New pecJ-n (n = 1, 2) Basis Sets for High-Quality Calculations of Indirect Nuclear Spin–Spin Coupling Constants Involving 31P and 29Si: The Advanced PEC Method
Abstract
1. Introduction
2. Results and Discussion
2.1. On the Creation of New pecJ-n (n = 1, 2) Basis Sets for Phosphorus and Silicon
2.2. The Performance of New pecJ-n (n = 1, 2) Basis Sets
3. Computational Details
4. Concluding Remarks
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rusakova, I.L. Quantum Chemical Approaches to the Calculation of NMR Parameters: From Fundamentals to Recent Advances. Magnetochemistry 2022, 8, 50. [Google Scholar] [CrossRef]
- Helgaker, T.; Coriani, S.; Jørgensen, P.; Kristensen, K.; Olsen, J.; Ruud, K. Recent advances in wave function-based methods of molecular-property calculations. Chem. Rev. 2012, 112, 543–631. [Google Scholar] [CrossRef] [PubMed]
- Kjær, H.; Sauer, S.P.A. Pople style basis sets for the calculation of NMR spin-spin coupling constants: The 6-31G-J and 6-311G-J basis sets. J. Chem. Theory Comput. 2011, 7, 4070–4076. [Google Scholar] [CrossRef] [PubMed]
- Oddershede, J.; Geertsen, J.; Scuseria, G.E. Nuclear spin-spin coupling constant of hydrogen molecule with deuterium (HD). J. Phys. Chem. 1988, 92, 3056–3059. [Google Scholar] [CrossRef]
- Geertsen, J.; Oddershede, J.; Raynes, W.T.; Scuseria, G.E. Nuclear spin-spin coupling in the methane isotopomers. J. Magn. Reson. 1991, 93, 458–471. [Google Scholar] [CrossRef]
- Geertsen, J.; Oddershede, J.; Scuseria, G.E. Spin–spin coupling constants of CO and N2. J. Chem. Phys. 1987, 87, 2138–2142. [Google Scholar] [CrossRef]
- Provasi, P.F.; Sauer, S.P.A. Optimized basis sets for the calculation of indirect nuclear spin-spin coupling constants involving the atoms B, Al, Si, P, and Cl. J. Chem. Phys. 2010, 133, 054308. [Google Scholar] [CrossRef]
- Jensen, F. The basis set convergence of spin–spin coupling constants calculated by density functional methods. J. Chem. Theory Comput. 2006, 2, 1360–1369. [Google Scholar] [CrossRef]
- Enevoldsen, T.; Oddershede, J.; Sauer, S.P.A. Correlated calculations of indirect nuclear spin-spin coupling constants using second-order polarization propagator approximations: SOPPA and SOPPA(CCSD). Theor. Chem. Acc. 1998, 100, 275–284. [Google Scholar] [CrossRef]
- Helgaker, T.; Jaszuński, M.; Ruud, K. Ab Initio Methods for the Calculation of NMR Shielding and Indirect Spin−Spin Coupling Constants. Chem. Rev. 1999, 99, 293–352. [Google Scholar] [CrossRef]
- Helgaker, T.; Jaszuński, M.; Ruud, K.; Górska, A. Basis-set dependence of nuclear spin-spin coupling constants. Theor. Chem. Acc. 1998, 99, 175–182. [Google Scholar] [CrossRef]
- Provasi, P.F.; Aucar, G.A.; Sauer, S.P.A. The effect of lone pairs and electronegativity on the indirect nuclear spin–spin coupling constants in CH2X (X=CH2, NH, O, S): Ab initio calculations using optimized contracted basis sets. J. Chem. Phys. 2001, 115, 1324–1334. [Google Scholar] [CrossRef]
- Barone, V.; Provasi, P.F.; Peralta, J.E.; Snyder, J.P.; Sauer, S.P.A.; Contreras, R.H. Substituent effects on scalar 2J(19F,19F) and 3J(19F,19F) NMR couplings: A comparison of SOPPA and DFT methods. J. Phys. Chem. A 2003, 107, 4748–4754. [Google Scholar] [CrossRef]
- Sauer, S.P.A.; Raynes, W.T. Unexpected differential sensitivity of nuclear spin-spin-coupling constants to bond stretching in BH4−, NH4+, and SiH4. J. Chem. Phys. 2000, 113, 3121–3129. [Google Scholar] [CrossRef]
- Sauer, S.P.A.; Raynes, W.T.; Nicholls, R.A. Nuclear spin-spin coupling in silane and its isotopomers: Ab initio calculation and experimental investigation. J. Chem. Phys. 2001, 115, 5994–6006. [Google Scholar] [CrossRef]
- Hedegård, E.D.; Kongsted, J.; Sauer, S.P.A. Optimized basis sets for calculation of electron paramagnetic resonance hyperfine coupling constants: Aug-cc-pVTZ-J for the 3d atoms Sc-Zn. J. Chem. Theory Comput. 2011, 7, 4077–4087. [Google Scholar] [CrossRef]
