Antioxidant and Antimicrobial Activities of Thai Edible Plant Extracts Prepared Using Different Extraction Techniques
Abstract
:1. Introduction
2. Results and Discussion
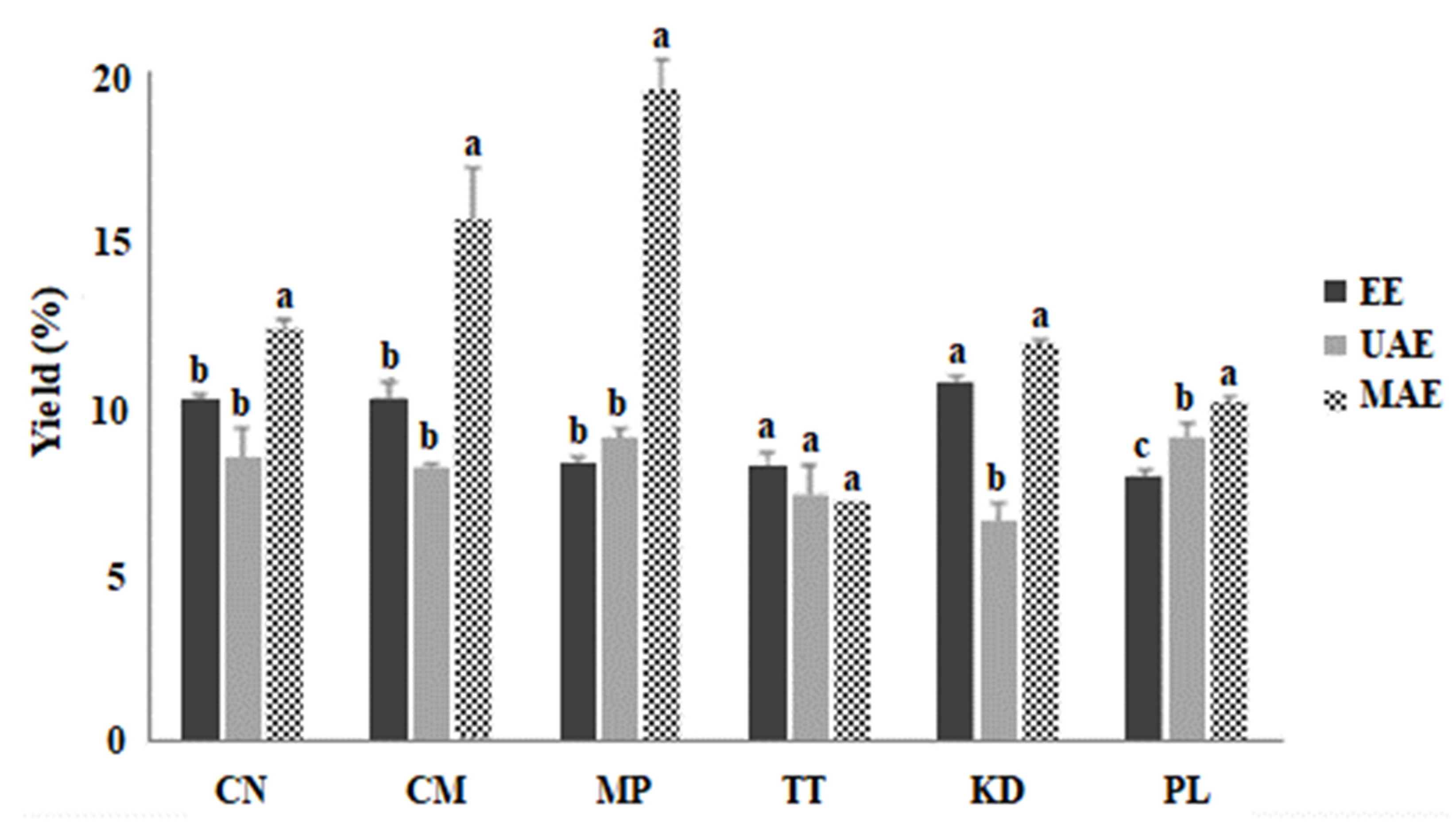
2.1. Extraction Efficiency of MAE, UAE, and EE
2.2. Total Phenolic and Flavonoid Contents of Plant Extracts Prepared by MAE, UAE, and EE Processes
2.3. Impact of MAE, UAE, and EE on the Antioxidant Activity of Plant Extracts
2.4. Disc Diffusion Test, MIC, and Minimum MBC of Thai Edible Plant Extracts Prepared by MAE, UAE, and EE
3. Materials and Methods
3.1. Materials
3.2. Preparation of Thai Edible Plant Extracts Using the Conventional EE, UAE, and MAE
3.3. Determination of Extraction Yield and Chemical Properties of Thai Edible Plant Extracts
3.4. Determination of Antimicrobial Properties of Edible Thai Plant Extracts
3.5. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Cowan, M.M. Plant products as antimicrobial agents. Clin. Microbiol. Rev. 1999, 12, 564–582. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- See, J.A.; Gall, M.M. Eating Plants for Health: From Fiber to Phytochemicals. J. Am. Diet. Assoc. 1997, 97 (Suppl. S9), A60. [Google Scholar] [CrossRef]
- Lee, J.H.; Cho, S.; Paik, H.D.; Choi, C.W.; Nam, K.T.; Hwang, S.G.; Kim, S.K. Investigation on antibacterial and antioxidant activities, phenolic and flavonoid contents of some thai edible plants as an alternative for antibiotics. Asian Australas J. Anim. Sci. 2014, 27, 1461–1468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sakunpak, A.; Panichayupakaranant, P. Antibacterial activity of Thai edible plants against gastrointestinal pathogenic bacteria and isolation of a new broad spectrum antibacterial polyisoprenylated benzophenone, chamuangone. Food Chem. 2012, 130, 826–831. [Google Scholar] [CrossRef]
- Campos-Vega, R.; Oomah, B.D. Chemistry and classification of phytochemicals. In Handbook of Plant Food Phytochemicals; Wiley-Blackwell: Chicester, UK, 2013; pp. 5–48. [Google Scholar]
- Pinchuk, I.; Shoval, H.; Dotan, Y.; Lichtenberg, D. Evaluation of antioxidants: Scope, limitations and relevance of assays. Chem. Phys. Lipids 2012, 165, 638–647. [Google Scholar] [CrossRef]
