Preparation and Characterization of Simvastatin-Loaded PCL/PEG Nanofiber Membranes for Drug Sustained Release
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Nanofiber Membrane
2.3. Characterization
2.4. In Vitro Drug Release Test
2.5. In Vitro Biocompatibility Test
3. Results and Discussion
3.1. Property Characterization of Spinning Solutions
3.2. Morphology Analysis of SIM and SA/SIM Particles
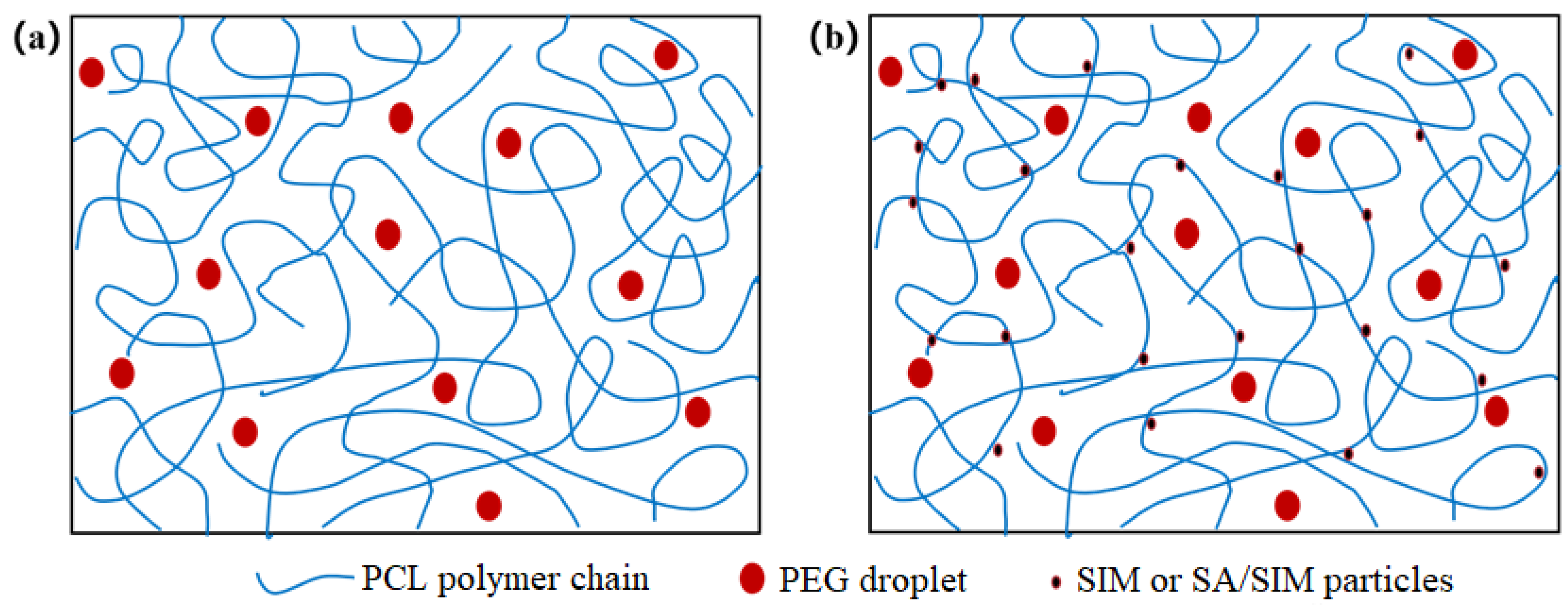
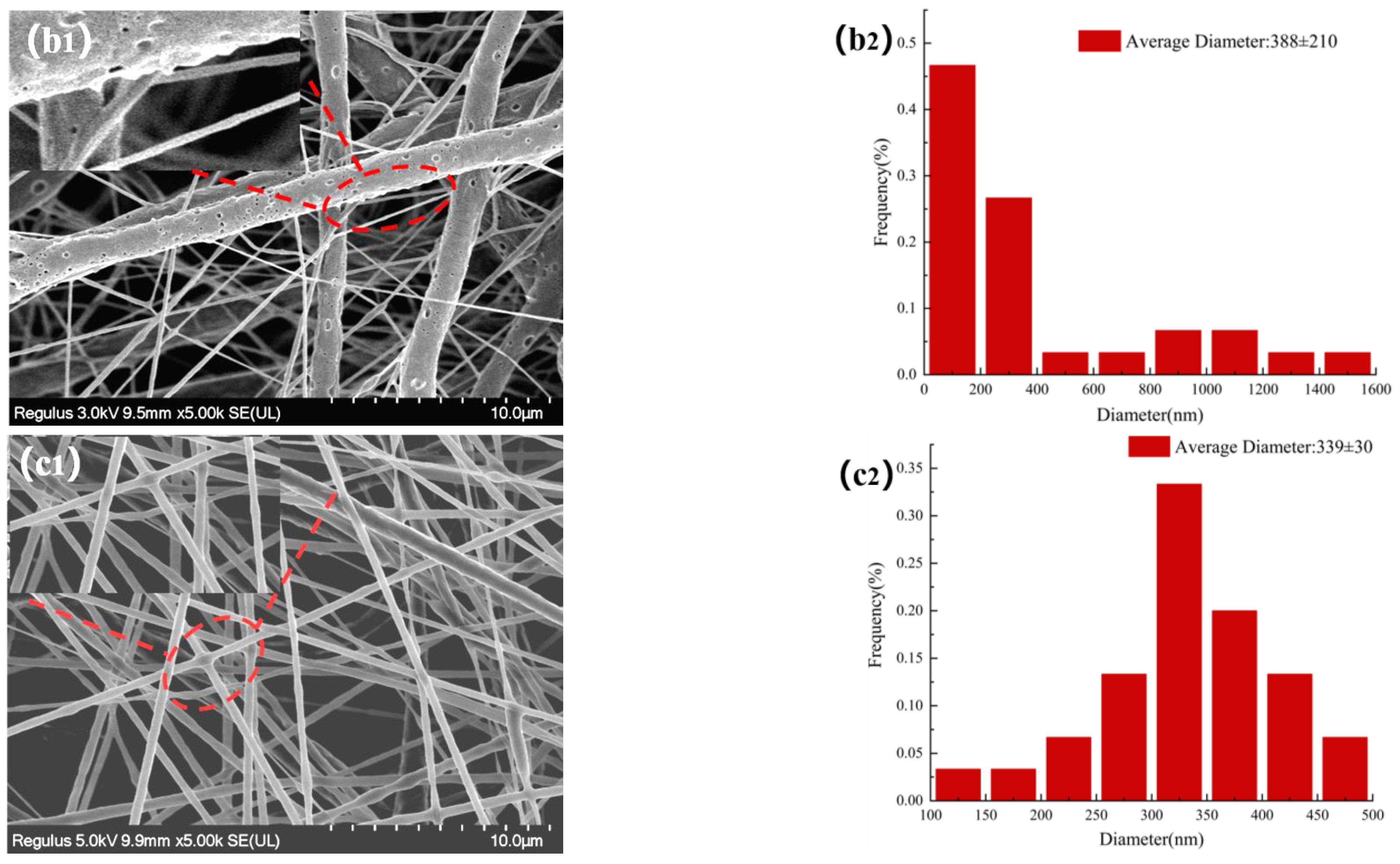
3.3. Morphology Characterization of NFMs
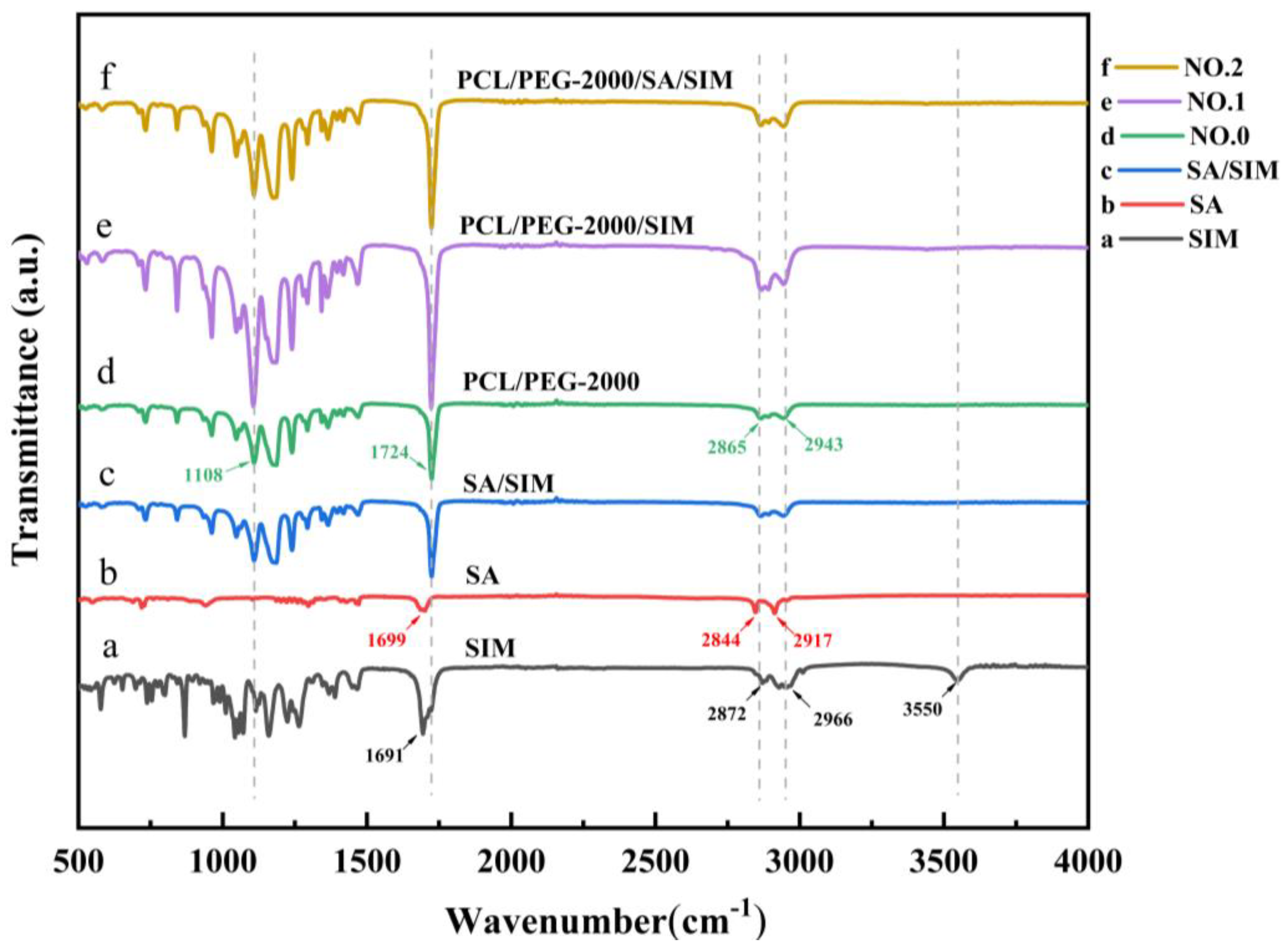
3.4. Fourier Transform Infrared (FTIR) Analysis
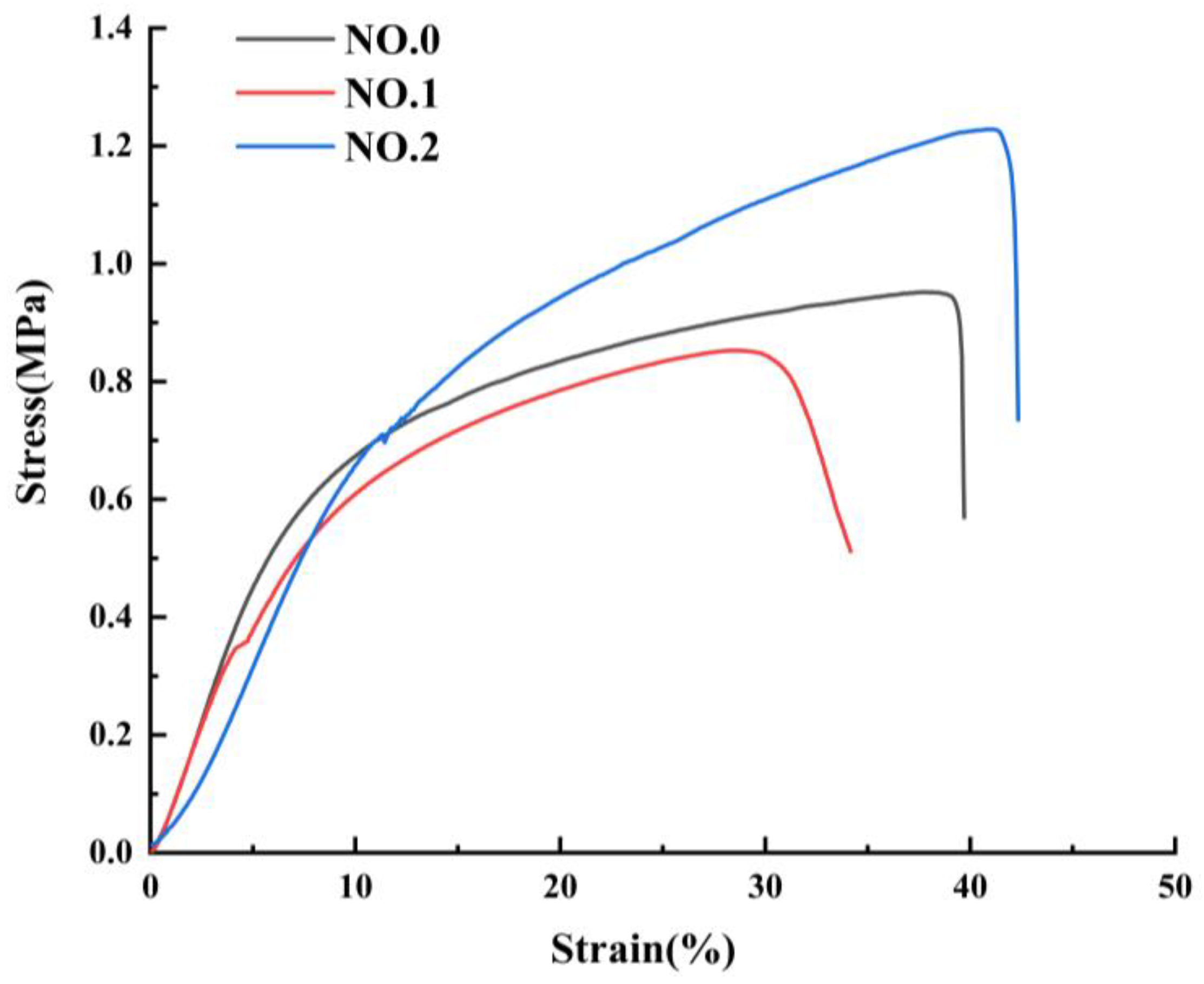
3.5. Mechanical Properties of NFMs
3.6. Wetting Properties of Nanofiber Membranes
3.7. Analysis of Drug Release Performance
3.8. Biocompatibility of NMFs
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Aminu, N.; Bello, I.; Umar, N.M.; Tanko, N.; Aminu, A.; Audu, M.M. The influence of nanoparticulate drug delivery systems in drug therapy. J. Drug Deliv. Sci. Technol. 2020, 60, 101961. [Google Scholar] [CrossRef]
- Yin, J.; Xu, L.; Ahmed, A. Batch preparation and characterization of electrospun porous polylactic acid-based nanofiber membranes for antibacterial wound dressing. Adv. Fiber Mater. 2022, 4, 832–844. [Google Scholar] [CrossRef]
- Xie, X.; Yu, J.; Zhao, Z.; Zheng, Z.; Xie, M.; Wang, X.; Han, Z.; Li, G. Fabrication and drug release properties of curcuminloaded silk fibroin nanofibrous membranes. Adsorpt. Sci. Technol. 2019, 37, 412–424. [Google Scholar] [CrossRef]
- Allafchian, A.; Hosseini, H.; Ghoreishi, S.M. Electrospinning of PVA carboxymethyl cellulose nanofibers for flufenamic acid drug delivery. Int. J. Biol. Macromol. 2020, 163, 1780–1786. [Google Scholar] [CrossRef]
