Quantitative Structure–Neurotoxicity Assessment and In Vitro Evaluation of Neuroprotective and MAO-B Inhibitory Activities of Series N′-substituted 3-(1,3,7-trimethyl-xanthin-8-ylthio)propanehydrazides
Abstract
:1. Introduction
2. Results
2.1. Chemistry
2.2. Neurotoxicity Evaluation
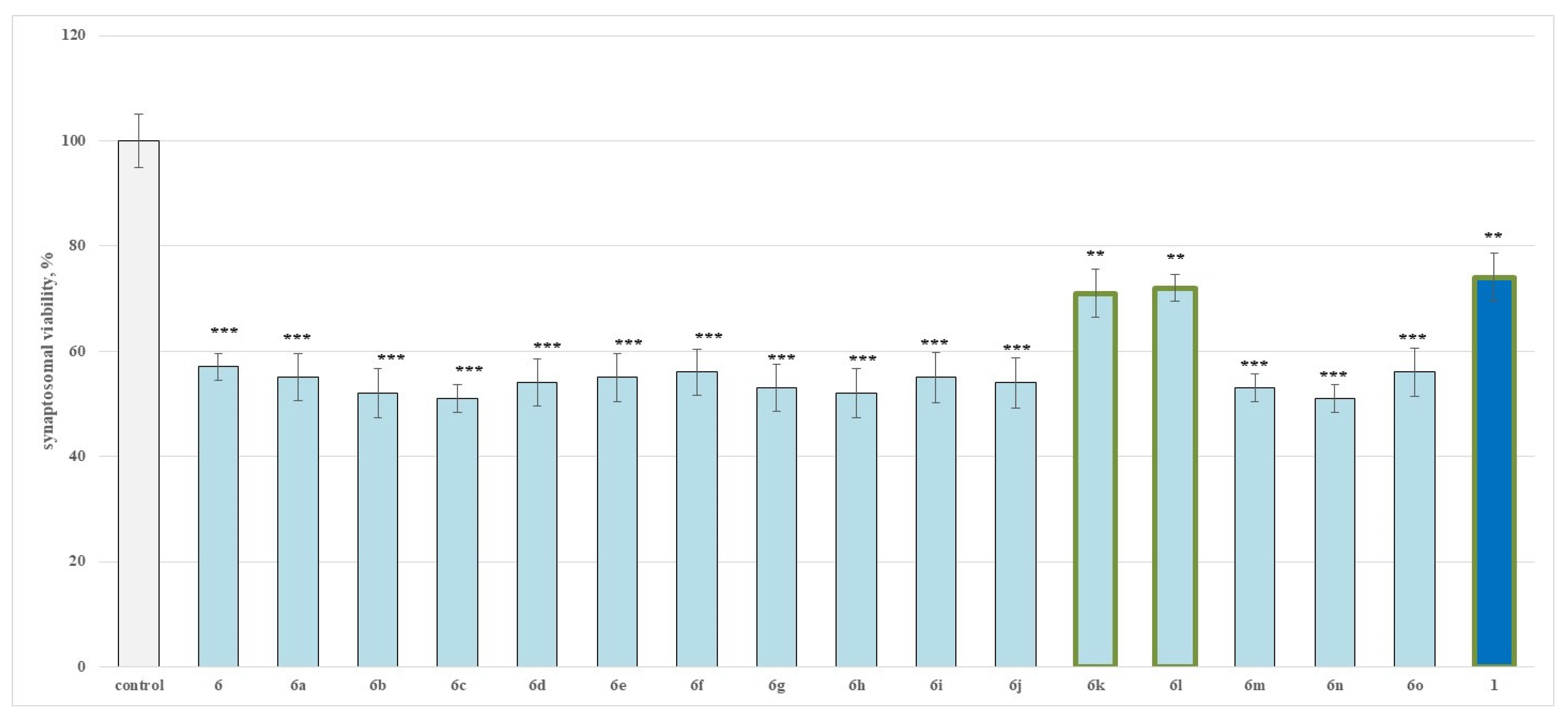
2.2.1. Effects of the Evaluated Hydrazide-Hydrazones on Isolated Rat Brain Synaptosomes
2.2.2. Effects of the Evaluated Hydrazide-Hydrazones on Glutathione (GSH) Level in Isolated Rat Brain Synaptosomes
2.2.3. Effects of the Evaluated Hydrazide-Hydrazones on Isolated Rat Brain Microsomes
2.2.4. Effects of the Evaluated Hydrazide-Hydrazones on Isolated Rat Brain Mitochondria
2.3. Quantitative Structure–Neurotoxicity Assessment for the Evaluated N′-Substituted Xanthinylthio Propanehydrazones
2.3.1. QSTR Model Based on Data for the Relative Synaptosomal Vitality (RV)
n = 15 r2 = 0.608 SEE = 0.063 F = 9.32 q2 = 0.523 r2scr = 0.144
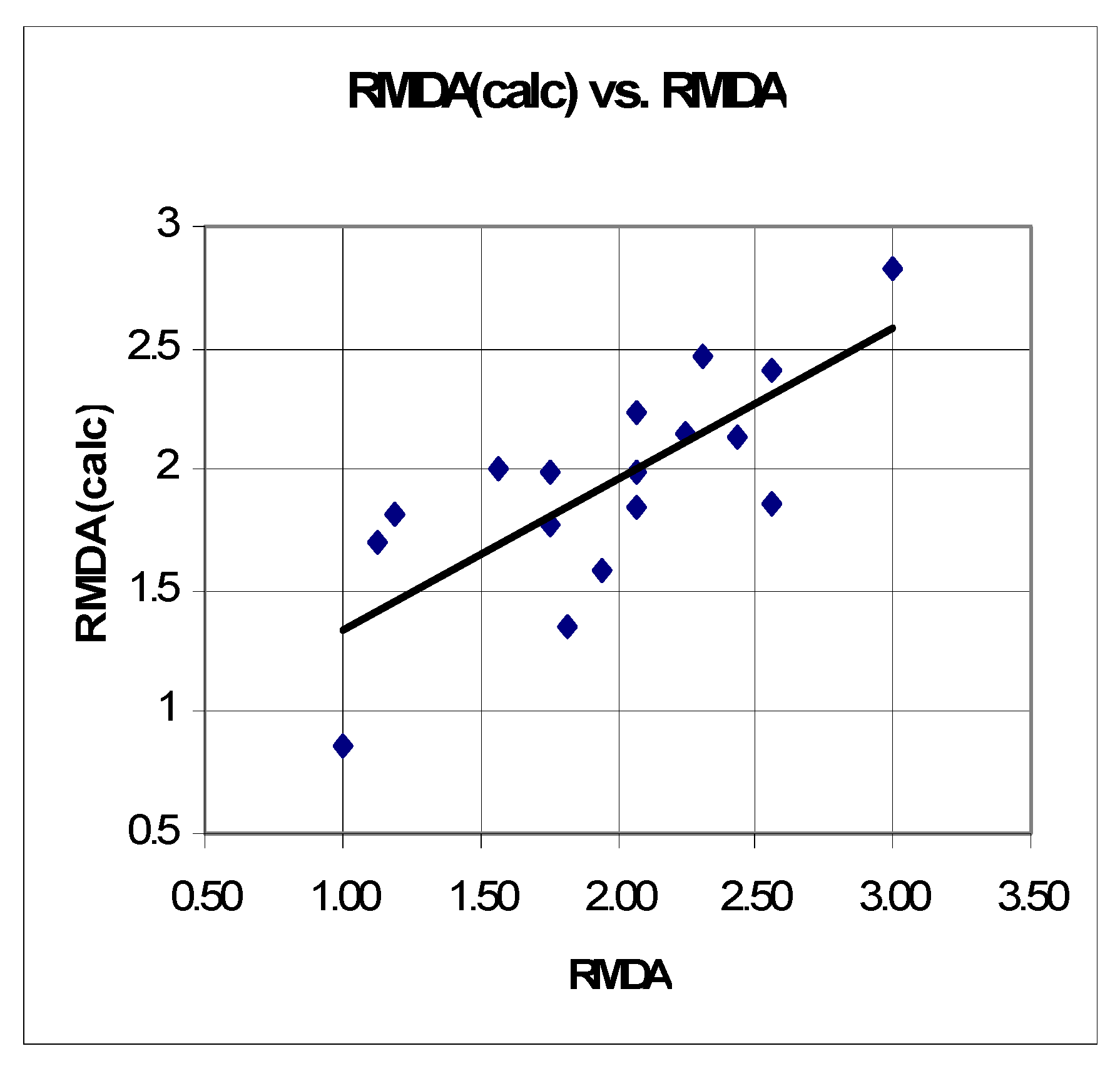
2.3.2. QSTR Model Based on Data for the Relative Increase in Microsomal MDA Production (RMDA)
n = 15 r2 = 0.600 SEE = 0.382 F = 9.751 q2 = 0.4553 r2scr = 0.153
2.4. Evaluation of the Neuroprotection Effects of the Studied N′-substituted 3-(1,3,7-trimethyl-xanthin-8-ylthio)propanehydrazides
2.4.1. Protective Effects in a Model of 6-OHDA-Induced Oxidative Stress in Isolated Rat Synaptosomes
2.4.2. Protective Effects in a Model of Iron Ascorbate (Fe2+/AA)-Induced Lipid Peroxidation in Isolated Rat Microsomes
2.4.3. Protective Effects in a Model of Tert-Butylhydroxyperoxide(t-BuOOH)-Induced Oxidative Stress in Isolated Rat Mitochondria
2.5. Investigation of the Influence of the Newly Obtained Derivatives on the Activity of Human Recombinant MAO-B Enzyme
3. Discussion
3.1. Neurotoxicity Evaluation
3.2. Quantitative Structure–Neurotoxicity Assessment for the Evaluated Xanthinylthio Propanehydrazones
3.3. Studies on the Neuroprotective Activity of Newly Synthesized Derivatives
3.4. Investigation of the Influence of the Newly Obtained Derivatives on the Activity of Human Recombinant MAO-B Enzyme
4. Materials and Methods
4.1. Chemistry
4.2. Biological Evaluation
4.2.1. Animals
4.2.2. Isolation and Incubation of Rat Brain Synaptosomes and Mitochondria
4.2.3. Synaptosomal Viability
4.2.4. Determination of Reduced Glutathione (GSH) in Brain Synaptosomes
4.2.5. Model of 6-OHDA-Induced Neurotoxicity in Synaptosomes
4.2.6. Tert-Butyl Hydroperoxide (t-BuOOH)-Induced Oxidative Stress in Isolated Brain Mitochondria
4.2.7. Measurement of GSH Content in Brain Mitochondria
4.2.8. Preparation of Brain Microsomes Using Ultracentrifugation
4.2.9. FeSO4/Ascorbic Acid-Induced Lipid Peroxidation in Isolated Brain Microsomes
