Honokiol and Alpha-Mangostin Inhibit Mayaro Virus Replication through Different Mechanisms
Abstract
:1. Introduction
2. Results
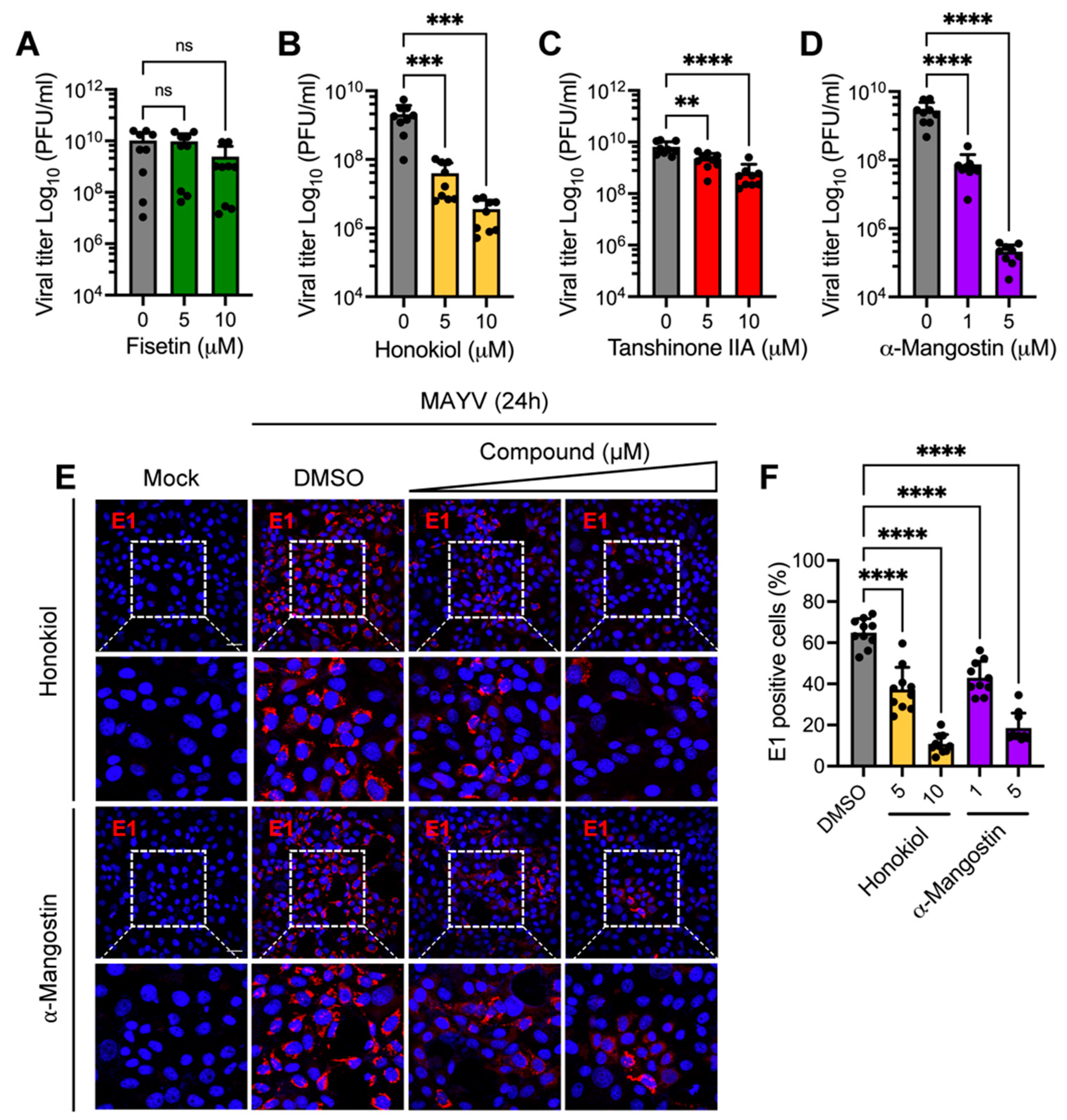
2.1. Honokiol and α-Mangostin Prevent MAYV-Induced Cytopathic Effects in Vero-E6 Cells in a Dose-Dependent Manner
2.2. Honokiol and α-Mangostin Reduce MAYV Replication in Vero-E6 Cells in a Dose-Dependent Manner
2.3. Honokiol and α-Mangostin Inhibit MAYV Progeny Production Independent of Virus Strain or Human Cell Line Tested
2.4. Pretreating HDFs with α-Mangostin, but Not Honokiol, Affects MAYV Progeny Production
2.5. Honokiol and α-Mangostin Treatment Disturb MAYV Infection at Different Stages of the Viral Life Cycle
2.6. Honokiol and α-Mangostin Downmodulate the Expression of MAYV E1 and nsP1 Proteins and Additionally, Affect Viral RNA Replication
2.7. Honokiol and α-Mangostin Also Inhibit the Una, Chikungunya, and Zika Arboviruses
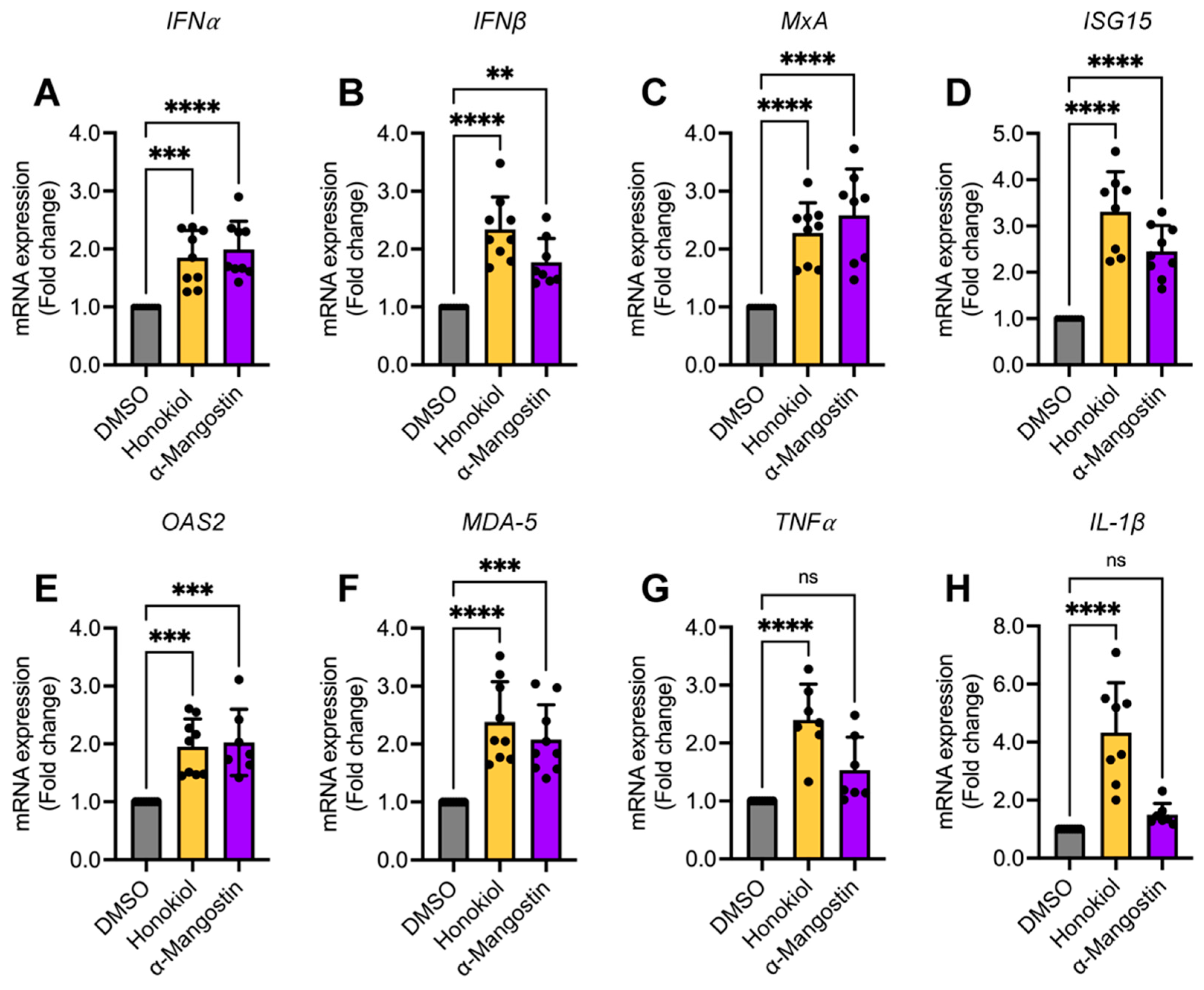
2.8. Honokiol and α-Mangostin Treatment Elicit the Expression of Type I Interferon and Specific Interferon-Stimulated Genes in HeLa Cells
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Reagents
4.2. Virus Strains and Propagation
4.3. Analysis of Cell Toxicity
4.4. Plaque-Forming Assay
4.5. Viral Infection Assay
4.6. Immunofluorescence Assay
4.7. Western Blot Assay
4.8. Gene Expression and Viral RNA Analysis by Quantitative RT-PCR
4.9. Data Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Yactayo, S.; Staples, J.E.; Millot, V.; Cibrelus, L.; Ramon-Pardo, P. Epidemiology of Chikungunya in the Americas. J. Infect. Dis. 2016, 214, S441–S445. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gubler, D.J.; Vasilakis, N.; Musso, D. History and Emergence of Zika Virus. J. Infect. Dis. 2017, 216, S860–S867. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ganjian, N.; Riviere-Cinnamond, A. Mayaro virus in Latin America and the Caribbean. Rev. Panam. Salud Publica 2020, 44, e14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aguilar-Luis, M.A.; Del Valle-Mendoza, J.; Sandoval, I.; Silva-Caso, W.; Mazulis, F.; Carrillo-Ng, H.; Tarazona-Castro, Y.; Martins-Luna, J.; Aquino-Ortega, R.; Pena-Tuesta, I.; et al. A silent public health threat: Emergence of Mayaro virus and co-infection with Dengue in Peru. BMC Res. Notes 2021, 14, 1–7. [Google Scholar] [CrossRef]
- Gonzalez-Escobar, G.; Churaman, C.; Rampersad, C.; Singh, R.; Nathaniel, S. Mayaro virus detection in patients from rural and urban areas in Trinidad and Tobago during the Chikungunya and Zika virus outbreaks. Pathog. Glob. Health 2021, 115, 188–195. [Google Scholar] [CrossRef]
- Mutricy, R.; Matheus, S.; Mosnier, E.; Martinez-Lorenzi, E.; De Laval, F.; Nacher, M.; Niemetzky, F.; Naudion, P.; Djossou, F.; Rousset, D.; et al. Mayaro virus infection in French Guiana, a cross sectional study 2003–2019. Infect. Genet. Evol. 2022, 99, 105243. [Google Scholar] [CrossRef]
