Design, Synthesis, Anticancer Activity and Molecular Docking of New 1,2,3-Triazole-Based Glycosides Bearing 1,3,4-Thiadiazolyl, Indolyl and Arylacetamide Scaffolds
Abstract
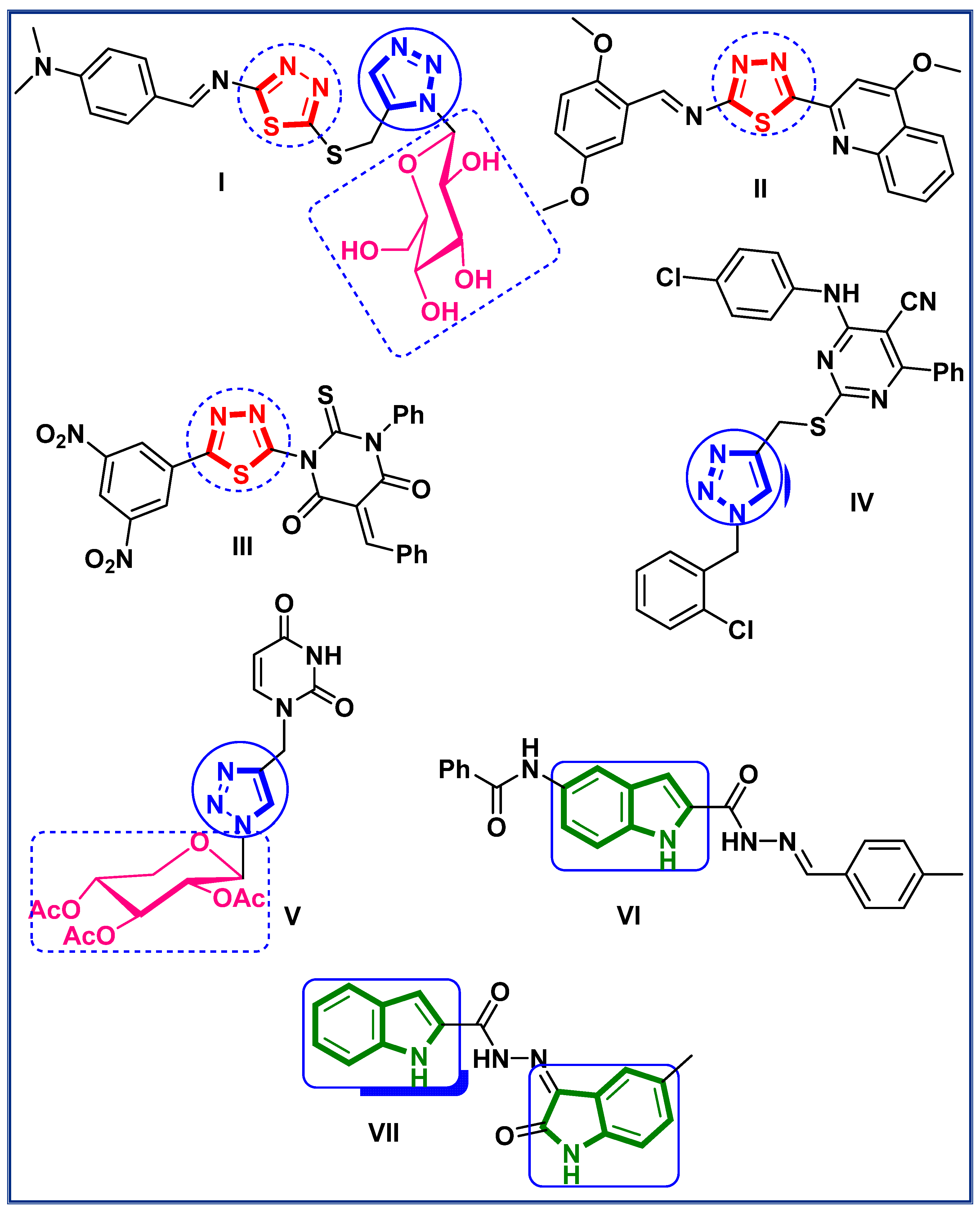
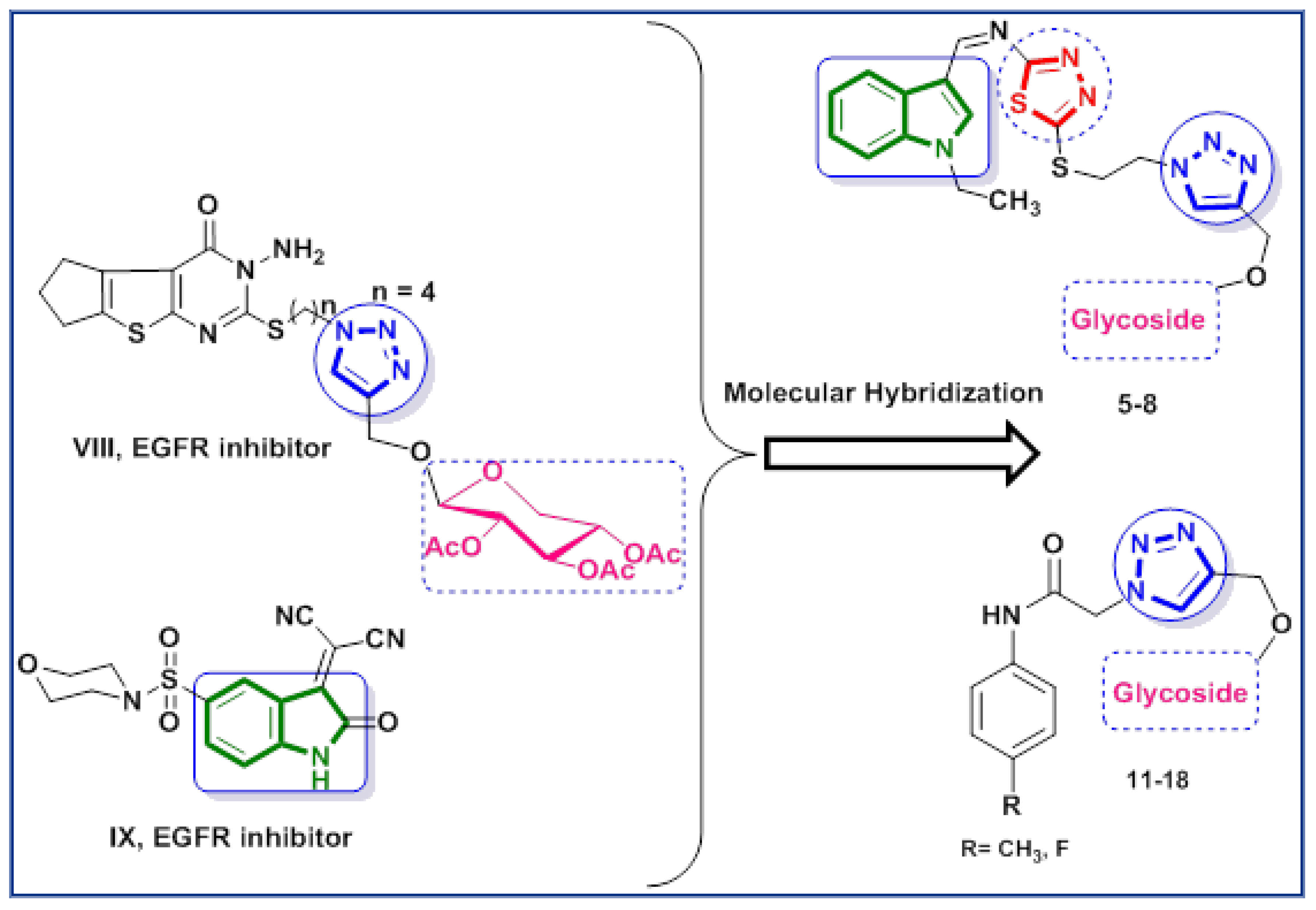
1. Introduction
2. Results and Discussion
2.1. Chemistry
2.2. Biological Evaluation
2.2.1. Cytotoxic Screening
2.2.2. Structure–Activity Relationship
2.2.3. In Vitro Inhibitory Assessment of EGFRWT, EGFRT790M and HER-2
2.2.4. Detection of Apoptosis and Cell Cycle Analysis
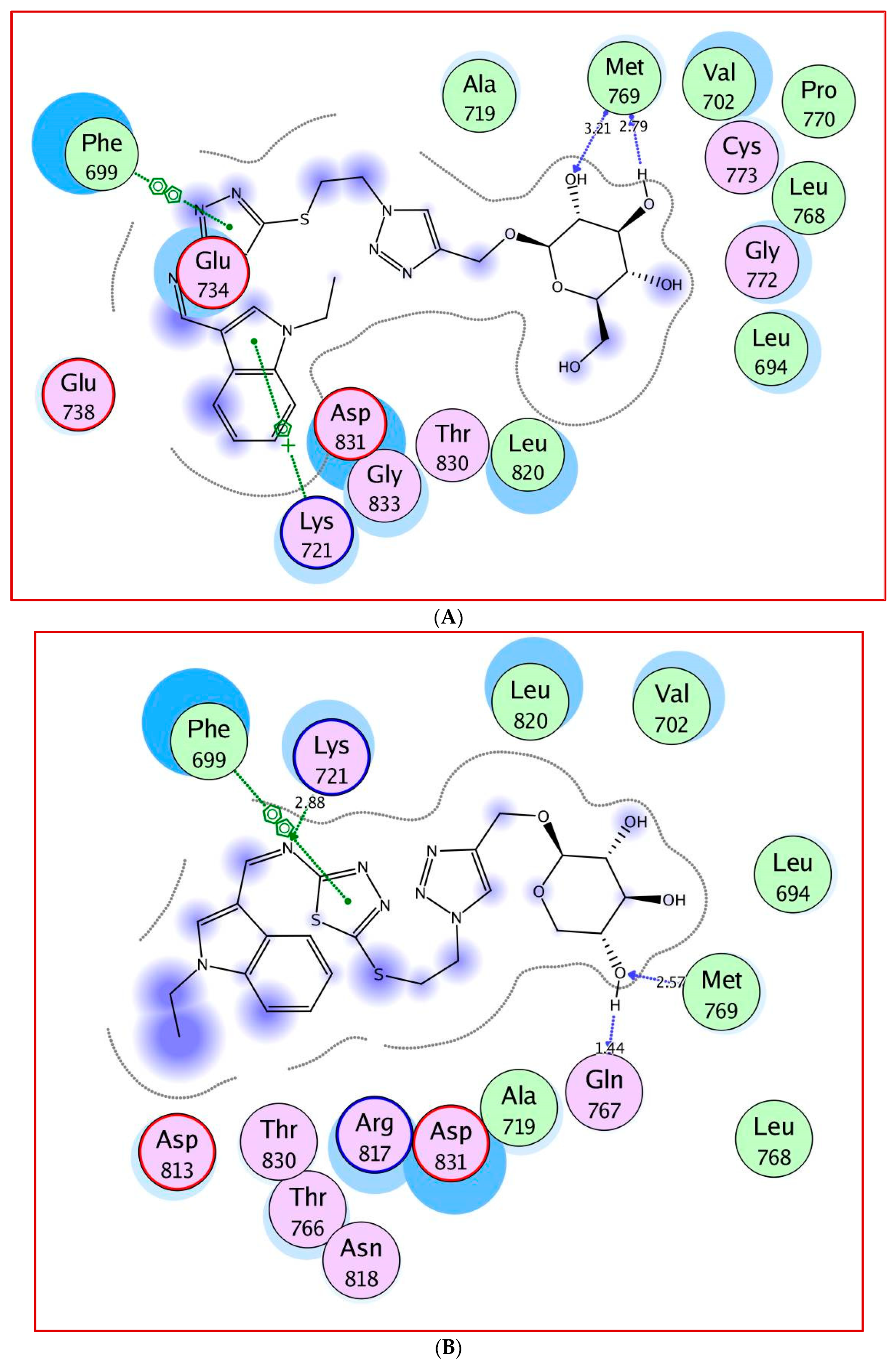
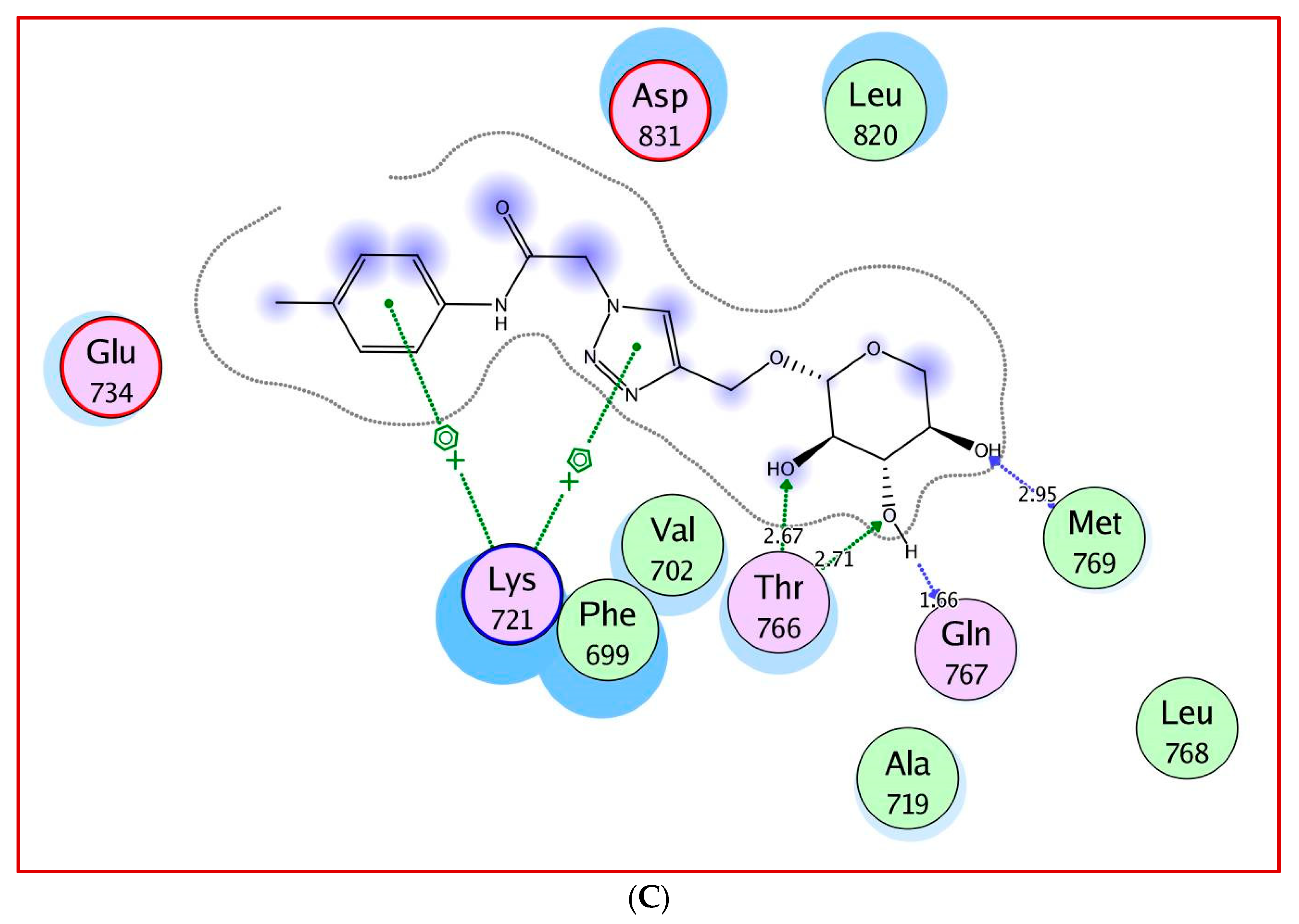
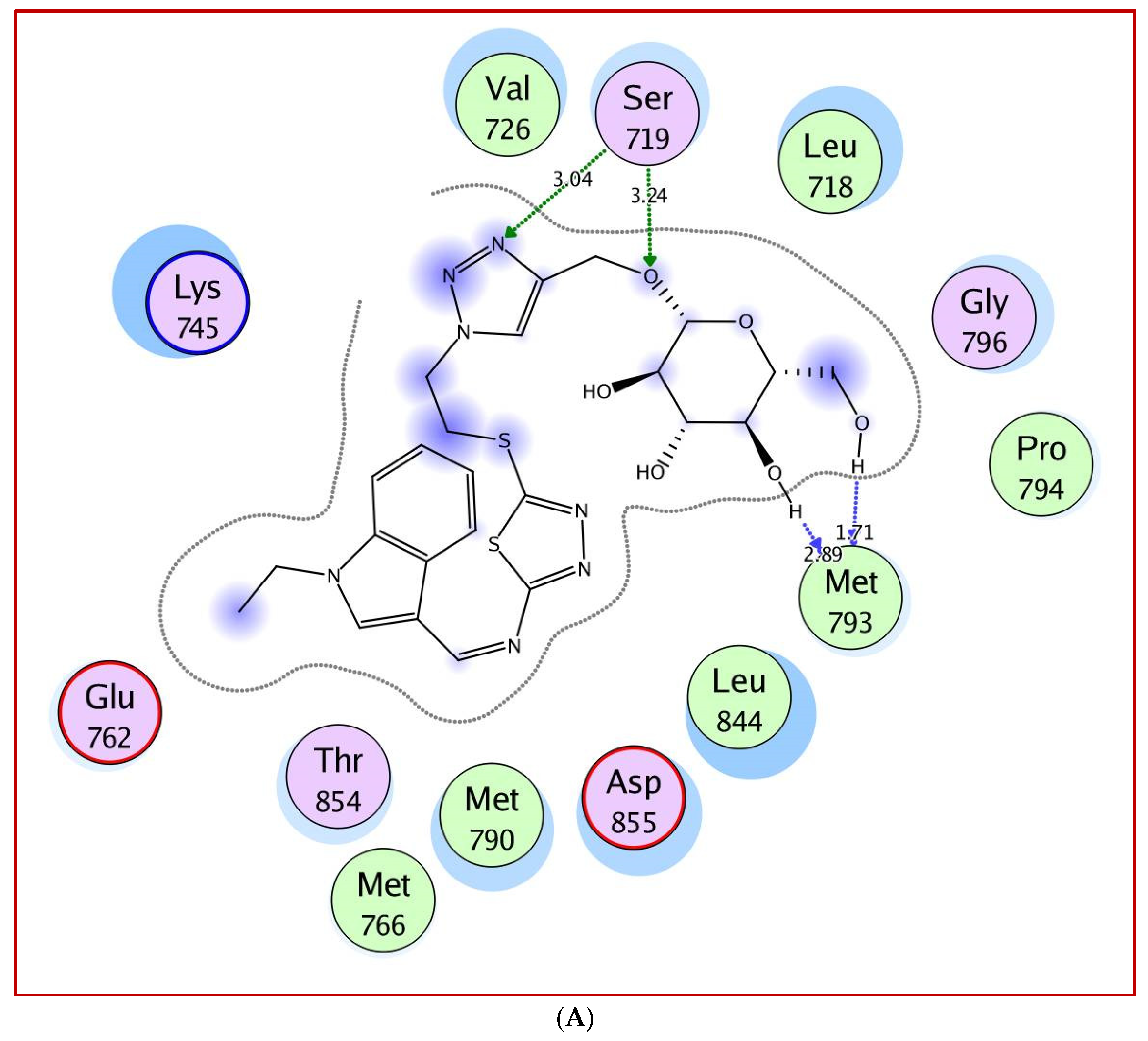
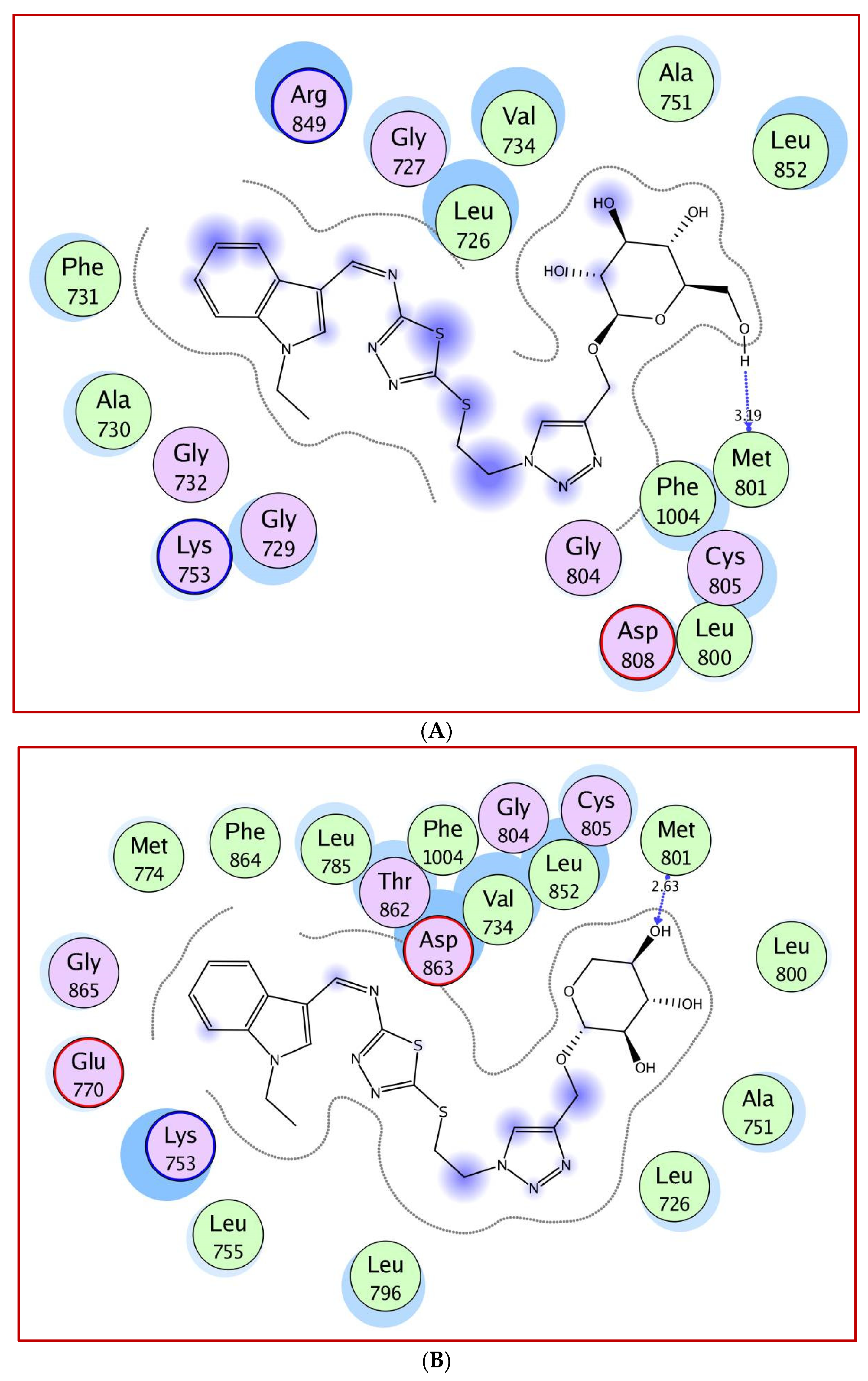
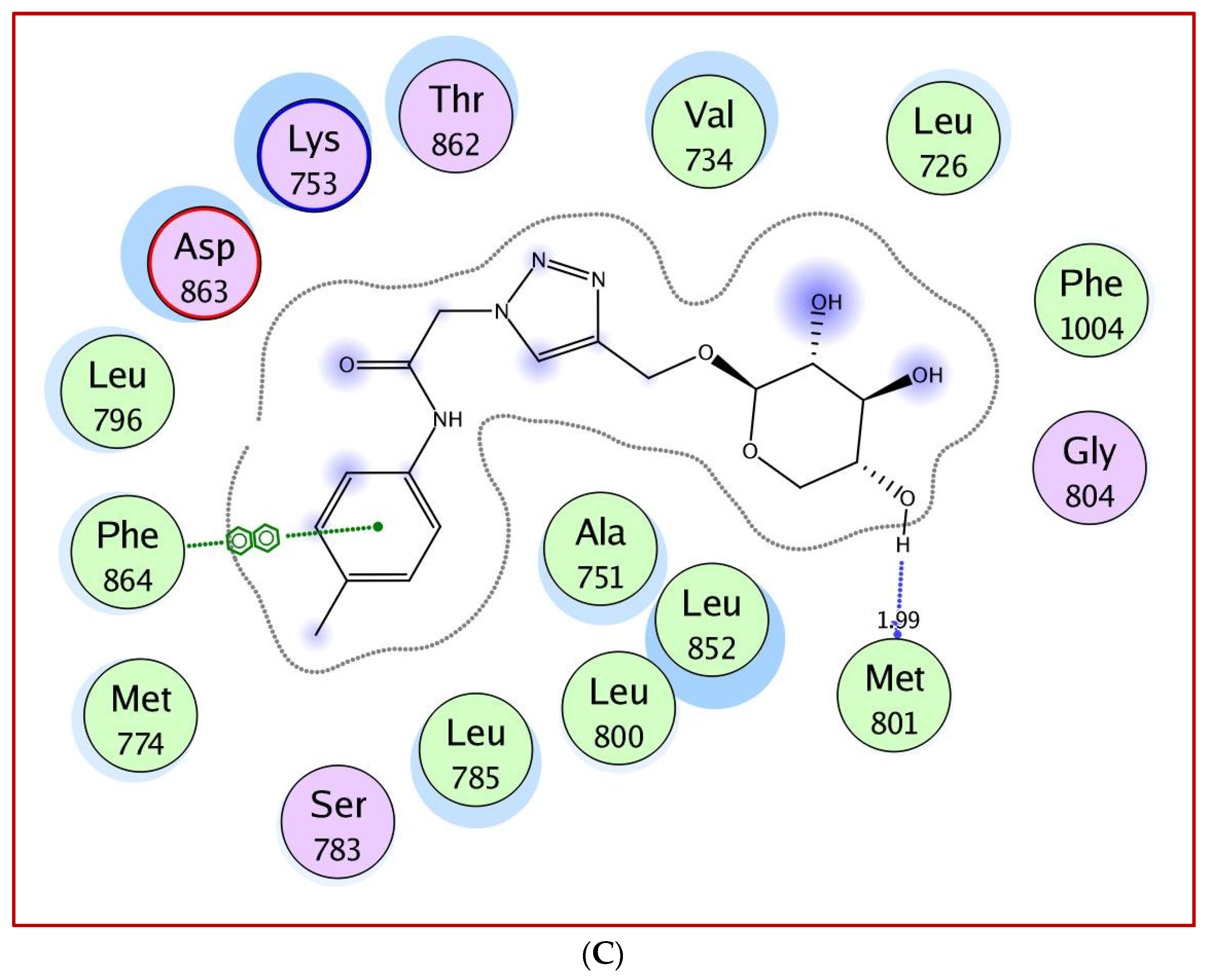
2.3. Molecular Docking Study
3. Experimental Section
3.1. General
3.2. Synthesis of Bromoethyl-1,3,4-Thidiazole Derivative 2 and Azide Derivative 3
3.3. Synthesis of Acetylated O-Glycosides (5,6)
3.4. Synthesis of Deacetylated O-Glycosides (7,8)
3.5. Synthesis of Acetylated O-Glycosides 11–14
3.6. Synthesis of Deacetylated O-Glycosides 15–18
4. Materials and Methods
4.1. In Vitro Cytotoxic Activity
4.2. MTT Cytotoxicity Assay
4.3. Statistical Analysis
4.4. EGFRWT, EGFRT790M and HER-2 Inhibition Assessment
4.5. Detection of Apoptosis and Cell Cycle Analysis
5. Molecular Docking Study
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Avin, B.R.V.; Thirusangu, P.; Ranganatha, V.L.; Firdouse, A.; Prabhakar, B.T.; Khanum, S.A. Synthesis and tumor inhibitory activity of novel coumarin analogs targeting angiogenesis and apoptosis. Eur. J. Med. Chem. 2014, 75, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Jiang, N.; Zhai, X.; Zhao, Y.; Liu, Y.; Qi, B.; Tao, H.; Gong, P. Synthesis and biological evaluation of novel 2-(2-arylmethylene)hydrazinyl-4-aminoquinazoline derivatives as potent antitumor agents. Eur. J. Med. Chem. 2012, 54, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.K.; Sharma, S.; Vaidya, A.; Ravichandran, V.; Agrawal, R.K. 1, 3, 4-thiadiazole and its derivatives: A review on recent progress in biological activities. Chem. Biol. Biol. Biol. Biol. Drug Des. 2013, 81, 557–576. [Google Scholar] [CrossRef]
- Kushwaha, N.; Kushwaha, S.K.S.; Rai, A.K. Biological activities of thiadiazole derivatives: A review. Int. J. Chem. Res. 2012, 4, 517–531. [Google Scholar]
