Development and Greenness Assessment of HPLC Method for Studying the Pharmacokinetics of Co-Administered Metformin and Papaya Extract
Abstract
:1. Introduction
2. Results and Discussion
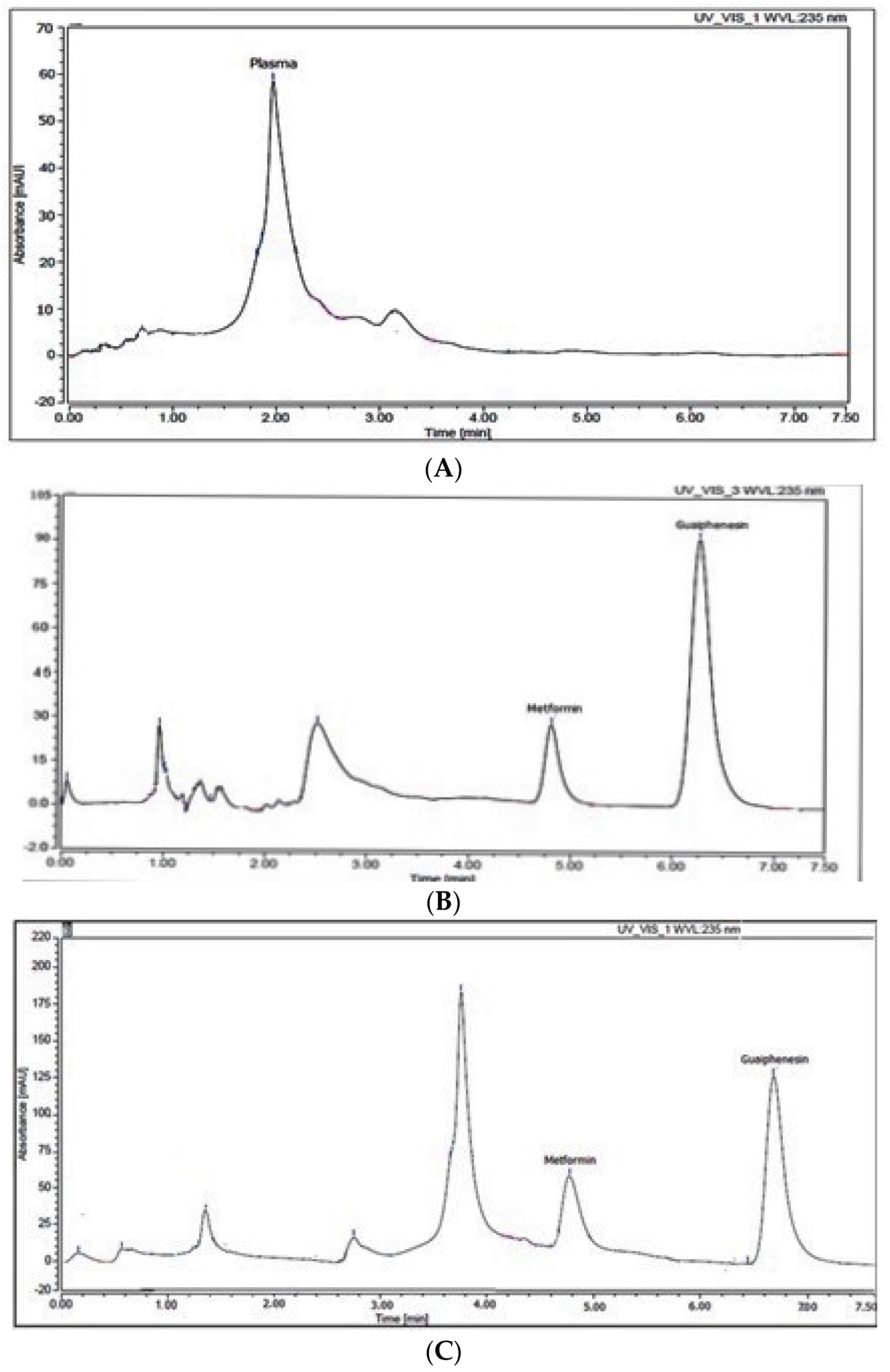
2.1. Optimization of the Developed Method
2.2. Method Validation
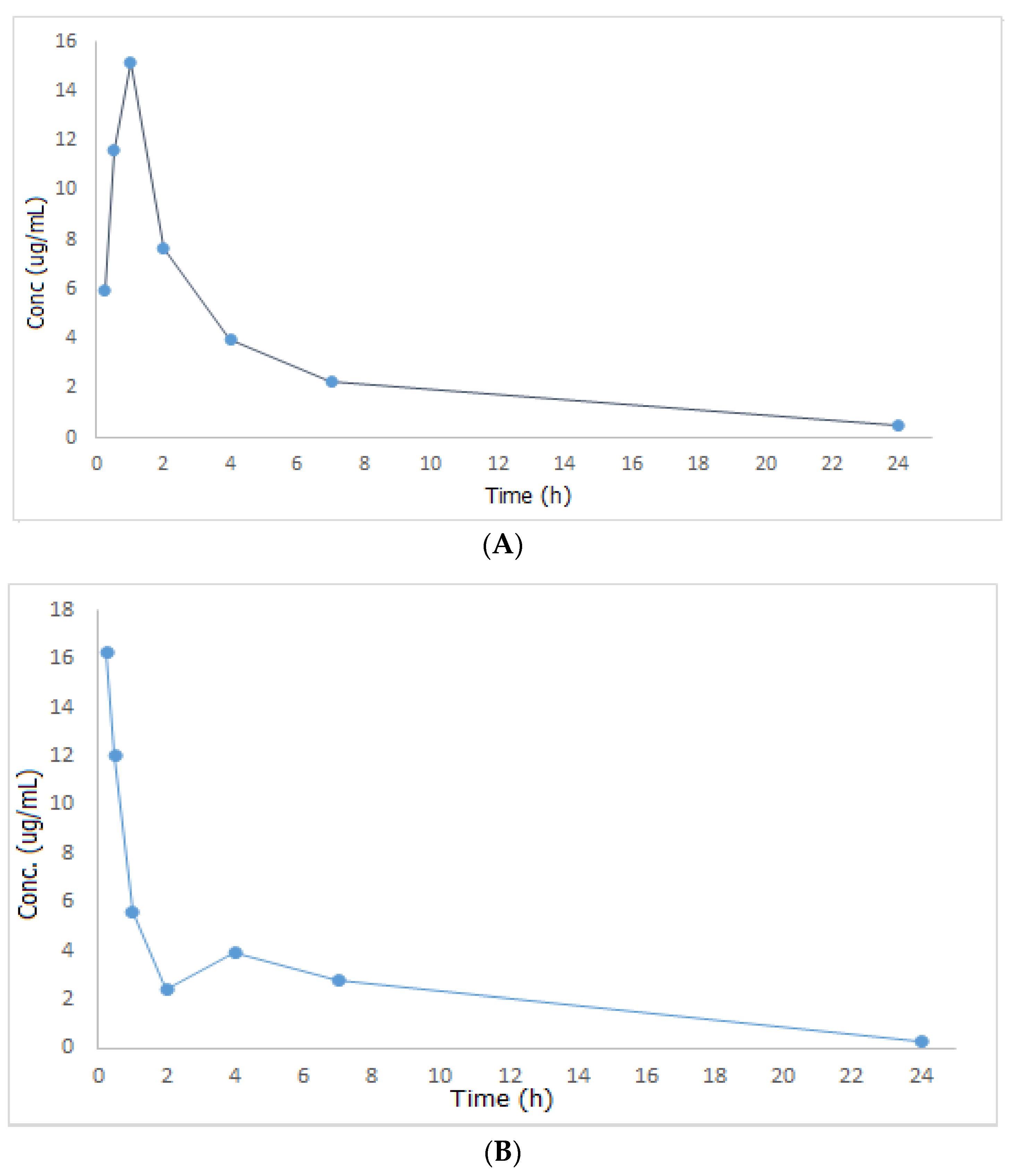
2.3. Pharmacokinetics Study Results
2.4. Evaluation of the Greenness of the Developed Method
3. Materials and Methods
3.1. Apparatuses
3.2. Chromatographic Conditions
3.3. Materials and Reagents
3.4. Solutions
3.5. Animals Used during the Study
3.6. Plant Material Extraction
3.7. Construction of Calibration Curve and Preparation of Quality Control Samples
3.8. Administration of Drug and Assembly of Plasma Samples
3.9. Pretreatment of the Collected Plasma Samples
3.10. Studying the Pharmacokinetics of the Chosen Drug
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
The Ethical Committee Approval
References
- Saleh, D.O.; Bayoumi, A.R.; El-Eraky, W.I.; El-Khatib, A.S. Streptozotocin-induced vascular and biochemical changes in rats: Effects of rosiglitazone vs. metformin. Bull. Fac. Pharm. Cairo Univ. 2013, 51, 131–138. [Google Scholar] [CrossRef] [Green Version]
- Tawfik, S.H.; Mahmoud, B.F.; Saad, M.I.; Shehata, M.; Kamel, M.A.; Helmy, M.H. Similar and additive effects of ovariectomy and diabetes on insulin resistance and lipid metabolism. Biochem. Res. Int. 2015, 2015, 567945. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruhe, R.C.; McDonald, R.B. Use of antioxidant nutrients in the prevention and treatment of type 2 diabetes. J. Am. Coll. Nutr. 2001, 20 (Suppl. S5), 363S–369S. [Google Scholar] [CrossRef] [PubMed]
- Agada, R.; Usman, W.A.; Shehu, S.; Thagariki, D. In vitro and in vivo inhibitory effects of Carica papaya seed on α-amylase and α-glucosidase enzymes. Heliyon 2020, 6, e03618. [Google Scholar] [CrossRef] [PubMed]
- Andrade-Cetto, A.; Heinrich, M. Mexican plants with hypoglycaemic effect used in the treatment of diabetes. J. Ethnopharmacol. 2005, 99, 325–348. [Google Scholar] [CrossRef]
- Oliveira, A.P.; Valentão, P.; Pereira, J.Á.; Silva, B.M.; Tavares, F.; Andrade, P.B. Ficus carica L: Metabolic and biological screening. Food Chem. Toxicol. 2009, 47, 2841–2846. [Google Scholar] [CrossRef]
- Runnie, I.; Salleh, M.N.; Mohamed, S.; Head, R.J.; Abeywardena, M.Y. Vasorelaxation induced by common edible tropical plant extracts in isolated rat aorta and mesenteric vascular bed. J. Ethnopharmacol. 2004, 92, 311–316. [Google Scholar] [CrossRef]
