Hepatitis C Virus NS3/4A Inhibition and Host Immunomodulation by Tannins from Terminalia chebula: A Structural Perspective
Abstract
1. Introduction
2. Materials and Methods
2.1. Retrieval of Phytocompounds
2.2. Target Identification
2.3. Gene Set Enrichment Analysis
2.4. Network Construction
2.5. Molecular Docking Studies
2.5.1. Preparation of Ligand and Protein
2.5.2. Assessment of Active Site Residues
2.5.3. Protein–Ligand Docking
2.6. Molecular Dynamics (MD) Simulation
2.7. Druglikeness, Side Effects, and Toxicity of Phytocompounds
3. Results
3.1. Retrieval of Phytocompounds and Target Identification
3.2. Gene Set Enrichment Analysis and Network Analysis
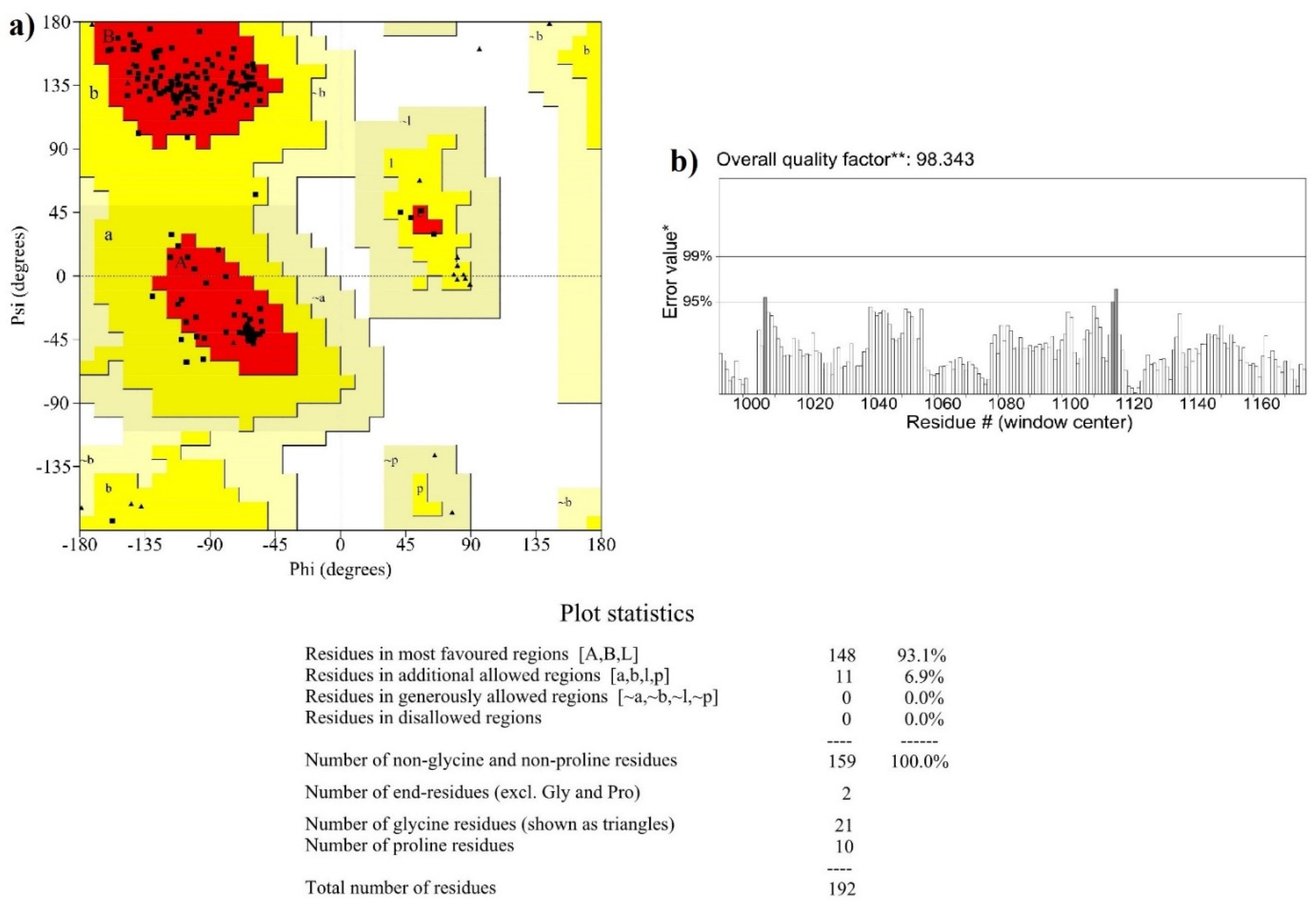
