1. Introduction
Over the past few decades, the world has become increasingly concerned about the danger of infections caused by pathogenic microorganisms. Current antibiotics are effective in the treatment of many infectious diseases, but their widespread use has led to the emergence of various multidrug-resistant strains and superbugs. There is an urgent need to develop new drugs and materials that can fight drug-resistant bacteria. New strategies and approaches are necessary to expand the spectrum of functional activity of new drugs without the development of drug resistance. One such strategy is chemotherapy with nitric oxide (“NO therapy”) based on a huge experimental material demonstrating NO’s effect both on the development of pathological processes [
1] and on the possibility of their correction by chemotherapeutic methods [
2].
The implementation of such a strategy includes the chemical synthesis of small molecules, and the use of experimental physical–chemical methods and methods of quantum chemical calculations to study their structure and properties in the solid phase and in solutions.
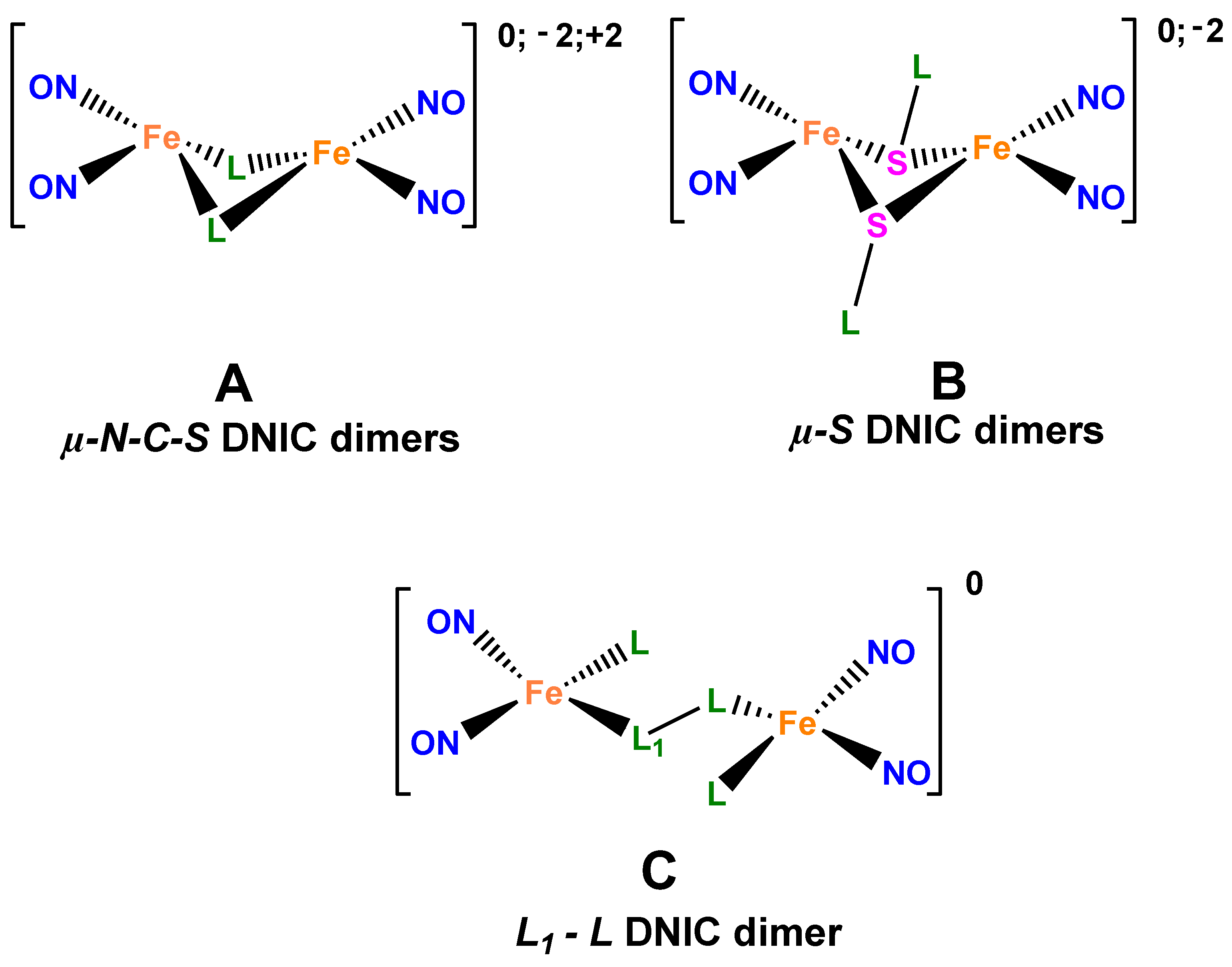
Being effective low-toxic exogenous nitric oxide (NO) donors complementary to a human organism, low-molecular dinitrosyl complexes of tetrahedral iron involving two nitrosyl and two sulfur-containing ligands, as structural analogs of the active sites of dinitrosyl [1Fe-2S] proteins, are promising for practical application in medicine, which is the reason for the increasing interest in fundamental studies of their structural features [
3,
4,
5]. As follows from our investigations, the activity of dinitrosyl complexes of tetrahedral iron (DNICs) and their dimers (
Figure 1A,B) as antibacterial agents is comparable and/or higher than that of antibiotics used in clinics [
6,
7,
8,
9,
10].
As a basic mechanism of the antimicrobial activity of NO donors of this class, the induction of oxidative–nitrosative stress is considered, which involves the interaction of NO generated by the nitrosyl complexes with oxygen and its active forms, thus leading to chemically active nitrogen forms arising that damage macromolecules, primarily, DNA [
11]. Concurrently, nitric oxide can cause the modification of thiol-containing amino acids in proteins, thus changing their activity [
12]. We have demonstrated that compounds of this family of NO donors cause nitrosylation and impair the functions of the NF-kB transcription factor [
13].