- Rusakov, Y.Y.; Krivdin, L.B.; Sauer, S.P.A.; Levanova, E.P.; Levkovskaya, G.G. Structural trends of 77Se-1H spin–spin coupling constants and conformational behavior of 2-substituted selenophenes. Magn. Reson. Chem. 2010, 48, 44–52. [Google Scholar] [CrossRef]
- Rusakov, Y.Y.; Krivdin, L.B.; Østerstrøm, F.F.; Sauer, S.P.A.; Potapov, V.A.; Amosova, S.V. First example of a high-level correlated calculation of the indirect spin–spin coupling constants involving tellurium: Tellurophene and divinyl telluride. Phys. Chem. Chem. Phys. 2013, 15, 13101–13107. [Google Scholar] [CrossRef]
- Rusakov, Y.Y.; Rusakova, I.L. Hierarchical basis sets for the calculation of nuclear magnetic resonance spin–spin coupling constants involving either selenium or tellurium nuclei. J. Phys. Chem. A 2019, 123, 6564–6571. [Google Scholar] [CrossRef]
- Rusakov, Y.Y.; Rusakova, I.L. Efficient J-oriented tin basis sets for the correlated calculations of indirect nuclear spin–spin coupling constants. Magn. Reson. Chem. 2021, 59, 713–722. [Google Scholar] [CrossRef]
- Helgaker, T.; Watson, M.; Handy, N.C. Analytical calculation of nuclear magnetic resonance indirect spin–spin coupling constants at the generalized gradient approximation and hybrid levels of density-functional theory. J. Chem. Phys. 2000, 113, 9402–9409. [Google Scholar] [CrossRef]
- Huzinaga, S. Gaussian-Type Functions for Polyatomic Systems. I. J. Chem. Phys. 1965, 42, 1293–1302. [Google Scholar] [CrossRef]
- Ruden, T.A.; Lutnæs, O.B.; Helgaker, T. Vibrational corrections to indirect nuclear spin–spin coupling constants calculated by density-functional theory. J. Chem. Phys. 2003, 118, 9572–9581. [Google Scholar] [CrossRef]
- Steinmann, C.; Sauer, S.P.A. The aug-cc-pVTZ-J basis set for the p-block fourth-row elements Ga, Ge, As, Se, and Br. Magn. Reson. Chem. 2021, 59, 1134–1145. [Google Scholar] [CrossRef]
- Jensen, F. The optimum contraction of basis sets for calculating spin–spin coupling constants. Theor. Chem. Acc. 2010, 126, 371–382. [Google Scholar] [CrossRef]
- Aggelund, P.A.; Sauer, S.P.A.; Jensen, F. Development of polarization consistent basis sets for spin-spin coupling constant calculations for the atoms Li, Be, Na, and Mg. J. Chem. Phys. 2018, 149, 044117. [Google Scholar] [CrossRef]
- Benedikt, U.; Auer, A.A.; Jensen, F. Optimization of augmentation functions for correlated calculations of spin-spin coupling constants and related properties. J. Chem. Phys. 2008, 129, 064111. [Google Scholar] [CrossRef]
- Rusakov, Y.Y.; Rusakov, I.L. An efficient method for generating property-energy consistent basis sets. New pecJ-n (n = 1, 2) basis sets for high-quality calculations of indirect nuclear spin–spin coupling constants involving 1H, 13C, 15N, and 19F nuclei. Phys. Chem. Chem. Phys. 2021, 23, 14925–14939. [Google Scholar] [CrossRef]
- Metropolis, N.; Ulam, S. The Monte Carlo Method. J. Am. Stat. Assoc. 1949, 44, 335–341. [Google Scholar] [CrossRef]
- Harrison, R.L. Introduction to Monte Carlo Simulation. AIP Conf. Proc. 2010, 1204, 17–21. [Google Scholar] [CrossRef]
- Del Moral, P.; Doucet, A.; Jasra, A. Sequential Monte Carlo samplers. J. R. Statist. Soc. B 2006, 68, 411–436. [Google Scholar] [CrossRef]
- Rusakov, Y.Y.; Krivdin, L.B.; Nosova, V.M.; Kisin, A.V. Benchmark calculations of 29Si–1H spin–spin coupling constants across double bond. Magn. Reson. Chem. 2012, 50, 278–283. [Google Scholar] [CrossRef] [PubMed]
- Rusakov, Y.Y.; Krivdin, L.B. One-bond 29Si-1H spin-spin coupling constants in the series of halosilanes: Benchmark SOPPA and DFT calculations, relativistic effects, and vibrational corrections. Magn. Reson. Chem. 2013, 51, 557–561. [Google Scholar] [CrossRef]
- Krivdin, L.B.; Rusakov, Y.Y. Structural and Stereochemical Applications of Computational NMR Using 29Si–1H and 77Se–1H Indirect Spin–Spin Coupling Constants. Emagres 2014, 3, 87–110. [Google Scholar] [CrossRef]
- Rusakova, I.L.; Rusakov, Y.Y.; Krivdin, L.B. Nonempirical calculations of the one-bond 29Si–13C spin–spin coupling constants taking into account relativistic and solvent corrections. Magn. Reson. Chem. 2014, 52, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Garbacz, P.; Makulski, W.; Jaszuński, M. The NMR spin–spin coupling constant 1J(31P,1H) in an isolated PH3 molecule. Phys. Chem. Chem. Phys. 2014, 16, 21559–21563. [Google Scholar] [CrossRef]
