Investigation of a Complex Reaction Pathway Network of Isobutane/2-Butene Alkylation by CGC–FID and CGC-MS-DS
Abstract
:1. Introduction
2. Results and Discussion
2.1. Optimization of the CGC-FID Analysis Conditions
2.2. Quantitative Analysis Results of the Peak Area Normalization Method
2.2.1. Relative Correction Factors
2.2.2. Comparison of the Peak Area Normalization Method and the Corrected Peak Area Normalization Method
2.2.3. Precision
2.2.4. Recovery
2.3. Chemical Composition of the C4 Alkylation Products
2.4. Alkylation Reaction Pathway Network
2.4.1. Changes in the Composition of the Reaction Process
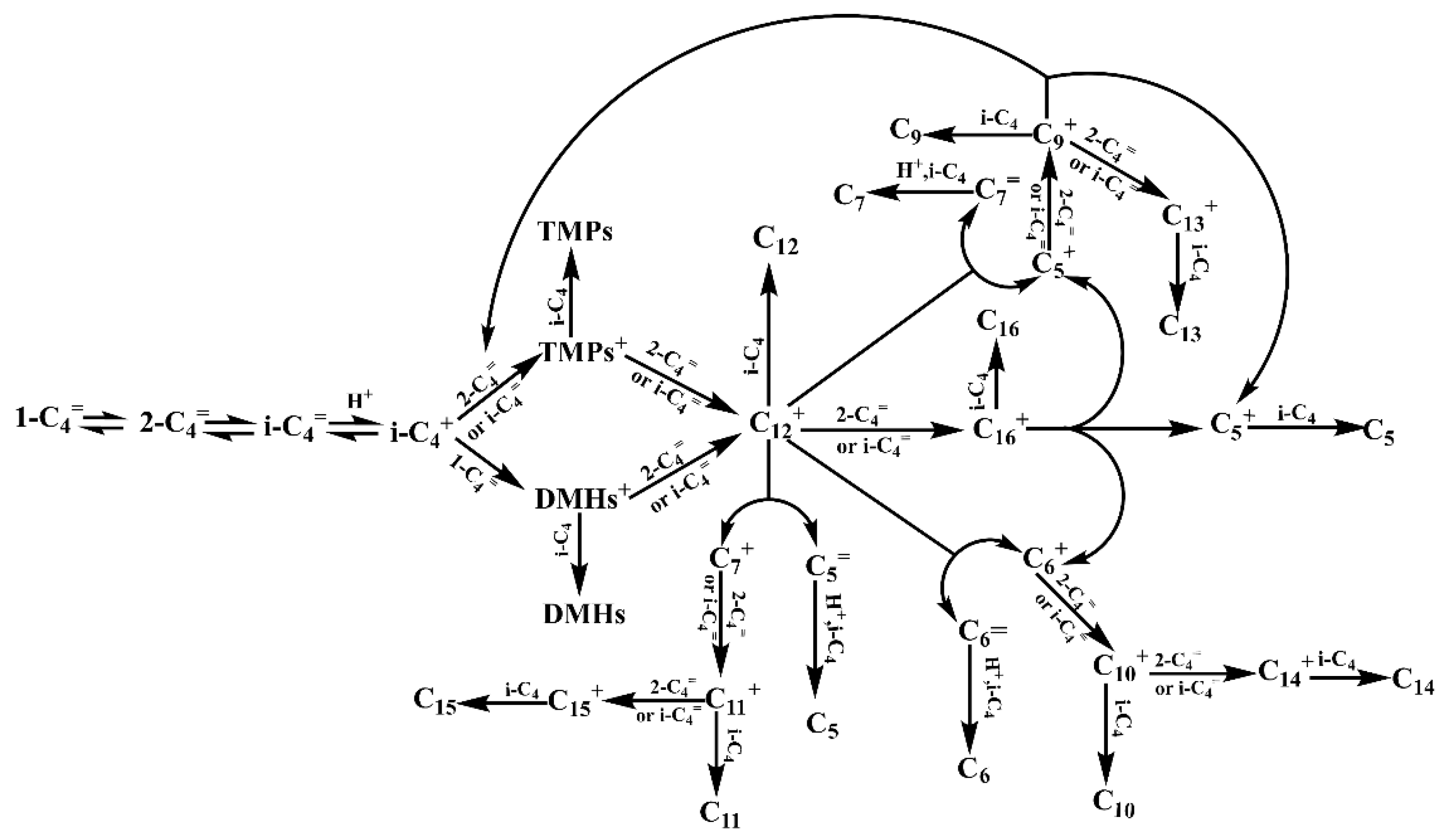
2.4.2. Reaction Pathway Network Construction
3. Experimental Section
3.1. Materials
3.2. Experimental Equipment
3.3. Quantitative Analysis
3.3.1. CGC-FID Analysis Conditions
3.3.2. Determination of the Relative Correction Factors
3.3.3. Determining the Mass% of the SAMPLE Components
3.4. Qualitative Analysis
CGC-MS-DS Analysis Conditions
3.5. C4 Alkylation Reaction
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Singhal, S.; Agarwal, S.; Singh, M.; Rana, S.; Arora, S.; Singhal, N. Ionic liquids: Green catalysts for alkene-isoalkane alkylation. J. Mol. Liq. 2019, 285, 299–313. [Google Scholar] [CrossRef]
- Zheng, W.; Wang, H.; Xie, W.; Zhao, L.; Sun, W. Understanding interfacial behaviors of isobutane alkylation with C4 olefin catalyzed by sulfuric acid or ionic liquids. AIChE J. 2018, 64, 950–960. [Google Scholar] [CrossRef]
- Zheng, W.; Wang, Z.; Sun, W.; Zhao, L.; Qian, F. H2SO4-catalyzed isobutane alkylation under low temperatures promoted by long-alkyl-chain surfactant additives. AIChE J. 2021, 67, e17349. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, T.; Yang, Y.; Chen, S.; Lv, L.; Tang, S. Intensification of isobutane/1-butene alkylation process in a micromixing microreactor catalyzed by ILs/H2SO4. Chem. Eng. Process. Process Intensif. 2022, 174, 108865. [Google Scholar] [CrossRef]
- Hommeltoft, S.I. Isobutane alkylation: Recent developments and future perspectives. Appl. Catal. A Gen. 2001, 221, 421–428. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, T.; Meng, X.; Wang, H.; Zhang, Y.; Wang, H.; Zhang, S. Improvement of product distribution through enhanced mass transfer in isobutane/butene alkylation. Chem. Eng. Res. Des. 2019, 143, 190–200. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, T.; Yang, Y.; Tang, S. Simulation and design microreactor configured with micromixers to intensify the isobutane/1-butene alkylation process. J. Taiwan Inst. Chem. Eng. 2019, 98, 53–62. [Google Scholar] [CrossRef]
- Gan, P.; Tang, S. Research progress in ionic liquids catalyzed isobutane/butene alkylation. Chin. J. Chem. Eng. 2016, 24, 1497–1504. [Google Scholar] [CrossRef]