- Fraga, C.G.; Galleano, M.; Verstraeten, S.V.; Oteiza, P.I. Basic biochemical mechanisms behind the health benefits of polyphenols. Mol. Aspects Med. 2010, 31, 435–445. [Google Scholar] [CrossRef]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell. Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef] [Green Version]
- Finley, J.W.; Kong, A.N.; Hintze, K.J.; Jeffery, E.H.; Ji, L.L.; Lei, X.G. Antioxidants in foods: State of the science important to the food industry. J. Agric. Food Chem. 2011, 59, 6837–6846. [Google Scholar] [CrossRef]
- Poljsak, B.; Kovač, V.; Milisav, I. Antioxidants, Food Processing and Health. Antioxidants 2021, 10, 433. [Google Scholar] [CrossRef]
- Mostafa, A.A.; Al-Askar, A.A.; Almaary, K.S.; Dawoud, T.M.; Sholkamy, E.N.; Bakri, M.M. Antimicrobial activity of some plant extracts against bacterial strains causing food poisoning diseases. Saudi. J. Biol. Sci. 2018, 25, 361–366. [Google Scholar] [CrossRef]
- Santajit, S.; Indrawattana, N. Mechanisms of Antimicrobial Resistance in ESKAPE Pathogens. Biomed. Res. Int. 2016, 2016, 2475067. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anand, G.; Ravinanthan, M.; Basaviah, R.; Shetty, A.V. In vitro antimicrobial and cytotoxic effects of Anacardium occidentale and Mangifera indica in oral care. J. Pharm. Bioallied. Sci. 2015, 7, 69. [Google Scholar] [PubMed]
- Stalikas, C.D. Extraction, separation, and detection methods for phenolic acids and flavonoids. J. Sep. Sci. 2007, 30, 3268–3295. [Google Scholar] [CrossRef]
- Gupta, A.; Naraniwal, M.; Kothari, V. Modern extraction methods for preparation of bioactive plant extracts. Int. J. Appl. Nat. Sci. 2012, 1, 8–26. [Google Scholar]
- Dahmoune, F.; Nayak, B.; Moussi, K.; Remini, H.; Madani, K. Optimization of microwave-assisted extraction of polyphenols from Myrtus communis L. leaves. Food Chem. 2015, 166, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Carreira-Casais, A.; Otero, P.; Garcia-Perez, P.; Garcia-Oliveira, P.; Pereira, A.G.; Carpena, M.; Soria-Lopez, A.; Simal-Gandara, J.; Prieto, M.A. Benefits and drawbacks of ultrasound-assisted extraction for the recovery of bioactive compounds from marine algae. Int. J. Environ. Res. Public Health 2021, 18, 9153. [Google Scholar] [CrossRef]