In the majority of works, aliphatic and aromatic thiols are used as ligands for the synthesis of DNICs and their dimers. Thioamide-based DNICs are virtually unknown. Due to a unique electron donor nature of heterocyclic thioamides, they possess a wide range of properties that are of great practical application in various fields of clinical chemistry and pharmacology. Given that thioamides have a higher biologic activity, including a higher antibacterial activity, than the corresponding amides [
14,
15,
16] and some of them are actively used as antituberculosis drugs [
17] and antimicrobial agents [
18,
19,
20], the high activity of DNIC with thioamides can be expected, which will be comparable to the activity of clinical antibiotics in the absence of pathogen resistance. In this regard, thioamides, on the one hand, are of interest as sulfur-containing ligands for the synthesis of nitrosyl iron complexes, i.e., two potentially functional blocks, NO groups and thioamides, can be combined in one molecule to yield a new family of prospective antibacterial compounds. On the other hand, it is interesting to investigate the bond’s nature and the nature of intermolecular interactions, as well as the electronic structure of the {Fe(NO)
2}
9 dinitrosyl fragment in the thioamide DNICs, which continues to be the subject of active discussion since general acceptance of the formalism [
21] to the present time [
22].
In this work, a new nitrosyl iron complex of [Fe(C
6H
5C-SNH
2)
2(NO)
2][Fe(C
6H
5C-SNH
2)(S
2O
3)(NO)
2] composition (complex
I) (
Figure 1C) was first synthesized. Experimental and theoretical investigations of its structure and antibacterial activity were performed, as well as correlations of “structure-activity” analyzed, taking into account the nature of intermolecular interactions that cause the emergence of the NO…NO contacts.
3. Discussion
Thioamides, being the thioanalogues of amides of carboxylic acids, are usually described by structure
a (
Figure 8). The structure of these compounds is favorable for donating the lone electron pair of the nitrogen atom to the π-antibonding orbital of the double bond, thus stabilizing the molecule planar structure. Due to such a structure,
N-monosubstituted and asymmetrical
N-disubstituted thioamides should possess a capability of geometrical isomerism, i.e., they can exist in one of the two (or in both) forms
a and
b. Theoretically, for primary and secondary thioamides, there is a possibility to transform into thioamide form
d. The existence of
d ↔
c equilibrium has often been suggested, based mainly on chemical data; however, X-ray analysis, and IR and NMR spectroscopy demonstrated no measurable concentrations of
d both in solutions and the solid state. At very high values of α-CH-acidity, the existence of
f tautomer was observed.