- Akhmadova, K.K.; Magomadova, M.K.; Syrkin, A.M.; Egutkin, N.L. History, Current State, and Prospects for Development of Isobutane Alkylation with Olefins. Theor. Found. Chem. Eng. 2019, 53, 643–655. [Google Scholar] [CrossRef]
- Kim, D.; Andersen, M.E.; Pleil, J.D.; Nylander-French, L.A.; Prah, J.D. Refined PBPK model of aggregate exposure to methyl tertiary-butyl ether. Toxicol. Lett. 2007, 169, 222–235. [Google Scholar] [CrossRef]
- Prah, J.; Ashley, D.; Blount, B.; Case, M.; Leavens, T.; Pleil, J.; Cardinali, F. Dermal, Oral, and Inhalation Pharmacokinetics of Methyl Tertiary Butyl Ether (MTBE) in Human Volunteers. Toxicol. Sci. 2004, 77, 195–205. [Google Scholar] [CrossRef] [Green Version]
- Silva, L.K.; Espenship, M.F.; Pine, B.N.; Ashley, D.L.; De Jesús, V.R.; Blount, B.C. Methyl Tertiary-Butyl Ether Exposure from Gasoline in the U.S. Population, NHANES 2001–2012. Environ. Health Perspect. 2019, 127, 127003. [Google Scholar] [CrossRef]
- Li, L.; Zhang, J.; Wang, K.; Luo, G. Caprolactam as a New Additive to Enhance Alkylation of Isobutane and Butene in H2SO4. Ind. Eng. Chem. Res. 2016, 55, 12818–12824. [Google Scholar] [CrossRef]
- Li, L.; Zhang, J.; Du, C.; Wang, K.; Luo, G. Kinetics Study of Sulfuric Acid Alkylation of Isobutane and Butene Using a Microstructured Chemical System. Ind. Eng. Chem. Res. 2018, 58, 1150–1158. [Google Scholar] [CrossRef]
- Ivashkina, E.; Ivanchina, E.; Dolganov, I.; Chuzlov, V.; Kotelnikov, A.; Dolganova, I.; Khakimov, R. Nonsteady-state mathematical modelling of H2SO4-catalysed alkylation of isobutane with alkenes. Oil Gas Sci. Technol. Rev. d’IFP Energies Nouv. 2021, 76, 36. [Google Scholar] [CrossRef]
- Höpfl, V.B.; Schachtl, T.; Liu, Y.; Lercher, J.A. Pellet Size-Induced Increase in Catalyst Stability and Yield in Zeolite-Catalyzed 2-Butene/Isobutane Alkylation. Ind. Eng. Chem. Res. 2022, 61, 330–338. [Google Scholar] [CrossRef]
- Huang, Q.; Zhao, G.; Zhang, S.; Yang, F. Improved Catalytic Lifetime of H2SO4 for Isobutane Alkylation with Trace Amount of Ionic Liquids Buffer. Ind. Eng. Chem. Res. 2015, 54, 1464–1469. [Google Scholar] [CrossRef]
- Wu, C.; Chen, X.; Fu, J.; Zhou, J.; Liang, J.; Wei, X.; Wang, L. ZIF-derived Co/NCNTs as a superior catalyst for aromatic hydrocarbon resin hydrogenation: Scalable green synthesis and insight into reaction mechanism. Chem. Eng. J. 2022, 443, 136193. [Google Scholar] [CrossRef]
- Schmerling, L. The Mechanism of the Alkylation of Paraffins. II. Alkylation of Isobutane with Propene, 1-Butene and 2-Butene. J. Am. Chem. Soc. 1946, 68, 275–281. [Google Scholar] [CrossRef]