4.2.10. MDA Assay in Brain Microsomes and Mitochondria
4.2.11. Measurement of Monoamine Oxidase B Activity
4.3. Quantitative Structure–Neurotoxicity Assessment for the Evaluated Xanthinylthio Propanehydrazones
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Drukarch, B.; van Muiswinkel, F.L. Drug Treatment of Parkinson’s Disease. Biochem. Pharmacol. 2000, 59, 1023–1031. [Google Scholar] [CrossRef]
- Strydom, B.; Bergh, J.J.; Petzer, J.P. 8-Aryl- and alkyloxycaffeine analogues as inhibitors of monoamine oxidase. Eur. J. Med. Chem. 2011, 46, 3474–3485. [Google Scholar] [CrossRef] [PubMed]
- Silvestri, R.; La Regina, G.; De Martino, G.; Artico, M.; Befani, O.; Palumbo, M.; Agostinelli, E.; Turini, P. Simple, Potent, and Selective Pyrrole Inhibitors of Monoamine Oxidase Types A and B. J. Med. Chem. 2003, 46, 917–920. [Google Scholar] [CrossRef] [PubMed]
- Youdim, M.B.H.; Bakhle, Y.S. Monoamine oxidase: Isoforms and inhibitors in Parkinson’s disease and depressive illness. Br. J. Pharmacol. 2009, 147, S287–S296. [Google Scholar] [CrossRef] [Green Version]
- Hagenow, J.; Hagenow, S.; Grau, K.; Khanfar, M.; Hefke, L.; Proschak, E.; Stark, H. Reversible Small Molecule Inhibitors of MAO A and MAO B with Anilide Motifs. Drug Des. Dev. Ther. 2020, 14, 371–393. [Google Scholar] [CrossRef] [Green Version]
- Fredholm, B.B.; Bättig, K.; Holmén, J.; Nehlig, A.; Zvartau, E.E. Actions of caffeine in the brain with special reference to factors that contribute to its widespread use. Pharmacol. Rev. 1999, 51, 83–133. [Google Scholar]
- Van der Walt, E.M.; Milczek, E.M.; Malan, S.F.; Edmondson, D.E.; Castagnoli, N., Jr.; Bergh, J.J.; Petzer, J.P. Inhibition of monoamine oxidase by (E)-styrylisatin analogues. Bioorganic Med. Chem. Lett. 2009, 19, 2509–2513. [Google Scholar] [CrossRef] [Green Version]
- Boia, R.; Ambrósio, A.F.; Santiago, A.R. Therapeutic Opportunities for Caffeine and A2A Receptor Antagonists in Retinal Diseases. Ophthalmic Res. 2016, 55, 212–218. [Google Scholar] [CrossRef]
- Xu, K.; Di Luca, D.; Orrú, M.; Xu, Y.; Chen, J.-F.; Schwarzschild, M. Neuroprotection by caffeine in the MPTP model of parkinson’s disease and its dependence on adenosine A 2A receptors. Neuroscience 2016, 322, 129–137. [Google Scholar] [CrossRef] [Green Version]
- Ferré, S.; Bonaventura, J.; Tomasi, D.; Navarro, G.; Moreno, E.; Cortés, A.; Lluís, C.; Casadó, V.; Volkow, N.D. Allosteric mechanisms within the adenosine A2A-dopamine D2 receptor heterotetramer. Neuropharmacology 2016, 104, 154–160. [Google Scholar] [CrossRef] [Green Version]