The vibration of the C=S group in IR spectra has a frequency ν(CS) equal to 1275 cm
−1 [
26]. In the IR spectra of thioamides, the characteristic band of an individual valence vibration of the C=S group is normally absent, in contrast to thioureas, for example (1100–1400 cm
−1) [
27]. Vibrations of the S-H group are known to lie in the range of 2500–2600 cm
−1, and they are not observed in the IR spectrum of the initial thiobenzamide either (
Figure S1 of Supplementary Materials).
In the IR spectrum of complex
I, sin-phase vibrations of the NO group (νNO-sym) are observed at ~1793 cm
−1, and antiphase vibrations (νNO-asym) at ~1723 cm
−1. The intensity of the bands of the NO groups’ valence vibrations is higher than that for other bands in the spectrum of
I. The difference between νNO-sym and νNO-asym (ΔνNO) is 70 cm
−1. This value of splitting of the νNO vibration doublet suggests non-equivalence of the NO groups, as with dimers of 1,2,4-triazolethiolyls of µ-NCS structural type [
10,
28], which appears in the inconsiderable change in the N-O and Fe-N bond lengths within the margin of error.
The molecular structure of
I is shown in
Figure 2. Coordination centers Fe(1) and Fe(2) have a distorted tetrahedral coordination of the iron atoms. The Fe-N bond lengths in the cation and anion are practically identical and equal to: Fe(1)-N(1) = 1.689(3) Å, Fe(1)-N(2) = 1.681(3) Å and Fe(2)-N(5) = 1.688(3) Å, Fe(2)-N(6) = 1.681(3) Å, respectively. The bond length Fe(1)-S(1) 2.3394(9) Å in the Fe(1) coordination center is elongated by 0.0494 Å as compared to the bond length Fe(1)-S(2) 2.2900(9) Å, and by 0.0152 Å as compared to the bond length Fe(2)-S(3) = 2.3242(9) Å in the Fe(2) coordination center. The Fe(2)-S(4) bond length is the least of all the Fe-S bonds (2.2650(9) Å). Formerly, in the structure of the neutral mononuclear iron complex with 1,2,4-triazole-3-thione, a considerable non-equivalence of the Fe-S bonds was revealed, 2.3015(2) and 2.3219(2) Å (the difference 0.02 Å), which was due to different types of chemical binding between the iron and sulfur atoms, both at room and low (100 K) temperatures [
29,
30]. In the studied compound, this tendency is enhanced (the difference in the lengths of the Fe-S bonds is 0.0494 Å in the Fe(1) coordination center and 0.0592 Å in the Fe(2)coordination center). The bond lengths of the NO groups practically do not differ from the previous studies [
31,
32,
33,
34], but they differ slightly from each other: in the Fe(1) center: O(1)-N(1) = 1.170(4) Å, O(2)-N(2) = 1.170(3)Å, and O(3)-N(5) = 1.166(3) Å, O(4)-N(6) = 1.174(3) Å in the Fe(2) center. The carbon–sulfur bond lengths of the thioamide ligand are S(1)-C(1) = 1.697(3) Å, S(2)-C(8) = 1.717(3) Å and S(3)-C(15) = 1.702(3) Å, which correspond to similar bonds in mononuclear complexes [
30], and slightly exceed the length of S=C equal to 1.684 Å [
35], but they are shorter than the single bond length S-C 1.719 Å in ligands with a thiol form. Thus, it can be concluded that the ligand coordinates the iron atoms in the thion form (structure
c in
Figure 8).
The lengths of the amide C-N bonds in the three ligands are reduced (1.304 Å average) due to the amide n–π*conjugation, but remain longer than double C=N bonds.
In the complex anion, unlike a cation, an intramolecular hydrogen bond of the N-H…O type is realized, and between the cation and the anion, an intermolecular hydrogen bond of the N-H…O type is also realized (
Table 2).