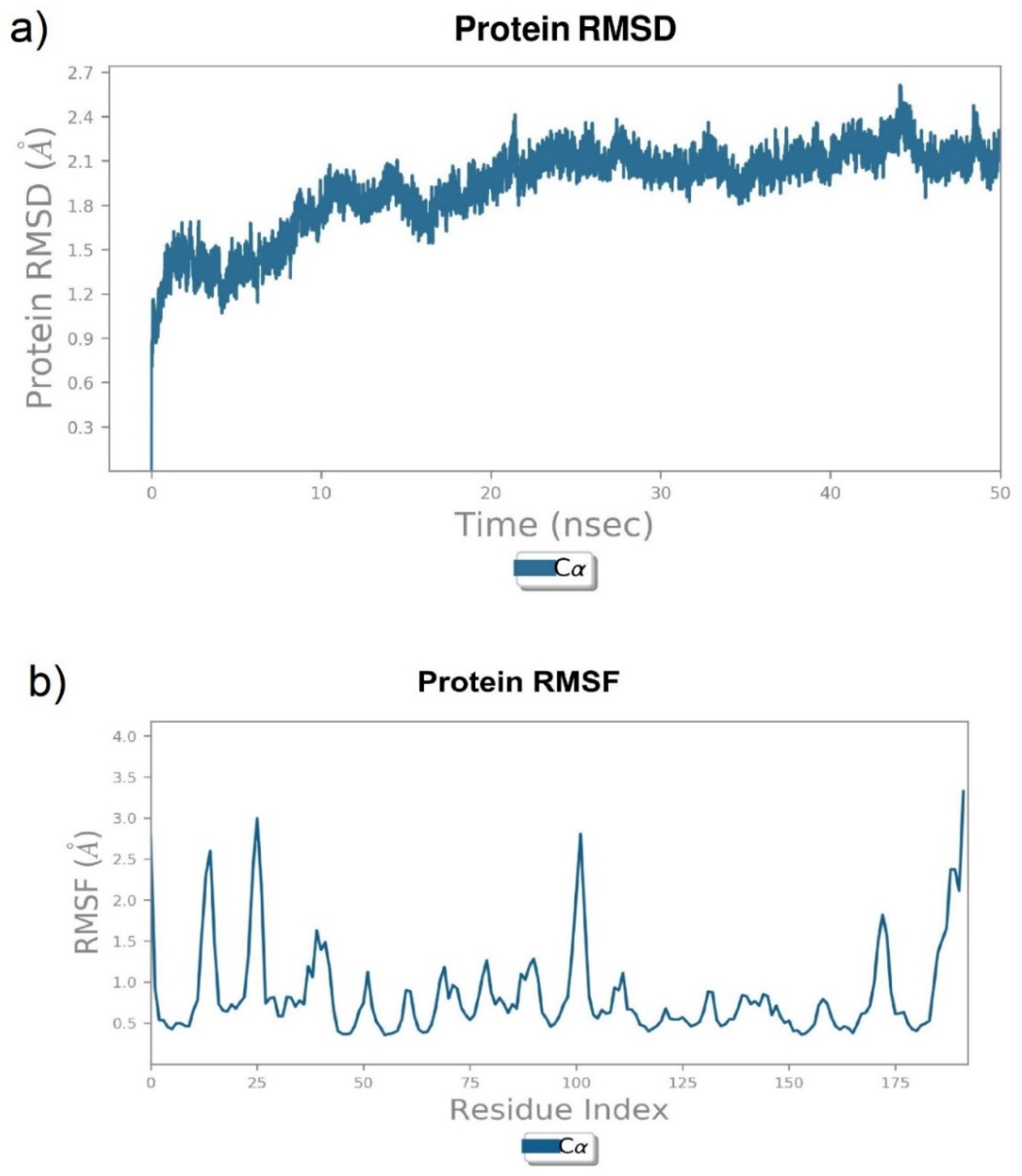
3.3. Protein Quality Check and Stability Analysis by Molecular Dynamics Simulation
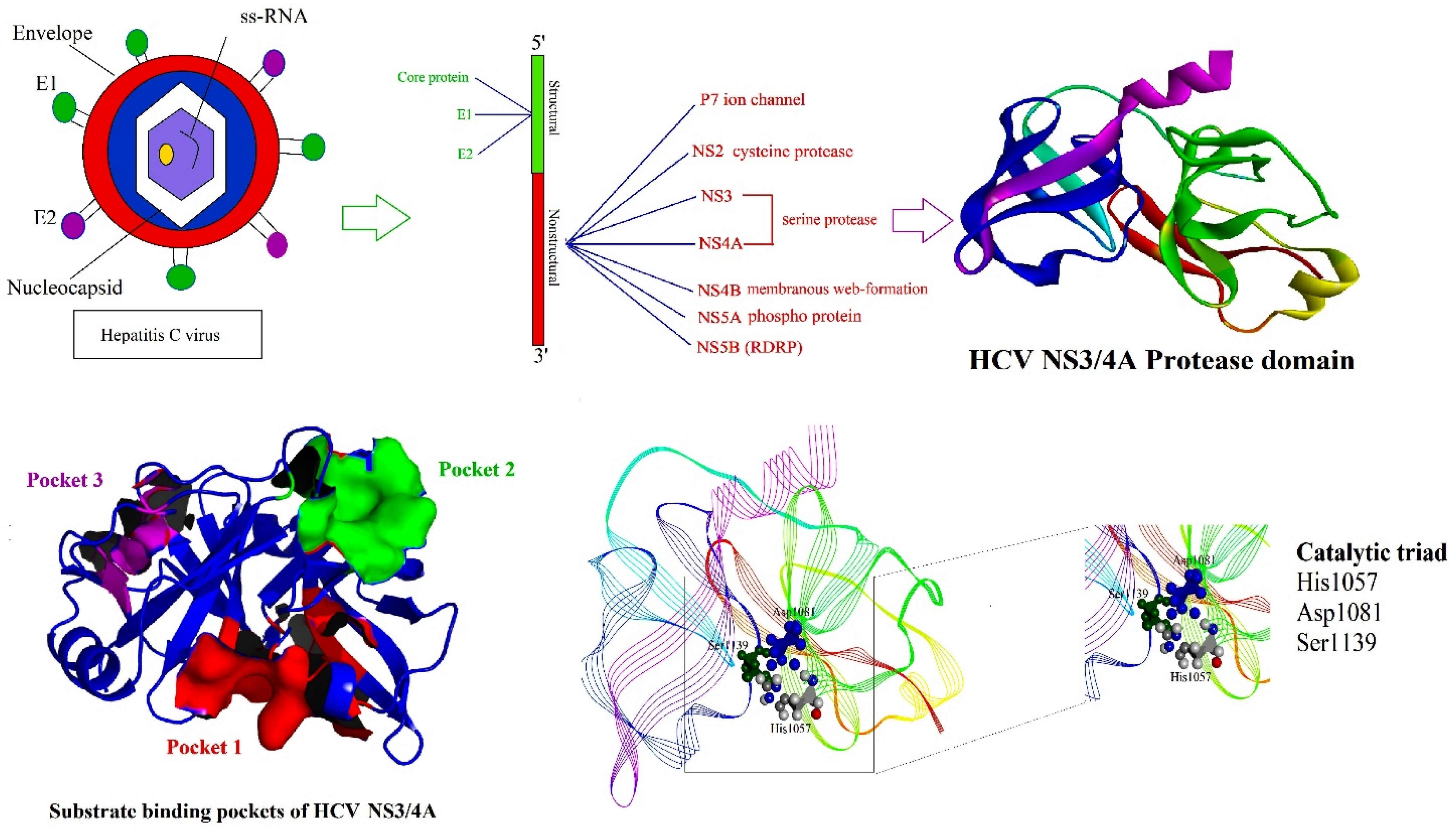
3.4. Assessment of Active Site Residues
3.5. Ligand–Protein Docking
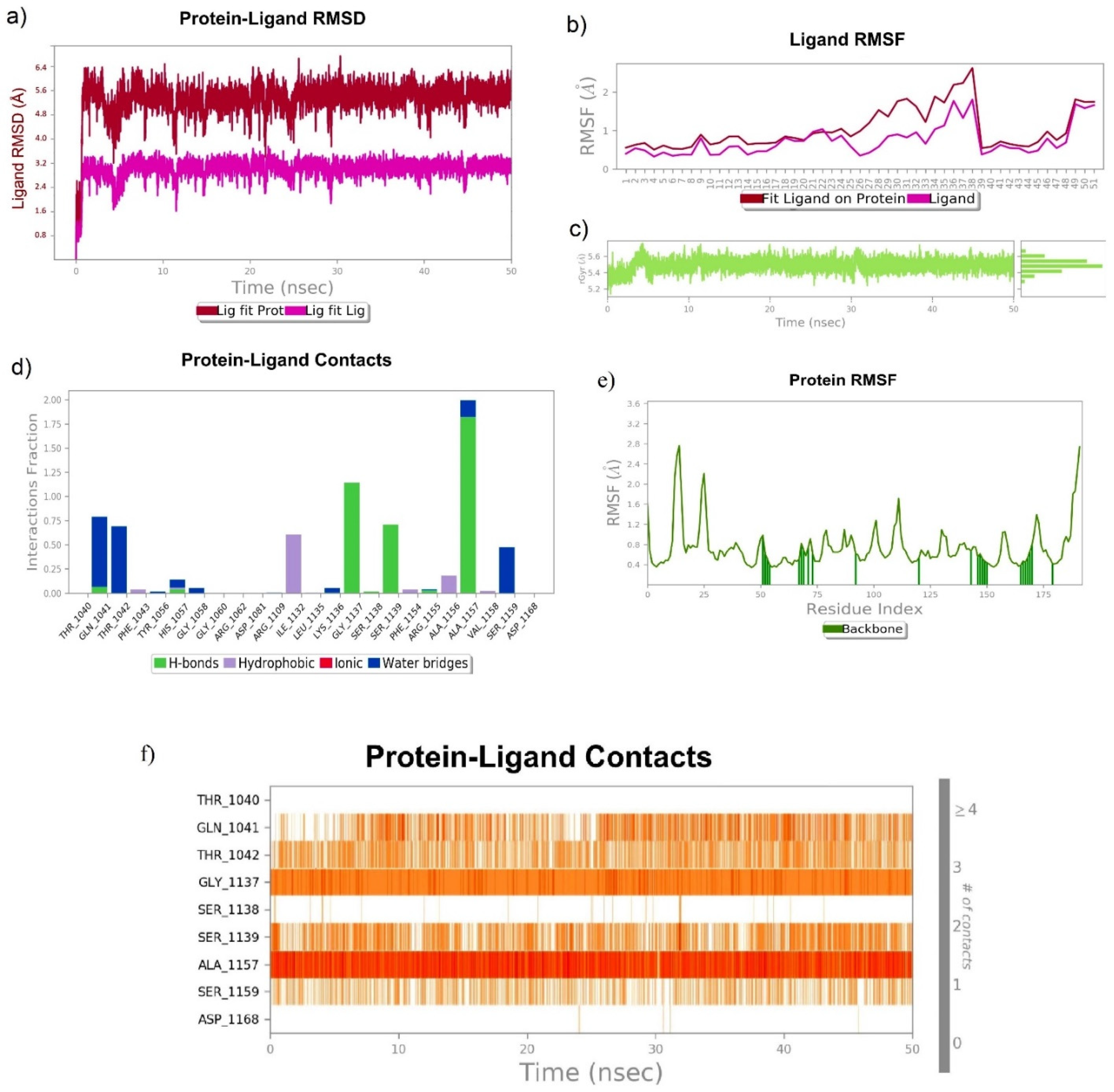