- Gomha, S.M.; Salah, T.A.; Abdelhamid, A.O. Synthesis, characterization, and pharmacological evaluation of some novel thiadiazoles and thiazoles incorporating pyrazole moiety as anticancer agents. Monatsh. Chem. 2015, 146, 149–158. [Google Scholar] [CrossRef]
- Gomha, S.M.; Abdel-aziz, H.M. Synthesis and antitumor activity of 1,3,4-thiadiazole derivatives bearing coumarine ring. Heterocycles 2015, 91, 583–592. [Google Scholar] [CrossRef]
- Siddiqui, N.; Ahuja, P.; Ahsan, W.; Pandeya, S.N.; Alam, M.S. Thiadiazoles: Progress report on biological activities. J. Chem. Pharm. Res. 2009, 1, 19–30. [Google Scholar]
- Foroumadi, A.; Kargar, Z.; Sakhteman, A.; Sharifzadeh, Z.; Feyzmohammadi, R.; Kazemi, M.; Shafiee, A. Synthesis and antimycobacterial activity of some alkyl [5-(nitroaryl)-1,3,4-thiadiazol-2-ylthio]propionates. Bioorganic Med. Chem. Lett. 2006, 16, 1164–1167. [Google Scholar] [CrossRef] [PubMed]
- Kumar, D.; Kumar, N.M.; Chang, K.H.; Shah, K. Synthesis and anticancer activity of 5-(3-indolyl)-1,3,4-thiadiazoles. Eur. J. Med. Chem. 2010, 45, 4664–4668. [Google Scholar] [CrossRef] [PubMed]
- Ay, K.; Ispartaloglu, B.; Halay, E.; Ay, E.; Yasa, I.; Karayıldırım, T. Synthesis and antimicrobial evaluation of sulfanilamide- and carbohydrate-derived 1,4-disubstitued-1,2,3-triazoles via click chemistry. Med. Chem. Res. 2017, 26, 1497–1505. [Google Scholar] [CrossRef]
- Subhashini, N.J.P.; Kumar, E.P.; Gurrapu, N.; Yerragunta, V. Design and synthesis of imidazolo-1,2,3-triazoles hybrid compounds by microwave-assisted method: Evaluation as an antioxidant and antimicrobial agents and molecular docking studies. J. Mol. Struct. 2019, 1180, 618–628. [Google Scholar] [CrossRef]
- Saeedi, M.; Mohammadi-Khanaposhtani, M.; Pourrabia, P.; Razzaghi, N.; Ghadimi, R.; Imanparast, S.; Rastegar, H. Design and synthesis of novel quinazolinone-1,2,3-triazole hybrids as new anti-diabetic agents: In vitro α-glucosidase inhibition, kinetic, and docking study. Bioorg. Chem. 2019, 83, 161–169. [Google Scholar] [CrossRef]
- Gholampour, M.; Ranjbar, S.; Edraki, N.; Mohabbati, M.; Firuzi, O.; Khosh-neviszadeh, M. Click chemistry-assisted synthesis of novel aminonaphtho- quinone-1,2,3-triazole hybrids and investigation of their cytotoxicity and cancer cell cycle alterations. Bioorg. Chem. 2019, 88, 102967. [Google Scholar] [CrossRef] [PubMed]