- Wang, F.; Sun, Z.; Yin, J.; Xu, L. Preparation, characterization and properties of porous PLA/PEG/Curcumin composite nanofibers for antibacterial application. Nanomaterials 2019, 9, 508. [Google Scholar] [CrossRef]
- Rasouli, M.; Pirsalami, S.; Zebarjad, S.M. Study on the formation and structural evolution of bead-on-string in electrospun polysulfone mats. Polym. Int. 2020, 69, 822–832. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, L. Preparation and characterization of porous core-shell fibers for slow release of tea polyphenols. Polymers 2018, 10, 144. [Google Scholar] [CrossRef]
- Lu, J.; Liu, J.G.; Song, X.F.; Chen, X.S.; Wu, X.D. Sandwich structure-like meshes fabricated via electrospinning for controllable release of zoledronic acid. Chem. Res. Chin. Univ. 2011, 27, 524–527. [Google Scholar]
- Luraghi, A.; Peri, F.; Moroni, L. Electrospinning for drug delivery applications: A review. J Control Release 2021, 334, 463–484. [Google Scholar] [CrossRef]
- Sung, Y.K.; Kim, S.W. Recent advances in polymeric drug delivery systems. Biomater. Res. 2020, 24, 12. [Google Scholar] [CrossRef]
- He, F.L.; Deng, X.; Zhou, Y.Q.; Zhang, T.D.; Liu, Y.L.; Ye, Y.J.; Yin, D.C. Controlled release of antibiotics from poly-epsilon-caprolactone/polyethylene glycol wound dressing fabricated by direct-writing melt electrospinning. Polym. Adv. Technol. 2019, 30, 425–434. [Google Scholar] [CrossRef]
- Yin, J.; Xu, L. Batch preparation of electrospun polycaprolactone/chitosan/aloe vera blended nanofiber membranes for novel wound dressing. Int. J. Biol. Macromol. 2020, 160, 352–363. [Google Scholar] [CrossRef] [PubMed]