- Huma, Z.-E.; Jayasena, V.; Abbas, S.; Imran, M.; Khan, M. Process optimization of polyphenol extraction from carob (Ceratonia siliqua) kibbles using microwave-assisted technique. J. Food Process. Preserv. 2017, 42, e13450. [Google Scholar] [CrossRef]
- Osorio-Tobón, J.F. Recent advances and comparisons of conventional and alternative extraction techniques of phenolic compounds. J. Food Sci. Technol. 2020, 57, 4299–4315. [Google Scholar] [CrossRef]
- Chan, C.-H.; Yeoh, H.K.; Yusoff, R.; Ngoh, G.C. A first-principles model for plant cell rupture in microwave-assisted extraction of bioactive compounds. J. Food Eng. 2016, 188, 98–107. [Google Scholar] [CrossRef]
- Chen, L.; Jin, H.; Ding, L.; Zhang, H.; Li, J.; Qu, C.; Zhang, H. Dynamic microwave-assisted extraction of flavonoids from Herba Epimedii. Sep. Purif. Technol. 2008, 59, 50–57. [Google Scholar] [CrossRef]
- Rahath Kubra, I.; Kumar, D.; Rao, L.J.M. Effect of microwave-assisted extraction on the release of polyphenols from ginger (Zingiber officinale). Int. J. Food Sci. Technol. 2013, 48, 1828–1833. [Google Scholar] [CrossRef]
- Vinatoru, M. An overview of the ultrasonically assisted extraction of bioactive principles from herbs. Ultrason. Sonochem. 2001, 8, 303–313. [Google Scholar] [CrossRef]
- Sun, C.; Wu, Z.; Wang, Z.; Zhang, H. Effect of ethanol/water solvents on phenolic profiles and antioxidant properties of Beijing propolis extracts. Evid Based Complement. Alternat. Med. 2015, 2, 595393. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, H.-F.; Zhang, X.; Yang, X.-H.; Qiu, N.-X.; Wang, Y.; Wang, Z.-Z. Microwave assisted extraction of flavonoids from cultivated Epimedium sagittatum: Extraction yield and mechanism, antioxidant activity and chemical composition. Ind. Crop. Prod. 2013, 50, 857–865. [Google Scholar] [CrossRef]
- Altemimi, A.; Watson, D.G.; Choudhary, R.; Dasari, M.R.; Lightfoot, D.A. Ultrasound assisted extraction of phenolic compounds from peaches and pumpkins. PLoS ONE 2016, 11, e0148758. [Google Scholar] [CrossRef] [Green Version]
- Büyüktunce, E.; Porgalı, E.; Çolak, C. Comparison of total phenolic content and total antioxidant activity in local red wines determined by spectrophotometric methods. Food Nutr. Sci. 2014, 2014, 1660–1667. [Google Scholar]