The oxygen atom O(1) of the NO group of the complex cation has a contact with the oxygen atom O(3A) {x + 1, y, z} of the NO group of the complex anion; the interatomic distance O(1)… O(3A) is equal to 2.787 Å (
Figure 9).
The crystal structure of complex
I is represented by alternating layers of cationic and anionic complexes. A fragment of the crystal structure of
I is shown in
Figure 10.
The shortest distances between the iron atoms in the crystalline packing of
I are Fe(1)… Fe(1)* {−x + 2, −y + 1, −z + 1} and Fe(1)… Fe(2)* {−x + 1, −y + 1, −z + 1} with lengths of 5.371(3) and 5.815(3) Å, respectively. In the crystal, the network of intermolecular hydrogen bonds of N-H…O type forms (
Figure 11). All three oxygen atoms of the S
2O
32− group are involved in the formation of these intermolecular hydrogen bonds (
Table 1). It should be noted that oxygen atoms O(5) and O(6) are involved in the formation of two hydrogen bonds of N-H…O type, one intramolecular and one intermolecular.
In this work we were mostly interested in the NO…NO interaction (the presence of critical points of (3, −1) type), which may stabilize the structure of the DNIC dimer. In addition, as stated earlier [
36], involvement of the NO groups’ orbitals in the intermolecular NO…NO interactions led to quenching of the orbital moment, while unquenched orbital moment of NO led to abnormal magnetic properties.
Intermolecular interactions along the first direction (the associate
I) are rather complicated, as follows from
Figure 3 where molecular graphs of the tetrameric associates are presented. QTAIM yields nine critical points along the bond lines of the atoms of two different dimeric molecules, and they are located almost symmetrically with regard to CP1 that is related to the NO…NO interaction we are interested in. Evidently, these bonds are related to rather weak intermolecular interactions; therefore their energy E
A-B can be estimated from the available correlation formula [
37] via potential energy density in the corresponding critical point ve(r): E
A-B ≈ 0.5ve(r).
The main contribution (11.0 kcal/mol) is made by hydrogen bonds between the oxygen atoms of the S2O3 groups and the hydrogen atoms of the NH2 groups of the ligands; the NO…NO contacts are much weaker but are slightly higher than the others. We can see that the NO groups form intermolecular contacts with the sulfur atoms of the S2O3 group at the expense of the oxygen atom. Generally, the S2O3 ligand easily forms contacts both at the expense of the oxygen, and the sulfur atoms.
We have performed NBO analysis of the tetrameric associate
I to reveal the nature of the O…O intermolecular interaction (CPP1). The bond order and Wiberg index for this bond are 0.0004 and 0.0001, respectively. To understand whether there is quenching of the orbital moment due to the involvement of NO π-orbitals in intermolecular NO…NO contacts [
36], the second order interactions were analyzed. Unexpectedly, no secondary interactions between the considered NO ligands were determined. The presence of the electron density in the CP1 critical point can be explained by delocalization of the electron density due to multiple secondary interactions of the NO groups with the sulfur and iron atoms of the neighboring molecule (
Table 4).
The structure of tetrameric associate
II along the other axis in the crystal differs essentially from that of
I. First, from QTAIM analysis there is only one critical point (3, −1) corresponding to the intermolecular contact, and it lies on the bond length of two oxygen atoms in NO…NO. This bond energy calculated from the Espinosa–Lecomte formula is 2.48 kcal/mol. From NBO analysis of this tetrameric associate, the Wiberg index and the bond order are 0.0019 and 0.0023, respectively, which are higher than for associate
I. We also analyzed secondary interactions to reveal the nature of the donor and acceptor orbitals of this bond (
Table 5).
It can be seen that NBO analysis confirms the presence of NO…NO interaction in this case. Each oxygen atom donates the electronic density of the lone electron pair on π* (NO) of the neighboring molecule. This donation occurs easier along O → O-N with an 1800 angle than with a 900 angle. As we mentioned, two NO ligands are located perpendicular to each other, such interaction being called T-like according to the classification in [
38]. Moreover, electron exchange occurs between the π*-orbitals of the NO ligand. Thus, in complex
I there is an NO…NO contact provided by NO π-orbitals, which leads to quenching of the orbital moment.