- Croitoru, A.M.; Karacelebi, Y.; Saatcioglu, E.; Altan, E.; Ulag, S.; Aydoğan, H.K.; Sahin, A.; Motelica, L.; Oprea, O.; Tihauan, B.M.; et al. Electrically triggered drug delivery from novel electrospun poly(lactic acid)/graphene oxide/quercetin fibrous scaffolds for wound dressing applications. Pharmaceutics 2021, 13, 957. [Google Scholar] [CrossRef] [PubMed]
- Al-Baadani, M.A.; Yie, K.H.R.; Al-Bishari, A.M.; Alshobi, B.A.; Zhou, Z.; Fang, K.; Dai, B.; Shen, Y.; Ma, J.; Liu, J.; et al. Co-electrospinning polycaprolactone/gelatin membrane as a tunable drug delivery system for bone tissue regeneration. Mater. Des. 2021, 209, 109962. [Google Scholar] [CrossRef]
- Pant, B.; Park, M.; Park, S.J. Drug delivery applications of core-sheath nanofibers prepared by coaxial electrospinning: A review. Pharmaceutics 2019, 11, 305. [Google Scholar] [CrossRef]
- Shepa, I.; Mudra, E.; Dusza, J. Electrospinning through the Prism of Time. Mater. Today Chem. 2021, 21, 100543. [Google Scholar] [CrossRef]
- Xie, X.; Chen, Y.; Wang, X.; Xu, X.; Shen, Y.; Khan, A.U.R.; Aldalbahi, A.; Fetz, A.E.; Bowlin, G.L.; El-Newehy, M.; et al. Electrospinning Nanofiber Scaffolds for Soft and Hard Tissue Regeneration. J. Mater. Sci. Technol. 2020, 59, 243–261. [Google Scholar] [CrossRef]
- Hemamalini, T.; Giri Dev, V.R. Comprehensive Review on Electrospinning of Starch Polymer for Biomedical Applications. Int. J. Biol. Macromol. 2018, 106, 712–718. [Google Scholar] [CrossRef]
- Zeng, J.; Yang, L.; Liang, Q.; Zhang, X.; Guan, H.; Xu, X.; Chen, X.; Jing, X. Influence of the drug compatibility with polymer solution on the release kinetics of electrospun fiber formulation. J. Control. Release 2005, 105, 43–51. [Google Scholar] [CrossRef]
