A Review on the Synthesis of Fluorescent Five- and Six-Membered Ring Azaheterocycles
Abstract
:1. Introduction
2. Synthesis and Fluorescent Properties of Five- and Six-Membered Ring Azaheterocycles
2.1. Five Membered Ring Azaheterocycles
2.2. Six Membered Ring Azaheterocycles
2.3. Fused Azaheterocycles
2.4. Our Recent Contribution to the Field
3. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, Z.; Askim, J.R.; Suslick, K.S. The Optoelectronic Nose: Colorimetric and Fluorometric Sensor Arrays. Chem. Rev. 2019, 119, 231–292. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Qu, X. Cancer Biomarker Detection: Recent Achievements and Challenges. Chem. Soc. Rev. 2015, 44, 2963–2997. [Google Scholar] [CrossRef] [PubMed]
- Queralto, N.; Berliner, A.N.; Goldsmith, B.; Martino, R.; Rhodes, P.; Lim, S.H. Detecting Cancer by Breath Volatile Organic Compound Analysis: A Review of Array-Based Sensors. J. Breath Res. 2014, 8, 027112. [Google Scholar] [CrossRef] [PubMed]
- Gąsiorski, P.; Matusiewicz, M.; Gondek, E.; Pokladko-Kowar, M.; Armatys, P.; Wojtasik, K.; Danel, A.; Uchacz, T.; Kityk, A.V. Efficient green electroluminescence from 1,3-diphenyl-1H-pyrazolo[3,4-b]quinoxaline dyes in dye-doped polymer based electroluminescent devices. Dyes Pigments 2018, 151, 380–384. [Google Scholar] [CrossRef]
- Karnik, A.V.; Malviya, N.J.; Kulkarni, A.M.; Jadhav, B.L. Synthesis and in vitro antibacterial activity of novel heterocyclic derivatives of 18-nor-equilenin. Eur. J. Med. Chem. 2006, 41, 891–895. [Google Scholar] [CrossRef] [PubMed]
- Burbiel, J.; Bracher, F. Azasteroids as antifungals. Steroids 2003, 68, 587–594. [Google Scholar] [CrossRef]
- Sandeep, C.; Basavaraj, P.; Venugopala, K.N.; Rashmi, S.K.; Rashmi, V.; Odhav, B. Efficient synthesis and characterization of ethyl 7-acetyl-2-substituted 3-(substitutedbenzoyl) indolizine-1-carboxylates for in vitro anticancer activity. Asian J. Chem. 2016, 28, 1043–1048. [Google Scholar] [CrossRef]
- Chang, S.M.; Jain, V.; Chen, T.L.; Patel, A.S.; Pidugu, H.B.; Lin, Y.W.; Wu, M.H.; Huang, J.R.; Wu, H.C.; Shah, A.; et al. Design and Synthesis of 1,2-Bis(hydroxymethyl)pyrrolo[2,1-a]phthalazine Hybrids as Potent Anticancer Agents that Inhibit Angiogenesis and Induce DNA Interstrand Cross-links. J. Med. Chem. 2019, 62, 2404–2418. [Google Scholar] [CrossRef]
- Gundersen, L.L.; Charnock, C.; Negussie, A.H.; Rise, F.; Teklu, S. Synthesis of indolizine derivatives with selective antibacterial activity against Mycobacterium tuberculosis. Eur. J. Pharm. Sci. 2007, 30, 26–35. [Google Scholar] [CrossRef]
- Moraski, G.C.; Markley, L.D.; Chang, M.; Cho, S.; Franzblau, S.G.; Hwang, C.H.; Boshoff, H.; Millera, M.J. Generation and exploration of new classes of antitubercular agents: The optimization of oxazolines, oxazoles, thiazolines, thiazoles to imidazo[1,2-a]pyridines and isomeric 5,6-fused scaffolds. Bioorg. Med. Chem. 2012, 20, 2214–2220. [Google Scholar] [CrossRef] [Green Version]
- Narajji, C.; Karvekar, M.D.; Das, A.K. Synthesis and antioxidant activity of 3,3′-diselanediylbis (N,N-disubstituted indolizine-1-carboxamide) and derivatives. S. Afr. J. Chem. 2008, 61, 53–55. [Google Scholar]
- Huang, W.; Zuo, T.; Luo, X.; Jin, H.; Liu, Z.; Yang, Z.; Yu, X.; Zhang, L.; Zhang, L. Indolizine derivatives as HIV–1 VIF–ElonginC interaction inhibitors. Chem. Biol. Drug. Des. 2013, 81, 730–741. [Google Scholar] [CrossRef]
- Fu, Y.; Finney, N.S. Small-molecule fluorescent probes and their design. RSC Adv. 2018, 8, 29051–29061. [Google Scholar] [CrossRef]
- Bagnich, S.A.; Gulyakevich, O.V.; Mikhalchuk, A.L. Spectral-Luminescent Properties of 12-Oximino Derivatives of 8-AZA-D-Homogona-12,17a-Diones and their Concentration Dependence. J. Fluoresc. 2008, 18, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Borisevich, N.A.; Raichyonok, T.F.; Sukhodola, A.A.; Tolstorozhev, G.B. Delayed Fluorescence and Phosphorescence of 8-Aza-d-Homogonane in the Gas and Condensed Phases. J. Fluoresc. 2006, 16, 649–653. [Google Scholar] [CrossRef] [PubMed]
- Akhrem, A.A.; Borisevich, N.A.; Gulyakevich, O.V.; Mikhalchuk, A.L.; Raichyonok, T.F.; Tikhomirov, S.A.; Tolstorozhev, G.B. Specific fluorescence properties and picosecond transient absorption of 8-azasteroids. J. Fluoresc. 1999, 9, 357–361. [Google Scholar] [CrossRef]