- Carvalho, V.L.; Azevedo, R.S.S.; Carvalho, V.L.; Azevedo, R.S.; Henriques, D.F.; Cruz, A.C.R.; Vasconcelos, P.F.C.; Martins, L.C. Arbovirus outbreak in a rural region of the Brazilian Amazon. J. Clin. Virol. 2022, 150-151, 105155. [Google Scholar] [CrossRef]
- Acosta-Ampudia, Y.; Monsalve, D.M.; Rodriguez, Y.; Pacheco, Y.; Anaya, J.M.; Ramirez-Santana, C. Mayaro: An emerging viral threat? Emerg. Microbes Infect. 2018, 7, 1–11. [Google Scholar] [CrossRef]
- Aguilar-Luis, M.A.; Del Valle-Mendoza, J.; Silva-Caso, W.; Gil-Ramirez, T.; Levy-Blitchtein, S.; Bazan-Mayra, J.; Zavaleta-Gavidia, V.; Cornejo-Pacherres, D.; Palomares-Reyes, C.; Del Valle, L.J. An emerging public health threat: Mayaro virus increases its distribution in Peru. Int. J. Infect. Dis. 2020, 92, 253–258. [Google Scholar] [CrossRef] [Green Version]
- Suchowiecki, K.; Reid, S.P.; Simon, G.L.; Firestein, G.S.; Chang, A. Persistent Joint Pain Following Arthropod Virus Infections. Curr. Rheumatol. Rep. 2021, 23, 1–12. [Google Scholar] [CrossRef]
- Diagne, C.T.; Bengue, M.; Choumet, V.; Hamel, R.; Pompon, J.; Misse, D. Mayaro Virus Pathogenesis and Transmission Mechanisms. Pathogens 2020, 9, 738. [Google Scholar] [CrossRef]
- Dieme, C.; Ciota, A.T.; Kramer, L.D. Transmission potential of Mayaro virus by Aedes albopictus and Anopheles quadrimaculatus from the USA. Parasit Vectors 2020, 13, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Pereira, T.N.; Carvalho, F.D.; De Mendonca, S.F.; Rocha, M.N.; Moreira, L.A. Vector competence of Aedes aegypti, Aedes albopictus and Culex quinquefasciatus mosquitoes for Mayaro virus. PLoS Negl. Trop. Dis. 2020, 14, e0007518. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- da Silva Neves, N.A.; da Silva Ferreira, R.; Morais, D.O.; Pavon, J.A.R.; de Pinho, J.B.; Slhessarenko, R.D. Chikungunya, Zika, Mayaro and Equine Encephalitis virus detection in adult Culicinae from South Central Mato Grosso, Brazil, during the rainy season of 2018. Braz. J. Microbiol. 2022, 53, 63–70. [Google Scholar] [CrossRef] [PubMed]
- de Curcio, J.S.; Salem-Izacc, S.M.; Pereira Neto, L.M.; Nunes, E.B.; Anunciacao, C.E.; de Paula Silveira-Lacerda, E. Detection of Mayaro virus in Aedes aegypti mosquitoes circulating in Goiania-Goias-Brazil. Microbes Infect. 2022, 24, 104948. [Google Scholar] [CrossRef]
- Goh, V.S.L.; Mok, C.K.; Chu, J.J.H. Antiviral Natural Products for Arbovirus Infections. Molecules 2020, 25, 2796. [Google Scholar] [CrossRef]
- Thomas, E.; Stewart, L.E.; Darley, B.A.; Pham, A.M.; Esteban, I.; Panda, S.S. Plant-Based Natural Products and Extracts: Potential Source to Develop New Antiviral Drug Candidates. Molecules 2021, 26, 6197. [Google Scholar] [CrossRef]
- Mohan, S.; Elhassan Taha, M.M.; Makeen, H.A.; Alhazmi, H.A.; Al Bratty, M.; Sultana, S.; Ahsan, W.; Najmi, A.; Khalid, A. Bioactive Natural Antivirals: An Updated Review of the Available Plants and Isolated Molecules. Molecules 2020, 25, 4878. [Google Scholar] [CrossRef]