- Moharram, H.; Youssef, M. Methods for determining the antioxidant activity: A review. Alex. J. Food Sci. Tech. 2014, 11, 31–42. [Google Scholar]
- Ahmad Shiekh, K.; Odunayo Olatunde, O.; Zhang, B.; Huda, N.; Benjakul, S. Pulsed electric field assisted process for extraction of bioactive compounds from custard apple (Annona squamosa) leaves. Food Chem. 2021, 359, 129976. [Google Scholar] [CrossRef]
- Haro, G.; Utami, N.; Sitompul, E. Study of the antibacterial activities of Soursop (Annona muricata L.) leaves. Int. J. Pharmtech. Res. 2014, 6, 575–581. [Google Scholar]
- Solomon-Wisdom, G.; Ugoh, S.; Mohammed, B. Phytochemical screening and antimicrobial activities of Annona muricata (L) leaf extract. Am. J. Biol. Chem. Pharm. Sci. 2014, 2, 1–7. [Google Scholar]
- Daduang, J.; Daduang, S.; Hongsprabhas, P.; Boonsiri, P. High phenolics and antioxidants of some tropical vegetables related to antibacterial and anticancer activities. Afr. J. Pharm. Pharmacol. 2011, 5, 608–615. [Google Scholar] [CrossRef] [Green Version]
- Chanudom, L.; Bhoopong, P.; Khwanchuea, R.; Tangpong, J. Antioxidant and antimicrobial activities of aqueous & ethanol crude extracts of 13 Thai traditional plants. Int. J. Curr. Microbiol. Appl. Sci. 2014, 3, 549–558. [Google Scholar]
- Cetin-Karaca, H.; Newman, M.C. Antimicrobial efficacy of plant phenolic compounds against Salmonella and Escherichia Coli. Food Biosci. 2015, 11, 8–16. [Google Scholar] [CrossRef]
- Jarriyawattanachaikul, W.; Chaveerach, P.; Chokesajjawatee, N. Antimicrobial activity of Thai-Herbal plants against food-borne pathogens E. Coli, S. Aureus and C. Jejuni. Agric. Agric. Sci. Procedia 2016, 11, 20–24. [Google Scholar] [CrossRef]
- Moreno, S.; Scheyer, T.; Romano, C.S.; Vojnov, A.A. Antioxidant and antimicrobial activities of rosemary extracts linked to their polyphenol composition. Free Radic. Res. 2006, 40, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Alam, G.; Natsir, S.; Alfath, A. Comparison of microwave assisted extraction (MAE) with variations of power and infusion extraction method on antibacterial activity of rosella calyx extract (Hibiscus sabdariffa). J. Phys. Conf. Ser. 2019, 1341, 072002. [Google Scholar] [CrossRef]