- Otsuki, N.; Dang, N.H.; Kumagai, E.; Kondo, A.; Iwata, S.; Morimoto, C. Aqueous extract of Carica papaya leaves exhibits anti-tumor activity and immunomodulatory effects. Ethnopharmacology 2010, 127, 760–767. [Google Scholar] [CrossRef]
- Mahmood, T.; Rahman, M.H.; Stringam, G.R.; Raney, J.P.; Good, A.G. Molecular markers for seed colour in Brassica juncea. Genome 2005, 48, 755–760. [Google Scholar] [CrossRef]
- Corral-Aguayo, R.D.; Yahia, E.M.; Carrillo-López, A.; González-Aguilar, G. Correlation between some nutritional components and the total antioxidant capacity measured with six different assays in eight horticultural crops. J. Agric. Food Chem. 2008, 56, 10498–10504. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Shaw, P.N.; Parat, M.O.; Hewavitharana, A.K. Anticancer activity of Carica papaya: A review. Mol. Nutr. Food Res. 2013, 57, 153–164. [Google Scholar] [CrossRef]
- Aruoma, O.I.; Somanah, J.; Bourdon, E.; Rondeau, P.; Bahorun, T. Diabetes as a risk factor to cancer: Functional role of fermented papaya preparation as phytonutraceutical adjunct in the treatment of diabetes and cancer. Mutat. Res. 2014, 768, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Rahmani, A.H.; Aldebasi, Y.H. Potential role of carica papaya and their active constituents in the prevention and treatment of disease. Int. J. Pharm. Pharm. Sci. 2016, 8, 975–1491. [Google Scholar]
- Ningrum, V.D.A.; Wibowo, A.; Fuaida, I.; Ikawati, Z.; Sadewa, A.H.; Robikhul Ikhsan, M. Validation of an HPLC-UV method for the determination of metformin hydrochloride in spiked-human plasma for the application of therapeutic drug monitoring. Res. J. Pharm. Technol. 2018, 11, 2197–2202. [Google Scholar] [CrossRef]
- Harahap, Y.; Dianpratami, K.; Wulandari, M.; Rahmawati, R. Validation of metformin hydrochloride in human plasma by HPLC-photo diode array (PDA) for application of bioequivalence study. J. Life Sci. 2012, 6, 20–27. [Google Scholar]
- Porta, V.; Schramm, S.G.; Kano, E.K.; Koono, E.E.; Armando, Y.P.; Fukuda, K.; dos Reis Serra, C.H. HPLC-UV determination of metformin in human plasma for application in pharmacokinetics and bioequivalence studies. J. Pharm. Biomed. Anal. 2008, 46, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Chhetri, H.P.; Thapa, P.; Van Schepdael, A. Simple HPLC-UV method for the quantification of metformin in human plasma with one step protein precipitation. Saudi Pharm. J. 2014, 22, 483–487. [Google Scholar] [CrossRef] [Green Version]