- Chinthala, Y.; Thakur, S.; Tirunagari, S.; Chinde, S.; Domatti, A.K.; Arigari, N.K.; Tiwari, A. Synthesis, docking and ADMET studies of novel chalcone triazoles for anti-cancer and anti-diabetic activity. Eur. J. Med. Chem. 2015, 93, 564–573. [Google Scholar] [CrossRef]
- Rao, P.S.; Kurumurthy, C.; Veeraswamy, B.; Kumar, G.S.; Poornachandra, Y.; mar, C.G.K.; Vasamsetti, S.B.; Kotamraju, S.; Narsaiah, B. Synthesis of novel 1,2,3-triazole substituted- N -alkyl/aryl nitrone derivatives, their anti-inflammatory and anti-cancer activity. Eur. J. Med. Chem. 2014, 80, 184–191. [Google Scholar]
- Alminderej, F.M.; Elganzory, H.H.; El-Bayaa, M.N.; Awad, H.M.; El-Sayed, W.A. Synthesis and cytotoxic activity of new 1, 3, 4-thiadiazole thioglycosides and 1, 2, 3-triazolyl-1, 3, 4-thiadiazole N-glycosides. Molecules 2019, 24, 3738. [Google Scholar] [CrossRef] [PubMed]
- Gür, M.; Yerlikaya, S.; Şener, N.; Özkınalı, S.; Baloglu, M.C.; Gökçe, H.; Altunoglu, Y.C.; Demir, S.; Şener, İ. Antiproliferative-antimicrobial properties and structural analysis of newly synthesized Schiff bases derived from some 1, 3, 4-thiadiazole compounds. J. Mol. Struct. 2020, 1219, 128570. [Google Scholar] [CrossRef]
- El-Naggar, M.; Sallam, H.A.; Shaban, S.S.; Abdel-Wahab, S.S.; EAmr, A.E.G.; Azab, M.E.; Nossier, E.S.; Al-Omar, M.A. Design, synthesis, and molecular docking study of novel heterocycles incorporating 1, 3, 4-thiadiazole moiety as potential antimicrobial and anticancer agents. Molecules 2019, 24, 1066. [Google Scholar] [CrossRef] [PubMed]
- Khattab, R.R.; Alshamari, A.K.; Hassan, A.A.; Elganzory, H.H.; El-Sayed, W.A.; Awad, H.M.; Nossier, E.S.; Hassan, N.A. Click chemistry based synthesis, cytotoxic activity and molecular docking of novel triazole-thienopyrimidine hybrid glycosides targeting EGFR. J. Enzym. Inhib. Med. Chem. 2021, 36, 504–516. [Google Scholar] [CrossRef]
- Medvedev, A.; Buneeva, O.; Gnedenko, O.V.; Ershov, P.V.; Ivanov, A. Isatin, an endogenous nonpeptide biofactor: A review of its molecular targets, mechanisms of actions, and their biomedical implications. BioFactors 2018, 44, 95–108. [Google Scholar] [CrossRef]
- Hu, H.; Wu, J.; Ao, M.; Zhou, X.; Li, B.; Cui, Z.; Wu, T.; Wang, L.; Xue, Y.; Wu, Z.; et al. Design, synthesis and biological evaluation of methylenehydrazine-1-carboxamide derivatives with (5-((4-(pyridin-3-yl) pyrimidin-2-yl) amino)-1H-indole scaffold: Novel potential CDK9 inhibitors. Bioorg. Chem. 2020, 102, 104064. [Google Scholar] [CrossRef]