- Sagaya Jansi, R.; Khusro, A.; Agastian, P.; Alfarhan, A.; Al-Dhabi, N.A.; Arasu, M.V.; Rajagopal, R.; Barcelo, D.; Al-Tamimi, A. Emerging paradigms of viral diseases and paramount role of natural resources as antiviral agents. Sci. Total Environ. 2021, 759, 143539. [Google Scholar] [CrossRef]
- dos Santos, A.E.; Kuster, R.M.; Yamamoto, K.A.; Salles, T.S.; Campos, R.; de Meneses, M.D.; Soares, M.R.; Ferreira, D. Quercetin and quercetin 3-O-glycosides from Bauhinia longifolia (Bong.) Steud. show anti-Mayaro virus activity. Parasit Vectors 2014, 7, 130. [Google Scholar] [CrossRef]
- Ferraz, A.C.; Moraes, T.F.S.; Nizer, W.; Santos, M.D.; Totola, A.H.; Ferreira, J.M.S.; Vieira-Filho, S.A.; Rodrigues, V.G.; Duarte, L.P.; de Brito Magalhaes, C.L.; et al. Virucidal activity of proanthocyanidin against Mayaro virus. Antivir. Res. 2019, 168, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Ferraz, A.C.; Almeida, L.T.; da Silva Caetano, C.C.; da Silva Menegatto, M.B.; Souza Lima, R.L.; de Senna, J.P.N.; de Oliveira Cardoso, J.M.; Perucci, L.O.; Talvani, A.; Geraldo de Lima, W.; et al. Hepatoprotective, antioxidant, anti-inflammatory and antiviral activities of silymarin against mayaro virus infection. Antivir. Res. 2021, 194, 105168. [Google Scholar] [CrossRef] [PubMed]
- Campos, D.; Navarro, S.; Llamas-Gonzalez, Y.Y.; Sugasti, M.; Gonzalez-Santamaria, J. Broad Antiviral Activity of Ginkgolic Acid against Chikungunya, Mayaro, Una, and Zika Viruses. Viruses 2020, 12, 449. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Basu, P.; Kumar, G.S. Sanguinarine and Its Role in Chronic Diseases. Adv. Exp. Med. Biol. 2016, 928, 155–172. [Google Scholar] [CrossRef] [PubMed]
- Boulos, J.C.; Rahama, M.; Hegazy, M.F.; Efferth, T. Shikonin derivatives for cancer prevention and therapy. Cancer Lett. 2019, 459, 248–267. [Google Scholar] [CrossRef]
- Xu, Q.; Yi, L.T.; Pan, Y.; Wang, X.; Li, Y.C.; Li, J.M.; Wang, C.P.; Kong, L.D. Antidepressant-like effects of the mixture of honokiol and magnolol from the barks of Magnolia officinalis in stressed rodents. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2008, 32, 715–725. [Google Scholar] [CrossRef]
- Guo, R.; Li, L.; Su, J.; Li, S.; Duncan, S.E.; Liu, Z.; Fan, G. Pharmacological Activity and Mechanism of Tanshinone IIA in Related Diseases. Drug Des. Dev. Ther. 2020, 14, 4735–4748. [Google Scholar] [CrossRef]
- Chavan, T.; Muth, A. The diverse bioactivity of alpha-mangostin and its therapeutic implications. Future Med. Chem. 2021, 13, 1679–1694. [Google Scholar] [CrossRef]
- Pal, H.C.; Pearlman, R.L.; Afaq, F. Fisetin and Its Role in Chronic Diseases. Adv. Exp. Med. Biol. 2016, 928, 213–244. [Google Scholar] [CrossRef]
- Fu, C.; Guan, G.; Wang, H. The Anticancer Effect of Sanguinarine: A Review. Curr. Pharm. Des. 2018, 24, 2760–2764. [Google Scholar] [CrossRef]