- Al Matarneh, C.M.; Shova, S.; Mangalagiu, I.I.; Danac, R. Synthesis, structure, antimycobacterial and anticancer evaluation of new pyrrolo-(phenanthroline) derivatives. J. Enzym. Inhib. Med. Chem. 2016, 31, 470–480. [Google Scholar] [CrossRef]
- Thanikachalam, P.V.; Maurya, R.K.; Garg, V.; Monga, V. An insight into the medicinal perspective of synthetic analogs of indole: A review. Eur. J. Med. Chem. 2019, 180, 562–612. [Google Scholar] [CrossRef]
- Danac, R.; Al Matarneh, C.M.; Shova, S.; Daniloaia, T.; Balan, M.; Mangalagiu, I.I. New indolizines with phenanthroline skeleton: Synthesis, structure, antimycobacterial and anticancer evaluation. Bioorg. Med. Chem. 2015, 23, 2318–2327. [Google Scholar] [CrossRef]
- Naim, M.J.; Alam, O.; Nawaz, F.; Alam, M.J.; Alam, P. Current status of pyrazole and its biological activities. J. Pharm. Bioallied Sci. 2016, 8, 2–17. [Google Scholar] [CrossRef]
- Ali, I.; Lone, M.N.; Aboul-Enein, H.Y. Imidazoles as potential anticancer agents. MedChemComm 2017, 8, 1742–1773. [Google Scholar] [CrossRef] [PubMed]
- Mantu, D.; Antoci, V.; Moldoveanu, C.; Zbancioc, G.; Mangalagiu, I.I. Hybrid imidazole (benzimidazole)/pyridine (quinoline) derivatives and evaluation of theiranticancer and antimycobacterialactivity. J. Enzym. Inhib. Med. Chem. 2016, 31, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Bansal, Y.; Silakari, O. The therapeutic journey of benzimidazoles: A review. Bioorg. Med. Chem. 2012, 20, 6208–6236. [Google Scholar] [CrossRef]
- Bozorov, K.; Zhao, J.; Aisa, H.A. 1,2,3-Triazole-containing hybrids as leads in medicinal chemistry: A recent overview. Bioorg. Med. Chem. 2019, 27, 3511–3531. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, N.; Kumar, N.; Kumar, A.; Singh, U.K. Tetrazoles: Synthesis and biological activity. Immunol. Endocr. Metab. Agents Med. Chem. 2018, 18, 3–21. [Google Scholar] [CrossRef]
- Martínez-Lara, F.; Suárez, A.; Suárez-Pantiga, S.; Tapia, M.J.; Sanz, R. Straight access to highly fluorescent angular indolocarbazoles via merging Au- and Mo-catalysis. Org. Chem. Front. J. 2020, 7, 1869–1877. [Google Scholar] [CrossRef]
- Moseev, T.D.; Varaksin, M.V.; Gorlov, D.A.; Charushin, V.N.; Chupakhin, O.N. Transition-Metal-Free C–H/C–Li Coupling of Nonaromatic 2H-Imidazole 1-Oxides with Pentafluorophenyl Lithium in the Design of Novel Fluorophores with Intramolecular Charge Transfer Effect. J. Org. Chem. 2020, 85, 11124–11133. [Google Scholar] [CrossRef]
- Pu, S.; Zhang, C.; Fan, C.; Liu, G. Multi-controllable properties of an antipyrine-based diarylethene and its high selectivity for recognition of Al3+. Dyes Pigments 2016, 129, 24–33. [Google Scholar] [CrossRef]
- Gonçalves, T.; Sameiro, M. Fluorescent labeling of biomolecules with organic probes. Chem. Rev. 2009, 109, 190–212. [Google Scholar] [CrossRef]
- Grammel, M.; Hang, H.C. Chemical reporters for biological discovery. Nat. Chem. Biol. 2013, 9, 475–484. [Google Scholar] [CrossRef]
- Lang, K.; Chin, J.W. Cellular incorporation of unnatural amino acids and bioorthogonal labeling of proteins. Chem. Rev. 2014, 114, 4764–4806. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, S.; Zhang, H.; Xu, H. Design and application of receptor-targeted fluorescent probes based on small molecular fluorescent Dyes. Bioconj. Chem. 2021, 32, 4–24. [Google Scholar] [CrossRef] [PubMed]
- Algar, W.R.; Massey, M.; Rees, K.; Higgins, R.; Krause, K.D.; Darwish, G.H.; Peveler, W.J.; Xiao, Z.; Tsai, H.-Y.; Gupta, R.; et al. Photoluminescent nanoparticles for chemical and biological analysis and imaging. Chem. Rev. 2021, 121, 9243–9358. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Li, Y.; Chang, W.; Pang, S.; Li, X.; Duan, L.; Zhang, Z. Synthesis and Fluorescent Properties of Aminopyridines and the Application in “Click and Probing”. Molecules 2022, 27, 1596. [Google Scholar] [CrossRef] [PubMed]
- Piloto, A.M.; Hungerford, G.; Costa, S.P.G.; Gonçalves, M.S.T. Acridinyl methyl esters as photoactive precursors in the release of neurotransmitteramino acids. Photochem. Photobiol. Sci. 2013, 12, 339–347. [Google Scholar] [CrossRef]
- Tang, R.; Wang, X.; Zhang, W.; Zhuang, X.; Bi, S.; Zhanga, W.; Zhang, F. Aromatic azaheterocycle-cored luminogens with tunable physical properties via nitrogen atoms for sensing strong acids. J. Mater. Chem. C 2016, 4, 7640–7648. [Google Scholar] [CrossRef]