- Al-Warhi, T.; El Kerdawy, A.M.; Aljaeed, N.; Ismael, O.E.; Ayyad, R.R.; Eldehna, W.M.; Abdel-Aziz, H.A.; Al-Ansary, G.H. Synthesis, biological evaluation and in silico studies of certain oxindole–indole conjugates as anticancer CDK inhibitors. Molecules 2020, 25, 2031. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, A.M.; Al-Qalawi, H.R.; El-Sayed, W.A.; Arafa, W.A.; Alhumaimess, M.S.; Hassan, A.K. Anticancer activity of newly synthesized triazolopyrimidine derivatives and their nucleoside analogs. Acta Pol. Pharm. Drug Res. 2015, 72, 307–318. [Google Scholar]
- Rahman, A.A.; Nassar, I.F.; Shaban, A.K.; El-Kady, D.S.; Awad, H.M.; El Sayed, W.A. Synthesis, docking studies into CDK-2 and anticancer activity of new derivatives based pyrimidine scaffold and their derived glycosides. Mini. Rev. Med. Chem. 2019, 19, 1093–1110. [Google Scholar] [CrossRef]
- Halay, E.; Ay, E.; Şalva, E.; Ay, K.; Karayıldırım, T. Syntheses of 1,2,3-triazolebridged pyranose sugars with purine and pyrimidine nucleobases and evaluation of their anticancer potential. Nucleosides Nucleotides Nucleic Acids 2017, 36, 598–619. [Google Scholar]
- Ghorab, M.M.; Alsaid, M.S.; Soliman, A.M. Dual EGFR/HER2 inhibitors and apoptosis inducers: New benzo [g] quinazoline derivatives bearing benzenesulfonamide as anticancer and radiosensitizers. Bioorg. Chem. 2018, 80, 611–620. [Google Scholar] [CrossRef] [PubMed]
- Das, D.; Xie, L.; Wang, J.; Xu, X.; Zhang, Z.; Shi, J.; Le, X.; Hong, J. Discovery of new quinazoline derivatives as irreversible dual EGFR/HER2 inhibitors and their anticancer activities–Part 1. Bioorganic Med. Chem. Lett. 2019, 29, 591–596. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Sami, A.; Xiang, J. HER2-directed therapy: Current treatment options for HER2-positive breast cancer. Breast Cancer 2015, 22, 101–116. [Google Scholar] [CrossRef]
- Ross, J.S.; Slodkowska, E.A.; Symmans, W.F.; Pusztai, L.; Ravdin, P.M.; Hortobagyi, G.N. The HER-2 receptor and breast cancer: Ten years of targeted anti–HER-2 therapy and personalized medicine. Oncologist 2009, 14, 320–368. [Google Scholar] [CrossRef]