- Matkar, S.S.; Wrischnik, L.A.; Hellmann-Blumberg, U. Sanguinarine causes DNA damage and p53-independent cell death in human colon cancer cell lines. Chem. Biol. Interact. 2008, 172, 63–71. [Google Scholar] [CrossRef]
- Wang, Q.; Wang, J.; Wang, J.; Ju, X.; Zhang, H. Molecular mechanism of shikonin inhibiting tumor growth and potential application in cancer treatment. Toxicol. Res. 2021, 10, 1077–1084. [Google Scholar] [CrossRef] [PubMed]
- Xia, S.; Lin, H.; Liu, H.; Lu, Z.; Wang, H.; Fan, S.; Li, N. Honokiol Attenuates Sepsis-Associated Acute Kidney Injury via the Inhibition of Oxidative Stress and Inflammation. Inflammation 2019, 42, 826–834. [Google Scholar] [CrossRef] [PubMed]
- Munroe, M.E.; Arbiser, J.L.; Bishop, G.A. Honokiol, a natural plant product, inhibits inflammatory signals and alleviates inflammatory arthritis. J. Immunol. 2007, 179, 753–763. [Google Scholar] [CrossRef] [Green Version]
- Yin, Q.; Wu, Y.J.; Pan, S.; Wang, D.D.; Tao, M.Q.; Pei, W.Y.; Zuo, J. Activation of Cholinergic Anti-Inflammatory Pathway in Peripheral Immune Cells Involved in Therapeutic Actions of alpha-Mangostin on Collagen-Induced Arthritis in Rats. Drug Des. Dev. Ther. 2020, 14, 1983–1993. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.H.; Lu, M.H.; Guo, D.S.; Zhai, Y.Y.; Miao, D.; Yue, J.Y.; Yuan, C.H.; Zhao, M.M.; An, D.R. Antifungal Effect of Magnolol and Honokiol from Magnolia officinalis on Alternaria alternata Causing Tobacco Brown Spot. Molecules 2019, 24, 2140. [Google Scholar] [CrossRef] [Green Version]
- Ye, H.; Wang, Q.; Zhu, F.; Feng, G.; Yan, C.; Zhang, J. Antifungal Activity of Alpha-Mangostin against Colletotrichum gloeosporioides In Vitro and In Vivo. Molecules 2020, 25, 5335. [Google Scholar] [CrossRef]
- Kataoka, S.; Umemura, A.; Okuda, K.; Taketani, H.; Seko, Y.; Nishikawa, T.; Yamaguchi, K.; Moriguchi, M.; Kanbara, Y.; Arbiser, J.L.; et al. Honokiol Acts as a Potent Anti-Fibrotic Agent in the Liver through Inhibition of TGF-beta1/SMAD Signaling and Autophagy in Hepatic Stellate Cells. Int. J. Mol. Sci. 2021, 22, 13354. [Google Scholar] [CrossRef]
- Li, R.S.; Xu, G.H.; Cao, J.; Liu, B.; Xie, H.F.; Ishii, Y.; Zhang, C.F. Alpha-Mangostin Ameliorates Bleomycin-Induced Pulmonary Fibrosis in Mice Partly Through Activating Adenosine 5′-Monophosphate-Activated Protein Kinase. Front. Pharm. 2019, 10, 1305. [Google Scholar] [CrossRef]
- Sakaue, Y.; Domon, H.; Oda, M.; Takenaka, S.; Kubo, M.; Fukuyama, Y.; Okiji, T.; Terao, Y. Anti-biofilm and bactericidal effects of magnolia bark-derived magnolol and honokiol on Streptococcus mutans. Microbiol. Immunol. 2016, 60, 10–16. [Google Scholar] [CrossRef]
- Guo, N.; Liu, Z.; Yan, Z.; Liu, Z.; Hao, K.; Liu, C.; Wang, J. Subinhibitory concentrations of Honokiol reduce alpha-Hemolysin (Hla) secretion by Staphylococcus aureus and the Hla-induced inflammatory response by inactivating the NLRP3 inflammasome. Emerg. Microbes Infect. 2019, 8, 707–716. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, S.; Zhu, C.; Li, H.; Chen, Y.; Liu, S. Potent in vitro and in vivo antimicrobial activity of semisynthetic amphiphilic gamma-mangostin derivative LS02 against Gram-positive bacteria with destructive effect on bacterial membrane. Biochim. Bioph. Acta Biomembr. 2020, 1862, 183353. [Google Scholar] [CrossRef] [PubMed]