According to calculations (see the results of molecular modeling of complex
I), four symmetrical conformations of cation
1 (c2, c5, c15, c16) appeared to be the most stable (within 3 kJ/mol), as well as four asymmetrical ones, in which the orientation of the ligands corresponds to the combination of conformers c2, c5 and c12. For all low-lying conformers, the characteristic values of α and β angles correspond to the values (g+, tr, …). For anion
2, two conformations (c1 and c2) appeared to be the most stable, which are characterized by the deviation of the benzene ring from the S–C–N plane. The conformation observed in the crystal packing of the ion pair
1…
2 of complex
I, has a relative energy of 5.9 kJ/mol for
1 (
Table S1) and 1.24 kJ/mol for
2 (
Table S2).
Figure 12 shows overlapping of the calculated conformations
1 and
2. Optimized atomic coordinates for the conformers from
Tables S1 and S2 are presented in the
Supplementary Materials. Overall, the calculations showed a wide variety of thermally available structures for molecules
1 and
2.
The results of the conformational analysis are taken into account in the subsequent modeling of binding 1 and 2 with active centers of targets in vivo. It is also concluded that when choosing a method for binding calculations, one should focus on methods of flexible ligand docking.
Figure 5 presents the time dependencies of NO generated by complex
I. The kinetic dependence for complex
I goes to a “plateau” in 300 s, and the amount of NO formed (~5.0 nM) does not decrease by 500 s of the experiment, which indicates the formation of long-lived nitrosyl intermediates of this complex, which may be responsible for prolonged NO donor activity. Based on the data of X-ray analysis and molecular modeling for complex
I, it can be assumed that mononuclear dinitrosyl [1Fe-2S] intermediates are responsible for prolonged NO release, as with antibacterial DNIC dimers with 5-nitrophenyl-4 H-1,2,4-triazole-3-thiolyls [
10], which were studied earlier.
The antibacterial effect of
I (see
Figure 6) is similar to or higher than that of the comparator drugs, kanamycin and streptomycin, against gram-positive bacteria
M. luteus. For gram-negative bacteria
E. coli, the MIC value for
I was two times higher. The difference in the sensitivity of gram-negative and gram-positive bacteria can be explained by differences in the cell wall architecture. Gram-positive microorganisms are known to have one cell membrane consisting of peptidoglycan. The cell wall of gram-negative bacteria has a more complex organization, having internal, plasmatic and external membranes enriched with lipopolysaccharides. In addition, the cell wall of gram-negative bacteria has many dense intermediate layers. Such an arrangement of the cell wall forms a selective barrier that protects the cell from external influences, including antibiotic therapy [
39,
40,
41].
Complex
I exhibited a cytotoxic effect on Vero cells with IC
50 of appr. 90 μM (
Table 7). It should be noted that at a concentration of 62.5 μM, i.e., a concentration at which complex
I inhibits the growth and biofilm formation of
M. luteus, a decrease in cell viability was apparently low (appr. 25%).
Ligands present in the structure of complex
I do not exhibit antibacterial activity against both bacterial strains studied (
Table 6). No remarkable cytotoxicity to Vero cells was also detected for the ligands at concentrations up to 500 μM (
Figure 7). Thus, the antibacterial and cytotoxic effects of the complex are determined by the overall structure of the compound, but not by the ligands comprising its structure.
Comparison of the antibacterial, cytotoxic and NO-donating properties of complex
I and DNICs with thiolyls studied earlier [
9,
10,
42,
43,
44] leads to some important points.
In our previous work, sixteen nitrosyl iron complexes were found to inhibit the growth of gram-positive bacteria and, with less efficacy, gram-negative bacteria. Most of these complexes were efficient inhibitors of biofilm formation [
9]. Three DNIC dimers with 5-(nitrophenyl)-4-H-1,2,4-triazole-3-thiolyls exhibited an equal growth-inhibiting effect on
M. luteus and
E. coli and inhibited biofilm formation at a wide range of concentrations [
10]. As was found for complex
I, DNICs with nitrothiophenols or dimethylthiourea [
9] and 5-(nitrophenyl)-4-H-1,2,4-triazole-3-thiolyls [
10], the antibiofilm activity of DNICs is comparable to that of antibiotics. Thus, DNICs of different structures exert antibacterial activity.