- Rebecca, Y.M.; Sudha, V.; Kumar, A.K.H. Validated high performance liquid chromatography method for the determination of metformin in human plasma and its application to pharmacokinetic study. Chromatogr. Sep. Technol. J. 2019, 2, 119–124. [Google Scholar]
- Sher, N.; Fatima, N.; Perveen, S.; Siddiqui, F.A. Simultaneous determination of anti-diabetic drugs. Braz. J. Pharm. Sci. 2019, 55, e17394. [Google Scholar] [CrossRef]
- Liu, A.; Coleman, S.P. Determination of Metformin in Human Plasma Using Hydrophilic Interaction Liquid Chromatography—Tandem Mass Spectrometry. J. Chromatogr. B 2009, 877, 3695–3700. [Google Scholar] [CrossRef]
- Koseki, N.; Kawashita, H.; Niina, M.; Nagae, Y.; Masuda, N. Development and validation of high selective quantitative determination of metformin in human plasma by cation exchanging with normal-phase LC/MS/MS. J. Pharm. Biomed. Anal. 2005, 36, 1063–1072. [Google Scholar] [CrossRef]
- Michel, D.; Casey Gaunt, M.; Arnason, T.; El-Aneed, A. Development and validation of fast and simple flow injection analysis-tandem mass spectrometry (FIA-MS/MS) for the determination of metformin in dog serum. J. Pharm. Biomed. Anal. 2015, 107, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Swales, J.G.; Gallagher, R.; Peter, R.M. Determination of metformin in mouse, rat, dog and human plasma samples by laser diode thermal desorption/atmospheric pressure chemical ionization tandem mass spectrometry. J. Pharm. Biomed. Anal. 2010, 53, 740–744. [Google Scholar] [CrossRef] [PubMed]
- Kiran, C.; Jianmei, W.; Yong, X.; Ali, W.; Linshu, W.; Xiaowei, D.; Eric, Y.C.; Ran, L.; Shao-Hua, Y. Determination of metformin bio-distribution by LC-MS/MS in mice treated with a clinically relevant paradigm. PLoS ONE 2020, 15, e0234571. [Google Scholar]
- Zhang, W.; Han, F.; Zhao, H.; Lin, Z.J.; Huang, Q.M.; Weng, N. Determination of metformin in rat plasma by HILIC-MS/MS combined with Tecan automation and direct injection. Biomed. Chromatogr. 2012, 26, 1163–1169. [Google Scholar] [CrossRef]
- Uçaktürk, E. The development and validation of a gas chromatography-mass spectrometry method for the determination of metformin in human plasma. Anal. Methods 2013, 5, 4723–4730. [Google Scholar] [CrossRef]
- Balamurugan, K.; Kirtimaya, M.; Suresh, R. Simultaneous estimation of linagliptin and metformin HCl in human plasma by RP-HPLC method. Int. Res. J. Pharm. 2019, 10, 167–170. [Google Scholar]
- Shakoor, A.; Adnan, A.; Ahmed, M. Simultaneous determination of metformin and vildagliptin by HPLC in human plasma: Application to pharmacokinetic studies. Lat. Am. J. Pharm. 2019, 38, 1416–1423. [Google Scholar]