- Ferreira, D.G.; Batalha, V.L.; Vicente Miranda, H.; Coelho, J.E.; Gomes, R.; Gonçalves, F.Q.; Real, J.I.; Rino, J.; Albino-Teixeira, A.; Cunha, R.A.; et al. Adenosine A2A Receptors Modulate α-Synuclein Aggregation and Toxicity. Cereb Cortex 2017, 27, 718–730. [Google Scholar] [PubMed] [Green Version]
- de Souza, M.F.; Bispo, J.M.M.; Leal, P.C.; de Gois, A.M.; Dos Santos, J.R. Commentary: Adenosine A2A Receptor Blockade Prevents Rotenone-Induced Motor Impairment in a Rat Model of Parkinsonism. Front. Behav. Neurosci. 2017, 11, 93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernandez-Duenas, V.; Perez-Arevalo, A.; Altafaj, X.; Ferre, S.; Ciruela, F. Adenosine A1-A2A Receptor Heteromer as a Possible Target for Early-Onset Parkinson’s Disease. Front. Neurosci. 2017, 22, 652. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vorovenci, R.J.; Antonini, A. The efficacy of oral adenosine A(2A) antagonist istradefylline for the treatment of moderate to severe Parkinson’s disease. Expert Rev. Neurother. 2015, 15, 1383–1390. [Google Scholar] [CrossRef] [PubMed]
- Fathalla, A.M.; Soliman, A.M.; Moustafa, A.A. Selective A2A receptors blockade reduces degeneration of substantia nigra dopamine neurons in a rotenone-induced rat model of Parkinson’s disease: A histological study. Neurosci. Lett. 2017, 643, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Wang, X.; Li, L.; Zhang, X.; Li, Y.; Zhu, F.; Hou, T.; Zhen, X. Discovery of Novel and Selective Adenosine A2A Receptor Antagonists for Treating Parkinson’s Disease through Comparative Structure-Based Virtual Screening. J. Chem. Inf. Model. 2017, 57, 1474–1487. [Google Scholar] [CrossRef] [PubMed]
- Reyhani-Rad, S.; Mahmoudi, J. Effect of adenosine A2A receptor antagonists on motor disorders induced by 6-hydroxydopamine in rat. Acta Cir. Bras. 2016, 31, 133–137. [Google Scholar] [CrossRef] [Green Version]
- Booysen, H.P.; Moraal, C.; Terre’Blanche, G.; Petzer, A.; Bergh, J.J.; Petzer, J.P. Thio- and aminocaffeine analogues as inhibitors of human monoamine oxidase. Bioorganic Med. Chem. Lett. 2011, 19, 7507–7518. [Google Scholar] [CrossRef]
- Strydom, B.; Malan, S.F.; Castagnoli, N., Jr.; Bergh, J.J.; Petzer, J.P. Inhibition of monoamine oxidase by 8-benzyloxycaffeine analogues. Bioorganic Med. Chem. Lett. 2010, 18, 1018–1028. [Google Scholar] [CrossRef]
- Guglielmi, P.; Carradori, S.; Ammazzalorso, A.; Secci, D. Novel approaches to the discovery of selective human monoamine oxidase-B inhibitors: Is there room for improvement? Expert Opin. Drug Discov. 2019, 14, 995–1035. [Google Scholar] [CrossRef]
- Sellitepe, H.E.; Oh, J.M.; Doğan, İ.S.; Yildirim, S.; Aksel, A.B.; Jeong, G.S.; Khames, A.; Abdelgawad, M.A.; Gambacorta, N.; Nicolotti, O.; et al. Synthesis of N0-(4-/3-/2-/Nonsubstituted benzylidene)-4-[(4-methylphenyl)sulfonyloxy] Benzohydrazides and Evaluation of Their Inhibitory Activities against Monoamine Oxidases and β-Secretase. Appl. Sci. 2021, 11, 5830. [Google Scholar] [CrossRef]