The cytotoxicity of DNICs varies in a wide range, but comparison with corresponding MICs demonstrates that many DNICs exhibit higher activity on bacteria than on eucaryotic cells. For complex
I (
Table 6) and DNICs with 3-nitrothiophenol, dimethylthiourea [
9] and 5-(nitrophenyl)-4-H-1,2,4-triazole-3-thiolyls [
10], IC
50 values for Vero cells exceeds corresponding MICs for
M. luteus. IC
50 for DNICs with 5-(nitrophenyl)-4-H-1,2,4-triazole-3-thiolyls are also higher than MICs for
E. coli [
10]. Conversely, DNIC with 4-nitrothiophenol exerted cytotoxicity with IC
50 lower than MICs for both
M. luteus and
E. coli [
9]. Based on these in vitro data, one may conclude that different DNICs can be considered as antibacterial agents except 4-nitrothiophenol-bearing DNIC.
The NO generation kinetics of all DNICs under discussion have a plateau, which demonstrates the prolonged NO release. The maximum level of NO generated by complex
I is valued at 5 nM (
Figure 5). Other DNICs generate NO with greater levels at their plateau. The maximum generated NO reached a value of ~60 nM for the DNIC dimer with 3-nitrothiophenol [
42], ~25 nM for the DNIC dimer with 4-nitrothiophenol [
8] and 10 to 20 nM for DNICs with 5-(nitrophenyl)-4-H-1,2,4-triazole-3-thiolyls [
10].
From these data it can be seen that both the antibacterial activity and cytotoxicity of DNICs cannot be attributed solely to their NO generation features. It is important to note that biological activity can also not be attributed to ligands of DNICs since the antibacterial and cytotoxic effects of ligands were not detectable (
Table 6 and
Table 7).
Thus, the antibacterial properties of studied DNICs are not determined by their ligands or by the amount of generated NO, but rather by the overall structure and physicochemical properties. Since corresponding ligands do not exert the biological activity of DNICs, it can be assumed that intermediates formed upon DNIC decomposition can contribute to the biological effects of NO-donating DNICs. Further design of antibacterial agents of this family should be directed towards the synthesis of complexes with prolonged NO generation, and a detailed analysis of the structure of the resulting long-lived nitrosyl intermediates can give insights into the mechanisms of their antibacterial action.
4. Materials and Methods
All operations for the synthesis of complex
I were carried out in an inert atmosphere. Beforehand, argon (technical) was purged for 30 min through the bidistilled water used for the synthesis. For the synthesis of Na
2[Fe
2(S
2O
3)
2(NO)
4]·4H
2O, according to the procedure in [
43], the following reactants were used: FeSO
4·7H
2O, Na
2S
2O
3·5H
2O (Sigma-Aldrich, St. Louis, MO, USA). NO was prepared by the reaction of iron (II) sulfate and sodium nitrite solutions in an acidic medium. For this, FeSO
4·7H
2O, NaNO
2 (pure grade) and HCl (pure grade, conc.) were used.
Analysis of C, H, N, S, O elements in complex I was performed on a CHNS/O elemental analyzer “Vario El cube”. Fe was detected on the atomic absorption spectrometer “AAS-3”. A sample of polycrystals of complex I was preliminarily mineralized. IR spectra were recorded on a Fourier spectrometer (Bruker ALPHA, Ettlingen, Germany) in the frequency range of 400–4000 cm−1 in the ATP mode at room temperature.