- Menzel, R.; Kupfer, S.; Mede, R.; Weiß, D.; Görls, H.; González, L.; Beckert, R. Arylamine-Modified Thiazoles as Donor–Acceptor Dyes: Quantum Chemical Evaluation of the Charge-Transfer Process and Testing as Ligands in Ruthenium(II) Complexes. Eur. J. Org. Chem. 2012, 27, 5231–5247. [Google Scholar] [CrossRef]
- Rajbongshi, B.K.; Bhattacharyya, H.P.; Choitanya; Pegu, D.; Sharma, S.; Baruah, P.K.; Sarma, M. Ultra-High Stokes Shift in Polycyclic Chromeno[2,3-b]Indoles. Polycycl. Aromat. Compd. 2022, 42, 1710–1727. [Google Scholar] [CrossRef]
- Li, Q.; Gong, J.; Li, Y.; Zhang, R.; Wang, H.; Zhang, J.; Yan, H.; Lam, J.W.Y.; Sung, H.H.Y.; Williams, I.D.; et al. Unusual light-driven amplification through unexpected regioselective photogeneration of five-membered azaheterocyclic AIEgen. Chem. Sci. 2021, 12, 709–717. [Google Scholar] [CrossRef]
- Zbancioc, A.M.; Miron, A.; Tuchilus, C.; Rotinberg, P.; Mihai, C.T.; Mangalagiu, I.; Zbancioc, G. Synthesis and in vitro analysis of novel dihydroxyacetophenone derivatives with antimicrobial and antitumor activities. Med. Chem. 2014, 10, 476–483. [Google Scholar] [CrossRef]
- Antoci, V.; Oniciuc, L.; Amariucai-Mantu, D.; Moldoveanu, C.; Mangalagiu, V.; Amarandei, A.M.; Lungu, C.N.; Dunca, S.; Mangalagiu, I.I.; Zbancioc, G. Benzoquinoline Derivatives: A Straightforward and Efficient Route to Antibacterial and Antifungal Agents. Pharmaceuticals 2021, 14, 335. [Google Scholar] [CrossRef] [PubMed]
- Popovici, L.; Amarandi, R.M.; Mangalagiu, I.I.; Mangalagiu, V.; Danac, R. Synthesis, molecular modelling and anticancer evaluation of new pyrrolo[1,2-b]pyridazine and pyrrolo[2,1-a]phthalazinederivatives. J. Enzym. Inhib. Med. Chem. 2019, 34, 230–243. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Li, Y.; Min, T.; Gong, J.; Du, L.; Phillips, D.L.; Liu, J.; Lam, J.W.Y.; Sung, H.H.Y.; Williams, I.D.; et al. Time-Dependent Photodynamic Therapy for Multiple Targets: A Highly Efficient AIE-Active Photosensitizer for Selective Bacterial Elimination and Cancer Cell Ablation. Angew. Chem. Int. Ed. 2020, 59, 9470–9477. [Google Scholar] [CrossRef]
- Han, Y.R.; Shim, S.-H.; Kim, D.-S.; Jun, C.-H. Synthesis of Benzoquinolizinium Salts by Rh(III)-Catalyzed Cascade Double N-Annulation Reactions of Allylamines, Diarylacetylenes, and HBF4. Org. Lett. 2017, 19, 2941–2944. [Google Scholar] [CrossRef] [PubMed]
- Wojtasik, K.; Danel, A.; Wojtasik, M.; Lukasiewicz, M. Synthesis of 1H-Pyrazolo[3,4-b]quinoxaline Derivatives by Modification of the Regiospecific Reaction–the Influence of the Microwave Field. ChemistrySelect 2021, 6, 4330–4335. [Google Scholar] [CrossRef]
- Goya, T.; Zimmermann Crocomo, P.; Hosono, T.; Minakata, S.; de Sousa, L.E.; de Silva, P.; Data, P.; Takeda, Y. A New Entry to Purely Organic Thermally Activated Delayed Fluorescence Emitters Based on Pyrido[2,3-b]pyrazine-Dihydrophenazasilines Donor-Acceptor Dyad. Asian J. Org. Chem. 2022, 11, e202100780. [Google Scholar] [CrossRef]
- Yin, L.; Liebscher, J. Palladium-Catalyzed Cross-Coupling Reactions of 7-Bromo-2,3-diphenylpyrido[2,3-b]pyrazine. Synthesis 2005, 8, 1345–1349. [Google Scholar] [CrossRef]
- Kaur, G.; Singh, A.; Kaur, N.; Banerjee, B. A general method for the synthesis of structurally diverse quinoxalines and pyrido-pyrazine derivatives using camphor sulfonic acid as an efficient organo-catalyst at room temperature. Synth. Commun. 2021, 51, 1121–1131. [Google Scholar] [CrossRef]
- Tatu, L.; Georgescu, E.; Boscornea, C.; Ungureanu, E.M. Synthesis and fluorescence of 1-[7-(4-nitrobenzoyl)-3-(biphenyl-4-yl)pyrrolo[1,2-c]pyrimidin-5-yl]ethanone. UPB Bull. Sci. 2015, 77, 49–58. [Google Scholar]
- Tatu, M.L.; Georgescu, E.; Boscornea, C.; Popa, M.M. Ungureanu, E.M. Synthesis and fluorescence of new 3-biphenylpyrrolo[1,2-c]pyrimidines. Arab. J. Chem. 2017, 10, 643–652. [Google Scholar] [CrossRef]
- Ungureanu, E.M.; Tatu, M.L.; Georgescu, E.; Boscornea, C.; Popa, M.M.; Stanciu, G. Influence of the chemical structure and solvent polarity on the fluorescence of 3-aryl-7-benzoyl-pyrrolo [1,2-c]pyrimidines. Dyes Pigments 2020, 174, 108023. [Google Scholar] [CrossRef]
- Tomashenko, O.A.; Khlebnikov, A.F.; Mosiagin, I.P.; Novikov, M.S.; Grachova, E.V.; Shakirova, J.R.; Tunik, S.P. A new heterocyclic skeleton with highly tunable absorption/emission wavelength via H-bonding. RSC Adv. 2015, 5, 94551–94561. [Google Scholar] [CrossRef]