- Eroglu, Z.; Tagawa, T.; Somlo, G. Human epidermal growth factor receptor familytargeted therapies in the treatment of HER2-overexpressing breast cancer. Oncologist 2014, 19, 135–150. [Google Scholar] [CrossRef]
- Hsu, J.L.; Hung, M.-C. The role of HER2, EGFR, and other receptor tyrosine kinases in breast cancer. Cancer Metastasis Rev. 2016, 35, 575–588. [Google Scholar] [CrossRef]
- Elferink, L.A.; Resto, V.A. Receptor-Tyrosine-Kinase-Targeted Therapies for Head and Neck Cancer. J. Signal Transduct. 2011, 2011, 982879. [Google Scholar] [CrossRef]
- Schuler, M.; Yang, J.H.; Park, K.; Kim, J.H.; Bennouna, J.; Chen, Y.M.; Chouaid, C.; De Marinis, F.; Feng, J.F.; Grossi, F.; et al. Afatinib beyond progression in patients with non-small-cell lung cancer following chemotherapy, erlotinib/gefitinib and afatinib: Phase III randomized LUX-Lung 5 trial. Ann. Oncol. 2016, 27, 417–423. [Google Scholar] [CrossRef]
- El-Sharief, A.M.S.; Ammar, Y.A.; Belal, A.; El-Sharief, M.A.S.; Mohamed, Y.A.; Mehany, A.B.; Ali, G.A.E.; Ragab, A. Design, synthesis, molecular docking and biological activity evaluation of some novel indole derivatives as potent anticancer active agents and apoptosis inducers. Bioorg. Chem. 2019, 85, 399–412. [Google Scholar] [CrossRef]
- Nagarsenkar, A.; Prajapti, S.K.; Guggilapu, S.D.; Birineni, S.; Kotapalli, S.S.; Ummanni, R.; Babu, B.N. Investigation of triazole-linked indole and oxindole glycoconjugates as potential anticancer agents: Novel Akt/PKB signaling pathway inhibitors. Med. Chem. Comm. 2016, 7, 646–653. [Google Scholar] [CrossRef]
- Abbas, H.S.; el Sayed, W.A.; Fathy, N.M. Synthesis and antitumor activity of new dihydropyridine thioglycosides and their corresponding dehydrogenated forms. Eur. J. Med. Chem. 2010, 45, 973–982. [Google Scholar] [CrossRef]
- Nassar, I.F.; el Kady, D.S.; Awad, H.M.; El-Sayed, W.A. Design, Synthesis, and Anticancer Activity of New Oxadiazolyl-Linked and Thiazolyl-Linked Benzimidazole Arylidines, Thioglycoside, and Acyclic Analogs. J. Heterocycl. Chem. 2019, 56, 1086–1100. [Google Scholar] [CrossRef]
- Kassem, A.F.; Abbas, E.M.H.; El-Kady, D.S.; Awad, H.M.; El-Sayed, W.A. Design, synthesis and anticancer activity of new thiazole-tetrazole or triazole hybrid glycosides targeting CDK-2 via structure-based virtual screening. Mini Rev. Med. Chem. 2019, 19, 933–948. [Google Scholar] [CrossRef]