- Latha, C. Microwave-assisted extraction of embelin from Embelia ribes. Biotechnol. Lett. 2007, 29, 319–322. [Google Scholar] [CrossRef]
- Costa, A.R.; de Lima Silva, J.R.; de Oliveira, T.J.S.; da Silva, T.G.; Pereira, P.S.; de Oliveira Borba, E.F.; de Brito, E.S.; Ribeiro, P.R.V.; Almeida-Bezerra, J.W.; Júnior, J.T.C. Phytochemical profile of Anacardium occidentale L. (cashew tree) and the cytotoxic and toxicological evaluation of its bark and leaf extracts. S. Afr. J. Bot. 2020, 135, 355–364. [Google Scholar] [CrossRef]
- Phukhatmuen, P.; Raksat, A.; Laphookhieo, S.; Charoensup, R.; Duangyod, T.; Maneerat, W. Bioassay-guided isolation and identification of antidiabetic compounds from Garcinia cowa leaf extract. Heliyon 2020, 6, e03625. [Google Scholar] [CrossRef]
- Nagappan, R. Evaluation of aqueous and ethanol extract of bioactive medicinal plant, Cassia didymobotrya (Fresenius) Irwin & Barneby against immature stages of filarial vector, Culex quinquefasciatus Say (Diptera: Culicidae). Asian Pac. Trop. Biomed. 2012, 2, 707–711. [Google Scholar]
- Kazibwe, Z.; Kim, D.-H.; Chun, S.; Gopal, J. Ultrasonication assisted ultrafast extraction of Tagetes erecta in water: Cannonading antimicrobial, antioxidant components. J. Mol. Liq. 2017, 229, 453–458. [Google Scholar] [CrossRef]
- Piovesan, N.; Viera, V.; Mello, R.d.O.; dos Santos, R.; Vaucher, R.d.A.; Dressler, V.; Bizzi, C.; Fries, L. Microwave-assisted extraction of bioactive compounds from blueberry (Vaccinium ashei Reade) and their antioxidant and antimicrobial capacity. Int. Food Res. J. 2017, 24, 2526–2533. [Google Scholar]
- Lu, Q.; Lv, S.; Peng, Y.; Zhu, C.; Pan, S. Characterization of phenolics and antioxidant abilities of red navel orange “Cara Cara” harvested from five regions of China. Int. J. Food Prop. 2018, 21, 1107–1116. [Google Scholar] [CrossRef]
- Olatunde, O.O.; Benjakul, S.; Vongkamjan, K. Antioxidant and antibacterial properties of guava leaf extracts as affected by solvents used for prior dechlorophyllization. J. Food Biochem. 2018, 42, e12600. [Google Scholar] [CrossRef]