- Mosby, J.F.; Albright, L.F. Alkylation of Isobutane with 1-Butene Using Sulfuric Acid as Catalyst at High Rates of Agitation. I&EC Prod. Res. Dev. 1966, 5, 183–190. [Google Scholar] [CrossRef]
- Li, K.W.; Eckert, R.E.; Albright, L.F. Alkylation of Isobutane with Light Olefins Using Sulfuric Acid. Operating Variables Affecting Physical Phenomena Only. Ind. Eng. Chem. Process Des. Dev. 1970, 9, 434–440. [Google Scholar] [CrossRef]
- Langley, J.R.; Pike, R.W. The kinetics of alkylation of isobutane with propylene. AIChE J. 1972, 18, 698–705. [Google Scholar] [CrossRef]
- Sun, W.; Shi, Y.; Chen, J.; Xi, Z.; Zhao, L. Alkylation Kinetics of Isobutane by C4 Olefins Using Sulfuric Acid as Catalyst. Ind. Eng. Chem. Res. 2013, 52, 15262–15269. [Google Scholar] [CrossRef]
- Likić, V.A. Extraction of pure components from overlapped signals in gas chromatography-mass spectrometry (GC-MS). BioData Min. 2009, 2, 6–11. [Google Scholar] [CrossRef] [Green Version]
- Bhardwaj, S.K.; Dwivedi, K.; Agarwal, D. A review: GC method development and validation. Int. J. Anal. Bioanal. Chem 2016, 6, 1–7. [Google Scholar]
- Santana, G.M.; Akgerman, A. Alkylation of Isobutane with 1-Butene on a Solid Acid Catalyst in Supercritical Reaction Media. Ind. Eng. Chem. Res. 2001, 40, 3879–3882. [Google Scholar] [CrossRef]
- Tammekivi, E.; Vahur, S.; Kekišev, O.; van der Werf, I.D.; Toom, L.; Herodes, K.; Leito, I. Comparison of derivatization methods for the quantitative gas chromatographic analysis of oils. Anal. Methods 2019, 11, 3514–3522. [Google Scholar] [CrossRef]
- Cao, Y.; Zhu, S.; Zhang, L.; Cui, Q.; Wang, H. Qualitative and quantitative determination of trace aldehydes and ketones in food preservative propionic acid for quality improvement. Anal. Methods 2021, 13, 2989–2996. [Google Scholar] [CrossRef] [PubMed]
- Bollenbach, A.; Baskal, S.; Mels, C.; Kruger, R.; Tsikas, D. Unusual Derivatization of Methylmalonic Acid with Pentafluorobenzyl Bromide to a Tripentafluorobenzyl Derivative and Its Stable-Isotope Dilution GC-MS Measurement in Human Urine. Molecules 2022, 27, 5202. [Google Scholar] [CrossRef] [PubMed]
- Schmerling, L. The Mechanism of the Alkylation of Paraffins. J. Am. Chem. Soc. 1945, 67, 1778–1783. [Google Scholar] [CrossRef]
- Hofmann, J.E.; Schriesheim, A. Ionic Reactions Occurring During Sulfuric Acid Catalyzed Alkylation. II. Alkylation of Isobutane with C14-Labeled Butenes. J. Am. Chem. Soc. 1962, 84, 957–961. [Google Scholar] [CrossRef]
- Schmerling, L. Reactions of Hydrocarbons. Ind. Eng. Chem. 1953, 45, 1447–1455. [Google Scholar] [CrossRef]
- Cao, P.; Zheng, L.; Sun, W.; Zhao, L. Multiscale Modeling of Isobutane Alkylation with Mixed C4 Olefins Using Sulfuric Acid as Catalyst. Ind. Eng. Chem. Res. 2019, 58, 6340–6349. [Google Scholar] [CrossRef]