- Del Valle, L.J.; Camps, R.; Diaz, A.; Franco, L.; Rodriguez-Galan, A.; Puiggali, J. Electrospinning of polylactide and polycaprolactone mixtures for preparation of materials with tunable drug release properties. J Polym. Res. 2011, 18, 1903–1917. [Google Scholar] [CrossRef]
- Rathinavel, S.; Korrapati, P.S.; Kalaiselvi, P.; Dharmalingam, S. Mesoporous silica incorporated PCL/curcumin nanofiber for wound healing application. Eur. J. Pharm. Sci. 2021, 167, 106021. [Google Scholar] [CrossRef] [PubMed]
- Yousefi, P.; Dini, G.; Movahedi, B.; Vaezifar, S.; Mehdikhani, M. Polycaprolactone/chitosan core/shell nanofibrous mat fabricated by electrospinning process as carrier for rosuvastatin drug. Polym. Bull. 2022, 79, 1627–1645. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhu, J.; Zhang, J.; Chen, Z.; Li, W.; Deng, L.; Chen, K.; Wan, H.; Li, J.; Li, R. Optimization of biodegradable PEG/PLGA nanofiber mats electrospinning process for anti-adhesion application: Research article. J. Appl. Polym. Sci. 2018, 135, 46282. [Google Scholar] [CrossRef]
- Bui, H.T.; Chung, O.H.; Dela Cruz, J.; Park, J.S. Fabrication and characterization of electrospun curcumin-loaded polycaprolactone-polyethylene glycol nanofibers for enhanced wound healing. Macromol. Res. 2014, 22, 1288–1296. [Google Scholar] [CrossRef]
- Riaz, T.; Khenoussi, N.; Rata, D.M.; Atanase, L.I.; Adolphe, D.C.; Delaite, C. Blend electrospinning of poly(ɛ-caprolactone) and poly(ethylene glycol-400) nanofibers loaded with ibuprofen as a potential drug delivery system for wound dressings. Autex Res. J. 2021; ahead of print. [Google Scholar] [CrossRef]
- Dehghan, F.; Gholipour-Kanani, A.; Kamali Dolatabadi, M.; Bahrami, S.H. Nanofibrous composite from polycaprolactone-polyethylene glycol-aloe vera as a promising scaffold for bone repairing. J. Appl. Polym. Sci. 2022, 139, e52463. [Google Scholar] [CrossRef]