- Tok, F.; Sağlık, B.N.; Özkay, Y.; Ilgın, S.; Kaplancıklı, Z.A.; Koçyiğit-Kaymakçıoğlu, B. Synthesis of new hydrazone derivatives and evaluation of their monoamine oxidase inhibitory activity. Bioorganic Chem. 2021, 114, 105038. [Google Scholar] [CrossRef] [PubMed]
- Mitkov, J.; Kondeva-Burdina, M.; Georgieva, M.; Zlatkov, A. Synthesis, in silico prediction of sites of metabolism and in-vitro hepatotoxicity evaluationofnew series N′-substituted 3-(1,3,7-trimethyl-xanthin-8-ylthio)propanehydrazides. Pharmacia 2022, 69, 351–361. [Google Scholar] [CrossRef]
- Golbraikh, A.; Tropsha, A. Predictive QSAR modeling based on diversity sampling of experimental datasets for the training and test set selection. J. Comput.-Aided Mol. Des. 2002, 16, 357–369. [Google Scholar] [CrossRef] [PubMed]
- Rudin, D.; Liechti, M.E.; Luethi, D. Molecular and clinical aspects of poten-tial neurotoxicity induced by new psychoactive stimulants and psychedelics. Exp. Neurol. 2021, 343, 113778. [Google Scholar] [CrossRef] [PubMed]
- Evans, G.J. The synaptosome as a model system for studying synaptic physiology. Cold Spring Harb. Protoc. 2015, 2015, 421–424. [Google Scholar] [CrossRef] [Green Version]
- Ansari, M.; Scheff, S. Oxidative stress in the progression of Alzheimer disease in the frontal cortex. J. Neuropathol. Exp. Neurol. 2010, 69, 155–167. [Google Scholar] [CrossRef] [Green Version]
- Parmentier, Y.; Bossant, M.-J.; Bertrand, M.; Walther, B. In Vitro Studies of Drug Metabolism. In Comprehensive Medicinal Chemistry II. 5; Taylor, J.B., Triggle, D.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2007; pp. 231–257. [Google Scholar]
- Pourahmad, J.; Hosseini, M.J. Application of isolated mitochondria in toxicological and clinical studies. Iran. J Pharm. Res. 2012, 11, 703–704. [Google Scholar]
- Gramatica, P. Principles of QSAR models validation: Internal and external. QSAR Comb. Sci. 2007, 26, 694–701. [Google Scholar] [CrossRef]
- Casson, R.J.; Chidlow, G.; Ebneter, A.; Wood, J.P.M.; Crowston, J.; Goldberg, I. Translational neuroprotection research in glaucoma: A review of definitions and principles. Clin. Exp. Ophthalmol. 2011, 40, 350–357. [Google Scholar] [CrossRef]
- Seidl, S.E.; Potashkin, J.A. The Promise of Neuroprotective Agents in Parkinson’s Disease. Front. Neurol. 2011, 2, 68. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dunnett, S.B.; Björklund, A. Prospects for new restorative and neuroprotective treatments in Parkinson’s disease. Nature 1999, 399, A32–A39. [Google Scholar] [CrossRef] [PubMed]