- Pecul, M.; Urbańczyk, M.; Wodyński, A.; Jaszuńskib, M. DFT calculations of 31P spin–spin coupling constants and chemical shift in dioxaphosphorinanes. Magn. Reson. Chem. 2011, 49, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Fedorov, S.V.; Krivdin, L.B.; Rusakov, Y.Y.; Ushakov, I.A.; Istomina, N.V.; Belogorlova, N.A.; Malysheva, S.F.; Gusarova, N.K.; Trofimov, B.A. Conformational analysis and stereochemical dependences of 31P–1H spin–spin coupling constants of bis(2-phenethyl)vinylphosphine and related phosphine chalcogenides. Magn. Reson. Chem. 2009, 47, 288–299. [Google Scholar] [CrossRef] [PubMed]
- Fedorov, S.V.; Krivdin, L.B.; Chernysheva, N.A.R.; Mikhailenko, V.L. Trivinylphosphine and trivinylphosphine chalcogenides: Stereochemical trends of 31P–1H spin–spin coupling constants. Magn. Reson. Chem. 2010, 48, S48–S55. [Google Scholar] [CrossRef]
- Rusakov, Y.Y.; Rusakova, I.L.; Fedorov, S.V.; Gray, G.A.; Krivdin, L.B. Stereochemical Dependences of 31P−13C Spin−Spin Coupling Constants of Heterocyclic Phosphines. J. Phys. Chem. A 2019, 123, 6298–6303. [Google Scholar] [CrossRef]
- Rusakova, I.L.; Rusakov, Y.Y. Correlated ab initio calculations of one-bond 31P−77Se and 31P−125Te spin–spin coupling constants in a series of P=Se and P=Te systems accounting for relativistic effects (part 2). Magn. Reson. Chem. 2020, 58, 929–940. [Google Scholar] [CrossRef]
- Kjær, H.; Sauer, S.P.A.; Kongsted, J. Benchmarking NMR indirect nuclear spin-spin coupling constants: SOPPA, SOPPA(CC2), and SOPPA(CCSD) versus CCSD. J. Chem. Phys. 2010, 133, 144106. [Google Scholar] [CrossRef] [PubMed]
- Malkina, O.L.; Salahub, D.R.; Malkin, V.G. Nuclear magnetic resonance spin–spin coupling constants from density functional theory: Problems and results. J. Chem. Phys. 1996, 105, 8793–8800. [Google Scholar] [CrossRef]
- Jensen, F. Method calibration or data fitting? J. Chem. Theory Comput. 2018, 14, 4651–4661. [Google Scholar] [CrossRef]
- Almlöf, J.; Taylor, P.R. Atomic Natural Orbital (ANO) basis sets for quantum chemical calculations. Adv. Quantum Chem. 1991, 22, 301–373. [Google Scholar] [CrossRef]
- Raffenetti, R.C. General contraction of Gaussian atomic orbitals: Core, valence, polarization, and diffuse basis sets; molecular integral evaluation. J. Chem. Phys. 1973, 58, 4452–4458. [Google Scholar] [CrossRef]
- Dunning, T.H., Jr.; Hay, P.J. Methods of electronic structure theory. In Modern Theoretical Chemistry, 1st ed.; Schaefer, H.F., III, Ed.; Springer, Science & Business Media: New York, NY, USA, 1977; Volume 3, pp. 1–27. [Google Scholar]
- Rusakov, Y.Y.; Rusakova, I.L. New pecS-n (n = 1, 2) basis sets for quantum chemical calculations of the NMR chemical shifts of H, C, N, and O nuclei. J. Chem. Phys. 2022, 156, 244112. [Google Scholar] [CrossRef] [PubMed]
- Sekino, H.; Bartlett, R.J. Nuclear spin–spin coupling constants evaluated using many body methods. J. Chem. Phys. 1986, 85, 3945–3949. [Google Scholar] [CrossRef]
- Bartlett, R.J. Many-Body Perturbation Theory and Coupled Cluster Theory for Electron Correlation in Molecules. Annu. Rev. Phys. Chem. 1981, 32, 359–401. [Google Scholar] [CrossRef]
- Reeves, C.M.; Fletcher, R. Use of Gaussian functions in the calculation of wavefunctions for small molecules. III. The orbital basis and its effect on valence. J. Chem. Phys. 1965, 42, 4073–4081. [Google Scholar] [CrossRef]
- Van Mourik, T.; Dunning, T.H. Gaussian basis sets for use in correlated molecular calculations. VIII. Standard and augmented sextuple zeta correlation consistent basis sets for aluminum through argon. Int. J. Quantum Chem. 2000, 76, 205–221. [Google Scholar] [CrossRef]
- Woon, D.E.; Dunning, T.H. Gaussian basis sets for use in correlated molecular calculations. III. The atoms aluminum through argon. J. Chem. Phys. 1993, 98, 1358–1371. [Google Scholar] [CrossRef]