- Yuan, X.; Zhang, M.; Wang, Y.; Zhao, H.; Sun, D. Using co-axial electrospray deposition to eliminate burst release of simvastatin from microparticles and to enhance induced osteogenesis. J. Biomater. Sci. Polym. Ed. 2019, 30, 355–375. [Google Scholar] [CrossRef]
- Hu, F.Q.; Liu, L.N.; Du, Y.Z.; Yuan, H. Synthesis and antitumor activity of doxorubicin conjugated stearic acid-g-chitosan oligosaccharide polymeric micelles. Biomaterials 2009, 30, 6955–6963. [Google Scholar] [CrossRef]
- Hajiabbas, M.; Alemzadeh, I.; Vossoughi, M. Hybrid silk fibroin-gelatin nanofibrous sheet for drug delivery and regenerative medicine: In-vitro characterization and controlled release of simvastatin/protein. Polym. Advan. Technol. 2020, 32, 1333–1344. [Google Scholar] [CrossRef]
- Betha, S.; Reddy, B.P.; Varma, M.M.; Raju, D.B.; Kolapalli, V.R.M. Development of simvastatin electrospun fibers: A novel approach for sustained drug delivery. J. Pharm. Investig. 2015, 45, 13–22. [Google Scholar] [CrossRef]
- Pan, L.; Cheng, T.T.; Xu, L. Preparation of polycaprolactone/polyethylene glycol nanofiber membranes with large pore sizes and its application for tissue engineering scaffold. J. Text. Res. 2020, 41, 167–173. [Google Scholar]
- Maccaferri, E.; Cocchi, D.; Mazzocchetti, L.; Benelli, T.; Brugo, T.M.; Giorgini, L.; Zucchelli, A. How nanofibers carry the load: Toward a universal and reliable approach for tensile testing of polymeric nanofibrous membranes. Macromol. Mater. Eng. 2021, 306, 2100183. [Google Scholar] [CrossRef]