- Yousif, M.N.M.; El-Sayed, W.A.; Abbas, H.A.S.; Awad, H.M.; Yousif, N.M. Anticancer activity of new substituted pyrimidines, their thioglycosides and thiazolopyrimidine derivatives. J. Appl. Pharm. Sci. 2019, 7, 21–32. [Google Scholar]
- Flefel, E.E.; Tantawy, W.A.; El-Sayed, W.A.; Sayed, H.H.; Fathy, N.M. Synthesis and Anticancer Activity of New Substituted Pyrazoles and Their Derived 1,2,4-Triazoles and Their Sugar Derivatives. J. Hetererocyclic Chem. 2013, 50, 344–350. [Google Scholar] [CrossRef]
- Abdel-Aal, M.T.; El-Sayed, W.A.; AH, A.A.; El Ashry, E.S. Synthesis of some functionalized arylaminomethyl-1, 2, 4-triazoles, 1, 3, 4-oxa-and thiadiazoles. Die Pharmazie 2003, 58, 788–792. [Google Scholar]
- Srour, A.M.; El-Bayaa, M.N.; Omran, M.M.; Sharaky, M.M.; El-Sayed, W.A. Synthesis and Cytotoxic Properties of New Substituted Glycosides-Indole Conjugates as Apoptosis Inducers in Cancer Cells. Anti-Cancer Agents Med. Chem. 2021, 20, 450–463. [Google Scholar] [CrossRef] [PubMed]
- Hassan, A.S.; Moustafa, G.O.; Awad, H.M.; Nossier, E.S.; Mady, M.F. Design, Synthesis, Anticancer Evaluation, Enzymatic Assays, and a Molecular Modeling Study of Novel Pyrazole–Indole Hybrids. ACS Omega 2021, 6, 12361–12374. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Meguid, E.A.; Moustafa, G.O.; Awad, H.M.; Zaki, E.R.; Nossier, E.S. Novel benzothiazole hybrids targeting EGFR: Design, synthesis, biological evaluation and molecular docking studies. J. Mol. Struct. 2021, 1240, 130595. [Google Scholar] [CrossRef]
- Othman, I.M.; Gad-Elkareem, M.A.; Amr, A.E.G.E.; Al-Omar, M.A.; Nossier, E.S.; Elsayed, E.A. Novel heterocyclic hybrids of pyrazole targeting dihydrofolate reductase: Design, biological evaluation and in silico studies. J. Enzym. Inhib. Med. Chem. 2020, 35, 1491–1502. [Google Scholar] [CrossRef] [PubMed]
- Hashem, H.E.; Amr, A.E.G.E.; Nossier, E.S.; Elsayed, E.A.; Azmy, E.M. Synthesis, antimicrobial activity and molecular docking of novel thiourea derivatives tagged with thiadiazole, imidazole and triazine moieties as potential DNA gyrase and topoisomerase IV inhibitors. Molecules 2020, 25, 2766. [Google Scholar] [CrossRef]
- Khattab, R.R.; Hassan, A.A.; AOsman, D.A.; Abdel-Megeid, F.M.; Awad, H.M.; Nossier, E.S.; El-Sayed, W.A. Synthesis, anticancer activity and molecular docking of new triazolo [4, 5-d] pyrimidines based thienopyrimidine system and their derived N-glycosides and thioglycosides. Nucleosides Nucleotides Nucleic Acids 2021, 40, 1090–1113. [Google Scholar] [CrossRef]