- Zugravescu, I.; Petrovanu, M. N-Ylid Chemistry; McGraw Hill Higher Education: New York, NY, USA, 1977; ISBN-13: 9780070730809. [Google Scholar]
- Zbancioc, A.M.; Zbancioc, G.; Tanase, C.; Miron, A.; Ursu, C.; Mangalagiu, I.I. Design, synthesis and in vitro anticancer activity of a new class of dual DNA intercalators. Lett. Drug Des. Discov. 2010, 7, 644–649. [Google Scholar] [CrossRef]
- Al Matarneh, C.M.; Amarandi, R.M.; Craciun, A.M.; Mangalagiu, I.I.; Zbancioc, G.; Danac, R. Design, Synthesis, Molecular Modelling and Anticancer Activities of New Fused Phenanthrolines. Molecules 2020, 25, 527. [Google Scholar] [CrossRef] [PubMed]
- Antoci, V.; Mantu, D.; Cozma, D.G.; Usru, C.; Mangalagiu, I.I. Hybrid anticancer 1,2-diazine derivatives with multiple mechanism of action. Part 3. Med. Hypotheses 2014, 82, 11–15. [Google Scholar] [CrossRef]
- Lungu, C.N.; Bratanovici, B.I.; Grigore, M.M.; Antoci, V.; Mangalagiu, I.I. Hybrid Imidazole-Pyridine Derivatives: An Approach to Novel Anticancer DNA Intercalators. Curr. Med. Chem. 2020, 27, 154–169. [Google Scholar] [CrossRef]
- Al Matarneh, C.; Ciobanu, C.I.; Mangalagiu, V.; Zbancioc, G.; Danac, R. Microwave assisted synthesis of six member ring azaheterocycles and their antimycobacterial and anticancer evaluation. Rev. Chim. 2020, 71, 287–293. [Google Scholar] [CrossRef]
- Mantu, D.; Antoci, V.; Mangalagiu, I.I. Design, synthesis and antituberculosis activity of some new pyridazine derivatives: Bis-pyridazine. Part IV. Infect. Disord.-Drug Targets 2013, 13, 344–351. [Google Scholar] [CrossRef]
- Antoci, V.; Cucu, D.; Zbancioc, G.; Moldoveanu, C.; Mangalagiu, V.; Amariucai-Mantu, D.; Aricu, A.; Mangalagiu, I.I. Bis-(imidazole/benzimidazole)-pyridine derivatives: Synthesis, structure and antimycobacterial activity. Part XII. Future Med. Chem. 2020, 12, 207–222. [Google Scholar] [CrossRef]
- Mantu, D.; Luca, M.C.; Moldoveanu, C.; Zbancioc, G.; Mangalagiu, I.I. Synthesis and antituberculosis activity of some new pyridazine derivatives. Part II. Eur. J. Med. Chem. 2010, 45, 5164–5168. [Google Scholar] [CrossRef]
- Mangalagiu, V.; Oniciuc, L.; Cucu, D.; Antoci, V.; Mangalagiu, I.I.; Cojean, S.; Loiseau, P. Azine, New Molecules with Azaheterocycles Skeleton of Potential Interest in Leishmaniasis. Org. Chem. Curr. Res. 2018, 7, 114. [Google Scholar] [CrossRef]
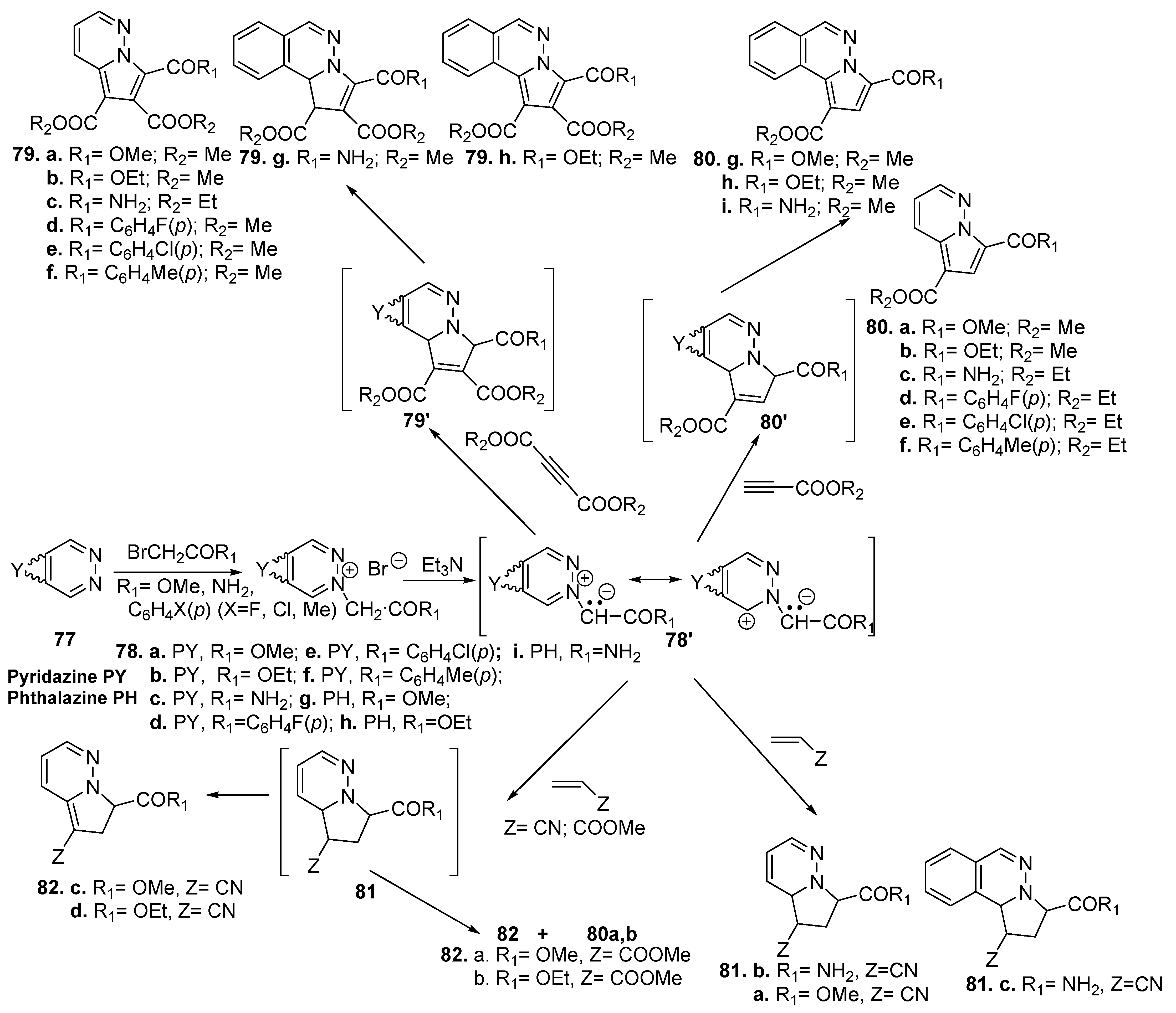
- Zbancioc, G.; Mangalagiu, I.I. Microwave-Assisted Synthesis of Highly Fluorescent Pyrrolopyridazine Derivatives. Synlett 2006, 5, 0804–0806. [Google Scholar] [CrossRef]
- Zbancioc, G.; Huhn, T.; Groth, U.; Deleanu, C.; Mangalagiu, I.I. Pyrrolodiazine derivatives as blue organic luminophores: Synthesis and properties. Part 3. Tetrahedron 2010, 66, 4298–4306. [Google Scholar] [CrossRef]
- Maftei, D.; Zbancioc, G.; Humelnicu, I.; Mangalagiu, I. Conformational effects on the lowest excited states of benzoyl-pyrrolopyridazine: Insights from PCM time-dependent DFT. J. Phys. Chem. A 2013, 117, 3165–3175. [Google Scholar] [CrossRef] [PubMed]
- Zbancioc, G.; Mangalagiu, I.I. Pyrrolopyridazine derivatives as blue organic luminophores: Synthesis and properties. Part 2. Tetrahedron 2010, 66, 278–282. [Google Scholar] [CrossRef]
- Moldoveanu, C.; Amariucai-Mantu, D.; Mangalagiu, V.; Antoci, V.; Maftei, D.; Mangalagiu, I.I.; Zbancioc, G. Microwave Assisted Reactions of Fluorescent Pyrrolodiazine Building Blocks. Molecules 2019, 24, 3760. [Google Scholar] [CrossRef]
- Moldoveanu, C.; Mangalagiu, I.; Zbancioc, G. Fluorescent Azasteroids through Ultrasound Assisted Cycloaddition Reactions. Molecules 2021, 26, 5098. [Google Scholar] [CrossRef]
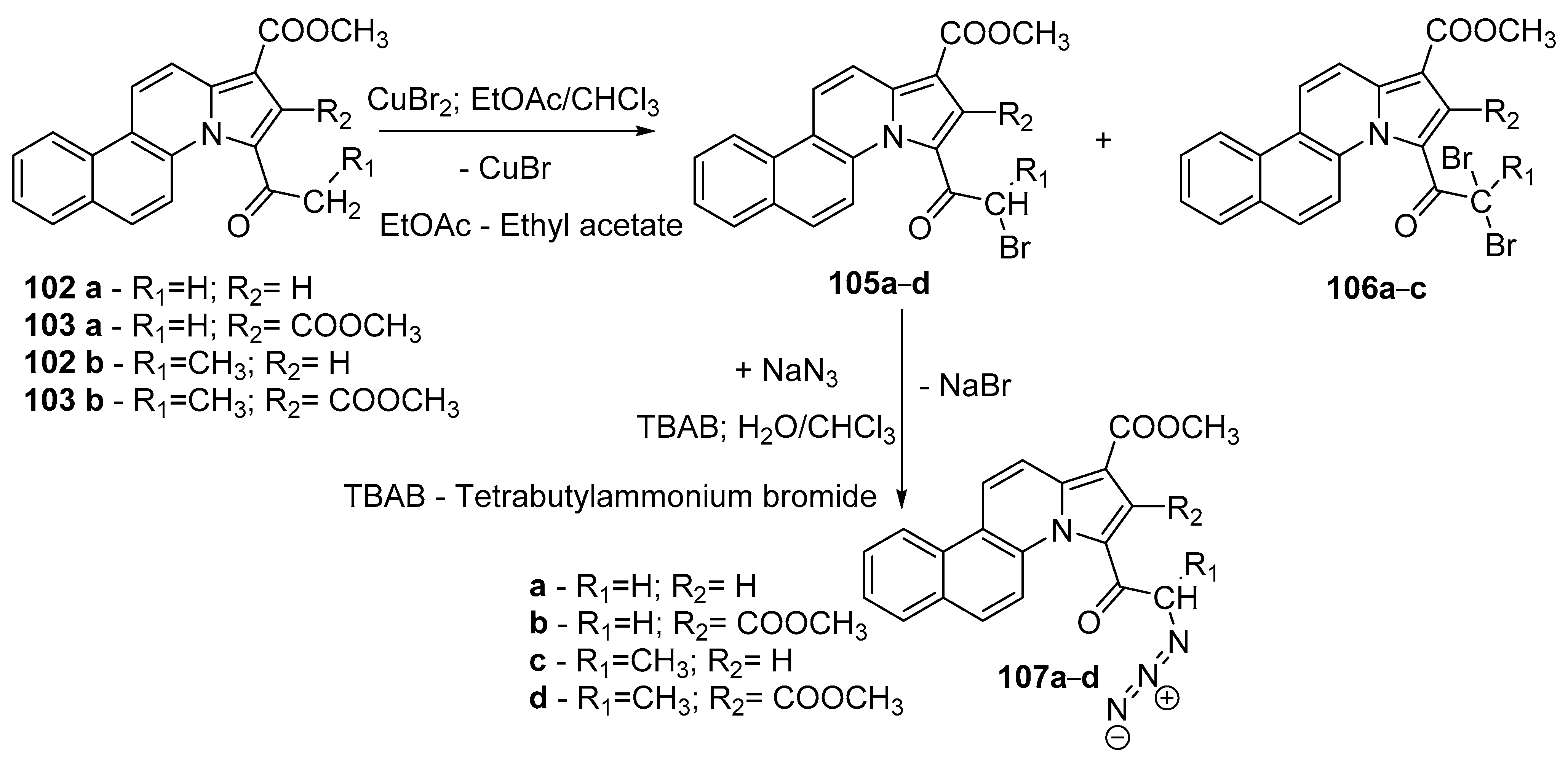
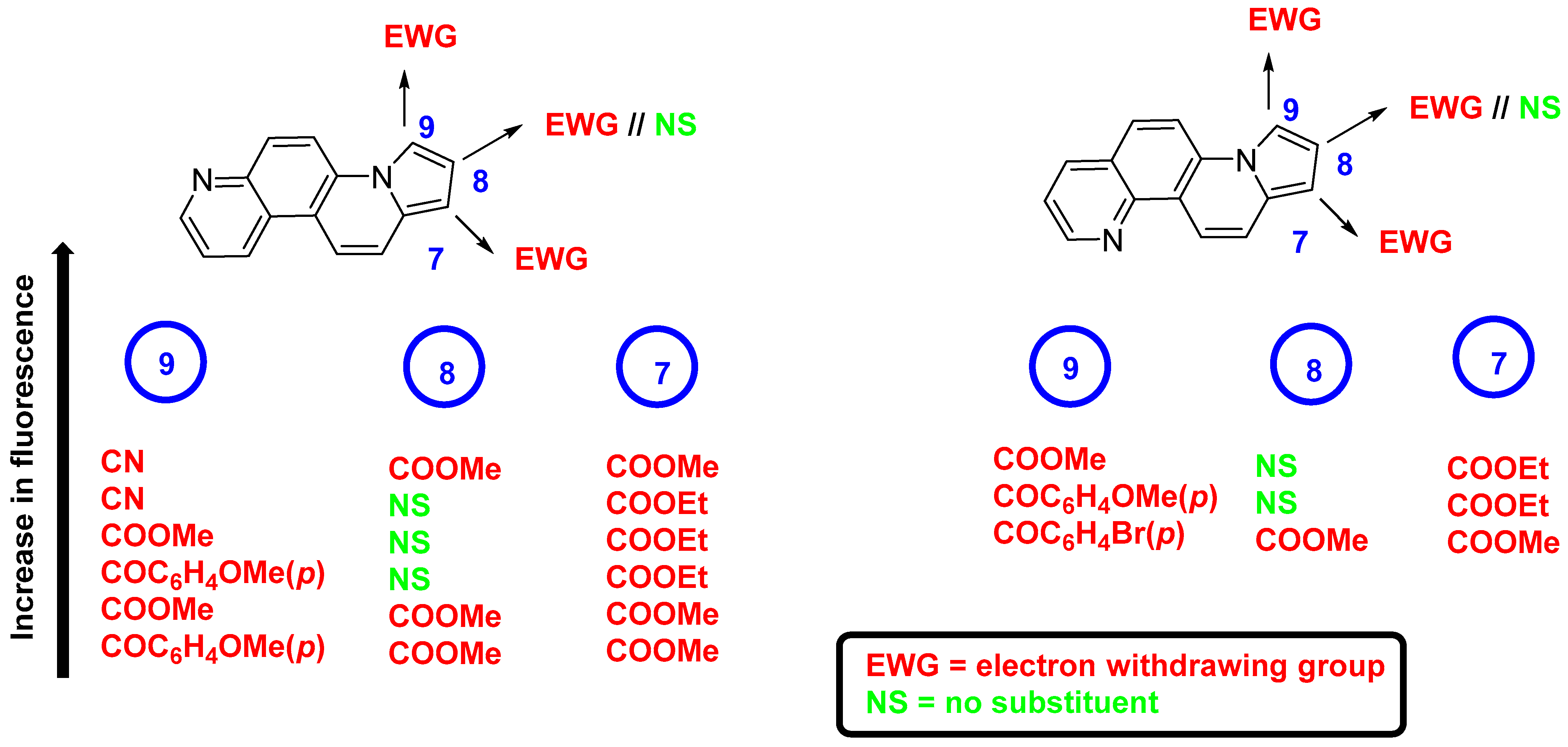
- Zbancioc, G.; Ciobanu, C.-I.; Mangalagiu, I.I.; Moldoveanu, C. Ultrasound-Assisted Synthesis of Fluorescent Azatetracyclic Derivatives: An Energy-Efficient Approach. Molecules 2022, 27, 3180. [Google Scholar] [CrossRef]
- Gherasim, C.; Airinei, A.; Tigoianu, R.; Craciun, A.M.; Danac, R.; Nicolescu, A.; Isac, D.; Mangalagiu, I.I. Synthesis and photophysical insights of new fused N-heterocyclic derivatives with isoquinoline skeleton. J. Mol. Liq. 2020, 310, 113196. [Google Scholar] [CrossRef]