- Yang, B.; Xu, L.; Wang, Q.; Li, S. Modulation of the wettability of excipients by surfactant and its impacts on the disintegration and release of tablets. Drug Dev. Ind. Pharm. 2016, 42, 1945–1955. [Google Scholar] [CrossRef] [PubMed]
- Aluigi, A.; Varesano, A.; Vineis, C.; Del Rio, A. Electrospinning of immiscible systems: The wool keratin/polyamide-6 case study. Mater. Des. 2017, 127, 144–153. [Google Scholar] [CrossRef]
- Maccaferri, E.; Mazzocchetti, L.; Benelli, T.; Brugo, T.M.; Zucchelli, A.; Giorgini, L. Rubbery nanofibers by co-electrospinning of almost immiscible NBR and PCL blends. Mater. Des. 2020, 186, 108210. [Google Scholar] [CrossRef]
- Maccaferri, E.; Mazzocchetti, L.; Benelli, T.; Brugo, T.M.; Zucchelli, A.; Giorgini, L. Self-assembled NBR/nomex nanofibers as lightweight rubbery nonwovens for hindering delamination in epoxy CFRPs. ACS Appl. Mater. Interfaces 2022, 14, 1885–1899. [Google Scholar] [CrossRef]
- Tipduangta, P.; Belton, P.; Fábián, L.; Wang, L.Y.; Tang, H.; Eddleston, M.; Qi, S. Electrospun polymer blend nanofibers for tunable drug delivery: The role of transformative phase separation on controlling the release rate. Mol. Pharm. 2016, 13, 25–39. [Google Scholar] [CrossRef] [PubMed]
- Walsh, D.J. Polymer blends. In Comprehensive Polymer Science and Supplements; Geoffrey, A., John, C.B., Eds.; Elsevier: Amsterdam, The Netherlands, 1989; Volume 2, pp. 135–154. [Google Scholar]
- Lin, W.-J.; Flanagan, D.R.; Linhardt, R.J. A novel fabrication of poly(ε-caprolactone) microspheres from blends of poly(ε-caprolactone) and poly(ethylene glycol)s. Polymer 1999, 40, 1731–1735. [Google Scholar] [CrossRef]
- Rostami, F.; Tamjid, E.; Behmanesh, M. Drug-eluting PCL/graphene oxide nanocomposite scaffolds for enhanced osteogenic differentiation of mesenchymal stem cells. Mater. Sci. Eng. C 2020, 115, 111102. [Google Scholar] [CrossRef]
