10.8 | |||
| Method 1 | Method 2 | Method 1 | Method 2 | |
| (8.96 ± 0.8) × 105 | (2.14 ± 0.3) × 106 | (5.1 ± 0.4) × 106 | (5.1 ± 0.09) × 106 | |
| (5.1 ± 0.2) × 107 | (2.95 ± 0.8) × 109 | (1.1 ± 0.04) × 108 | (7.2 ± 0.8) × 107 | |
| Rate constant, dm3/mol s |  |  | ||
| pH = 7 | pH = 10.8 | |||
| Equation (26) | ||||
| Method 1 | Method 2 | Method 1 | Method 2 | |
| (1.31 ± 0.1) × 106 | (2.2 ± 0.4) × 106 | (6.2 ± 0.4) × 106 | (6.3 ± 0.07) × 106 | |
| (8.8 ± 0.3) × 107 | (1.9 ± 0.1) × 109 | (1.4 ± 0.3) × 108 | (1.5 ± 0.2) × 108 | |
| no quenching | (5.5 ± 1.1) × 107 | no quenching | (3.2 ± 0.4) × 106 | |
| Rate constant, dm3/mol s 1 | Equation (27) | |||
| pH = 7 | pH = 10.8 | |||
| Method 1 | Method 2 | Method 1 | Method 2 | |
| (1.31 ± 0.1) × 106 | (2.6 ± 0.2) × 106 | (6.20 ± 0.2) × 106 | (6.21 ± 0.8) × 106 | |
| (8.8 ± 0.3) × 107 | (2.4 ± 0.1) × 109 | (1.4 ± 0.3) × 108 | (7.9 ± 0.1) × 107 | |
| Rate constant, dm3/mol s | 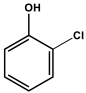 |  | ||
| pH = 7 | pH = 10.8 | |||
| Equation (26) | ||||
| Method 1 | Method 2 | Method 1 | Method 2 | |
| (2.5 ± 0.3) × 106 | (3.4 ± 0.1) × 106 | (1.22 ± 0.02) × 108 | (1.15 ± 0.3) × 108 | |
| (2.9 ± 0.4) × 107 | (6.8 ± 0.2) × 107 | (2.03 ± 0.2) × 108 | (1.88 ± 0.02) × 108 | |
| no quenching | (3.0 ± 0.7) × 106 | (2.4 ± 0.08) × 109 | (2.22 ± 0.03) × 109 | |
| Compound | krArOH dm3/mol s | krArO− dm3/mol s | ktArOH dm3/mol s | ktArO− dm3/mol s |
|---|---|---|---|---|
| phenol | (2.6 ± 4.0) × 106 (a) | (1.55 ± 0.05) × 108 (a) | - | - |
| 4′-Hydroxy acetophenone | (1.5 ± 0.1) × 106 (a) | (2.36 ± 0.01) × 107 (a) | - | - |
| 4-hydroxyphenol | (3.8 ± 5.5) × 107 (a) | - | - | - |
| 2-benzylphenol | - | 4.4 × 107 (b) | - | - |
| salicylic acid methyl ester | - | <2 × 106 (c) | 1.2 × 108 (c) | |
| 4-chlorophenol | (6.0 ± 3.6) × 106 (a) | (1.93 ± 0.04) × 108 (a) | - | - |
| 3-chlorophenol | (5.4 ± 1.0) × 106 (a) | (1.6 ± 0.02) × 108 (a) | - | - |
| 2-chlorophenol | (9.2 ± 9.4) × 106 (d) | (1.92 ± 0.1) × 108 (d) | - | - |
| - | 1.7 × 107 (d) | - | 2.3 × 108 (d) | |
| - | 2.2 × 108 (e) | - | 6.8 × 108 (e) | |
| 3.8 × 105 (e) | 2.05 × 108 (e) | 1.33 × 106 (e) | 2.36 × 109 (e) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gmurek, M. TPPS4—Sensitized Photooxidation of Micropollutants—Singlet Molecular Oxygen Kinetic Study. Molecules 2022, 27, 5260. https://doi.org/10.3390/molecules27165260
Gmurek M. TPPS4—Sensitized Photooxidation of Micropollutants—Singlet Molecular Oxygen Kinetic Study. Molecules. 2022; 27(16):5260. https://doi.org/10.3390/molecules27165260
Chicago/Turabian StyleGmurek, Marta. 2022. "TPPS4—Sensitized Photooxidation of Micropollutants—Singlet Molecular Oxygen Kinetic Study" Molecules 27, no. 16: 5260. https://doi.org/10.3390/molecules27165260