- Ruden, T.A.; Ruud, K. Calculation of NMR and EPR Parameters. In Theory and Applications; Kaupp, M., Bühl, M., Malkin, V.G., Eds.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2004; Chapter 10; pp. 153–173. [Google Scholar]
- Adamo, C.; Barone, V. Toward reliable density functional methods without adjustable parameters: The PBE0 model. J. Chem. Phys. 1999, 110, 6158–6170. [Google Scholar] [CrossRef]
- Perdew, J.P.; Ernzerhof, M.; Burke, K. Rationale for mixing exact exchange with density functional approximations. J. Chem. Phys. 1996, 105, 9982–9985. [Google Scholar] [CrossRef]
- Tomasi, J.; Mennucci, B.; Cancès, E. The IEF version of the PCM solvation method: An overview of a new method addressed to study molecular solutes at the QM ab initio level. J. Mol. Struct. THEOCHEM 1999, 464, 211–226. [Google Scholar] [CrossRef]
- Tomasi, J.; Mennucci, B.; Cammi, R. Quantum Mechanical Continuum Solvation Models. Chem. Rev. 2005, 105, 2999–3094. [Google Scholar] [CrossRef]
- Dyall, K.G. Relativistic double-zeta, triple-zeta, and quadruple-zeta basis sets for the light elements H–Ar. Theor. Chem. Acc. 2016, 135, 128. [Google Scholar] [CrossRef]
- Grant, I.P.; Quiney, H.M. Application of relativistic theories and quantum electrodynamics to chemical problems. Int. J. Quantum Chem. 2000, 80, 283–297. [Google Scholar] [CrossRef]
- Liu, W. Ideas of relativistic quantum chemistry. Mol. Phys. 2010, 108, 1679–1706. [Google Scholar] [CrossRef]
- Dyall, K.G.; Faegri, K. Kinetic balance and variational bounds failure in the solution of the Dirac equation in a finite Gaussian basis set. Chem. Phys. Lett. 1990, 174, 25–32. [Google Scholar] [CrossRef]
- Ashe, A.J., III; Sharp, R.R.; Tolan, J.W. The nuclear magnetic resonance spectra of phosphabenzene, arsabenzene, and stibabenzene. J. Am. Chem. Soc. 1976, 98, 5451–5456. [Google Scholar] [CrossRef]
- Guillemin, J.-C.; Janati, T.; Denis, J.-M. A Simple Route to Kinetically Unstabilized Phosphaalkynes. J. Org. Chem. 2001, 66, 7864–7868. [Google Scholar] [CrossRef] [PubMed]
- Vongehr, M.; Marsmann, H.C. Die Anwendung von Parametern der gegenseitigen Wechselwirkung in der 29Si-Resonanz. Z. Naturforsch. 1976, 31, 1423–1424. [Google Scholar] [CrossRef]
- Jackowski, K. Gas–Phase Studies of Spin–Spin Coupling Constants. Int. J. Mol. Sci. 2003, 4, 135–142. [Google Scholar] [CrossRef]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 1988, 37, 785–789. [Google Scholar] [CrossRef]
- Becke, A.D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Noga, J.; Bartlett, R.J. The full CCSDT model for molecular electronic structure. J. Chem. Phys. 1987, 86, 7041–7050. [Google Scholar] [CrossRef]
- Scuseria, G.E.; Schaefer, H.F. A new implementation of the full CCSDT model for molecular electronic structure. Chem. Phys. Lett. 1988, 152, 382–386. [Google Scholar] [CrossRef]
- Stanton, J.F.; Gauss, J.; Cheng, L.; Harding, M.E.; Matthews, D.A.; Szalay, P.G.; Auer, A.A.; Bartlett, R.J.; Benedikt, U.; Berger, C.; et al. CFOUR, a Quantum Chemical Program Package. Available online: http://www.cfour.de (accessed on 15 September 2022).
- Dunning, T.H. Gaussian basis sets for use in correlated molecular calculations. I. The atoms boron through neon and hydrogen. J. Chem. Phys. 1989, 90, 1007–1023. [Google Scholar] [CrossRef]
- Kendall, R.A.; Dunning, T.H.; Harrison, R.J. Electron affinities of the first-row atoms revisited. Systematic basis sets and wave functions. J. Chem. Phys. 1992, 96, 6796–6806. [Google Scholar] [CrossRef]
- Aidas, K.; Angeli, C.; Bak, K.L.; Bakken, V.; Bast, R.; Boman, L.; Christiansen, O.; Cimiraglia, R.; Coriani, S.; Dahle, P.; et al. The Dalton quantum chemistry program system. WIREs Comput. Mol. Sci. 2014, 4, 269–284. [Google Scholar] [CrossRef] [PubMed]
- Gomes, A.S.P.; Saue, T.; Visscher, L.; Jensen, H.J.A.; Bast, R.; Aucar, A.; Bakken, V.; Dyall, K.G.; Dubillard, S.; Ekström, U.; et al. DIRAC, a Relativistic Ab Initio Electronic Structure Program, Release DIRAC19. 2019. Available online: http://www.diracprogram.org (accessed on 15 September 2022).