- Molnar, K.; Vas, L.M.; Czigany, T. Determination of tensile strength of electrospun single nanofibers through modeling tensile behavior of the nanofibrous mat. Compos. Part B-Eng. 2012, 43, 15–21. [Google Scholar] [CrossRef]
- Maccaferri, E.; Mazzocchetti, L.; Benelli, T.; Brugo, T.M.; Zucchelli, A.; Giorgini, L. Rubbery-modified CFRPs with improved mode I fracture toughness: Effect of nanofibrous mat grammage and positioning on tan delta behaviour. Polymers 2021, 13, 1918. [Google Scholar] [CrossRef] [PubMed]
- Maccaferri, E.; Ortolani, J.; Mazzocchetti, L.; Benelli, T.; Brugo, T.M.; Zucchelli, A.; Giorgini, L. New application field of polyethylene oxide: PEO nanofibers as epoxy toughener for effective CFRP delamination resistance improvement. ACS Omega 2022, 7, 23189–23200. [Google Scholar] [CrossRef]
- Li, W.; Zong, Y.; Liu, Q.; Sun, Y.; Li, Z.; Wang, H.; Li, Z. A highly stretchable and biodegradable superamphiphobic fluorinated polycaprolactone nanofibrous membrane for antifouling. Prog. Org. Coat. 2020, 147, 105776. [Google Scholar] [CrossRef]
- Yang, X.; Pu, Y.; Zhang, Y.; Liu, X.; Li, J.; Yuan, D.; Ning, X. Multifunctional composite membrane based on BaTiO3@PU/PSA nanofibers for high-efficiency PM2.5 removal. J. Hazard. Mater. 2020, 391, 122254. [Google Scholar] [CrossRef] [PubMed]
- Salam, A.; Khan, M.Q.; Hassan, T.; Hassan, N.; Nazir, A.; Hussain, T.; Azeem, M.; Kim, I.S. In-vitro assessment of appropriate hydrophilic scaffolds by co-electrospinning of poly(1,4 cyclohexane isosorbide terephthalate)/polyvinyl alcohol. Sci. Rep. 2020, 10, 19751. [Google Scholar] [CrossRef] [PubMed]


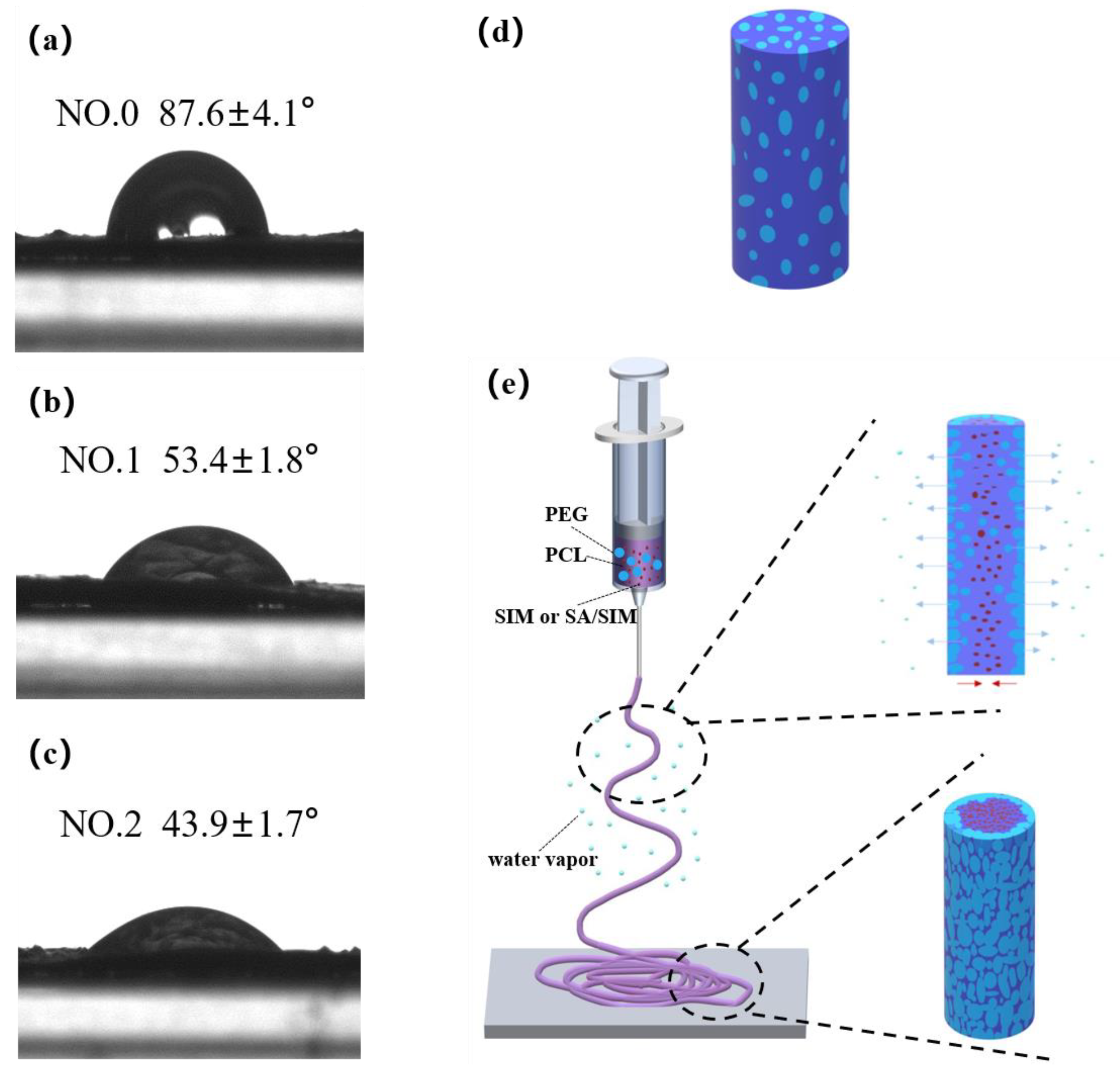