- Airinei, A.; Tigoianu, R.; Danac, R.; Al Matarneh, C.M.; Isac, D.L. Steady state and time resolved fluorescence studies of new indolizine with phenanthroline skeleton derivatives. J. Lumin. 2018, 199, 6–12. [Google Scholar] [CrossRef]
- Al Matarneh, C.; Rosca, I.; Shova, S.; Danac, R. Synthesis and properties of new fused pyrrolo-1,10-phenanthroline type derivatives. J. Serb. Chem. Soc. 2021, 86, 901–915. [Google Scholar] [CrossRef]
- Doroftei, C.; Carlescu, A.; Leontie, L.; Danac, R.; Al-Matarneh, C.M. Structural, Electrical and Optical Properties of Pyrrolo[1,2-i][1,7] Phenanthroline-Based Organic Semiconductors. Materials 2022, 15, 1684. [Google Scholar] [CrossRef] [PubMed]
- Marangoci, N.L.; Popovici, L.; Ursu, E.L.; Danac, R.; Clima, L.; Cojocaru, C.; Coroaba, A.; Neamtu, A.; Mangalagiu, I.I.; Pinteala, M.; et al. Pyridyl-indolizine derivatives as DNA binders and pH-sensitive fluorescent dyes. Tetrahedron 2016, 72, 8215–8222. [Google Scholar] [CrossRef]
- Gradinaru, R.V.; Luca, A.M.; Cretescu, I.; Danac, R. Fluorescent Conjugates: pH Stability, Dye-DNA Interaction and Biological Activity. Rev. Chim. 2010, 61, 903–906. [Google Scholar]
- Pricope, G.; Ursu, E.L.; Sardaru, M.; Cojocaru, C.; Clima, L.; Marangoci, N.; Danac, R.; Mangalagiu, I.I.; Simionescu, B.C.; Pinteala, M.; et al. Novel cyclodextrin-based pH-sensitive supramolecular host–guest assembly for staining acidic cellular organelles. Polym. Chem. 2018, 9, 968–975. [Google Scholar] [CrossRef]
- Sardaru, M.-C.; Carp, O.; Ursu, E.-L.; Craciun, A.-M.; Cojocaru, C.; Silion, M.; Kovalska, V.; Mangalagiu, I.; Danac, R.; Rotaru, A. Cyclodextrin Encapsulated pH Sensitive Dyes as Fluorescent Cellular Probes: Self-Aggregation and In Vitro Assessments. Molecules 2020, 25, 4397. [Google Scholar] [CrossRef]
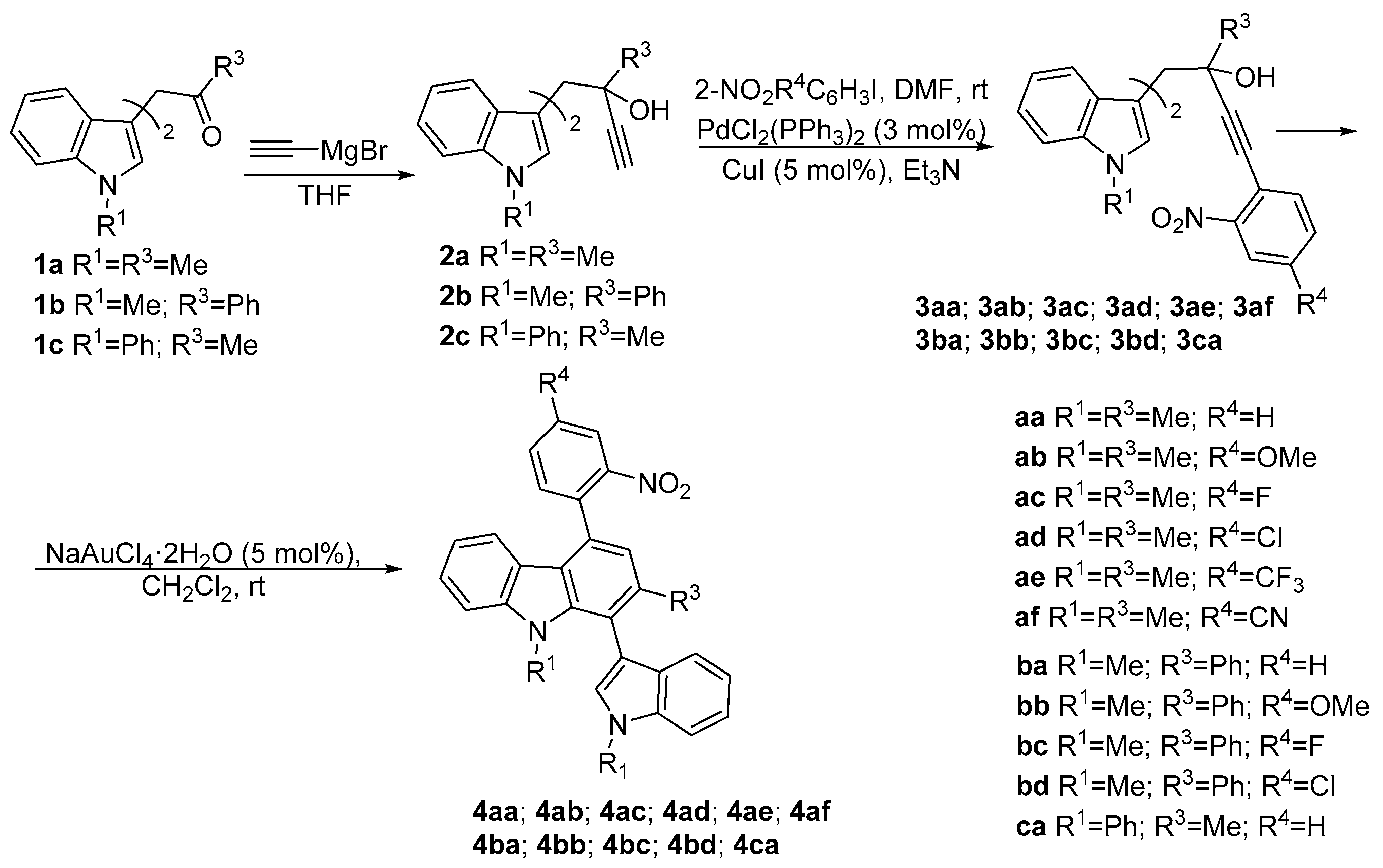

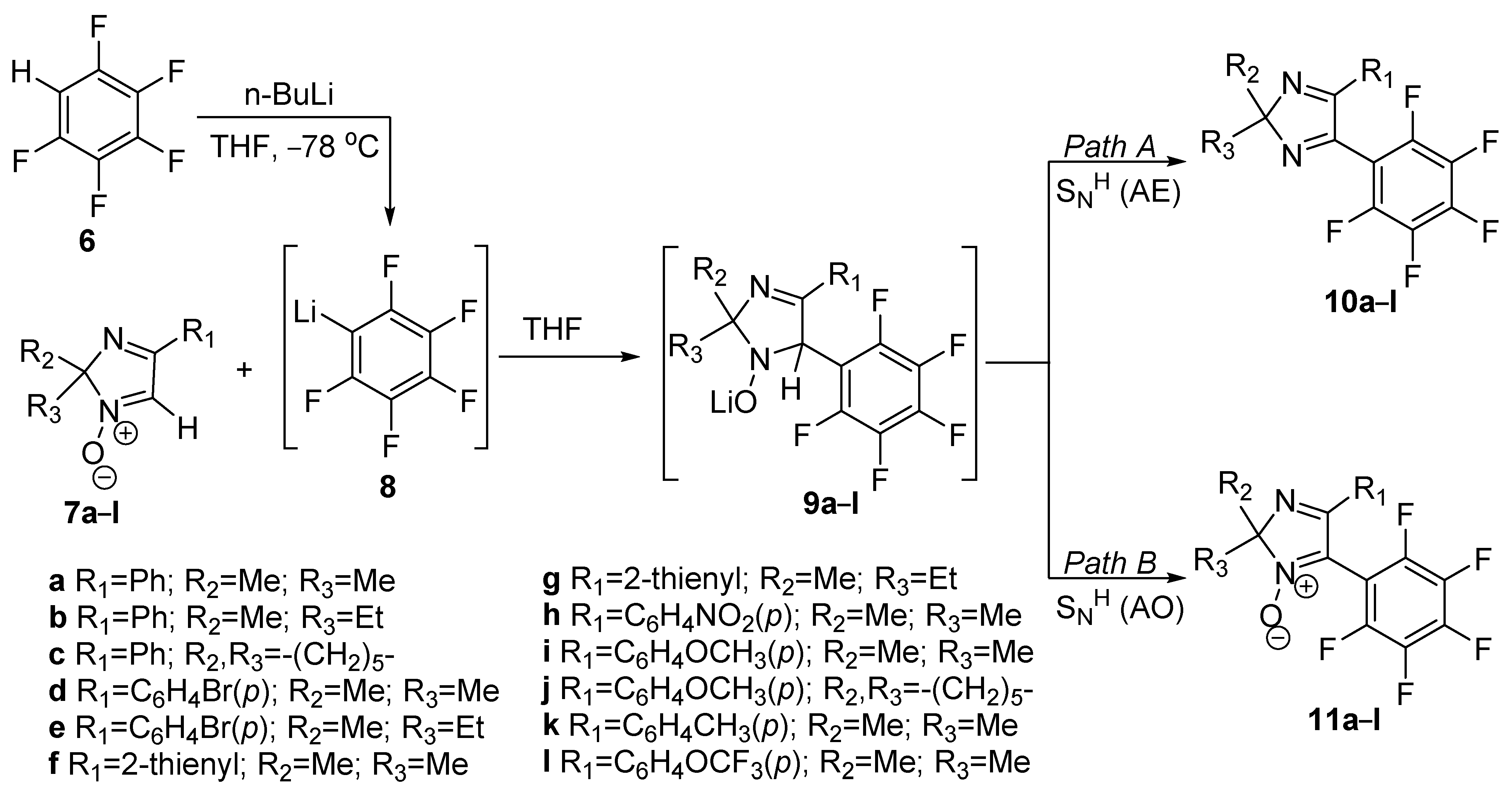
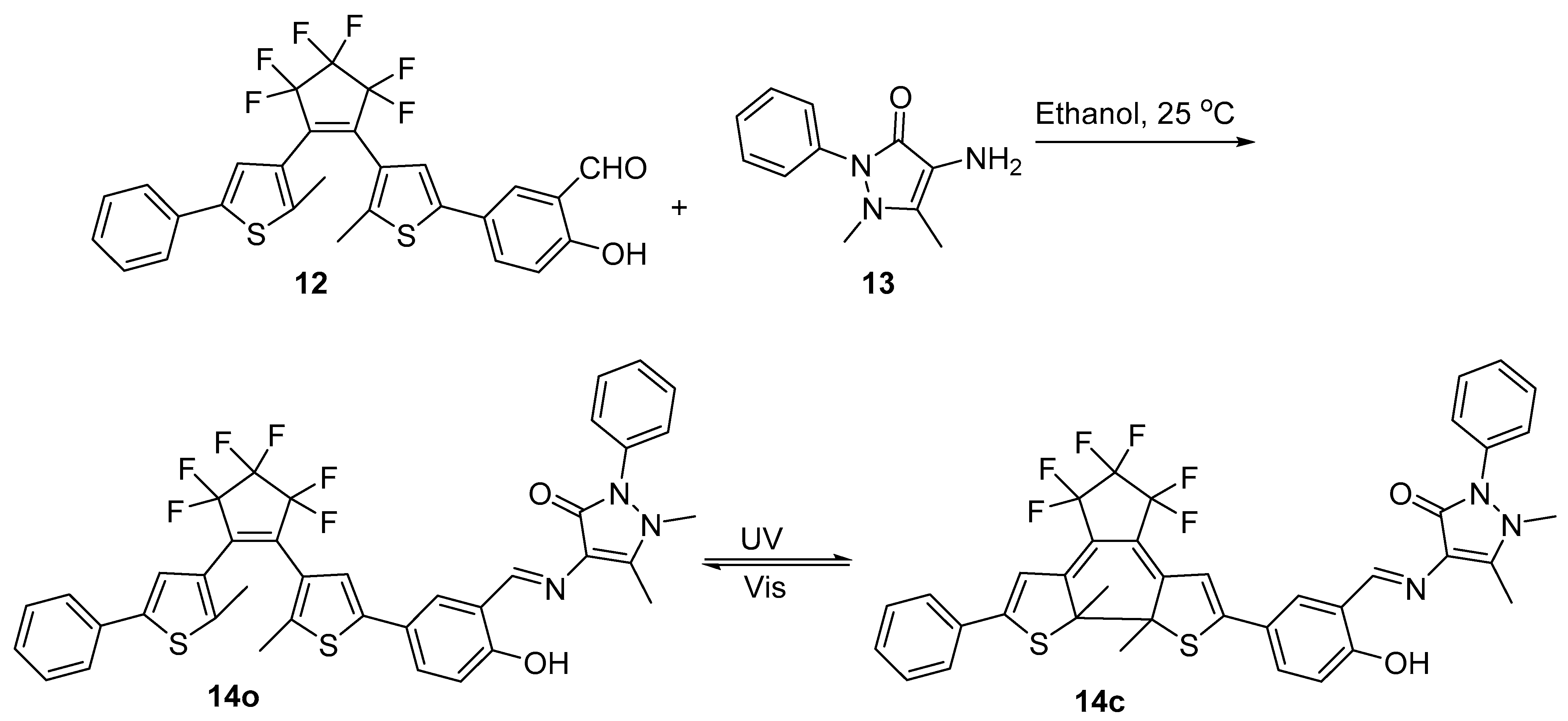





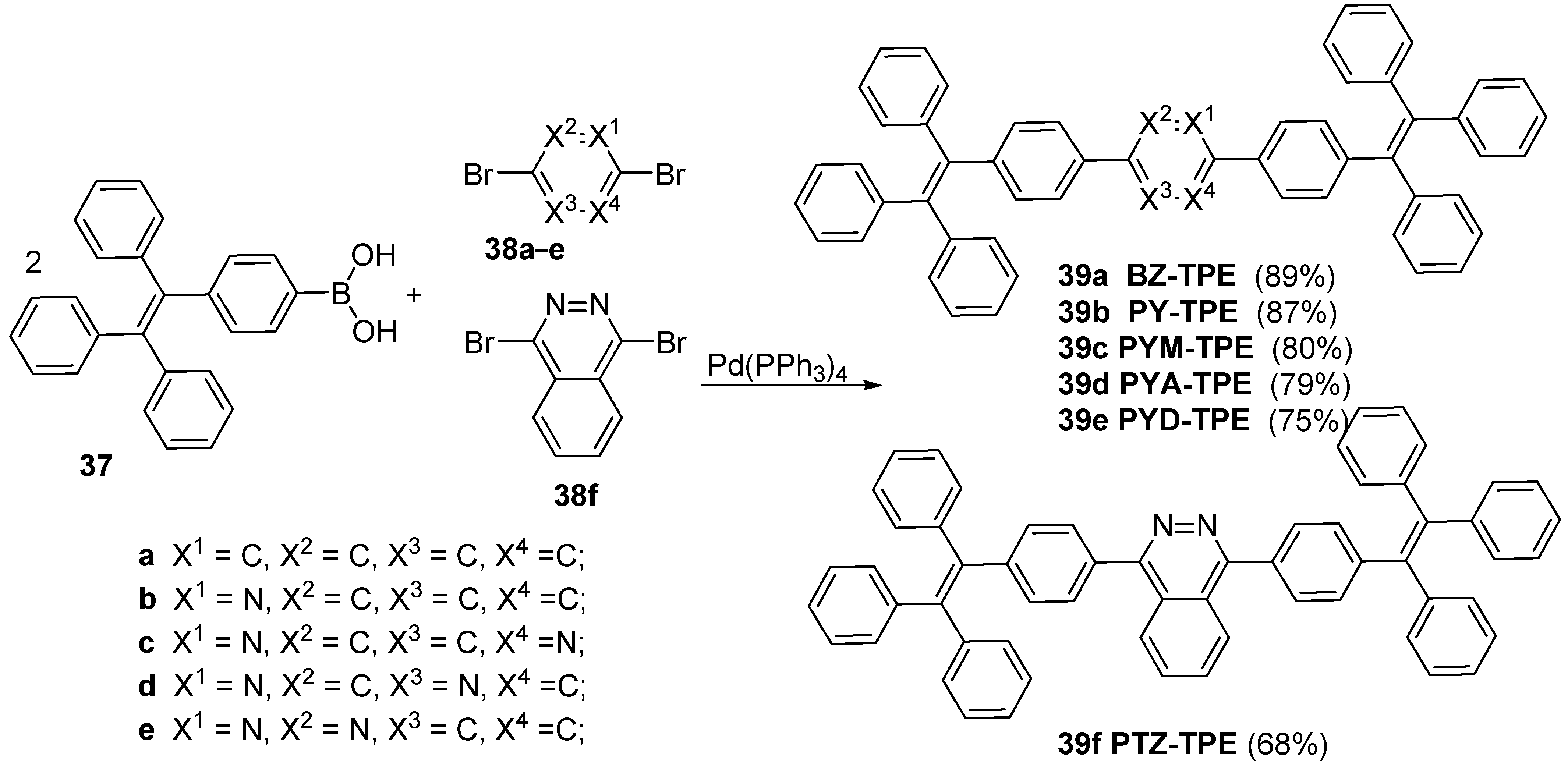
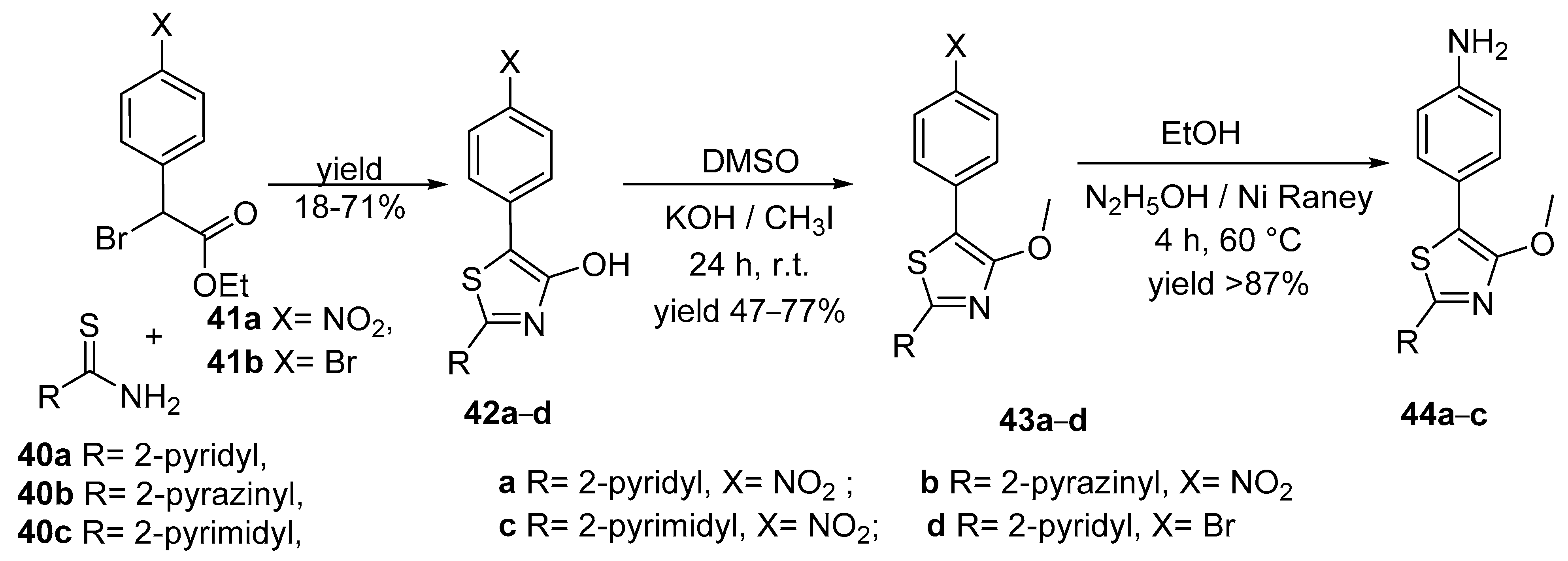


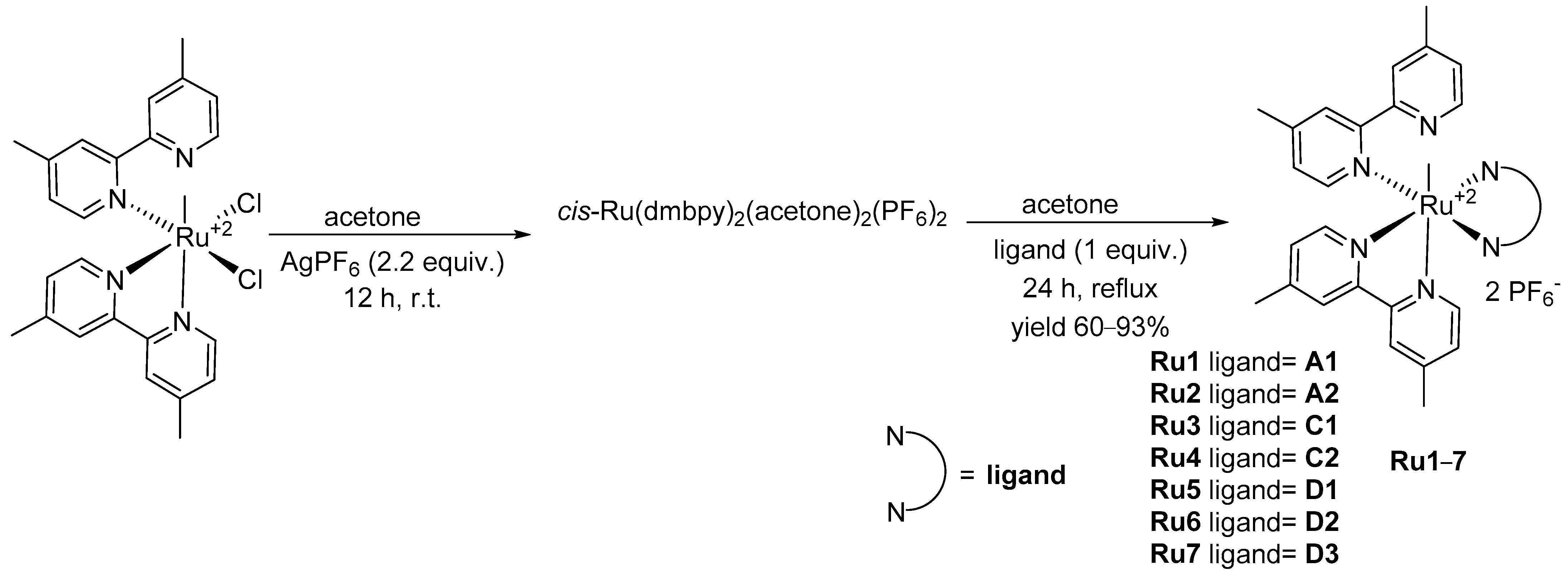

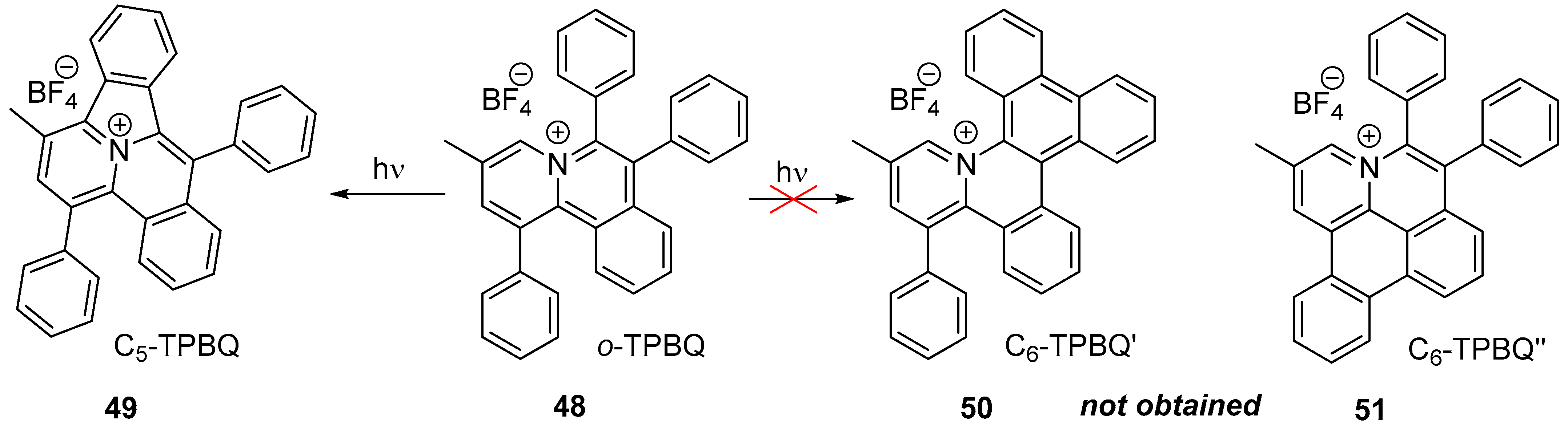



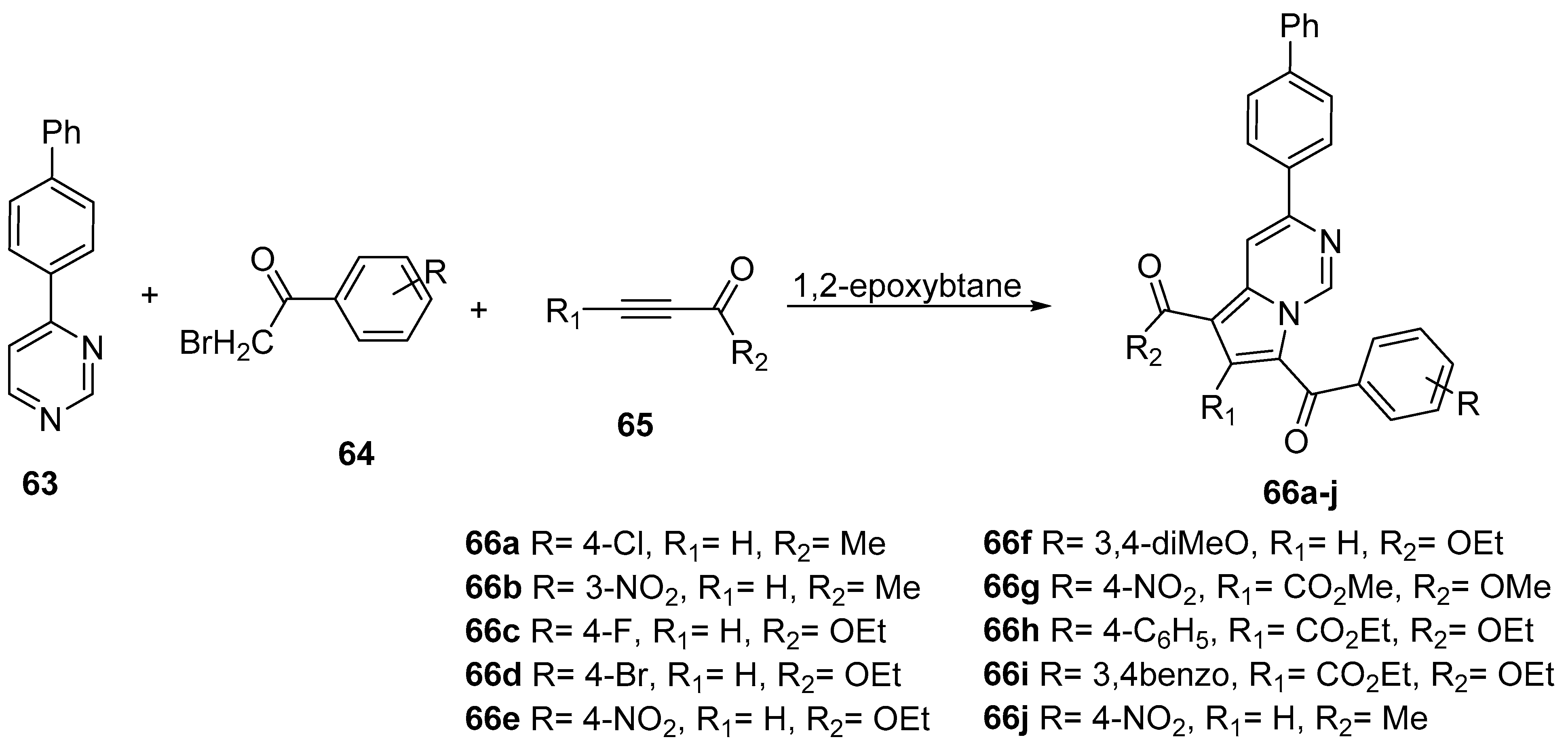













| Compound | λabs (nm) | εmax (L·mol−1·cm−1) | λem (nm) | Φfl (%) | τ (ns) | kf (107·s−1) | Knr (107·s−1) |
|---|---|---|---|---|---|---|---|
| 5aa | 338 | 20,700 ± 300 | 407 | 72 ± 4 | 7.4 ± 0.1 | 9.54 ± 0.10 | 3.89 ± 0.04 |
| 351 | 23,700 ± 300 | ||||||
| 377 | 6900 ± 200 | ||||||
| 397 | 8200 ± 200 | ||||||
| 5ba | 342 | 16,700 ± 200 | 424 | 69 ± 2 | 6.4 ± 0.1 | 10.77 ± 0.05 | 4.84 ± 0.02 |
| 354 | 20,000 ± 200 | ||||||
| 384 | 6500 ± 100 | ||||||
| 402 | 7500 ± 100 |
| Compound | λabs (nm) a | λem (nm) b | Φfl (%) c | Compound | λabs (nm) a | λem (nm) b | Φfl (%) c |
|---|---|---|---|---|---|---|---|
| 10a | 259 | 316 | <0.1 | 11a | 281 | 316 | <0.1 |
| 10b | 266 | 316 | <0.1 | 11b | 278 | 317 | <0.1 |
| 10c | 263 | 317 | <0.1 | 11c | 279 | 316 | <0.1 |
| 10d | 272 | 316 | <0.1 | 11d | 282 | 316 | <0.1 |
| 10e | 272 | 317 | <0.1 | 11e | 283 | 316 | <0.1 |
| 10f | 261, 302 | 333, 420 (br) | <0.1 | 11f | 297 | 322 | <0.1 |
| 10g | 304 | 333, 420 (br) | <0.1 | 11g | 297 | 321 | <0.1 |
| 10h | 272 | 316 | 0.6 | 11h | 272 | 317 | <0.1 |
| 10i | 305 | 331, 475 (br) | 0.1 | 11i | 298 | 320 | <0.1 |
| 10j | 305 | 335, 464 (br) | 0.2 | 11j | 297 | 325 | <0.1 |
| 10k | 277 | 317, 352 (br) | <0.1 | 11k | 288 | 317 | <0.1 |
| 10l | 256 | 316 | 1.9 | 11l | 277 | 317 | <0.1 |
| Compound | Yield [%] | λabs [nm] | λex [nm] | λem [nm] | Φfl [%] |
|---|---|---|---|---|---|
| 71 | 270 | 390 | 480 | 34 | |
| 18 | 24 | 270 | 390 | 480 | 31 |
| 19 | 65 | 270 | 390 | 480 | 44 |
| 20 | 52 | 270 | 390 | 485 | 31 |
| 21 | 60 | 270 | 390 | 485 | 27 |
| 22 | 52 | 270 | 390 | 485 | 32 |
| 23 | 53 | 270 | 390 | 485 | 22 |
| 24 | 17 | 258 | 345 | 455 | 2 |
| 25 | 67 | 270 | 390 | 480 | 35 |
| 26 | 57 | 270 | 390 | 480 | 30 |
| Compound | Yield [%] | EtOH | MeOH-HEPES (80:20) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| λabs [nm] | log ε | λem [nm] | Φfl [%] | Δλ [nm] | λabs [nm] | log ε | λem [nm] | Φfl [%] | Δλ [nm] | |||
| 36a | Boc-Gly-OAcm | 69 | 360 | 3.90 | 435 | 9 | 75 | 362 | 3.48 | 423 | 12 | 61 |
| 36b | Boc-Ala-OAcm | 93 | 361 | 3.93 | 411 | 11 | 50 | 360 | 3.69 | 412 | 15 | 52 |
| 36c | Boc-Glu-OAcm | 19 | 361 | 4.04 | 433 | 8 | 72 | 362 | 3.57 | 433 | 11 | 71 |
| 36d | Boc-β-Ala-OAcm | 72 | 361 | 3.91 | 412 | 11 | 51 | 361 | 3.79 | 417 | 14 | 56 |