- Van Rossum, G.; Drake, F.L. Python 3 Reference Manual; CreateSpace: Scotts Valley, CA, USA, 2009; ISBN 978-1-4414-1269-0. [Google Scholar]
- Irkutsk Supercomputer Center of SB RAS. Irkutsk: ISDCT SB RAS. Available online: https://hpc.icc.ru (accessed on 1 August 2022).
| Original dyall.aae4z | dyall.aae4z+ | Additional ζi |
|---|---|---|
| H: (12s, 4p, 3d, 2f) | +3s | ζ1 = 1.42009326 × 106 ζ2 = 2.05353505 × 105 ζ3 = 2.96952766 × 104 |
| C: (19s, 11p, 6d, 4f, 2g) | +2s | ζ1 = 6.64165270 × 107 ζ2 = 9.15768223 × 106 |
| Si, P: (25s, 15p, 10d, 7f, 4g) | +1s | Si: ζ1 = 3.37124315 × 108 P: ζ1 = 3.68247433 × 108 |
| Basis Set | Simple Contraction Scheme | Extended Contraction Scheme | Nc/Nuc |
|---|---|---|---|
| pecJ-1 | (14s, 8p, 3d|8s, 5p, 3d) | 8s: (8, 8, 1, 1, 1, 1, 1, 1) 5p: (5, 5, 1, 1, 1) 3d: (1, 1, 1) | 38/53 |
| pecJ-2 | (17s, 9p, 4d, 1f|10s, 6p, 4d, 1f) | 10s: (9, 9, 1, 1, 1, 1, 1, 1, 1, 1) 6p: (5, 5, 1, 1, 1, 1) 4d: (1, 1, 1, 1) 1f: (1) | 55/71 |
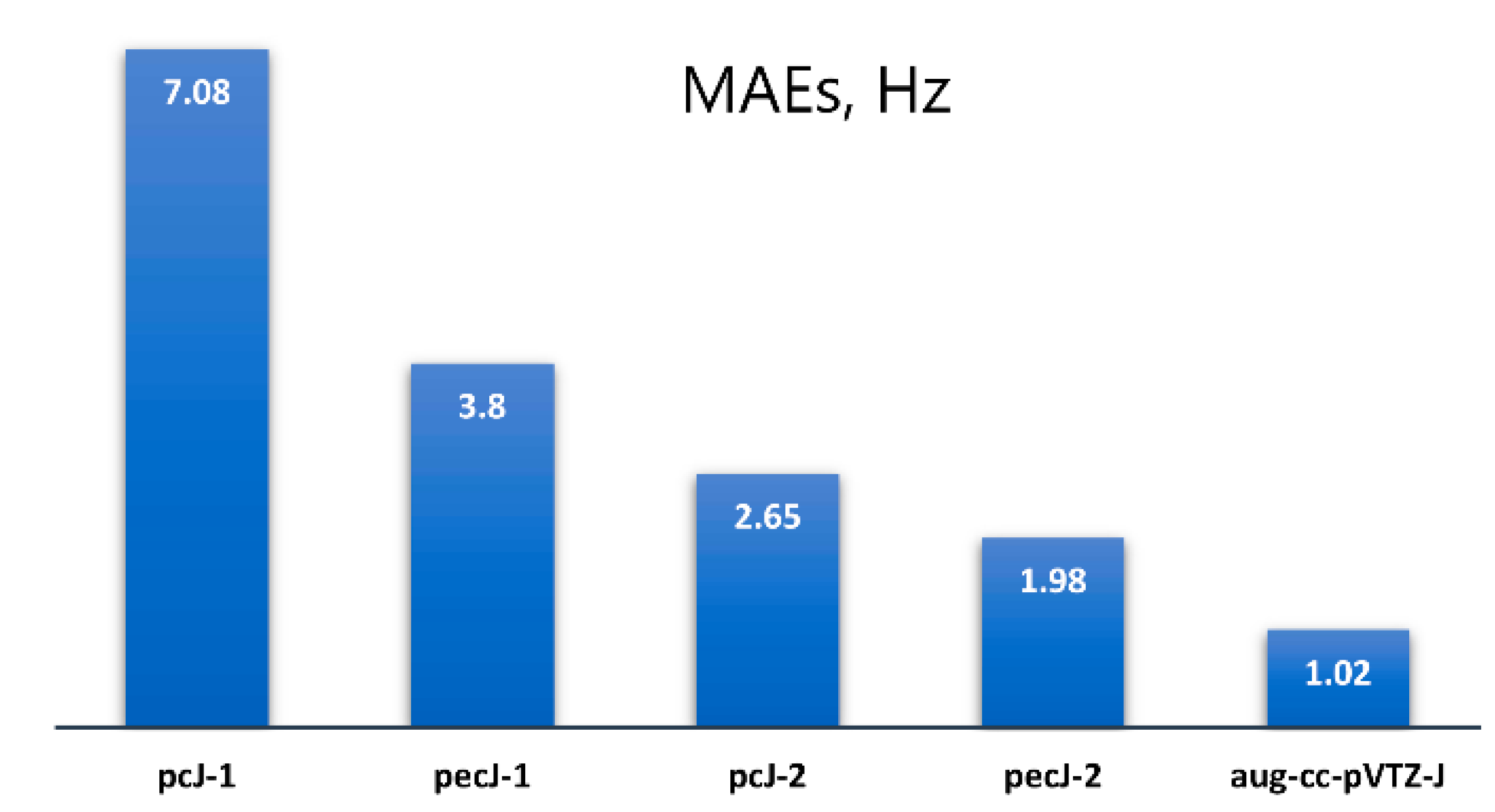
| # | Molecule | SSCC 1 | dyall.aae4z+ 2 | pecJ-1 | pecJ-2 | pcJ-1 | pcJ-2 | aug-cc-pVTZ-J |
|---|---|---|---|---|---|---|---|---|
| 1 |  Phosphabenzene | 1J(P,C2) | −46.27 | −37.68 | −43.13 | −29.57 | −42.41 | −42.82 |
| 2J(P,C3) | −19.88 | −19.72 | −19.87 | −20.23 | −20.16 | −19.99 | ||
| 3J(P,C4) | 29.52 | 28.78 | 29.27 | 28.46 | 29.44 | 28.40 | ||
| 2J(P,H2) | 32.96 | 30.81 | 32.34 | 30.81 | 32.05 | 33.28 | ||
| 3J(P,H3) | 9.49 | 9.61 | 9.27 | 9.66 | 9.40 | 9.49 | ||
| 4J(P,H4) | −7.01 | −7.13 | −6.74 | −6.89 | −7.08 | −6.84 | ||
| 2 | P≡CH Phosphaethyne | 1J(P,C) | 78.11 | 74.25 | 76.46 | 90.63 | 73.94 | 76.83 |
| 2J(P,H) | 53.72 | 50.97 | 53.03 | 51.87 | 52.34 | 52.59 | ||
| 3 | H2N–PH2 Phosphanamine | 1J(P,N) | −3.73 | 0.93 | −2.65 | 1.71 | −2.50 | −3.85 |
| 1J(P,H) | 191.34 | 188.90 | 190.64 | 189.73 | 191.82 | 191.61 | ||
| 2J(P,H) | 11.81 | 10.91 | 11.49 | 11.98 | 11.35 | 11.61 | ||
| 4 | O=PH3 Phosphine oxide | 1J(P,H) | 445.05 | 441.47 | 441.86 | 439.92 | 446.41 | 445.73 |
| 5 | PF3 Phosphorus trifluoride | 1J(P, F) | −1409.57 | −1430.81 | −1381.37 | −1337.65 | −1382.72 | −1402.76 |
| 6 |  Methylphosphane | 1J(P,C) | 0.00 | 5.84 | 2.00 | 10.03 | 3.43 | 0.73 |
| 1J(P,Hα) | 195.19 | 194.72 | 194.30 | 190.91 | 195.49 | 195.81 | ||
| 2J(P,Hβ) | 7.56 | 6.24 | 6.94 | 6.65 | 6.94 | 7.26 | ||
| 2J(P,Hβ’) | −8.81 | −9.08 | −9.07 | −9.05 | −9.16 | −9.24 | ||