- Rebaya, A.; Belghith, S.I.; Baghdikian, B.; Leddet, V.M.; Mabrouki, F.; Olivier, E.; Cherif, J.; Ayadi, M.T. Total phenolic, total flavonoid, tannin content, and antioxidant capacity of Halimium halimifolium (Cistaceae). J. App. Pharm. Sci. 2014, 5, 52–57. [Google Scholar]
- Molyneux, P. The use of the stable radical Diphenylpicrylhydrazyl (DPPH) for estimating antioxidant activity. Songklanakarin J. Sci. Technol. 2003, 26, 211–219. [Google Scholar]
- Tagrida, M.; Benjakul, S. Betel (Piper betle L.) leaf ethanolic extracts dechlorophyllized using different methods: Antioxidant and antibacterial activities, and application for shelf-life extension of Nile tilapia (Oreochromis niloticus) fillets. RSC Adv. 2021, 11, 17630–17641. [Google Scholar] [CrossRef] [PubMed]
- Sadeghi, Z.; Valizadeh, J.; Shermeh, O.A.; Akaberi, M. Antioxidant activity and total phenolic content of Boerhavia elegans (choisy) grown in Baluchestan, Iran. Avicenna J. Phytomed. 2015, 5, 1. [Google Scholar]
- Bauer, A.W.; Kirby, W.M.; Sherris, J.C.; Turck, M. Antibiotic susceptibility testing by a standardized single disk method. Am. J. Clin. Pathol. 1966, 45, 493–496. [Google Scholar] [CrossRef]
- Mosaddik, M.; Banbury, L.; Forster, P.; Booth, R.; Markham, J.; Leach, D.; Waterman, P.G. Screening of some Australian Flacourtiaceae species for in vitro antioxidant, cytotoxic and antimicrobial activity. Phytomedicine 2004, 11, 461–466. [Google Scholar] [CrossRef]


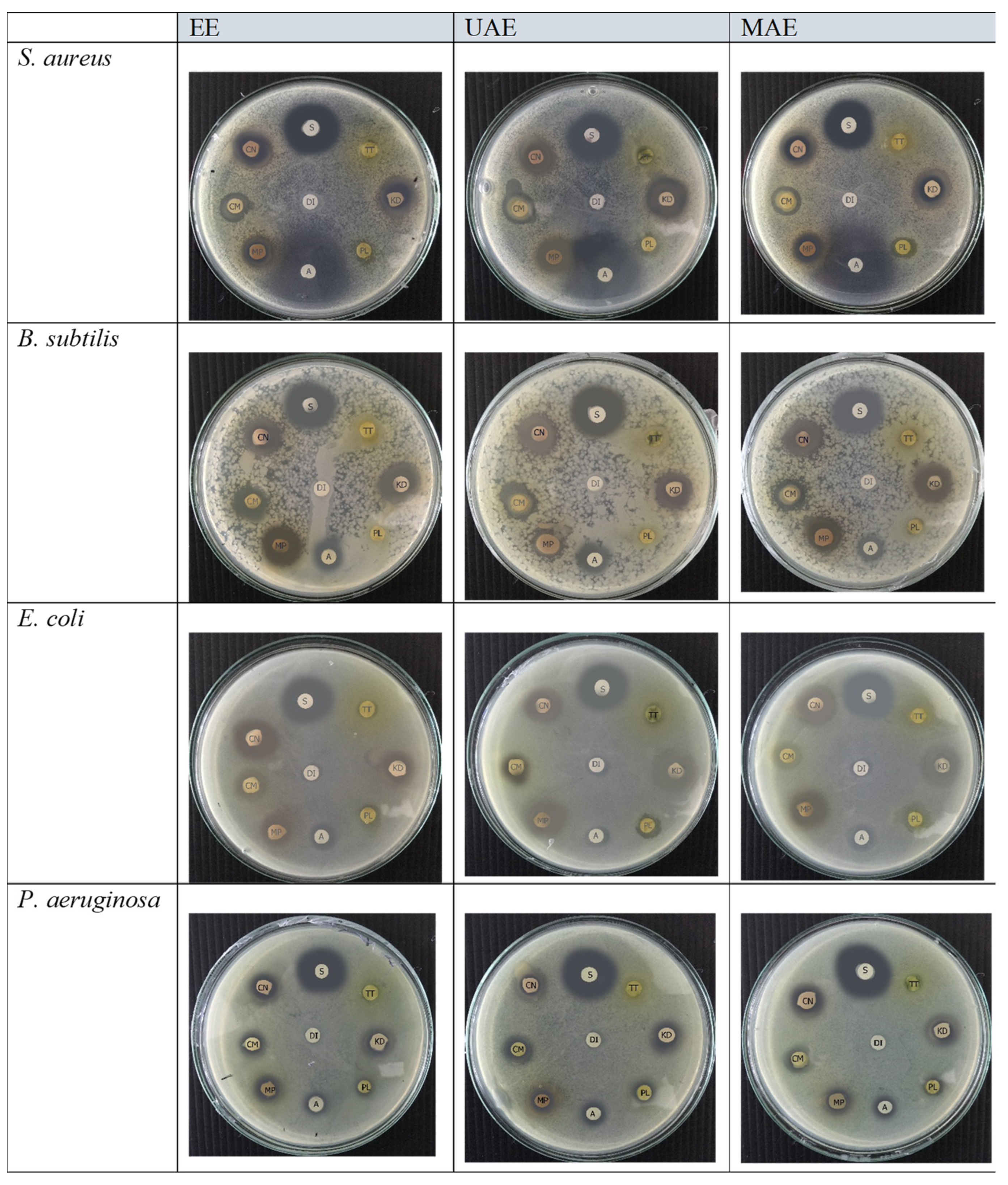
| Bacterial Strains | Samples | Zone of Inhibition (mm) | ||||
|---|---|---|---|---|---|---|
| EE | UAE | MAE | Ampicillin | Streptomycin | ||
| S. aureus | 29 | 25 | ||||
| CN | 16 | 14 | 17 | |||
| CM | 14 | 14 | 14 | |||
| MP | 16 | 14 | 14 | |||
| TT | NI | NI | NI | |||
| KD | 17 | 17 | 18 | |||
| PL | 10 | 10 | 11 | |||
| B. subtilis | 12 | 23 | ||||
| CN | 15 | 16 | 16 | |||
| CM | 14 | 15 | 15 | |||
| MP | 15 | 16 | 16 | |||
| TT | NI | NI | NI | |||
| KD | 17 | 17 | 17 | |||
| PL | NI | NI | NI | |||
| E. coli | 13 | 22 | ||||
| CN | 12 | 14 | 15 | |||
| CM | 11 | 11 | 11 | |||
| MP | 10 | 15 | 15 | |||
| TT | NI | NI | NI | |||
| KD | 11 | 15 | 16 | |||
| PL | 9 | 10 | 11 | |||
| P. aeruginosa | 12 | 26 | ||||
| CN | 14 | 15 | 16 | |||
| CM | 12 | 13 | 12 | |||
| MP | 13 | 13 | 14 | |||