- Guillermo-Lagae, R.; Santha, S.; Thomas, M.; Zoelle, E.; Stevens, J.; Kaushik, R.S.; Dwivedi, C. Antineoplastic Effects of Honokiol on Melanoma. BioMed Res. Int. 2017, 2017, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Lu, C.H.; Chen, S.H.; Chang, Y.S.; Liu, Y.W.; Wu, J.Y.; Lim, Y.P.; Yu, H.I.; Lee, Y.R. Honokiol, a potential therapeutic agent, induces cell cycle arrest and program cell death in vitro and in vivo in human thyroid cancer cells. Pharm. Res. 2017, 115, 288–298. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Tan, Y.P.; Zhao, L.; Wang, L.; Fu, N.J.; Zheng, S.P.; Shen, X.F. Anticancer activity of dietary xanthone alpha-mangostin against hepatocellular carcinoma by inhibition of STAT3 signaling via stabilization of SHP1. Cell Death Dis. 2020, 11, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Xu, Q.; Ma, J.; Lei, J.; Duan, W.; Sheng, L.; Chen, X.; Hu, A.; Wang, Z.; Wu, Z.; Wu, E.; et al. alpha-Mangostin suppresses the viability and epithelial-mesenchymal transition of pancreatic cancer cells by downregulating the PI3K/Akt pathway. BioMed Res. Int. 2014, 2014, 1–12. [Google Scholar] [CrossRef]
- Dikalov, S.; Losik, T.; Arbiser, J.L. Honokiol is a potent scavenger of superoxide and peroxyl radicals. Biochem. Pharm. 2008, 76, 589–596. [Google Scholar] [CrossRef] [Green Version]
- Pedraza-Chaverri, J.; Reyes-Fermin, L.M.; Nolasco-Amaya, E.G.; Orozco-Ibarra, M.; Medina-Campos, O.N.; Gonzalez-Cuahutencos, O.; Rivero-Cruz, I.; Mata, R. ROS scavenging capacity and neuroprotective effect of alpha-mangostin against 3-nitropropionic acid in cerebellar granule neurons. Exp. Toxicol. Pathol. 2009, 61, 491–501. [Google Scholar] [CrossRef]
- Jalali, A.; Firouzabadi, N.; Zarshenas, M.M. Pharmacogenetic-based management of depression: Role of traditional Persian medicine. Phytother. Res. 2021, 35, 5031–5052. [Google Scholar] [CrossRef]
- Hoi, C.P.; Ho, Y.P.; Baum, L.; Chow, A.H. Neuroprotective effect of honokiol and magnolol, compounds from Magnolia officinalis, on beta-amyloid-induced toxicity in PC12 cells. Phytother. Res. 2010, 24, 1538–1542. [Google Scholar] [CrossRef]
- Liu, J.; Tang, M.; Li, T.; Su, Z.; Zhu, Z.; Dou, C.; Liu, Y.; Pei, H.; Yang, J.; Ye, H.; et al. Honokiol Ameliorates Post-Myocardial Infarction Heart Failure Through Ucp3-Mediated Reactive Oxygen Species Inhibition. Front. Pharm. 2022, 13, 811682. [Google Scholar] [CrossRef]
- Tiwari, A.; Khera, R.; Rahi, S.; Mehan, S.; Makeen, H.A.; Khormi, Y.H.; Rehman, M.U.; Khan, A. Neuroprotective Effect of alpha-Mangostin in the Ameliorating Propionic Acid-Induced Experimental Model of Autism in Wistar Rats. Brain Sci. 2021, 11, 288. [Google Scholar] [CrossRef] [PubMed]