| 7 | H2P–F Fluorophosphane | 1J(P,F) | −798.09 | −821.89 | −790.47 | −736.60 | −782.17 | −810.23 |
| 1J(P,H) | 191.59 | 190.72 | 192.66 | 193.93 | 192.24 | 191.96 | ||
| 8 | PH3 Phosphane | 1J(P,H) | 190.63 | 190.52 | 190.46 | 184.99 | 190.56 | 191.18 |
| 9 | 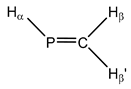 Methylenephosphane | 1J(P,C) | −25.56 | −15.30 | −22.50 | −7.66 | −21.78 | −21.93 |
| 1J(P,Hα) | 131.63 | 126.48 | 132.19 | 126.73 | 129.69 | 132.85 | ||
| 2J(P,Hβ) | −33.11 | −31.93 | −32.87 | −31.12 | −33.73 | −33.84 | ||
| 2J(P,Hβ’) | 24.18 | 21.36 | 23.02 | 23.38 | 22.95 | 24.04 | ||
| 10 | F2P–H Difluorophosphane | 1J(P,H) | 201.52 | 198.47 | 202.60 | 209.59 | 202.51 | 201.56 |
| 1J(P,F) | −1152.93 | −1168.95 | −1133.17 | −1074.49 | −1124.99 | −1152.84 | ||
| 11 | H2C=SiH2 Methylenesilane | 1J(Si,C) | −130.07 | −132.52 | −131.53 | −137.73 | −132.39 | −132.47 |
| 1J(Si,H) | −241.84 | −241.10 | −240.39 | −244.05 | −244.16 | −241.78 | ||
| 2J(Si,H) | 4.85 | 7.40 | 5.58 | 8.28 | 5.91 | 5.21 | ||
| 12 | CH≡SiH Methylidynesilane | 1J(Si,C) | −378.85 | −375.00 | −377.57 | −381.61 | −380.36 | −382.16 |
| 1J(Si,H) | −445.90 | −445.84 | −443.48 | −452.96 | −450.82 | −446.72 | ||
| 2J(Si,H) | −112.00 | −104.22 | −109.70 | −98.79 | −108.23 | −111.40 | ||
| 13 |  Silanamine | 1J(Si,N) | −15.26 | −16.17 | −15.61 | −16.56 | −15.96 | −15.67 |
| 1J(Si,Hα) | −199.09 | −197.59 | −197.50 | −199.06 | −200.47 | −199.21 | ||
| 1J(Si,Hα’) | −188.85 | −186.41 | −187.08 | −186.78 | −189.67 | −188.83 | ||
| 2J(Si,Hβ) | −1.11 | −0.80 | −1.04 | −1.05 | −1.10 | −1.04 | ||
| 14 | 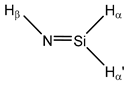 Silanimine | 1J(Si,N) | −4.53 | −5.37 | −4.73 | −6.39 | −4.81 | −5.02 |
| 1J(Si,Hα) | −214.71 | −208.67 | −211.00 | −209.13 | −213.74 | −214.03 | ||
| 1J(Si,Hα’) | −273.58 | −271.32 | −271.49 | −275.74 | −276.00 | −273.58 | ||
| 2J(Si,Hβ) | 0.88 | 2.38 | 1.36 | 2.15 | 1.26 | 1.15 | ||
| 15 | H3Si–CH3 Methylsilane | 1J(Si,C) | −54.02 | −55.82 | −54.77 | −58.66 | −55.72 | −54.93 |
| 1J(Si,H) | −184.74 | −183.56 | −183.57 | −184.81 | −186.05 | −184.78 | ||
| 2J(Si,H) | 8.30 | 8.96 | 8.42 | 9.27 | 8.59 | 8.53 | ||
| 16 | H3Si–F Fluorosilane | 1J(Si,H) | −217.11 | −216.95 | −216.31 | −217.40 | −218.39 | −217.41 |
| 1J(Si,F) | 248.88 | 257.46 | 249.12 | 245.43 | 247.17 | 247.66 | ||
| 17 | SiH4 Silane | 1J(Si,H) | −191.02 | −190.88 | −190.15 | −192.1 | −192.76 | −191.33 |
| 18 | F3Si–H Trifluorosilane | 1J(Si,H) | −344.22 | −345.76 | −344.27 | −345.12 | −343.74 | −343.93 |