| TT | NI | NI | NI | |||
| KD | 13 | 14 | 16 | |||
| PL | 9 | 10 | 10 | |||
| Bacterial Strains | Plant | MIC (mg/mL) | MBC (mg/mL) | ||||
|---|---|---|---|---|---|---|---|
| EE | UAE | MAE | EE | UAE | MAE | ||
| Staphylococcus aureus | CN | 1.56 | <1.56 | 0.78 | 25 | 25 | 12.5 |
| CM | 3.12 | <1.56 | 0.78 | 25 | 25 | 25 | |
| MP | <1.56 | <1.56 | 0.78 | 25 | 25 | 25 | |
| TT | N/D | N/D | N/D | N/D | N/D | N/D | |
| KD | <1.56 | <1.56 | <1.56 | 25 | 25 | 25 | |
| PL | 50 | 50 | 12.5 | 50 | 50 | 50 | |
| Bacillus subtilis | CN | 1.56 | <1.56 | <1.56 | 50 | 50 | 25 |
| CM | 1.56 | <1.56 | <1.56 | 50 | 50 | 50 | |
| MP | <1.56 | <1.56 | <1.56 | 50 | 50 | 25 | |
| TT | N/D | N/D | N/D | N/D | N/D | N/D | |
| KD | <1.56 | <1.56 | <1.56 | 50 | 50 | 25 | |
| PL | 25 | 12.5 | 6.25 | 50 | 50 | 50 | |
| Escherichia coli | CN | 1.56 | 1.56 | 1.56 | 50 | 50 | 25 |
| CM | 12.5 | 12.5 | 12.5 | 100 | 100 | 100 | |
| MP | 6.25 | 3.12 | 3.12 | 50 | 50 | 50 | |
| TT | N/D | N/D | N/D | N/D | N/D | N/D | |
| KD | 3.12 | 3.12 | 1.56 | 50 | 50 | 50 | |
| PL | 50 | 50 | 50 | 100 | 100 | 100 | |
| Pseudomonas aeruginosa | CN | 1.56 | 1.56 | 1.56 | 50 | 50 | 25 |
| CM | 12.5 | 12.5 | 12.5 | 100 | 100 | 50 | |
| MP | 3.12 | 6.25 | 3.12 | 50 | 50 | 25 | |
| TT | N/D | N/D | N/D | N/D | N/D | N/D | |
| KD | 1.56 | 1.56 | 1.56 | 50 | 25 | 25 | |
| PL | 50 | 25 | 25 | 100 | 100 | 100 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Junsathian, P.; Nakamura, S.; Katayama, S.; Rawdkuen, S. Antioxidant and Antimicrobial Activities of Thai Edible Plant Extracts Prepared Using Different Extraction Techniques. Molecules 2022, 27, 6489. https://doi.org/10.3390/molecules27196489
Junsathian P, Nakamura S, Katayama S, Rawdkuen S. Antioxidant and Antimicrobial Activities of Thai Edible Plant Extracts Prepared Using Different Extraction Techniques. Molecules. 2022; 27(19):6489. https://doi.org/10.3390/molecules27196489
Chicago/Turabian StyleJunsathian, Pimmada, Soichiro Nakamura, Shigeru Katayama, and Saroat Rawdkuen. 2022. "Antioxidant and Antimicrobial Activities of Thai Edible Plant Extracts Prepared Using Different Extraction Techniques" Molecules 27, no. 19: 6489. https://doi.org/10.3390/molecules27196489