- Liang, L.; Liu, Y.; Jiao, W.; Zhang, Q.; Zhang, C. Octane compositions in sulfuric acid catalyzed isobutane/butene alkylation products: Experimental and quantum chemistry studies. Front. Chem. Sci. Eng. 2021, 15, 1229–1242. [Google Scholar] [CrossRef]
- Chen, Z.; Gao, F.; Ren, K.; Wu, Q.; Luo, Y.; Zhou, H.; Zhang, M.; Xu, Q. Mechanism of byproducts formation in the isobutane/butene alkylation on HY zeolites. RSC Adv. 2018, 8, 3392–3398. [Google Scholar] [CrossRef] [Green Version]
- Boronat, M.; Viruela, P.; Corma, A. Theoretical study of the mechanism of branching rearrangement of carbenium ions. Appl. Catal. A Gen. 1996, 146, 207–223. [Google Scholar] [CrossRef]
- Albright, L.F.; Goldsby, A.R. Industrial and Laboratory Alkylations; American Chemical Society: Washington, DC, USA, 1977; p. 0841203857. [Google Scholar]
- Liu, G.; Wu, G.; Liu, Y.; Hu, R.; Gao, G. Theoretical study on the C4 alkylation mechanism catalyzed by Cu-containing chloroaluminate ionic liquids. Fuel 2022, 310, 122379. [Google Scholar] [CrossRef]



| Component | Relative Correction Factors (fis) | |||
|---|---|---|---|---|
| 1 | 2 | 3 | Mean Value | |
| 2,3-Dimethylbutane | 1.0381 | 1.0243 | 1.0040 | 1.02 |
| 2-Methylpentane | 1.0193 | 1.0236 | 1.0165 | 1.02 |
| 3-Methylpentane | 1.0121 | 0.9946 | 1.0057 | 1.00 |
| 2,4-Dimethylpentane | 1.0048 | 0.9717 | 1.0044 | 0.99 |
| 2,2,3-Trimethylbutane | 1.0402 | 1.0541 | 1.0479 | 1.05 |
| 2-Methylhexane | 0.9846 | 0.9856 | 1.0098 | 0.99 |
| 2,3-Dimethylpentane | 0.9939 | 0.9943 | 0.9846 | 0.99 |
| 3-Methylhexane | 1.0581 | 1.0351 | 1.0566 | 1.05 |
| 2,2,4-Trimethylpentane | 1.0000 | 1.0000 | 1.0000 | 1.00 |
| 2,4-Dimethylhexane | 1.0022 | 1.0076 | 0.9938 | 1.00 |
| 2,2,3-Trimethylpentane | 1.0070 | 1.0465 | 1.0610 | 1.04 |
| 2,3,4-Trimethylpentane | 1.0074 | 1.0017 | 1.0399 | 1.02 |
| 2,3-Dimethylhexane | 1.0389 | 1.0839 | 1.0848 | 1.07 |
| 3,4-Dimethylhexane | 1.0013 | 1.0356 | 1.0518 | 1.03 |
| 2,2,5-Trimethylhexane | 0.9701 | 0.9923 | 1.0096 | 0.99 |
| Component | Serial Number | Initial Concentration (wt%) | Concentration Measurement for the Quantitative Analytical Method | |||
|---|---|---|---|---|---|---|
| Peak Area Normalization (wt%) | RD (%) | Corrected Peak Area Normalization (wt%) | RD (%) | |||
| 2,3-Dimethylbutane | 1 | 3.5872 | 3.5260 | 1.71 | 3.5563 | 1.02 |
| 2 | 8.0537 | 7.9519 | 1.26 | 8.0048 | 0.61 | |
| 3 | 4.4130 | 4.3533 | 1.35 | 4.3821 | 0.70 | |
| 2-Methylpentane | 1 | 5.6946 | 5.6813 | 0.23 | 5.7167 | 0.15 |
| 2 | 6.6314 | 6.6641 | 0.49 | 6.6959 | 0.93 | |
| 3 | 5.2425 | 5.1777 | 1.24 | 5.1999 | 0.81 | |
| 3-Methylpentane | 1 | 7.1512 | 7.0880 | 0.88 | 7.0226 | 1.99 |
| 2 | 5.0339 | 5.0358 | 0.04 | 5.0005 | 1.02 | |
| 3 | 6.7699 | 6.7055 | 0.95 | 6.6308 | 2.06 | |