| 36e | Boc-GABA-OAcm | 49 | 361 | 3.97 | 424 | 4 | 63 | 360 | 3.94 | 424 | 11 | 64 |
| Compound | UV-Vis Absorption | Fluorescence | |||||
|---|---|---|---|---|---|---|---|
| λabs [nm] | log ε | λem [nm] | Φfl [%] | τ [ns] | kf 107 [s−1] | knf 107 [s−1] | |
| 39a BZ-TPE | 332 | 4.43 | 480 | 53.7 | 3.5 | 15.3 | 13.2 |
| 39b PY-TPE | 340 | 4.57 | 481 | 42.6 | 3.3 | 12.1 | 16.3 |
| 39c PYM-TPE | 346 | 4.42 | 482 | 31.2 | 3.0 | 10.4 | 22.9 |
| 39d PYA-TPE | 363 | 4.51 | 484 | 14.6 | 2.0 | 7.3 | 42.7 |
| 39e PYD-TPE | 343 | 4.24 | 450 | 0.6 | 1.3 | 0.46 | 76.2 |
| 39f PTZ-TPE | 323 | 4.45 | 480 | 1.6 | 1.7 | 0.94 | 57.8 |
| Dye | λabs [nm] | log ε | λabs [nm] | log ε | λem [nm] | Φfl [%] | τ [ns] | |
|---|---|---|---|---|---|---|---|---|
| A1 | 301 | 4.33 | 405 | 4.44 | 547 | 47 | 3.83 | |
| A2 | 299 | 4.35 | 417 | 4.43 | 605 | <1 | 0.26 | |
| A3 | 307 | 4.43 | 433 | 4.36 | n.d. | n.d. | n.d. | |
| B1 | 303 | 4.40 | 425 | 4.47 | 593 | 40 | 2.99 | |
| B2 | 302 | 4.40 | 439 | 4.46 | 611 | <1 | 0.13 | |
| C1 | 302 | 4.32 | 426 | 4.36 | 601 | 8 | 1.26 | |
| C2 | 301 | 4.38 | 433 | 4.46 | 630 | <1 | <0.1 * | |
| D1 | 292 | 4.11 | 380 | 4.24 | 478 | 90 | 3.12 | |
| D2 | 257 | 4.72 | 377 | 4.42 ** | 484 sh, | 507 | 2 | 0.11 |
| D3 | 240 | 4.77 | 373 | 4.40 | 455 sh, | 470 | <1 | 0.13 |
| Solvent | A2 | D1 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| λabs [nm] | log ε | λem [nm] | Φfl [%] | Stokes Shift [cm−1] | τ [ns] | λabs [nm] | log ε | λem [nm] | Φfl [%] | Stokes Shift [cm−1] | τ [ns] | |
| Heptane | 427 | 4.585 | 483, 512 | 63 | 3900 | 2.6 | 387 | 4.466 | 456, 464 | 97 | 4300 | 2.4 |
| Dioxane | 425 | 4.441 | 530 | 59 | 4700 | 3.8 | 385 | 4.399 | 472 | 100 | 4800 | 2.7 |
| CHCl3 | 428 | 4.422 | 561 | 40 | 5500 | 4.0 | 387 | 4.235 | 475 | 95 | 4800 | 2.9 |
| THF | 425 | 4.485 | 552 | 43 | 5400 | 3.8 | 384 | 4.474 | 473 | 100 | 4900 | 2.8 |
| MeOH | 421 | 4.456 | 581 | <1 | 6500 | 0.1 | 382 | * | 489 | 81 | 5700 | 3.3 |
| CH3CN | 417 | 4.434 | 603 | 1 | 7400 | 0.1 | 380 | 4.244 | 475 | 90 | 5300 | 3.1 |
| Compound | Fluorescence Quantum Yield | |||
|---|---|---|---|---|
| Acetonitrile | Ethyl Acetate | |||
| 280 nm | 313 nm | 280 nm | 313 nm | |
| 47a | 3.7 | 3.5 | 3.9 | 3.7 |
| 47b | 3.6 | 3.7 | 4.3 | 3.5 |
| 47c | 3.9 | 3.6 | 3.7 | 3.4 |
| Compound | Solvent | c [mol dm3] | λabs [nm] | ε(λabs) [M−1 cm−1] | λem [nm] | Φfl [%] |
|---|---|---|---|---|---|---|
| 53e | MCHX | 2.94 × 10−5 | 396 | 5315 | 436 | 1.0 |
| THF | 4.68 × 10−5 | 399 | 3816 | 459 | 1.5 | |
| ACN | 4.89 × 10−5 | 398 | 3865 | 461 | 2.1 | |
| 53f | MCHX | 3.68 × 10−5 | 402 | 5324 | 442 | 0.7 |
| THF | 3.83 × 10−5 | 404 | 5175 | 467 | 3.4 | |
| ACN | 4.02 × 10−5 | 406 | 4852 | 479 | 67.2 | |
| 53g | MCHX | 3.17 × 10−5 | 399 | 4051 | 443 | 1.0 |
| THF | 3.43 × 10−5 | 397 | 4055 | 480 | 6.8 | |
| ACN | 3.44 × 10−5 | 397 | 4156 | 483 | 11.6 |
| Compound | Host | λem [nm] | Φfl [%] | τPF [ns] | τDF [μs] | DF/PF | EA [eV] | S1 [eV] | T1 [eV] | ΔEST [eV] |
|---|---|---|---|---|---|---|---|---|---|---|
| 56 | Zeonex® | 527 | 25 | 13.24 ± 1.18 | 9.45 ± 0.98 | 6.08 | 0.024 | 2.46 | 2.27 | 0.19 |
| CBP | 555 | 34 | 25.28 ± 1.06 | 1.06 ± 0.11 | 1.57 | 0.031 | 2.24 | 2.12 | 0.12 | |
| TCTA | 555 | 26 | 32.29 ± 0.57 | 2.10 ± 0.44 | 1.20 | 0.026 | 2.25 | 2.07 | 0.19 | |
| 57 | Zeonex® | 521 | 8 | 8.68 ± 0.09 | 0.7 ± 0.06 | 5.16 | 0.062 | 2.37 | 2.26 | 0.11 |
| CPB | 553 | 29 | 17.67 ± 0.18 | 2.7 ± 0.24 | 1.99 | 0.033 | 2.28 | 2.05 | 0.22 | |
| TCTA | 551 | 14 | 26.81 ± 0.83 | 3.92 ± 0.32 | 1.14 | 0.037 | 2.26 | 2.05 | 0.21 | |
| 58 | Zeonex® | 507 | 6 | 7.27 ± 0.32 | 9.90 ± 0.76 | 3.15 | 0.061 | 2.44 | 2.34 | 0.10 |
| CPB | 535 | 20 | 11.84 ± 0.42 | 10.9 ± 0.66 | 0.78 | 0.036 | 2.33 | 2.07 | 0.27 | |
| TCTA | 529 | 10 | 32.45 ± 1.23 | 5.26 ± 0.71 | 1.17 | 0.034 | 2.34 | 2.09 | 0.25 |
| Compound | Absorption | Emission | Reference | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| λabs1 [nm] | ε1 [L/(mol cm)] | λabs2 [nm] | ε2 [L/(mol cm)] | λex1 [nm] | λem1 [nm] | λex2 [nm] | λem2 [nm] | Δν1 [cm−1] | Δν2 [cm−1] | Φfl [%] | ||
| 66a | 293 | 49,337 | 386 | 105,456 | 268 | 453 | 383 | 454 | 15,238 | 4083 | 4.64 | [50] |
| 66b | 263 | 25,640 | 389 | 34,200 | 262 | 459 | 354 | 466 | 16,381 | 6789 | 6.42 | [50] |
| 66c | 262 | 87,480 | 381 | 144,430 | 262 | 462 | 381 | 463 | 16,552 | 4648 | 19.02 | [50] |