- Devi Sampath, P.; Vijayaraghavan, K. Cardioprotective effect of alpha-mangostin, a xanthone derivative from mangosteen on tissue defense system against isoproterenol-induced myocardial infarction in rats. J. Biochem. Mol. Toxicol. 2007, 21, 336–339. [Google Scholar] [CrossRef] [PubMed]
- Fang, C.Y.; Chen, S.J.; Wu, H.N.; Ping, Y.H.; Lin, C.Y.; Shiuan, D.; Chen, C.L.; Lee, Y.R.; Huang, K.J. Honokiol, a Lignan Biphenol Derived from the Magnolia Tree, Inhibits Dengue Virus Type 2 Infection. Viruses 2015, 7, 4894–4910. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, S.; Li, L.; Tan, L.; Liang, X. Inhibition of Herpes Simplex Virus-1 Replication by Natural Compound Honokiol. Virol. Sin. 2019, 34, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Lim, C.Y.; Chung, M.S. Magnolia officinalis and Its Honokiol and Magnolol Constituents Inhibit Human Norovirus Surrogates. Foodborne Pathog. Dis. 2021, 18, 24–30. [Google Scholar] [CrossRef]
- Panda, K.; Alagarasu, K.; Patil, P.; Agrawal, M.; More, A.; Kumar, N.V.; Mainkar, P.S.; Parashar, D.; Cherian, S. In Vitro Antiviral Activity of alpha-Mangostin against Dengue Virus Serotype-2 (DENV-2). Molecules 2021, 26, 3016. [Google Scholar] [CrossRef] [PubMed]
- Patil, P.; Agrawal, M.; Almelkar, S.; Jeengar, M.K.; More, A.; Alagarasu, K.; Kumar, N.V.; Mainkar, P.S.; Parashar, D.; Cherian, S. In vitro and in vivo studies reveal alpha-Mangostin, a xanthonoid from Garcinia mangostana, as a promising natural antiviral compound against chikungunya virus. Virol. J. 2021, 18, 47. [Google Scholar] [CrossRef]
- Barroso, M.M.; Lima, C.S.; Silva-Neto, M.A.; Da Poian, A.T. Mayaro virus infection cycle relies on casein kinase 2 activity. Biochem. Bioph. Res. Commun. 2002, 296, 1334–1339. [Google Scholar] [CrossRef]
- Sugasti-Salazar, M.; Llamas-Gonzalez, Y.Y.; Campos, D.; Gonzalez-Santamaria, J. Inhibition of p38 Mitogen-Activated Protein Kinase Impairs Mayaro Virus Replication in Human Dermal Fibroblasts and HeLa Cells. Viruses 2021, 13, 1156. [Google Scholar] [CrossRef]
- Stetson, D.B.; Medzhitov, R. Type I interferons in host defense. Immunity 2006, 25, 373–381. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.; Hu, Y.; Shan, L.; Yu, X.; Hao, K.; Wang, G.X. Magnolol and honokiol from Magnolia officinalis enhanced antiviral immune responses against grass carp reovirus in Ctenopharyngodon idella kidney cells. Fish Shellfish Immunol. 2017, 63, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Sun, Z.; Pei, J.; Luo, Q.; Zeng, X.; Li, Q.; Yang, Z.; Quan, J. Identification of alpha-Mangostin as an Agonist of Human STING. ChemMedChem 2018, 13, 2057–2064. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Zhang, H.; Zhu, M.; Pu, Q.; Li, J.; Hu, X. beta-Hydroxyisovaleryl-Shikonin Exerts an Antitumor Effect on Pancreatic Cancer Through the PI3K/AKT Signaling Pathway. Front. Oncol. 2022, 12, 904258. [Google Scholar] [CrossRef] [PubMed]