- Sebaiy, M.M.; El-Adl, S.M.; Baraka, M.M.; Hassan, A.A. Rapid RP-HPLC method for simultaneous estimation of metformin, pioglitazone, and glimepiride in human plasma. Acta Chromatogr. 2020, 32, 16–21. [Google Scholar] [CrossRef]
- Ranetti, M.C.; Ionescu, M.; Hinescu, L.; Ionică, E.; Anuţa, V.; Ranetti, A.E.; Stecoza, C.E.; Mircioiu, C. Validation of a HPLC method for the simultaneous analysis of metformin and gliclazide in human plasma. Farmacia 2009, 57, 728–735. [Google Scholar]
- Yardimci, C.; Ozaltin, N.; Gurlek, A. Simultaneous determination of rosiglitazone and metformin in plasma by gradient liquid chromatography with UV detection. Talanta 2007, 72, 1416–1422. [Google Scholar] [CrossRef] [PubMed]
- Shakoora, A.; Ahmedb, M.; Ikramc, R.; Hussaina, S.; Tahird, A.; Janc, B.M.; Adnan, A. Stability-indicating RP-HPLC method for simultaneous determination of metformin hydrochloride and vildagliptin in tablet and biological samples. Acta Chromatogr. 2020, 32, 39–43. [Google Scholar] [CrossRef]
- Elgawish, M.S.; Nasser, S.; Salama, I.; Abbasc, A.M.; Mostafaa, S.M. Liquid chromatography tandem mass spectrometry for the simultaneous determination of metformin and pioglitazone in rat plasma: Application to pharmacokinetic and drug-drug interaction studies. J. Chromatogr. B 2019, 1124, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.Y.; Peng, Y.; Wan, P.; Yin, L.; Wang, G.J.; Sun, J.G. Simultaneous determination and pharmacokinetic study of metformin and pioglitazone in dog plasma by LC-MS-MS. J. Chromatogr. Sci. 2014, 52, 52–58. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jagadeesh, B.; Bharathi, D.V.; Pankaj, C.; Narayana, V.S.; Venkateswarulu, V. Development and validation of highly selective and robust method for simultaneous estimation of pioglitazone, hydroxypioglitazone and metformin in human plasma by LC-MS/MS: Application to a pharmacokinetic study. J. Chromatogr. B 2013, 930, 136–145. [Google Scholar] [CrossRef]
- Polagani, S.R.; Pilli, N.R.; Gajula, R.; Gandu, V. Simultaneous determination of atorvastatin, metformin and glimepiride in human plasma by LC–MS/MS and its application to a human pharmacokinetic study. J. Pharm. Anal. 2013, 3, 9–19. [Google Scholar] [CrossRef] [Green Version]
- Li, N.; Deng, Y.; Qin, F.; Yu, J.; Li, F. Simultaneous quantification of metformin and glipizide in human plasma by high-performance liquid chromatography-tandem mass spectrometry and its application to a pharmacokinetic study. Biomed. Chromatogr. 2013, 27, 191–196. [Google Scholar] [CrossRef]