| 1J(Si,F) | 239.41 | 252.97 | 241.19 | 243.83 | 238.43 | 237.91 | ||
| 19 |  Silylphosphane | 1J(P,Si) | 6.82 | −5.39 | 4.72 | −3.32 | −1.34 | 4.95 |
| 1J(P,Hα) | 189.24 | 186.39 | 186.98 | 181.46 | 187.49 | 189.96 | ||
| 1J(Si,Hβ) | −195.27 | −192.96 | −193.79 | −195.05 | −196.09 | −195.27 | ||
| 1J(Si,Hγ) | −201.51 | −198.33 | −199.60 | −198.99 | −201.44 | −201.50 | ||
| 2J(P,Hβ) | 25.41 | 24.92 | 24.84 | 24.42 | 25.00 | 25.27 | ||
| 2J(P,Hγ) | −5.16 | −3.44 | −4.70 | −4.42 | −5.17 | −5.40 | ||
| 2J(Si,Hα) | 8.32 | 9.44 | 8.58 | 10.09 | 8.87 | 8.78 | ||
| 20 |  Silylidenephosphane | 1J(P,Si) | 125.09 | 111.02 | 125.27 | 110.88 | 113.64 | 119.58 |
| 1J(P,Hα) | 135.77 | 132.54 | 133.50 | 126.41 | 130.96 | 136.70 | ||
| 1J(Si,Hβ) | −227.93 | −223.38 | −224.92 | −227.49 | −227.66 | −227.47 | ||
| 1J(Si,Hβ’) | −221.37 | −219.50 | −219.69 | −225.64 | −223.94 | −221.05 | ||
| 2J(P,Hβ) | −31.96 | −29.86 | −31.28 | −31.31 | −32.09 | −32.12 | ||
| 2J(P,Hβ’) | 36.13 | 34.66 | 35.64 | 35.60 | 35.42 | 36.13 | ||
| 2J(Si,Hα) | 23.50 | 24.30 | 24.24 | 25.91 | 24.51 | 24.57 |
| Basis Set | H | C, N, F | P, Si |
|---|---|---|---|
| pcJ-1/pcJ-1(uc) | 10/12 | 27/34 | 31/50 |
| pecJ-1/pecJ-1(uc) | 11/13 | 27/35 | 38/53 |
| pcJ-2/pcJ-2(uc) | 24/27 | 51/62 | 54/77 |
| pecJ-2/pecJ-2(uc) | 20/22 | 43/51 | 55/71 |
| aug-cc-pVTZ-J/aug-cc-pVTZ-J(uc) | 20/24 | 46/55 | 68/87 |
| Molecule | SSCC | Basis Set | CCSD | Δvib 1 | Δrel 2 | Δsol 3 | Jtot | Jexp4 | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| JFC | JSD | JPSO | JDSO | Jbasic | ||||||||
 1 | 1J(P,C2) | pecJ-1 | −10.14 | 5.09 | −36.72 | 0.20 | −41.57 | −4.33 | −4.91 | 2.95 | −47.86 | (−)53.0 |
| pecJ-2 | −12.23 | 5.12 | −37.5 | 0.20 | −44.41 | 3.40 | −50.25 | |||||
| 2J(P,C3) | pecJ-1 | −13.51 | −3.48 | 0.92 | −0.05 | −16.12 | −1.38 | 0.00 | −0.09 | −17.59 | −14.0 | |
| pecJ-2 | −13.56 | −3.42 | 0.93 | −0.05 | −16.10 | −0.26 | −17.74 | |||||
| 3J(P,C4) | pecJ-1 | 12.57 | 8.81 | 1.67 | −0.04 | 23.01 | 3.39 | −0.24 | 0.55 | 26.71 | 22.0 | |
| pecJ-2 | 12.21 | 9.04 | 1.78 | −0.04 | 22.99 | 0.68 | 26.82 | |||||
| 2J(P,H2) | pecJ-1 | 42.46 | −0.40 | −7.07 | −0.41 | 34.58 | −0.41 | 1.47 | −0.66 | 34.98 | 38.0 | |
| pecJ-2 | 43.42 | −0.3 | −7.09 | −0.42 | 35.61 | −0.75 | 35.92 | |||||
| 3J(P,H3) | pecJ-1 | 8.71 | −0.21 | 0.65 | −0.69 | 8.46 | 0.45 | −0.45 | 0.72 | 9.18 | 8.0 | |
| pecJ-2 | 8.58 | −0.28 | 0.70 | −0.70 | 8.30 | 0.77 | 9.07 | |||||