- Lan, K.H.; Wang, Y.W.; Lee, W.P.; Lan, K.L.; Tseng, S.H.; Hung, L.R.; Yen, S.H.; Lin, H.C.; Lee, S.D. Multiple effects of Honokiol on the life cycle of hepatitis C virus. Liver Int. 2012, 32, 989–997. [Google Scholar] [CrossRef] [PubMed]
- Diaz, Y.; Carrera, J.P.; Cerezo, L.; Arauz, D.; Guerra, I.; Cisneros, J.; Armien, B.; Botello, A.M.; Arauz, A.B.; Gonzalez, V.; et al. Chikungunya virus infection: First detection of imported and autochthonous cases in Panama. Am. J. Trop. Med. Hyg. 2015, 92, 482–485. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Godugu, C.; Doddapaneni, R.; Singh, M. Honokiol nanomicellar formulation produced increased oral bioavailability and anticancer effects in triple negative breast cancer (TNBC). Colloids Surf. B Biointerfaces 2017, 153, 208–219. [Google Scholar] [CrossRef] [Green Version]
- Ben-Shabat, S.; Yarmolinsky, L.; Porat, D.; Dahan, A. Antiviral effect of phytochemicals from medicinal plants: Applications and drug delivery strategies. Drug Deliv. Transl. Res. 2020, 10, 354–367. [Google Scholar] [CrossRef] [Green Version]
- Guo, Y.; Meng, J.R.; Liu, J.Z.; Xu, T.; Zheng, Z.Y.; Jiang, Z.H.; Bai, L.P. Synthesis and Biological Evaluation of Honokiol Derivatives Bearing 3-((5-phenyl-1,3,4-oxadiazol-2-yl)methyl)oxazol-2(3H)-ones as Potential Viral Entry Inhibitors against SARS-CoV-2. Pharmaceuticals 2021, 14, 885. [Google Scholar] [CrossRef]
- Hu, X.; Liu, C.; Wang, K.; Zhao, L.; Qiu, Y.; Chen, H.; Hu, J.; Xu, J. Multifunctional Anti-Alzheimer’s Disease Effects of Natural Xanthone Derivatives: A Primary Structure-Activity Evaluation. Front. Chem. 2022, 10, 842208. [Google Scholar] [CrossRef]
- Powers, A.M.; Aguilar, P.V.; Chandler, L.J.; Brault, A.C.; Meakins, T.A.; Watts, D.; Russell, K.L.; Olson, J.; Vasconcelos, P.F.; Da Rosa, A.T.; et al. Genetic relationships among Mayaro and Una viruses suggest distinct patterns of transmission. Am. J. Trop. Med. Hyg. 2006, 75, 461–469. [Google Scholar] [CrossRef]
- Llamas-Gonzalez, Y.Y.; Campos, D.; Pascale, J.M.; Arbiza, J.; Gonzalez-Santamaria, J. A Functional Ubiquitin-Proteasome System is Required for Efficient Replication of New World Mayaro and Una Alphaviruses. Viruses 2019, 11, 370. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Devhare, P.B.; Chatterjee, S.N.; Arankalle, V.A.; Lole, K.S. Analysis of antiviral response in human epithelial cells infected with hepatitis E virus. PLoS ONE 2013, 8, e63793. [Google Scholar] [CrossRef] [Green Version]
- Stevenson, N.J.; Murphy, A.G.; Bourke, N.M.; Keogh, C.A.; Hegarty, J.E.; O’Farrelly, C. Ribavirin enhances IFN-alpha signalling and MxA expression: A novel immune modulation mechanism during treatment of HCV. PLoS ONE 2011, 6, e27866. [Google Scholar] [CrossRef] [PubMed]