- Andersen, J.K. Oxidative stress in neurodegeneration: Cause or consequence? Nat. Med. 2004, 10, S18–S25. [Google Scholar] [CrossRef] [PubMed]
- Diler, A.; Ziya, Y.; Üzüm, G. N-methyl-d-aspartate Receptors Are involved in Caffeine-Induced Facilitation on Memory Retention of Passive Avoidance Learning in Rats. J. Caffeine Res. 2013, 3, 169–174. [Google Scholar] [CrossRef]
- Kim, I.Y.; O’Reilly, É.J.; Hughes, K.C.; Gao, X.; Schwarzschild, M.A.; McCullough, M.L.; Hannan, M.T.; Betensky, R.A.; Ascherio, A. Interaction Between Caffeine and Polymorphisms of Glutamate Inotropic Receptor NMDA Type Subunit 2A (GRIN2A) and Cytochrome P450 1A2 (CYP1A2) on Parkinson’s Disease Risk. Mov. Disord. 2018, 33, 414–420. [Google Scholar] [CrossRef]
- Villanueva-García, D.; Mota-Rojas, D.; Miranda-Cortés, A.; Mora-Medina, P.; Hernández-Avalos, I.; Casas-Alvarado, A.; Olmos-Hernández, A.; Martínez-Burnes, J. Neurobehavioral and neuroprotector effects of caffeine in animal models. J. Anim. Behav. Biometeorol. 2020, 8, 298–307. [Google Scholar] [CrossRef]
- Stokes, A.H.; Freeman, W.M.; Mitchell, S.G.; Burnette, T.A.; Hellmann, G.M.; Vrana, K.E. Induction of GADD45 and GADD153 in Neuroblastoma Cells by Dopamine-Induced Toxicity. NeuroToxicology 2002, 23, 675–684. [Google Scholar] [CrossRef]
- Metro, D.; Cernaro, V.; Santoro, D.; Papa, M.; Buemi, M.; Benvenga, S.; Manasseri, L. Beneficial effects of oral pure caffeine on oxidative stress. J. Clin. Transl. Endocrinol. 2017, 10, 22–27. [Google Scholar] [CrossRef]
- Aoyama, K.; Matsumura, N.; Watabe, M.; Wang, F.; Kikuchi-Utsumi, K.; Nakaki, T. Caffeine and uric acid mediate glutathione synthesis for neuroprotection. Neuroscience 2011, 181, 206–215. [Google Scholar] [CrossRef]
- Gáspárová, Z.; Ondrejičková, O.; Gajdošíková, A.; Gajdošík, A.; Šnirc, V.; Štolc, S. Oxidative stress induced by the Fe2+/ascorbic acid system or model ischemia in vitro: Effect of carvedilol and pyridoindole antioxidant SMe1EC2 in young and adult rat brain tissue. Interdiscip. Toxicol. 2010, 3, 122–126. [Google Scholar] [CrossRef]
- Yara, S.; Lavoie, J.-C.; Beaulieu, J.-F.; Delvin, E.; Amre, D.; Marcil, V.; Seidman, E.; Levy, E. Iron-Ascorbate-Mediated Lipid Peroxidation Causes Epigenetic Changes in the Antioxidant Defense in Intestinal Epithelial Cells: Impact on Inflammation. PLoS ONE 2013, 8, e63456. [Google Scholar] [CrossRef] [PubMed]
- Varma, S.D.; Hegde, K.R. Prevention of Oxidative Damage to Lens by Caffeine. J. Ocul. Pharmacol. Ther. 2010, 26, 73–78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gazdag, Z.; Máté, G.; Čertik, M.; Türmer, K.; Virág, E.; Pócsi, I.; Pesti, M. tert-Butyl hydroperoxide-induced differing plasma membrane and oxidative stress processes in yeast strains BY4741 and erg5D. J. Basic Microbiol. 2014, 54, S50–S62. [Google Scholar] [CrossRef]