| 4J(P,H4) | pecJ-1 | −4.33 | −0.29 | 0.55 | −0.64 | −4.71 | −0.69 | 0.32 | −0.03 | −5.11 | −3.5 | |
| pecJ-2 | −4.17 | −0.15 | 0.61 | −0.64 | −4.35 | −0.07 | −4.79 | |||||
| P≡CH 2 | 1J(P,C) | pecJ-1 | 19.52 | 40.96 | 11.51 | 0.00 | 71.99 | −5.47 | −4.50 | −6.66 | 55.36 | 56.0 |
| pecJ-2 | 19.85 | 43.26 | 13.67 | 0.00 | 76.78 | −5.93 | 60.88 | |||||
| 2J(P,H) | pecJ-1 | 27.88 | 3.28 | 21.48 | −1.46 | 51.18 | −4.16 | 0.24 | 1.37 | 48.63 | 44.0 | |
| pecJ-2 | 25.73 | 5.36 | 23.46 | −1.47 | 53.08 | 1.17 | 50.33 | |||||
| PH3 8 | 1J(P,H) | pecJ-1 | 179.64 | −0.78 | 5.37 | 0.03 | 184.26 | −9.34 | −2.63 | Gas phase | 172.29 | 176.2 |
| pecJ-2 | 182.68 | −0.91 | 5.93 | 0.00 | 187.70 | 175.73 | ||||||
| H3Si–F 16 | 1J(Si,H) | pecJ-1 | −215.64 | −0.08 | 1.14 | −0.22 | −214.80 | −8.82 | −3.30 | −1.66 | −228.58 | (−)233.6 |
| pecJ-2 | −213.72 | −0.17 | 1.11 | −0.21 | −212.99 | −1.55 | −226.66 | |||||
| 1J(Si,F) | pecJ-1 | 212.82 | −7.32 | 58.57 | −0.08 | 263.99 | 8.69 | 10.43 | −3.82 | 279.29 | 278.7 | |
| pecJ-2 | 203.39 | −7.78 | 58.88 | −0.06 | 254.43 | −3.62 | 269.93 | |||||
| SiH4 17 | 1J(Si,H) | pecJ-1 | −189.89 | −0.02 | 0.48 | −0.03 | −189.46 | −7.27 | −2.96 | Gas phase | −199.69 | (−)201.9 |
| pecJ-2 | −188.71 | −0.07 | 0.43 | −0.02 | −188.37 | −198.60 | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rusakov, Y.Y.; Rusakova, I.L. New pecJ-n (n = 1, 2) Basis Sets for High-Quality Calculations of Indirect Nuclear Spin–Spin Coupling Constants Involving 31P and 29Si: The Advanced PEC Method. Molecules 2022, 27, 6145. https://doi.org/10.3390/molecules27196145
Rusakov YY, Rusakova IL. New pecJ-n (n = 1, 2) Basis Sets for High-Quality Calculations of Indirect Nuclear Spin–Spin Coupling Constants Involving 31P and 29Si: The Advanced PEC Method. Molecules. 2022; 27(19):6145. https://doi.org/10.3390/molecules27196145
Chicago/Turabian StyleRusakov, Yuriy Yu., and Irina L. Rusakova. 2022. "New pecJ-n (n = 1, 2) Basis Sets for High-Quality Calculations of Indirect Nuclear Spin–Spin Coupling Constants Involving 31P and 29Si: The Advanced PEC Method" Molecules 27, no. 19: 6145. https://doi.org/10.3390/molecules27196145
APA StyleRusakov, Y. Y., & Rusakova, I. L. (2022). New pecJ-n (n = 1, 2) Basis Sets for High-Quality Calculations of Indirect Nuclear Spin–Spin Coupling Constants Involving 31P and 29Si: The Advanced PEC Method. Molecules, 27(19), 6145. https://doi.org/10.3390/molecules27196145