- Bektas, N.; Noetzel, E.; Veeck, J.; Press, M.F.; Kristiansen, G.; Naami, A.; Hartmann, A.; Dimmler, A.; Beckmann, M.W.; Knuchel, R.; et al. The ubiquitin-like molecule interferon-stimulated gene 15 (ISG15) is a potential prognostic marker in human breast cancer. Breast Cancer Res. 2008, 10, R58. [Google Scholar] [CrossRef] [Green Version]
- Hamel, R.; Dejarnac, O.; Wichit, S.; Ekchariyawat, P.; Neyret, A.; Luplertlop, N.; Perera-Lecoin, M.; Surasombatpattana, P.; Talignani, L.; Thomas, F.; et al. Biology of Zika Virus Infection in Human Skin Cells. J. Virol. 2015, 89, 8880–8896. [Google Scholar] [CrossRef] [Green Version]
- Chen, F.; Zhang, G.; Yu, L.; Feng, Y.; Li, X.; Zhang, Z.; Wang, Y.; Sun, D.; Pradhan, S. High-efficiency generation of induced pluripotent mesenchymal stem cells from human dermal fibroblasts using recombinant proteins. Stem Cell Res. Ther. 2016, 7, 99. [Google Scholar] [CrossRef]








| Gene | Primer Sequences (5′-3′) | References |
|---|---|---|
| IFN⍺ | Forward: GCCTCGCCCTTTGCTTTACT | [74] |
| Reverse: CTGTGGGTCTCAGGGAGATCA | ||
| IFNβ | Forward: ATGACCAACAAGTGTCTCCTCC | [74] |
| Reverse: GCTCATGGAAAGAGCTGTAGTG | ||
| MxA | Forward: GGTGGTGGTCCCCAGTAATG | [75] |
| Reverse: ACCACGTCCACAACCTTGTCT | ||
| ISG15 | Forward: GAGAGGCAGCGAACTCATCT | [76] |
| Reverse: CTTCAGCTCTGACACCGACA | ||
| OAS2 | Forward: AAACCAGGCCTGTGATCTTG | [77] |
| Reverse: GGGCTATTTCCAGACAACGC | ||
| MDA-5 | Forward: GCCATTGCAGATGCAACCAG | [77] |
| Reverse: TTGCGATTTCCTTCTTTTGCAG | ||
| TNF⍺ | Forward: CAGAGGGAAGAGTTCCCCAGGGACC | [74] |
| Reverse: CCTTGGTCTGGTAGGAGACGG | ||
| IL-1β | Forward: AACCTCTTCGAGGCACAAGG | [77] |
| Reverse: GTCCTGGAAGGAGCACTTCAT | ||
| β-actin | Forward: AGAGCTACGAGCTGCCTGAC | [78] |
| Reverse: AGCACTGTGTTGGCGTACAG | ||
| MAYV | Forward: CATGGCCTACCTGTGGGATAATA | |
| Reverse: GCACTCCCGACGCTCACTG |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valdés-Torres, P.; Campos, D.; Bhakta, M.; Galán-Jurado, P.E.; Durant-Archibold, A.A.; González-Santamaría, J. Honokiol and Alpha-Mangostin Inhibit Mayaro Virus Replication through Different Mechanisms. Molecules 2022, 27, 7362. https://doi.org/10.3390/molecules27217362
Valdés-Torres P, Campos D, Bhakta M, Galán-Jurado PE, Durant-Archibold AA, González-Santamaría J. Honokiol and Alpha-Mangostin Inhibit Mayaro Virus Replication through Different Mechanisms. Molecules. 2022; 27(21):7362. https://doi.org/10.3390/molecules27217362
Chicago/Turabian StyleValdés-Torres, Patricia, Dalkiria Campos, Madhvi Bhakta, Paola Elaine Galán-Jurado, Armando A. Durant-Archibold, and José González-Santamaría. 2022. "Honokiol and Alpha-Mangostin Inhibit Mayaro Virus Replication through Different Mechanisms" Molecules 27, no. 21: 7362. https://doi.org/10.3390/molecules27217362
APA StyleValdés-Torres, P., Campos, D., Bhakta, M., Galán-Jurado, P. E., Durant-Archibold, A. A., & González-Santamaría, J. (2022). Honokiol and Alpha-Mangostin Inhibit Mayaro Virus Replication through Different Mechanisms. Molecules, 27(21), 7362. https://doi.org/10.3390/molecules27217362