| 2,4-Dimethylpentane | 1 | 4.9740 | 5.0375 | 1.28 | 4.9389 | 0.85 |
| 2 | 5.9555 | 5.9559 | 0.01 | 5.8615 | 2.14 | |
| 3 | 8.5898 | 8.6865 | 1.13 | 8.4999 | 1.05 | |
| 2,2,3-Trimethylbutane | 1 | 4.1528 | 4.1006 | 1.26 | 4.2379 | 1.94 |
| 2 | 5.1249 | 5.0591 | 1.28 | 5.1933 | 1.83 | |
| 3 | 8.1348 | 8.0010 | 1.64 | 8.2529 | 1.45 | |
| 2-Methylhexane | 1 | 7.1202 | 7.1621 | 0.59 | 7.0197 | 1.49 |
| 2 | 4.3302 | 4.4232 | 2.15 | 4.3520 | 0.08 | |
| 3 | 6.4961 | 6.6022 | 1.63 | 6.4584 | 0.58 | |
| 2,3-Dimethylpentane | 1 | 7.0737 | 7.1637 | 1.27 | 7.0045 | 1.04 |
| 2 | 8.3446 | 8.4279 | 1.00 | 8.2767 | 1.44 | |
| 3 | 7.5703 | 7.7102 | 1.85 | 7.5242 | 0.61 | |
| 3-Methylhexane | 1 | 5.3537 | 5.2108 | 2.67 | 5.3983 | 0.78 |
| 2 | 9.7008 | 9.5368 | 1.69 | 9.8088 | 1.65 | |
| 3 | 8.3076 | 8.1247 | 2.20 | 8.4007 | 1.12 | |
| 2,2,4-Trimethylpentane | 1 | 13.6205 | 13.8235 | 1.49 | 13.6397 | 0.17 |
| 2 | 9.7481 | 9.8102 | 0.64 | 9.7042 | 0.89 | |
| 3 | 11.6904 | 11.8166 | 1.08 | 11.6369 | 0.46 | |
| 2,4-Dimethylhexane | 1 | 7.5308 | 7.7190 | 2.50 | 7.6254 | 0.95 |
| 2 | 12.0867 | 12.1274 | 0.34 | 12.0074 | 1.07 | |
| 3 | 5.2154 | 5.3102 | 1.82 | 5.2357 | 0.39 | |
| 2,2,3-Trimethylpentane | 1 | 7.0814 | 6.9541 | 1.80 | 7.1235 | 1.17 |
| 2 | 3.9665 | 3.9703 | 0.10 | 4.0466 | 2.34 | |
| 3 | 3.6356 | 3.5926 | 1.18 | 3.6729 | 1.03 | |
| 2,3,4-Trimethylpentane | 1 | 7.9879 | 7.9515 | 0.14 | 7.9739 | 0.06 |
| 2 | 5.3991 | 5.4484 | 0.91 | 5.4595 | 1.00 | |
| 3 | 5.9442 | 5.9357 | 0.46 | 5.9409 | 0.04 | |
| 2,3-Dimethylhexane | 1 | 4.3620 | 4.2262 | 3.11 | 4.4585 | 2.30 |
| 2 | 4.3818 | 4.2664 | 2.63 | 4.4528 | 2.52 | |
| 3 | 5.9779 | 5.8033 | 2.92 | 6.1105 | 2.22 | |
| 3,4-Dimethylhexane | 1 | 6.5313 | 6.5465 | 0.28 | 6.6504 | 1.10 |
| 2 | 5.0972 | 5.0845 | 0.25 | 5.1479 | 1.14 | |
| 3 | 4.4632 | 4.4506 | 0.23 | 4.5125 | 1.95 | |
| 2,2,5-Trimethylhexane | 1 | 7.7787 | 7.8093 | 0.39 | 7.6336 | 1.69 |
| 2 | 6.1456 | 6.2382 | 1.51 | 6.1248 | 0.97 | |
| 3 | 7.5493 | 7.7298 | 2.39 | 7.5412 | 0.11 | |
| Component | Measured Value (wt%) | Mean Value (wt%) | RSD (%) | ||||
|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | |||
| 2,3-Dimethylbutane | 4.7289 | 4.6799 | 4.6630 | 4.6613 | 4.6298 | 4.6726 | 0.78 |
| 2-Methylpentane | 7.7475 | 7.6278 | 7.6606 | 7.6680 | 7.6258 | 7.6659 | 0.64 |
| 3-Methylpentane | 3.5913 | 3.5486 | 3.5586 | 3.5540 | 3.5390 | 3.5583 | 0.56 |
| 2,4-Dimethylpentane | 6.2783 | 6.2489 | 6.2517 | 6.2546 | 6.2558 | 6.2578 | 0.19 |
| 2,2,3-Trimethylbutane | 3.9137 | 3.8916 | 3.9033 | 3.9071 | 3.9019 | 3.9035 | 0.21 |
| 2-Methylhexane | 6.8789 | 6.8749 | 6.8655 | 6.8750 | 6.8775 | 6.8743 | 0.08 |
| 2,3-Dimethylpentane | 6.4327 | 6.4289 | 6.4326 | 6.4426 | 6.4411 | 6.4356 | 0.09 |