- Mowaka, S.; Elkady, E.F.; Elmazar, M.M.; Ayoub, B. Enhanced LC-MS/MS determination of alogliptin and metformin in plasma: Application to a pharmacokinetic study. Microchem. J. 2017, 130, 360–365. [Google Scholar] [CrossRef]
- Mohamed, D.; Elshahed, M.S.; Aboutaleb, T.N.; Zakaria, N.O. Novel LC–MS/MS method for analysis of metformin and canagliflozin in human plasma: Application to a pharmacokinetic study. BMC Chem. 2019, 13, 82–92. [Google Scholar] [CrossRef]
- Wattamwar, T.; Mungantiwar, A.; Halde, S.; Pandita, N. Development of Simultaneous Determination of Empagliflozin and Metformin in Human Plasma Using Liquid Chromatography—Mass Spectrometry and Application to Pharmacokinetics. Eur. J. Mass Spectrom. 2020, 26, 117–130. [Google Scholar] [CrossRef]
- Mohamed, A.M.I.; Mohamed, F.A.; Ahmed, S.; Mohamed, Y.A. An efficient hydrophilic interaction liquid chromatographic method for the simultaneous determination of metformin and pioglitazone using high-purity silica column. J. Chromatogr. B 2015, 997, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Pontarolo, R.; Gimenez, A.C.; Francisco, T.M.G.; Ribeiro, R.P.; Pontes, F.L.D.; Gasparetto, J.C. Simultaneous determination of metformin and vildagliptin in human plasma by a HILIC-MS/MS method. J. Chromatogr. B 2014, 965, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Abdelwahab, N.S.; Mokhtar, H.M.; Mostafa, Y.A.; Morsi, A.; Aboul Magd, A.M. Ecological HPLC method for analysis of anti-diabetic drug in real rat plasma samples; studying the effect of concurrent administered fenugreek extract on its pharmacokinetics. RSC Adv. 2021, 11, 4740–4750. [Google Scholar] [CrossRef]
- Francisco, P.; Wojciech, W.; Marek, T. AGREE—Analytical GREEnness metric approach and software. Anal. Chem. 2020, 92, 10076–10082. [Google Scholar]
- Paweł, M.N.; Paweł, K. What color is your method? Adaptation of the RGB Additive Color Model to analytical method evaluation. Anal. Chem. 2019, 91, 10343–10352. [Google Scholar]
- Sureshkumar, D.; Begum, S.; Johannah, N.M.; Maliakel, B.; Krishnakumar, I.M. Toxicological evaluation of a saponin-rich standardized extract of fenugreek seeds (FenuSMART®): Acute, sub-chronic and genotoxicity studies. Tox. Rep. 2018, 5, 1060–1068. [Google Scholar] [CrossRef] [PubMed]
- Boyea, A.; Acheampongb, D.O.; Gyamerahc, E.O.; Asiamahd, E.A.; Addoe, J.K.; Mensaha, D.A.; Brahb, A.S.; Ayikua, P.J. Glucose lowering and pancreato-protective effects of Abrus Precatorius (L.) leaf extract in normoglycemic and STZ/Nicotinamide—Induced diabetic rats. J. Ethnopharmacol. 2020, 258, 112918. [Google Scholar] [CrossRef]