- Yoshikawa, S.; Kukimoto-Niino, M.; Parker, L.; Handa, N.; Terada, T.; Fujimoto, T.; Terazawa, Y.; Wakiyama, M.; Sato, M.; Sano, S.; et al. Structural basis for the altered drug sensitivities of non-small cell lung cancer-associated mutants of human epidermal growth factor receptor. Oncogene 2013, 32, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, T.; Seto, M.; Banno, H.; Kawakita, Y.; Oorui, M.; Taniguchi, T.; Ohta, Y.; Tamura, T.; Nakayama, A.; Miki, H.; et al. Design and synthesis of novel human epidermal growth factor receptor 2 (HER2)/epidermal growth factor receptor (EGFR) dual inhibitors bearing a pyrrolo [3, 2-d] pyrimidine scaffold. J. Med. Chem. 2011, 54, 8030–8050. [Google Scholar] [CrossRef]
- Abuelizz, H.A.; Marzouk, M.; Bakheit, A.H.; Awad, H.M.; Soltan, M.M.; Naglah, A.M.; Al-Salahi, R. Antiproliferative and Antiangiogenic Properties of New VEGFR-2-targeting 2-thioxobenzo [g] quinazoline Derivatives (In Vitro). Molecules 2020, 25, 5944. [Google Scholar] [CrossRef] [PubMed]
- Nossier, E.S.; El-Hallouty, S.M.; Zaki, E.R. Synthesis, anticancer evaluation and molecular modeling of some substituted thiazolidinonyl and thiazolyl pyrazole derivatives. Int. J. Pharm. Sci. 2015, 7, 353–359. [Google Scholar]
- Dawood, D.H.; Nossier, E.S.; Ali, M.M.; Mahmoud, A.E. Synthesis and molecular docking study of new pyrazole derivatives as potent anti-breast cancer agents targeting VEGFR-2 kinase. Bioorg. Chem. 2020, 101, 103916. [Google Scholar] [CrossRef]
- Othman, I.M.; Alamshany, Z.M.; Tashkandi, N.Y.; Gad-Elkareem, M.A.; Anwar, M.M.; Nossier, E.S. New pyrimidine and pyrazole-based compounds as potential EGFR inhibitors: Synthesis, anticancer, antimicrobial evaluation and computational studies. Bioorg. Chem. 2021, 114, 105078. [Google Scholar] [CrossRef] [PubMed]





| Compound No. | IC50 (µM) ± SD | |
|---|---|---|
| HCT-116 | MCF-7 | |
| 1 | 46.9 ± 0.5 | 18.1 ± 0.2 |
| 2 | 52.7 ± 0.6 | 14.1 ± 0.3 |
| 3 | 30.8 ± 0.4 | 1.1 ± 0.1 |
| 5 | 22.6 ± 0.2 | 13.9 ± 0.2 |
| 6 | 20.8 ± 0.2 | 10.8 ± 1.5 |
| 7 | 15.5 ± 0.2 | 0.5 ± 0.0 |
| 8 | 4.6 ± 0.1 | 4.2 ± 0.1 |
| 9a | 42.3 ± 0.6 | 24.1 ± 0.3 |
| 9b | 33.3 ± 0.4 | 26.4 ± 0.3 |