| 3-Methylhexane | 10.9570 | 10.9688 | 10.9536 | 10.9686 | 10.9730 | 10.9642 | 0.08 |
| 2,2,4-Trimethylpentane | 23.2735 | 23.3669 | 23.3195 | 23.3433 | 23.3566 | 23.3320 | 0.16 |
| 2,4-Dimethylhexane | 4.3548 | 4.3370 | 4.3977 | 4.3747 | 4.3748 | 4.3678 | 0.53 |
| 2,2,3-Trimethylpentane | 2.9461 | 2.9966 | 2.9515 | 2.9656 | 2.9797 | 2.9679 | 0.70 |
| 2,3,4-Trimethylpentane | 5.8892 | 5.9298 | 5.9284 | 5.9132 | 5.9313 | 5.9184 | 0.30 |
| 2,3-Dimethylhexane | 6.1666 | 6.2031 | 6.2007 | 6.1866 | 6.2028 | 6.1919 | 0.25 |
| 3,4-Dimethylhexane | 2.6111 | 2.6291 | 2.6386 | 2.6291 | 2.6371 | 2.6290 | 0.42 |
| 2,2,5-Trimethylhexane | 4.2305 | 4.2682 | 4.2747 | 4.2564 | 4.2741 | 4.2608 | 0.43 |
| Component | Initial Value (g) | Spiked Value (g) | Measured Value (g) | Recovery (%) |
|---|---|---|---|---|
| 2,3-Dimethylbutane | 0.1488 | 0.1091 | 0.2563 | 98.53 |
| 2-Methylpentane | 0.0926 | 0.0637 | 0.1555 | 98.74 |
| 3-Methylpentane | 0.0933 | 0.0946 | 0.1868 | 98.76 |
| 2,4-Dimethylpentane | 0.1085 | 0.1571 | 0.2688 | 102.05 |
| 2,2,3-Trimethylbutane | 0.1404 | 0.0613 | 0.2012 | 99.22 |
| 2-Methylhexane | 0.1179 | 0.1320 | 0.2498 | 99.86 |
| 2,3-Dimethylpentane | 0.1277 | 0.1836 | 0.3129 | 100.85 |
| 3-Methylhexane | 0.1163 | 0.1526 | 0.2670 | 98.76 |
| 2,2,4-Trimethylpentane | 0.1916 | 0.1208 | 0.3136 | 100.99 |
| 2,4-Dimethylhexane | 0.0746 | 0.0814 | 0.1584 | 102.85 |
| 2,2,3-Trimethylpentane | 0.0553 | 0.0277 | 0.0836 | 101.97 |
| 2,3,4-Trimethylpentane | 0.0883 | 0.0773 | 0.1647 | 98.86 |
| 2,3-Dimethylhexane | 0.0759 | 0.0755 | 0.1505 | 98.77 |
| 3,4-Dimethylhexane | 0.1003 | 0.0739 | 0.1731 | 98.53 |
| 2,2,5-Trimethylhexane | 0.0895 | 0.0928 | 0.1825 | 100.20 |
| Serial Number | Retention Time (min) | Peak Area (%) | Similarity (%) | Alkylate Components | Molecular Weight | Molecular Formula |
|---|---|---|---|---|---|---|
| 1 | 7.150 | 0.4114 | 95 | Isobutane | 58 | C4H10 |
| 2 | 8.292 | 1.4791 | 88 | 2-Methylbutane | 72 | C5H12 |
| 3 | 10.392 | 1.3210 | 90 | 2,3-Dimethylbutane | 86 | C6H14 |
| 4 | 10.470 | 1.2597 | 88 | 2,2-Dimethylbutane | 86 | C6H14 |
| 5 | 10.975 | 0.5609 | 95 | 3-Methylpentane | 86 | C6H14 |
| 6 | 12.95 | 2.1885 | 91 | 2,4-Dimethylpentane | 100 | C7H16 |
| 7 | 13.359 | 0.3170 | 95 | 2,2-Dimethylpentane | 100 | C7H16 |
| 8 | 14.998 | 0.8880 | 89 | 2-Methylhexane | 100 | C7H16 |
| 9 | 15.217 | 1.2700 | 93 | 2,3-Dimethylpentane | 100 | C7H16 |
| 10 | 15.578 | 0.6173 | 94 | 3-Methylhexane | 100 | C7H16 |
| 11 | 16.734 | 16.4986 | 90 | 2,2,4-Trimethylpentane | 114 | C8H18 |
| 12 | 19.879 | 4.4194 | 86 | 2,5-Dimethylhexane | 114 | C8H18 |
| 13 | 20.112 | 3.6922 | 88 | 2,4-Dimethylhexane | 114 | C8H18 |
| 14 | 20.255 | 2.2556 | 88 | 2,2,3-Trimethylpentane | 114 | C8H18 |