- Fares, M.Y.; Abdelwahab, N.S.; Abdelrahman, M.M.; Abdel-Rahman, H.M. Determination of sofosbuvir with two co-administered drugs; paracetamol and DL-methionine by two chromatographic methods. Application to a pharmacokinetic study. Bioanalysis 2019, 11, 349–364. [Google Scholar] [CrossRef]
- Zhang, Y.; Huo, M.; Zhou, J.; Xie, S. An add-in program for pharmacokinetic and pharmacodynamic data analysis in Microsoft Excel. Comp. Methods Prog. Biomed. 2010, 99, 306–314. [Google Scholar] [CrossRef]

| Solvent | Acetonitrile | Ethanol | Methanol | Aqueous Perchloric Acid |
|---|---|---|---|---|
| Extraction recovery | 78.45 ± 7.75 | 82.10 ± 6.74 | 87.56 ± 5.32 | 80.98 ± 2.98 |
| Concentration (µg mL−1) * | Intraday | Interday | ||||
|---|---|---|---|---|---|---|
| Recovery % | %RSD | %RE ** | Recovery % | %RSD | %RE ** | |
| 2.00 (LQC) | 100.99 | 0.27 | 0.99 | 100.30 | 0.57 | 0.30 |
| 15.00 (MQC) | 93.18 | 1.60 | −6.82 | 94.44 | 4.72 | −5.56 |
| 28.00 (HQC) | 99.30 | 0.08 | −0.70 | 99.30 | 0.15 | −0.70 |
| Concentration of the Analyte (µg mL−1) | Recovery % * | |
|---|---|---|
| 2.00 | 87.56 | |
| 15.00 | 82.90 | |
| 28.00 | 92.22 | |
| Mean ± %RSD | 87.56 ± 5.32 | |
| IS | 50.00 | 90.51 ± 3.82 |
| Recovery % * | ||||
|---|---|---|---|---|
| Concentration of the Analyte (µg mL−1) | Three Freeze Thaw Cycles | Bench Top Stability | Short Term Stability for 24 h | |
| 2.00 | 86.48 | 92.27 | 99.13 | |
| 15.00 | 101.17 | 99.36 | 113.77 | |
| 28.00 | 97.44 | 99.82 | 112.00 | |
| Mean ± %RSD | 95.03 ± 8.04 | 97.15 ± 4.35 | 108.30 ± 7.38 | |
| Parameter | Unit | Metformin (300 mg/kg) | Metformin + Papaya Extract 300 + 500 (mg/kg) |
|---|---|---|---|
| T1/2 | h | 2.95 | 3.30 |
| Tmax | h | 1.00 | 0.25 |
| Cmax | µg mL−1 | 15.14 | 16.25 |
| AUC0- | µg mL−1 h | 42.82 | 33.01 |
| AUC0-inf | µg mL−1 h | 52.60 | 46.34 |
| Mean residence time (MRT) | h | 4.01 | 5.18 |
| Volume of distribution (Vd/F) | L | 24.25 | 30.83 |
| Clearance (Cl/F) | L/h | 5.70 | 6.47 |
| Accuracy (Bias%) | Precision (%RSD) | Extraction Efficiency | Sensitivity | Selectivity | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| REDNESS (analytical performance) | W = 1 | |||||||||||
| CS: | 67.1% | LAV = 33.3 | 15 | 15 | 15 | 15 | 75 | 75 | L | L | L | L |
| LSV = 66.6 | 3 | 3 | 3 | 3 | 90 | 90 | H | H | H | H | ||
| Result | 2.51 | 2.51 | 1.23 | 1.23 | 87.5 | 87.5 | M | M | VH | VH | ||
| Score (0–100) | 70 | 70 | 75 | 75 | 65 | 65 | 50 | 50 | 80 | 80 | ||