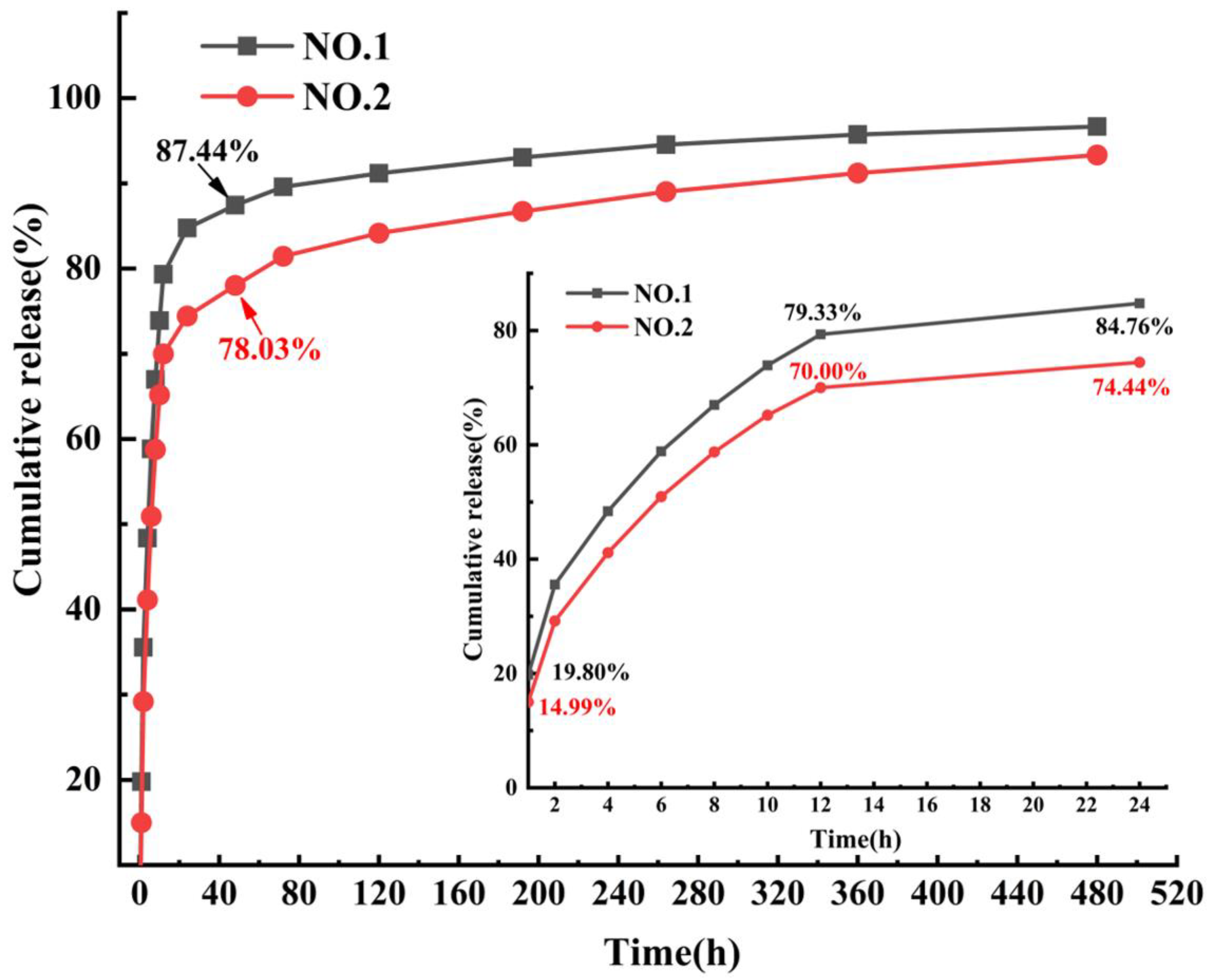
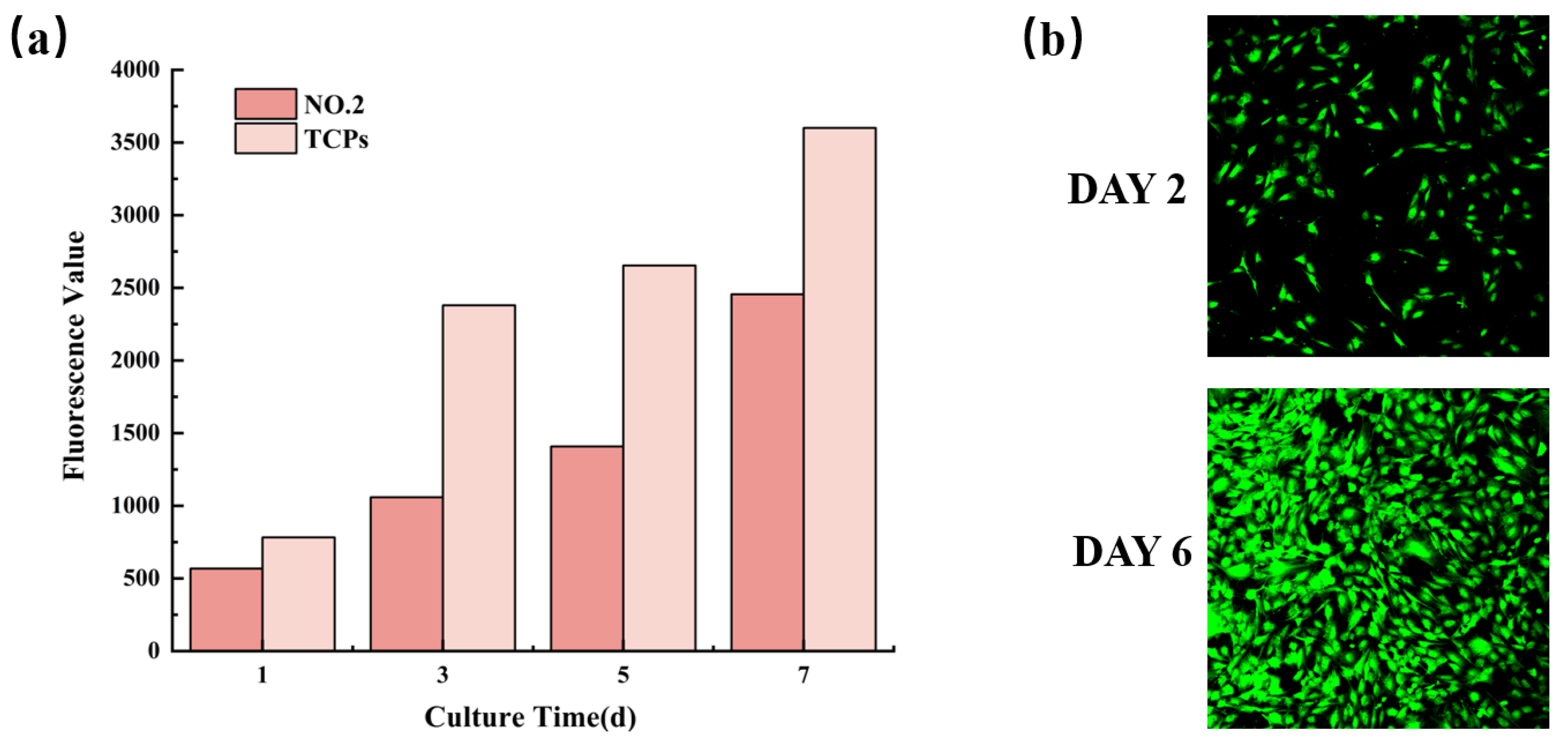
| Electrospun NFM Code | Spinning Solution Composition |
|---|---|
| NO.0 | 25 wt% PCL/PEG (PCL:PEG = 8:2) |
| NO.1 | 25 wt% PCL/PEG (PCL:PEG = 8:2) 0.16 g SIM |
| NO.2 | 25 wt% PCL/PEG (PCL:PEG = 8:2) 5 wt% SIM/3 wt% SA containing 0.16 g SIM |
| Spinning Solution Code | Viscositiy (mPa.S) | Conductitivity (μS/cm) |
|---|---|---|
| NO.0 | 307 | 7.79 |
| NO.1 | 275 | 8.15 |
| NO.2 | 126 | 14.19 |
| NFM | Thickness (mm) | Young’s Modulus (MPa) | Fracture Strength (MPa) | Elongation at Break (%) |
|---|---|---|---|---|
| NO.0 | 0.11 ± 0.03 | 0.10 ± 0.09 | 0.93 ± 0.05 | 35.27 ± 3.98 |
| NO.1 | 0.16 ± 0.04 | 0.09 ± 0.07 | 0.85 ± 0.09 | 31.04 ± 1.76 |
| NO.2 | 0.11 ± 0.03 | 0.06 ± 0.07 | 1.24 ± 0.03 | 43.89 ± 2.15 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pan, L.; Yang, J.; Xu, L. Preparation and Characterization of Simvastatin-Loaded PCL/PEG Nanofiber Membranes for Drug Sustained Release. Molecules 2022, 27, 7158. https://doi.org/10.3390/molecules27217158
Pan L, Yang J, Xu L. Preparation and Characterization of Simvastatin-Loaded PCL/PEG Nanofiber Membranes for Drug Sustained Release. Molecules. 2022; 27(21):7158. https://doi.org/10.3390/molecules27217158
Chicago/Turabian StylePan, Lu, Jiahao Yang, and Lan Xu. 2022. "Preparation and Characterization of Simvastatin-Loaded PCL/PEG Nanofiber Membranes for Drug Sustained Release" Molecules 27, no. 21: 7158. https://doi.org/10.3390/molecules27217158
APA StylePan, L., Yang, J., & Xu, L. (2022). Preparation and Characterization of Simvastatin-Loaded PCL/PEG Nanofiber Membranes for Drug Sustained Release. Molecules, 27(21), 7158. https://doi.org/10.3390/molecules27217158