| 66d | 266 | 43,920 | 384 | 68,180 | 2658 | 462 | 389 | 466 | 15,668 | 4536 | 6.35 | [50] |
| 66e | 270 | 18,880 | 399 | 20,706 | 270 | 457 | 400 | 462 | 15,155 | 3354 | 0.18 | [50] |
| 66f | 290 | 38,272 | 383 | 68,795 | 290 | 435 | 380 | 446 | 11,494 | 3894 | 55.27 | [50] |
| 66g | 268 | 34,200 | 398 | 44,780 | 262 | 451 | 360 | 463 | 15,994 | 6179 | 3.47 | [50] |
| 66h | 262 | 65,250 | 380 | 77,820 | 263 | 430 | 380 | 460 | 14,767 | 4576 | 8.91 | [50] |
| 66i | 292 | 39,784 | 380 | 34,836 | 264 | 455 | 380 | 464 | 15,900 | 4764 | 15.37 | [50] |
| 66j | 260 | 40,740 | 381 | 44,950 | 329 | 478 | 358 | 472 | 9860 | 6740 | 11.85 | [49] |
| Compound | R4 | Absorption | Emission | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| λabs1 [nm] | ε1 [L/(mol cm)] | λabs2 [nm] | ε2 [L/(mol cm)] | λex [nm] | λem [nm] | Δν1 [cm−1] | Δν2 [cm−1] | Φfl [%] | ||
| 69a | H | 268 | 23,300 | 396 | 39,100 | 396 | 462 | 15,668 | 3608 | 51.77 |
| 69b | H | 265 | 25,200 | 400 | 36,800 | 400 | 462 | 16,091 | 3355 | 38.26 |
| 69c | H | 275 | 29,090 | 397 | 47,900 | 397 | 458 | 14,530 | 3355 | 61.35 |
| 69d | H | 258 | 42,300 | 405 | 53,800 | 405 | 437 | 15,876 | 1808 | 42.40 |
| 69e | CO2Me | 261 | 44,620 | 386 | 46,960 | 386 | 463 | 16,716 | 6786 | 11.04 |
| 69f | CO2Et | 252 | 39,520 | 390 | 50,630 | 390 | 539 | 21,130 | 7088 | 2.64 |
| 69g | CO2Me | 242 | 42,770 | 392 | 57,930 | 392 | 440 | 18,595 | 6957 | 6.63 |
| 69h | CO2Et | 250 | 61,060 | 395 | 65,910 | 395 | 456 | 18,070 | 3387 | 2.99 |
| 69i | CO2Me | 242 | 25,600 | 396 | 27,850 | 396 | 450 | 19,100 | 3030 | 27.46 |
| 69j | CO2Me | 268 | 56,200 | 388 | 47,200 | 388 | 535 | 18,622 | 7116 | 3.24 |
| Compound | Solvent | Emission λmax nm | Excitation λmax nm | τ ns | Φfl % |
|---|---|---|---|---|---|
| 76a | MePh | 602 | 337, 430, 516 | 4.6 ± 0.5 | 14 |
| MeCN | 598 | 405, 494 | 3.9 ± 0.5 | 8 | |
| DCM | 590 | 329, 413, 502 | 5.5 ± 0.5 | 18 | |
| MeOH | 507 | 295, 428 | 9.0 ± 0.5 | 76 | |
| 76b | MePh | 640 | 427, 528 | 1.0 ± 0.2 | 7 |
| MeCN | 630 | 401, 498 | 1.0 ± 0.2 | 0.7 | |
| DCM | 628 | 438, 500 | 1.0 ± 0.2 | 3 | |
| MeOH | 530 | 292, 426 | 10.3 ± 0.5 | 47 | |
| 76c | MePh | 645 | 440, 539 | 1.4 ± 0.2 | 7.5 |
| MeCN | 642 | 417, 513 | 1.5 ± 0.2 | 2.5 | |
| DCM | 626 | 330, 420, 522 | 1.7 ± 0.2 | 9 | |
| MeOH | 537 | 443 | 7.5 ± 0.5 | 81 |
| Compound | Fluorescence (λmax, nm) (Quantum Yields (%)) | Absorption (λmax, nm) | ||||
|---|---|---|---|---|---|---|
| EtOH | CHCl3 | Cyclohexane | EtOH | CHCl3 | Cyclohexane | |
| 79a | 431 (70) | 430 (90) | 430 (83) | 343 | 351 | 355 |
| 79b | 431 (70) | 429 (90) | 430 (83) | 343 | 352 | 355 |
| 79c | 429 (63) | 431 (82) | 434 (76) | 356 | 362 | 366 |
| 79d | 446 (1) | 436 (8) | 433 (11) | 334 | 335 | 329 |
| 79e | 448 (2) | 439 (3) | 434 (-) | 334 | 334 | 329 |
| 79f | 451 (1) | 438 (7) | 436 (10) | 332 | 333 | 327 |
| 79g | 487 (1) | 471 (1) | insoluble | 374 | 375 | insoluble |
| 79h | 448 (4) | 431 (5) | 434 (8) | 314 | 319 | 322 |
| 80a | 430 (66) | 423 (91) | 416 (85) | 349 | 357 | 361 |
| 80b | 429 (66) | 422 (91) | 416 (85) | 349 | 358 | 361 |
| 80c | 436 (89) | 437 (76) | insoluble | 365 | 370 | insoluble |
| 80d | 442 (1) | 436 (5) | insoluble | 333 | 335 | insoluble |
| 80e | 430 (2) | 436 (7) | insoluble | 330 | 332 | insoluble |
| 80f | 433 (1) | 437 (4) | 432 (8) | 333 | 334 | 329 |
| 80g | 446 (4) | 430 (5) | 432 (9) | 315 | 319 | 322 |
| 80h | 446 (4) | 430 (4) | 432 (8) | 316 | 320 | 322 |
| 80i | 448 (3) | 432 (2) | insoluble | 319 | 322 | insoluble |
| 81b | 442 (2) | 439 (2) | insoluble | 326 | 321 | insoluble |
| 81c | 427 (1) | 439 (5) | insoluble | 316 | 304 | insoluble |
| 82a | 430 (9) | 427 (18) | 415 (40) | 470 | 479 | 496 |
| 82b | 430 (9) | 427 (18) | 415 (40) | 470 | 479 | 496 |
| 82c | 432 (3) | 424 (4) | 420 (4) | 485 | 488 | 508 |
| 82d | 433 (4) | 424 (4) | 419 (4) | 484 | 488 | 508 |
| Compound | λmax (nm) [ε (L mol−1 cm−1) × 103] | Fluorescence Intensity | ||||
|---|---|---|---|---|---|---|
| Number of Absorption Maxima | ||||||
| I | II | III | IV | V | ||
| 86a | 356.00 [2.65] | 347.00 [2.33] | 277.00 [30.33] | 264.00 [25.02] | 225.00 [11.30] | 6276.11 |
| 86b | 357.00 [2.93] | 337.00 [2.58] | 280.00 [32.95] | 263.00 [26.00] | 226.00 [11.90] | 6554.21 |
| 86c | 355.00 [2.93] | 341.00 [2.80] | 284.00 [30.23] | 265.00 [24.15] | 228.00 [11.91] | 7510.05 |