| Amount of chemicals | Toxicity | Waste | Occupational hazards | Energy | ||||||||
| GREENNESS (safety and eco-friendliness) | W = 1 | |||||||||||
| CS: | 70.4% | LAV = 33.3 | 50 | 50 | 10 pictograms in total | 10 pictograms in total | 20 | 20 | H | H | 1.5 | 1.5 |
| LSV = 66.6 | 15 | 15 | 5 pictograms in total | 5 pictograms in total | 10 | 10 | L | L | <1.5 | <1.5 | ||
| Result | 12.75 | 12.75 | 3 pictograms in total | 3 pictograms in total | 12.75 | 12.75 | no | no | <1.5 | <1.5 | ||
| Score (0–100) | 70 | 70 | 75 | 75 | 55 | 55 | 90 | 90 | 66.6 | 66.6 | ||
| Analysis cost | Sample throughout | Sample material consumption | ||||||||||
| BLUENESS (productivity/practical effectiveness) | W = 1 | |||||||||||
| CS: | 64.6% | LAV = 33.3 | H | H | H | H | 5 samples/h | 5 samples/h | 5 samples/h | H | H | H |
| LSV = 66.6 | M | M | M | M | 15 samples/h | 15 samples/h | 15 samples/h | M | M | M | ||
| Result | M | M | M | M | 8 sample/h | 8 sample/h | 8 sample/h | VL | VL | VL | ||
| Score (0–100) | 66.66 | 66.66 | 66.66 | 66.66 | 50 | 50 | 50 | 80 | 80 | 80 | ||
| FINAL COLOR: | REDNESS | GREENNESS | BLUENESS | BRILLIANCE (MB): | 67.3% | |||||||
| YELLOW | ≥33.3% | ≥66.6% | ≥33.3% | ≥66.6% | ≥33.3% | ≥66.6% | ||||||
| yes | yes | yes | yes | yes | no | |||||||
| Short annotation: 67.3 yellow | Long annotation: 67.3 yellow (67.1/1 red − 70.4/1 green − 64.6/1 blue) | |||||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdelgawad, M.A.; Elmowafy, M.; Musa, A.; Al-Sanea, M.M.; Nayl, A.A.; Ghoneim, M.M.; Ahmed, Y.M.; Hassan, H.M.; AboulMagd, A.M.; Salem, H.F.; et al. Development and Greenness Assessment of HPLC Method for Studying the Pharmacokinetics of Co-Administered Metformin and Papaya Extract. Molecules 2022, 27, 375. https://doi.org/10.3390/molecules27020375
Abdelgawad MA, Elmowafy M, Musa A, Al-Sanea MM, Nayl AA, Ghoneim MM, Ahmed YM, Hassan HM, AboulMagd AM, Salem HF, et al. Development and Greenness Assessment of HPLC Method for Studying the Pharmacokinetics of Co-Administered Metformin and Papaya Extract. Molecules. 2022; 27(2):375. https://doi.org/10.3390/molecules27020375
Chicago/Turabian StyleAbdelgawad, Mohamed A., Mohammed Elmowafy, Arafa Musa, Mohammad M. Al-Sanea, AbdElAziz A. Nayl, Mohammed M. Ghoneim, Yasmine M. Ahmed, Hossam M. Hassan, Asmaa M. AboulMagd, Heba F. Salem, and et al. 2022. "Development and Greenness Assessment of HPLC Method for Studying the Pharmacokinetics of Co-Administered Metformin and Papaya Extract" Molecules 27, no. 2: 375. https://doi.org/10.3390/molecules27020375