- Feng, J.; Chen, X.; Sun, X.; Wang, F.; Sun, X. Expression of endoplasmic reticulum stress markers GRP78 and CHOP induced by oxidative stress in blue light-mediated damage of A2E-containing retinal pigment epithelium cells. Ophthalmic Res. 2014, 52, 224–233. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.-W.; Chiang, M.-T.; Wang, C.-Y.; Lii, C.-K. Inhibition of tert-butyl hydroperox-ide-induced cell membrane bleb formation by α-tocopherol and glutathione. Food Chem. Toxicol. 2000, 38, 1089–1096. [Google Scholar] [CrossRef]
- Prasanthi, J.R.; Dasari, B.; Marwarha, G.; Larson, T.; Chen, X.; Geiger, J.D.; Ghribi, O. Caffeine protects against oxidative stress and Alzheimer’s disease-like pathology in rabbit hippocampus induced by cholesterol-enriched diet. Free. Radic. Biol. Med. 2010, 49, 1212–1220. [Google Scholar] [CrossRef] [Green Version]
- Devasagayam, T.P.; Kamat, J.P.; Mohan, H.; Kesavan, P.C. Caffeine as an antioxidant: In-hibition of lipid peroxidation induced by reactive oxygen species. Biochim. Biophys. Acta 1996, 1282, 63–70. [Google Scholar] [CrossRef] [Green Version]
- Lee, C. Antioxidant ability of caffeine and its metabolites based on the study of oxygen radical absorbing capacity and inhibition of LDL peroxidation. Clin. Chim. Acta 2000, 295, 141–154. [Google Scholar] [CrossRef]
- Shi, X.; Dalal, N.S.; Jain, A.C. Antioxidant behaviour of caffeine: Efficient scavenging of hydroxyl radicals. Food Chem. Toxicol. 1991, 29, 1–6. [Google Scholar] [CrossRef]
- Youdim, M.B.H.; Edmondson, D.; Tipton, K.F. The therapeutic potential of monoamine oxidase inhibitors. Nat. Rev. Neurosci. 2006, 7, 295–309. [Google Scholar] [CrossRef]
- Petzer, J.P.; Castagnoli, N.; Schwarzschild, M.A.; Chen, J.F.; Schyf, C.J. Dual-target-directed drugs that block monoamine oxidase B and adenosine A2A receptors for Parkinson’s disease. Neurotherapeutics 2009, 6, 141–151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jasiewicz, B.; Sierakowska, A. Caffeine and its analogues, antioxidants and applications. In Aging, 2nd ed.; Preedy, V.R., Patel, V.B., Eds.; Academic Press: Cambridge, MA, USA, 2020; pp. 155–164. [Google Scholar]
- Sims, N.R.; Anderson, M.F. Isolation of mitochondria from rat brain using Percoll density gradient centrifugation. Nat. Protoc. 2008, 3, 1228–1239. [Google Scholar] [CrossRef] [PubMed]
- Taupin, P.; Zini, S.; Cesselin, F.; Ben-Ari, Y.; Roisin, M.P. Subcellular Fractionation on Percoll Gradient of Mossy Fiber Synaptosomes: Morphological and Biochemical Characterization in Control and Degranulated Rat Hippocampus. J. Neurochem. 1994, 62, 1586–1595. [Google Scholar] [CrossRef] [PubMed]