4.1. Synthesis
Thiobenzamide powder (97%, Sigma-Aldrich, St. Louis, MO, USA) (0.48 g, 3.5 mmol) was dissolved in 0.5 mL of concentrated HCl and 5 mL of methanol. The acidified solution was stirred without heating and filtered. A dry mixture of 0.38 g (0.4 mmol) of Na2S2O3·5H2O and 0.44 g (0.77 mmol) of Na2[Fe2(S2O3)2(NO)4]·4H2O was dissolved in 5 mL of water and filtered. The filtrate was purged for 1–2 min in an inert gas current. To the filtrate of the acidified thiobenzamide solution in the argon current, the filtrate of the mixture of sodium thiosulfate and nitrosyl iron complex was quickly poured, with the inert gas still passing through the solution. The instantaneous precipitate was filtered, and the filtrate was stored in a closed flask at +5 °C. After 3 weeks, the formed single crystals of complex I as red-brown thin needles of ~0.2 mm were filtered and dried in argon.
For C21H21N7O7S5Fe2: Found, % C 33.54, H 2.80, N 12.95, O 14.79, S 21.17, Fe 14.75. Calculated, % C 33.40, H 2.78, N 12.98, O 14.83, S 21.22, Fe 14.79.
IR spectrum of I (cm−1): 3379 (v.w.), 3030 (w), 1793 (v.s.), 1723 (v.s), 1467 (m.), 1259 (m.), 1113 (m.), 1117 (m.), 997 (s.), 856 (m.), 760 (m.), 701 (m.), 680 (m.), 631 (m.), 524 (m.), 421 (m.), νNO = 1793 cm−1, 1723 cm−1.
4.2. X-ray Analysis
X-ray study of single crystals of
I was performed at 100 K on a “Belok” beamline diffractometer of the National Research Center “Kurchatov Institute” (Moscow, Russia) using a Rayonix SX165 detector at λ = 0.96990 Å. The data were indexed and integrated using an iMOSFLM utility in CCP4 program [
44], and then scaled with absorption correction using the Scala program [
45]. Crystallographic data and main refinement parameters are shown in
Table 8. The structure was solved by the direct method [
46]. Positions and temperature parameters of non-hydrogen atoms were refined in the anisotropic approximation by full-matrix least-squares method [
46]. Positions of all hydrogen atoms were calculated geometrically and then refined by imposing the restrictions of the “riding” model [
46]. All calculations were performed using SHELXTL program complex [
46]. The crystalline structure was deposited with the Cambridge Structural Database, CCDC, Deposition Number 2182839, and it can be freely received at
www.ccdc.cam.ac.uk/data_request/cif (accessed on 12 July 2022).
4.3. Quantum Chemical Calculations
We performed quantum chemical calculations of tetrameric associates
I and
II using GAUSSIAN-09 program [
47] without optimization of their geometrical structure, in tpssh/6–31G* approximation. The AIMAll program package (Version 15.05.18) [
48] was used to analyze the wave function by QTAIM methods. The NBO computational procedure implemented in the Gaussian 09 program was used.
Conformation properties, electronic structure and free energies of complex I hydration were studied by means of quantum mechanical calculations by density functional theory method using B3LYP functional and a set of basis functions 6–31 + G(d,p). The effect of water surrounding was taken into account in the frame of polarized continuum medium model (PCM) [
49] and SMD [
50]. A license program Gaussian-09 was used [
47].
4.4. Amperometric Determination of NO Generation
To determine NO amount generated by complex
I in solution, a sensor electrode “amiNO-700” of “Nitric oxide measurement system inNO-T” (Innovative Instruments, Inc., Tampa, FL, USA) was used. All the experiments were performed in aerobic 0.1% aqueous DMSO solution at 25 °C and pH 7.0. NO concentration was recorded for ~500 s (with 0.2 s pace). DMSO was purified according to the technique in [
51]. Commercial buffer Hydrion (Sigma-Aldrich, N 239089, St. Louis, MO, USA) was used (pH of the solutions was measured using a membrane pH-meter “HI 8314” (HANNA Instruments, Vöhringen, Germany)). A standard 100 μM NaNO
2 aqueous solution was added to the solution. The mixture of 0.12 M KI and 2 mL of 1 M H
2SO
4 in 18 mL of water was used for calibration of the electrochemical sensor.