| 15 | 21.804 | 5.5302 | 89 | 2,3,4-Trimethylpentane | 114 | C8H18 |
| 16 | 22.407 | 8.5216 | 89 | 2,3,3-Trimethylpentane | 114 | C8H18 |
| 17 | 22.678 | 1.6362 | 90 | 2,3-Dimethylhexane | 114 | C8H18 |
| 18 | 23.130 | 0.6178 | 92 | 2-Methylheptane | 114 | C8H18 |
| 19 | 23.298 | 0.1607 | 88 | 4-Methylheptane | 114 | C8H18 |
| 20 | 23.610 | 0.4025 | 94 | 3,4-Dimethylhexane | 114 | C8H18 |
| 21 | 23.892 | 0.4454 | 90 | 3-Methylheptane | 114 | C8H18 |
| 22 | 25.049 | 13.3359 | 94 | 2,2,5-Trimethylhexane | 128 | C9H20 |
| 23 | 25.748 | 0.2054 | 96 | 2,2,4-Trimethylhexane | 128 | C9H20 |
| 24 | 27.218 | 0.2218 | 90 | 2,4,4-Dimethylhexane | 128 | C9H20 |
| 25 | 27.894 | 1.6321 | 87 | 2-Methyloctane | 128 | C9H20 |
| 26 | 28.433 | 0.4001 | 91 | 4,4-Dimethylheptane | 128 | C9H20 |
| 27 | 28.954 | 0.7304 | 93 | 2-Methyloctane | 128 | C9H20 |
| 28 | 29.530 | 1.3327 | 88 | 2,5-Dimethylheptane | 128 | C9H20 |
| 29 | 29.655 | 0.1551 | 88 | 2,2,3-Trimethylhexane | 128 | C9H20 |
| 30 | 30.606 | 0.0958 | 92 | 2,3,4-Trimethylhexane | 128 | C9H20 |
| 31 | 30.971 | 0.1409 | 92 | 1,3,5-Trimethylcyclohexane | 126 | C9H18 |
| 32 | 31.194 | 0.2355 | 90 | 2,3-Dimethylheptane | 128 | C9H20 |
| 33 | 31.423 | 0.0970 | 92 | 3,4-Dimethylheptane | 128 | C9H20 |
| 34 | 32.305 | 0.1495 | 95 | 2-Methyloctane | 128 | C9H20 |
| 35 | 32.665 | 0.3004 | 92 | 4,4-Dimethyloctane | 142 | C10H22 |
| 36 | 32.791 | 0.3195 | unknown | C10H22 | ||
| 37 | 32.952 | 2.3452 | 87 | 2,2-Dimethyloctane | 142 | C10H22 |
| 38 | 33.089 | 0.3087 | 96 | 2,2,4-Trimethylheptane | 142 | C10H22 |
| 39 | 33.461 | 2.0970 | 87 | 3,4-Dimethyloctane | 142 | C10H22 |
| 40 | 34.393 | 1.1930 | 88 | 2,5,5-Trimethylheptane | 142 | C10H22 |
| 41 | 35.018 | 0.0851 | 87 | 1,1,3,5-Tetramethylcyclohexane | 140 | C10H22 |
| 42 | 35.269 | 0.1540 | 87 | 2,4,6-Trimethylheptane | 142 | C10H22 |
| 43 | 35.550 | 0.7600 | 90 | 2,3-Dimethyloctane | 142 | C10H22 |
| 44 | 35.706 | 0.1259 | 91 | 2,3,5-Trimethylheptane | 142 | C10H22 |
| 45 | 36.064 | 0.1843 | 87 | 2,5-Dimethyloctane | 142 | C10H22 |
| 46 | 36.556 | 0.1462 | 86 | 2,7-Dimethyloctane | 142 | C10H22 |
| 47 | 36.661 | 0.2365 | 94 | 3,6-Dimethyloctane | 142 | C10H22 |
| 48 | 37.957 | 0.0768 | 92 | 2,3-Dimethyloctane | 142 | C10H22 |
| 49 | 38.536 | 1.8619 | 90 | 3,8-Dimethylnonane | 156 | C11H24 |
| 50 | 39.154 | 0.4097 | 92 | 2,2,6,6-Tetramethylheptane | 156 | C11H24 |
| 51 | 39.320 | 0.7298 | 87 | 2,2,3,5-Tetramethylheptane | 156 | C11H24 |
| 52 | 39.463 | 0.1995 | 89 | 2,4,6-Trimethyloctane | 156 | C11H24 |
| 53 | 39.592 | 0.6486 | 88 | 3,6-Dimethyldecane | 170 | C12H24 |
| 54 | 39.679 | 0.4938 | 86 | 3-Methylundecane | 170 | C12H24 |
| 55 | 40.377 | 0.4704 | 86 | 3,8-Dimethyldecane | 170 | C12H24 |
| 56 | 40.503 | 0.1684 | 83 | 5-Methylundecane | 170 | C12H24 |