- Mungarro-Menchaca, X.; Ferrera, P.; Moran, J.; Arias, C. Beta-Amyloid Peptide Induces Ultrastructural Changes in Synaptosomes and Potentiates Mitochondrial Dysfunction in the Presence of Ryanodine. J. Neurosci. 2002, 68, 89–96. [Google Scholar]
- Robyt, F.; Ackerman, R.J.; Chittenden, C.G. Reaction of Protein Disulfide Groups with Ellman’s Reagent: A Case Study of the Number of Sulfhydryl and Disulfide Groups in Aspergillus Oryzae—Amylase, Papain, and Lysosome. Arch. Biochem. Biophys. 1971, 147, 262–269. [Google Scholar] [CrossRef]
- Kondeva-Burdina, M.; Voynova, M.; Aluani, D.; Tzankova, V.; Krasteva, I. Effects of Amanita muscaria extract on different in vitro neurotoxicity models at sub-cellular and cellular levels. Food Chem.Toxicol. 2019, 132, 110687. [Google Scholar] [CrossRef]
- Karlsson, J.; Emgard, M.; Brundin, P.; Burkitt, M.J. Trans-Resveratrol protects embryonic mesencephalic cells from tert-butylhydroperoxide: Electron paramagnetic resonances pintrapping evidence for radical scavenging mechanism. J. Neurochem. 2000, 75, 141–150. [Google Scholar] [CrossRef]
- Shirani, M.; Alizadeh, S.; Mahdavinia, M.; Dehghani, M.A. The ameliorative effect of quercetin on bisphenol A-induced toxicity in mitochondria isolated from rats. Environ. Sci. Pollut. Res. Int. 2019, 26, 7688–7696. [Google Scholar] [CrossRef]
- Ravindranath, V.; Anandatheerthavarada, H.K. Preparation of brain microsomes with cytochrome P450 activity using calcium aggregation method. Anal. Biochem. 1990, 187, 310–313. [Google Scholar] [CrossRef]
- Mansuy, D.; Sassi, A.; Dansette, P.M.; Plat, M. A new potent inhibitor of lipid peroxidation in vitro and in vivo, the hepatoprotective drug anisyldithiolthione. Biochem. Biophys. Res. Commun. 1986, 135, 1015–1021. [Google Scholar] [CrossRef]
- Bautista-Aguilera, O.M.; Esteban, G.; Bolea, I.; Nikolic, K.; Agbaba, D.; Moraleda, I.; Iriepa, I.; Samadi, A.; Soriano, E.; Unzeta, M.; et al. Design, synthesis, pharmacological evaluation, QSAR analysis, molecular modeling and ADMET of novel donepezil-indolyl hybrids as multipotent cholinesterase/monoamine oxidase inhibitors for the potential treatment of Alzheimer’s disease. J. Eur. J. Med. Chem. 2014, 75, 82–95. [Google Scholar] [CrossRef] [PubMed]
- Hocquet, A.; Langgård, M. An Evaluation of the MM+ Force Field. J. Mol. Med. 1998, 4, 94–112. [Google Scholar] [CrossRef]
- Dewar, M.; Zoebisch, E.; Healy, E.; Stewart, J. Development and use of quantum mechanical molecular models. 76. AM1: A new general purpose quantum mechanical molecular model. J. Am. Chem. Soc. 1985, 107, 3902–3909. [Google Scholar] [CrossRef]
- Leardi, R.; Boggia, R.; Terrile, M. Genetic algorithms as a strategy for feature selection. J. Chemom. 1992, 6, 267–281. [Google Scholar] [CrossRef]