4.5. In Vitro Antibacterial Activity
Antibacterial activity of
I was studied by the serial dilution method [
10]. Laboratory cultures of gram-negative
Escherichia coli bacteria (strain BB) and gram-positive Micrococcus luteus bacteria (strain 21/26) were used as bacterial agents. The bacteria were dispersed into 96-well flat-bottomed plates in the LB incubation medium (Peptone 1%, yeast extract 0.5%, NaCl 1%, glucose 0.1%, pH 6.8–7.0) of 100 μL each, and 100 μL of consecutive double dilutions of the test substances were introduced. The initial solution of the compounds was prepared in DMSO at a maximum concentration of 200 mM. Final concentrations of compounds in the samples ranged from 1000 μM to 0.47 μM. The final maximum concentration of DMSO in the samples was 10%. This concentration of DMSO does not affect the viability of the bacterial agent. The final concentration of the bacterial agent in the well was 5 × 10
5 CFU/mL.
As comparison drugs, antibiotics ampicillin (JSC “Sintez”, Kurgan, Russia), kanamycin (JSC “Sintez”, Kurgan, Russia), streptomycin (JSC “Biochemist”, Saransk, Russia) and ceftriaxone (JSC “Biochemist”, Saransk, Russia), dissolved in the LB incubation medium, were used. Final concentrations of antibiotics ranged from 1000 μM to 0.49 μM. Samples were incubated at 37 °C for 24 h after administration of the complex and antibiotics. Control samples were grown in the LB medium or in the LB medium with 10% DMSO at 37 °C. Negative control was grown in the LB medium at 4 °C. The minimum inhibitory concentration of the compound that inhibits the growth of microorganisms (MIC) was defined as the minimum concentration at which there was no visible growth of microorganisms compared to the control samples.
To obtain biofilms, gram-positive Micrococcus luteus bacteria (strain 21/26) were cultured in a 96-well plate at a concentration of 5 × 106 CFU/mL at 37 °C. For determination of the background staining, samples with LB medium without bacteria were used. Control samples contained bacteria in either LB medium or LB medium with solvent (10% DMSO). All compounds and comparator drugs were used at concentrations corresponding to MICs. After 24 h of incubation in the presence of studied compounds, the medium with planktonic cells was removed, the wells were washed with sterile PBS buffer (137 mM NaCl, 2.68 mM KCl, 4.29 mM Na2HPO4, 1.47 mM KH2PO4, pH 7.4) and dried at 37 °C for 60 min, then stained with 0.1% solution of crystal violet for 15 min. After staining, the wells were washed with water and dried. The dye bound to biofilms was dissolved in 95% ethanol, and optical density of the resulting solutions was determined photometrically at a wavelength of 570 nm. The optical density of the solution in control samples with the bacteria suspension without the test complexes was taken as 100%. Background staining of the substrate (empty wells) was subtracted from all samples.
4.6. In Vitro Cytotoxicity Assay
4.6.1. Cell Culture
As a model, a cell culture of Vero (African green monkey kidney epithelial cells) was used. Cells were grown DMEM incubation mediums supplemented with 10% fetal calf serum, penicillin (100 U/mL) and streptomycin (100 µg/mL). The cells were cultured at 37 °C in a humidified atmosphere with 5% CO2.
4.6.2. MTT Assay
To assess the cytotoxic effect of
I, the MTT test method was used [
52]. Vero cells (5 × 10
3 cells) were plated into 96-well plates in 100 µL aliquots and allowed to adhere for 24 h. All studied compounds were dissolved in DMSO and used immediately. The initial concentration of all compounds was 500 mM, and serial dilutions were prepared just before use. Incubation medium in all samples was aspirated and replaced by medium with studied compounds. DMSO concentration in all samples was 0.1%, the final concentration of complex
I and its ligands ranged from 7.81 to 500 µM. After 72 h of incubation, 10 µL of MTT solution (5 mg/mL) was added and plates were incubated for an additional 4 h at 37 °C, then the media removed and MTT formazan crystals were dissolved in 100 µL of 100% DMSO. The absorption of the samples was measured at 570 nm (reference wavelength 650 nm) on Spark 10 M multimode microplate reader (Tecan, Männedorf, Switzerland).