| 57 | 40.763 | 0.3103 | 81 | 2,8,8-Trimethyldecane | 184 | C13H28 |
| 58 | 41.564 | 0.1575 | 90 | 6,6-Dimethylundecane | 184 | C13H28 |
| 59 | 42.015 | 1.9689 | 86 | 3,9-Dimethylundecane | 184 | C13H28 |
| 60 | 42.447 | 0.4732 | 87 | 3,3-Dimethylundecane | 184 | C13H28 |
| 61 | 42.789 | 0.9040 | 89 | 3,6-Dimethylundecane | 184 | C13H28 |
| 62 | 43.579 | 0.3784 | 87 | 6-Methyldodecane | 184 | C13H28 |
| 63 | 43.765 | 0.5435 | 88 | 2,9-Dimethylundecane | 184 | C13H28 |
| 64 | 44.010 | 1.0890 | 86 | 5-Methyldodecane | 184 | C13H28 |
| 65 | 44.283 | 0.2509 | 90 | 2,5-Dimethylundecane | 184 | C13H28 |
| 66 | 44.699 | 1.2211 | 87 | 4,4-Dimethylundecane | 184 | C13H28 |
| 67 | 44.986 | 0.2573 | 90 | 5-Methyl-5-propylnonane | 184 | C13H28 |
| 68 | 45.138 | 0.2227 | 94 | 3-Methyldodecane | 184 | C13H28 |
| 69 | 45.311 | 0.2427 | 92 | 2,2,4-Trimethyldecane | 184 | C13H28 |
| 70 | 45.576 | 0.4157 | 93 | 2,6-Dimethylundecane | 184 | C13H28 |
| 71 | 46.537 | 0.2544 | 88 | 4,6-Dimethyldodecane | 198 | C14H30 |
| 72 | 46.713 | 0.7725 | 87 | unknown | ||
| 73 | 47.060 | 0.1439 | 92 | 5-Methyltridecane | 198 | C14H30 |
| 74 | 47.242 | 0.2518 | 90 | 2,6,10-Trimethyldodecane | 212 | C15H32 |
| 75 | 48.230 | 0.4782 | 92 | 4-Methyltetradecane | 212 | C15H32 |
| 76 | 48.714 | 0.8915 | 90 | 2,6,11-Trimethyldodecane | 212 | C15H32 |
| 77 | 48.779 | 0.6753 | 90 | 3-Methyltetradecane | 212 | C15H32 |
| 78 | 49.045 | 0.2249 | 86 | n-pentadecane | 212 | C15H32 |
| 79 | 49.355 | 0.1177 | 92 | 2,2-Dimethyltetradecane | 226 | C16H34 |
| 80 | 49.986 | 0.1678 | 92 | n-Hexadecane | 226 | C16H34 |
| 81 | 50.404 | 0.2211 | 92 | 2,2,11,11-Tetramethyldodecane | 226 | C16H34 |
| 82 | 51.043 | 0.1408 | unknown | |||
| 83 | 52.963 | 0.2317 | unknown | |||
| 84 | 53.307 | 0.2980 | unknown | |||
| 85 | 53.987 | 0.0631 | unknown | |||
| 86 | 54.134 | 0.0924 | unknown |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fu, K.; Liu, B.; Chen, X.; Chen, Z.; Liang, J.; Zhang, Z.; Wang, L. Investigation of a Complex Reaction Pathway Network of Isobutane/2-Butene Alkylation by CGC–FID and CGC-MS-DS. Molecules 2022, 27, 6866. https://doi.org/10.3390/molecules27206866
Fu K, Liu B, Chen X, Chen Z, Liang J, Zhang Z, Wang L. Investigation of a Complex Reaction Pathway Network of Isobutane/2-Butene Alkylation by CGC–FID and CGC-MS-DS. Molecules. 2022; 27(20):6866. https://doi.org/10.3390/molecules27206866
Chicago/Turabian StyleFu, Kaiwei, Bei Liu, Xiaopeng Chen, Zhiyu Chen, Jiezhen Liang, Zhongyao Zhang, and Linlin Wang. 2022. "Investigation of a Complex Reaction Pathway Network of Isobutane/2-Butene Alkylation by CGC–FID and CGC-MS-DS" Molecules 27, no. 20: 6866. https://doi.org/10.3390/molecules27206866