| 86d | 356.00 [3.00] | 340.00 [2.73] | 284.00 [31.68] | 264.00 [24.73] | 227.00 [11.75] | 8264.76 |
| 87a | 344.00 [3.50] | 340.00 [3.46] | 276.00 [31.15] | 264.00 [26.75] | 226.00 [7.45] | 5016.82 |
| 87b | 350.00 [3.18] | 335.00 [2.95] | 276.00 [32.38] | 264.00 [26.75] | 226.00 [11.51] | 5269.64 |
| 87c | 350.00 [3.18] | 335.00 [3.01] | 282.00 [30.21] | 265.00 [25.68] | 227.00 [6.00] | 6018.34 |
| 87d | 350.00 [3.31] | 334.00 [3.06] | 276.00 [34.46] | 264.00 [29.01] | 228.00 [12.33] | 6304.55 |
| Compound | Fluorescence (λmax, nm) (Quantum Yields (%)) | Absorption (λmax, nm) | ||
|---|---|---|---|---|
| Cyclohexane | Dichloromethane | Cyclohexane | Dichloromethane | |
| 91 | 426 (24) | 432 (21) | 360 | 358 |
| 92 | 422 (25) | 426 (26) | 356 | 354 |
| 93 | 428 (7) | 441 (5) | 327 | 325 |
| 94 | 439 (6) | 446 (4) | 324 | 322 |
| 95 | 491 | - | 374 | 372 |
| Compound | Fluorescence (λmax, nm) | Absorption (λmax, nm) | ||
|---|---|---|---|---|
| Cyclohexane | Trichloromethane | Cyclohexane | Trichloromethane | |
| 102a | 434 | 439 | 399 | 403 |
| 102b | 434 | 440 | 399 | 402 |
| 102c | 440 | 445 | 384 | 386 |
| 103a | 432 | 432 | 378 | 381 |
| 103b | 432 | 433 | 376 | 375 |
| 103c | 451 | 430 | 373 | 370 |
| 104c | 450 | 449 | 374 | 369 |
| Compound | Fluorescence (λmax, nm) | Absorption (λmax, nm) | Quantum Yields (%) |
|---|---|---|---|
| 105a | 443 | 394 | 22.11 |
| 105b | 431 | 391 | 18.64 |
| 105c | 444 | 412 | 15.92 |
| 105d | 433 | 393 | 17.09 |
| 107a | 430 | 406 | 9.34 |
| 107b | 435 | 387 | 7.17 |
| 107c | 430 | 409 | 8.99 |
| 107d | 445 | 393 | 5.30 |
| Compound | Solvent | λabs [nm] | λem [nm] | Φfl [%] | τ1 [ns] | τ2 [ns] |
|---|---|---|---|---|---|---|
| 112a | DCM | 352, 335, 320, 275, 244 | 356, 374, 392, 415sh | 28.37 | ||
| DMSO | 353, 336, 321, 276 | 359, 376, 395, 415sh | 63.47 | 0.069 | 3.115 | |
| 112b | DCM | 358, 341, 325, 274, 246 | 363, 381, 401, 425sh | 20.10 | ||
| DMSO | 358, 341, 325, 275 | 361, 382, 401, 425sh | 53.95 | 0.866 | 3.095 | |
| 112c | DCM | 358, 341, 324, 274, 247 | 363, 381, 401, 425sh | 31.28 | ||
| DMSO | 358, 341, 325sh, 275 | 364, 382, 400, 425sh | 55.76 | 0.112 | 3.219 | |
| 113a | DCM | 338, 323, 309, 252 | 340, 356, 375, 395sh | 41.04 | ||
| DMSO | 339, 324, 309 | 345, 361, 378 | 3.36 | 0.031 | 2.785 | |
| 113b | DCM | 338, 323, 309, 251 | 344, 360, 374, 400sh | 18.37 | ||
| DMSO | 338, 324, 309 | 346, 360, 374 | 7.96 | 0.060 | 2.700 | |
| 113c | DCM | 338, 323, 309, 252 | 345, 359, 375, 400sh | 29.41 | ||
| DMSO | 339, 323, 309 | 346, 359, 372 | 15.95 | 0.036 | 5.400 |
| Compound | Φfl [%] | τ1 [ns] | τ2 [ns] | τ3 [ns] | f1 | f2 | f3 |
|---|---|---|---|---|---|---|---|
| 114a | 38.71 | 4.41 | |||||
| 114b | 17.87 | 3.39 | |||||
| 114c | 16.89 | 0.48 | 9.35 | 0.23 | 0.77 | ||
| 114e | 75.42 | 4.00 | |||||
| 114f | 6.65 | 0.41 | 2.51 | 8.12 | 0.37 | 0.25 | 0.38 |
| 114g | 6.09 | 0.60 | 8.06 | 0.57 | 0.43 | ||
| 115a | 33.10 | 3.12 | |||||
| 115b | 6.16 | 0.76 | 4.45 | 0.94 | 0.06 | ||
| 115e | 4.43 | 3.73 |
| Compound | pH | λex [nm] | λem [nm] | Stokes Shift [cm−1] |
|---|---|---|---|---|
| 121 | 2.0 | 395 | 521 | 6123 |
| 5.0 | 539 | 6764 | ||
| 5.5 | 539 | 6764 | ||
| 6.0 | 539 | 6764 | ||
| 6.5 | 539 | 6764 | ||
| 7.0 | 539 | 6764 | ||
| 7.5 | 539 | 6764 | ||
| 8.0 | 539 | 6764 | ||
| 12.0 | 491, 526 | 4950, 6305 | ||
| 124 | 2.0 | 395 | 560 | 7459 |
| 5.0 | 486, 525 | 4740, 6269 | ||
| 5.5 | 480, 513 | 4483, 5823 | ||
| 6.0 | 480, 514 | 4483, 5861 | ||
| 6.5 | 480 | 4483 | ||
| 7.0 | 480 | 4483 | ||
| 7.5 | 481 | 4526 | ||
| 8.0 | 482 | 4570 | ||
| 12.0 | 482 | 4570 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zbancioc, G.; Mangalagiu, I.I.; Moldoveanu, C. A Review on the Synthesis of Fluorescent Five- and Six-Membered Ring Azaheterocycles. Molecules 2022, 27, 6321. https://doi.org/10.3390/molecules27196321
Zbancioc G, Mangalagiu II, Moldoveanu C. A Review on the Synthesis of Fluorescent Five- and Six-Membered Ring Azaheterocycles. Molecules. 2022; 27(19):6321. https://doi.org/10.3390/molecules27196321
Chicago/Turabian StyleZbancioc, Gheorghita, Ionel I. Mangalagiu, and Costel Moldoveanu. 2022. "A Review on the Synthesis of Fluorescent Five- and Six-Membered Ring Azaheterocycles" Molecules 27, no. 19: 6321. https://doi.org/10.3390/molecules27196321