| 11 | 15.4 ± 0.2 | 0.8 ± 0.0 |
| 12 | 2.2 ± 0.1 | 5.7 ± 0.1 |
| 13 | 38.2 ± 0.5 | 34.4 ± 0.4 |
| 14 | 33.8 ± 0.3 | 35.3 ± 0.3 |
| 15 | 11.4 ± 0.3 | 0.6 ±0.0 |
| 16 | 35.7 ± 0.4 | 20.3 ± 0.9 |
| 17 | 26.5 ± 0.3 | 30.1 ± 0.4 |
| 18 | 28.8 ± 0.1 | 31.6 ± 0.5 |
| Doxorubicin | 12.1 ± 0.2 | 9.4 ± 0.1 |
| Compounds | IC50 (Mean ± SEM) (µM) | ||
|---|---|---|---|
| EGFRWT | EGFRT790M | HER-2 | |
| 7 | 0.30 ± 0.01 | 1.12 ± 0.55 | 2.25 ± 0.30 |
| 8 | 0.38 ± 0.05 | 1.95 ± 0.02 | 2.77 ± 0.13 |
| 11 | 6.45 ± 0.10 | 11.80 ± 0.05 | 8.60 ± 0.20 |
| 12 | 12.70 ± 0.15 | 15.22 ± 0.20 | 18. 40 ± 0.12 |
| 15 | 0.21 ± 0.05 | 1.50 ± 0.11 | 2.18 ± 0.42 |
| Erlotinib | 0.13 ± 0.01 | 0.62 ± 0.10 | 1.40 ± 0.05 |
| Compd. | Apoptosis | Necrosis | ||
|---|---|---|---|---|
| Total | Early | Late | ||
| 7/MCF-7 | 34.61 | 4.39 | 18.81 | 11.41 |
| 15/MCF-7 | 42.45 | 2.69 | 25.48 | 14.28 |
| Cont./MCF-7 | 1.99 | 0.46 | 0.28 | 1.25 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elganzory, H.H.; Alminderej, F.M.; El-Bayaa, M.N.; Awad, H.M.; Nossier, E.S.; El-Sayed, W.A. Design, Synthesis, Anticancer Activity and Molecular Docking of New 1,2,3-Triazole-Based Glycosides Bearing 1,3,4-Thiadiazolyl, Indolyl and Arylacetamide Scaffolds. Molecules 2022, 27, 6960. https://doi.org/10.3390/molecules27206960
Elganzory HH, Alminderej FM, El-Bayaa MN, Awad HM, Nossier ES, El-Sayed WA. Design, Synthesis, Anticancer Activity and Molecular Docking of New 1,2,3-Triazole-Based Glycosides Bearing 1,3,4-Thiadiazolyl, Indolyl and Arylacetamide Scaffolds. Molecules. 2022; 27(20):6960. https://doi.org/10.3390/molecules27206960
Chicago/Turabian StyleElganzory, Hussein H., Fahad M. Alminderej, Mohamed N. El-Bayaa, Hanem M. Awad, Eman S. Nossier, and Wael A. El-Sayed. 2022. "Design, Synthesis, Anticancer Activity and Molecular Docking of New 1,2,3-Triazole-Based Glycosides Bearing 1,3,4-Thiadiazolyl, Indolyl and Arylacetamide Scaffolds" Molecules 27, no. 20: 6960. https://doi.org/10.3390/molecules27206960
APA StyleElganzory, H. H., Alminderej, F. M., El-Bayaa, M. N., Awad, H. M., Nossier, E. S., & El-Sayed, W. A. (2022). Design, Synthesis, Anticancer Activity and Molecular Docking of New 1,2,3-Triazole-Based Glycosides Bearing 1,3,4-Thiadiazolyl, Indolyl and Arylacetamide Scaffolds. Molecules, 27(20), 6960. https://doi.org/10.3390/molecules27206960