 | ||||||
|---|---|---|---|---|---|---|
| Compound | R1 | R2 | R3 | R4 | R5 | R6 |
| 6a | H | H | H | H | H | H |
| 6b | H | H | NO2 | H | H | H |
| 6c | Cl | H | H | H | Cl | H |
| 6d | H | H | H | Cl | H | H |
| 6e | H | H | Br | H | H | H |
| 6f | H | H | CF3 | H | H | H |
| 6g | H | OCH3 | OCH3 | H | H | H |
| 6h | H | OCH3 | OCH3 | OCH3 | H | H |
| 6i | H | OCH3 | OH | OCH3 | H | H |
| 6j | H | H | OH | OCH3 | H | H |
| 6k | OCH3 | OCH3 | H | H | H | H |
| 6l | OH | H | H | H | H | H |
| 6m | CH3 | H | CH3 | H | CH3 | H |
| 6n | OH | OCH3 | H | H | H | H |
| 6o | H | NO2 | H | H | H | H |
| 6p | H | NO2 | OH | OCH3 | H | H |
| Compound ID | RV | RMDA | LogP_ACD | Dipole (D) | xc3 |
|---|---|---|---|---|---|
| 1 | 1.000 | 1.000 | −0.13 | 2.657 | 1.253 |
| 6a | 0.743 | 1.938 | 3.410 | 8.639 | 1.869 |
| 6b | 0.703 | 2.063 | 3.330 | 8.706 | 2.369 |
| 6c | 0.689 | 1.563 | 4.800 | 11.438 | 2.272 |
| 6d | 0.730 | 1.750 | 4.210 | 7.708 | 2.158 |
| 6e | 0.743 | 2.563 | 4.340 | 9.912 | 2.158 |
| 6f | 0.757 | 3.000 | 4.380 | 9.161 | 3.379 |
| 6g | 0.716 | 2.438 | 3.530 | 16.350 | 2.202 |
| 6h | 0.703 | 2.563 | 3.400 | 20.764 | 2.338 |
| 6i | 0.743 | 2.313 | 2.790 | 21.067 | 2.39 |
| 6j | 0.730 | 2.063 | 3.040 | 17.462 | 2.271 |
| 6k | 0.959 | 1.188 | 3.130 | 9.319 | 2.134 |
| 6l | 0.973 | 1.125 | 3.490 | 7.687 | 2.067 |
| 6m | 0.716 | 2.250 | 4.790 | 8.943 | 2.561 |
| 6n | 0.689 | 2.063 | 3.360 | 8.788 | 2.191 |
| 6o | 0.757 | 1.750 | 3.510 | 8.591 | 2.369 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kondeva-Burdina, M.; Mitkov, J.; Valkova, I.; Peikova, L.; Georgieva, M.; Zlatkov, A. Quantitative Structure–Neurotoxicity Assessment and In Vitro Evaluation of Neuroprotective and MAO-B Inhibitory Activities of Series N′-substituted 3-(1,3,7-trimethyl-xanthin-8-ylthio)propanehydrazides. Molecules 2022, 27, 5321. https://doi.org/10.3390/molecules27165321
Kondeva-Burdina M, Mitkov J, Valkova I, Peikova L, Georgieva M, Zlatkov A. Quantitative Structure–Neurotoxicity Assessment and In Vitro Evaluation of Neuroprotective and MAO-B Inhibitory Activities of Series N′-substituted 3-(1,3,7-trimethyl-xanthin-8-ylthio)propanehydrazides. Molecules. 2022; 27(16):5321. https://doi.org/10.3390/molecules27165321
Chicago/Turabian StyleKondeva-Burdina, Magdalena, Javor Mitkov, Iva Valkova, Lily Peikova, Maya Georgieva, and Alexander Zlatkov. 2022. "Quantitative Structure–Neurotoxicity Assessment and In Vitro Evaluation of Neuroprotective and MAO-B Inhibitory Activities of Series N′-substituted 3-(1,3,7-trimethyl-xanthin-8-ylthio)propanehydrazides" Molecules 27, no. 16: 5321. https://doi.org/10.3390/molecules27165321