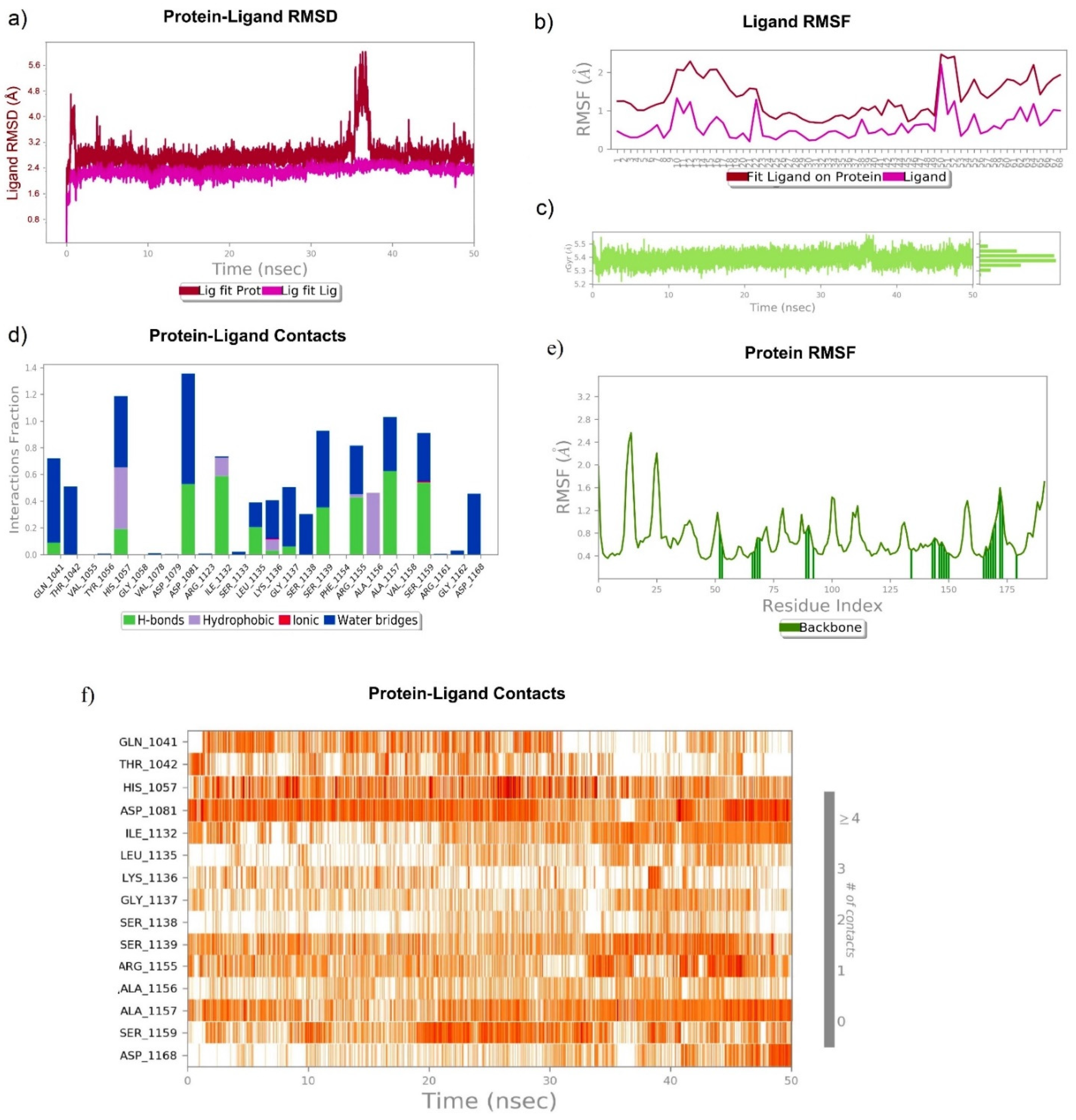
3.6. Molecular Dynamics Simulation of NS3/4A–Ligand Complexes
3.6.1. Asunaprevir–NS3/4A Complex
3.6.2. Chebulagic Acid–NS3/4A Complex
3.6.3. 1,2,3,4,6-Pentagalloyl Glucose–NS3/4A Complex
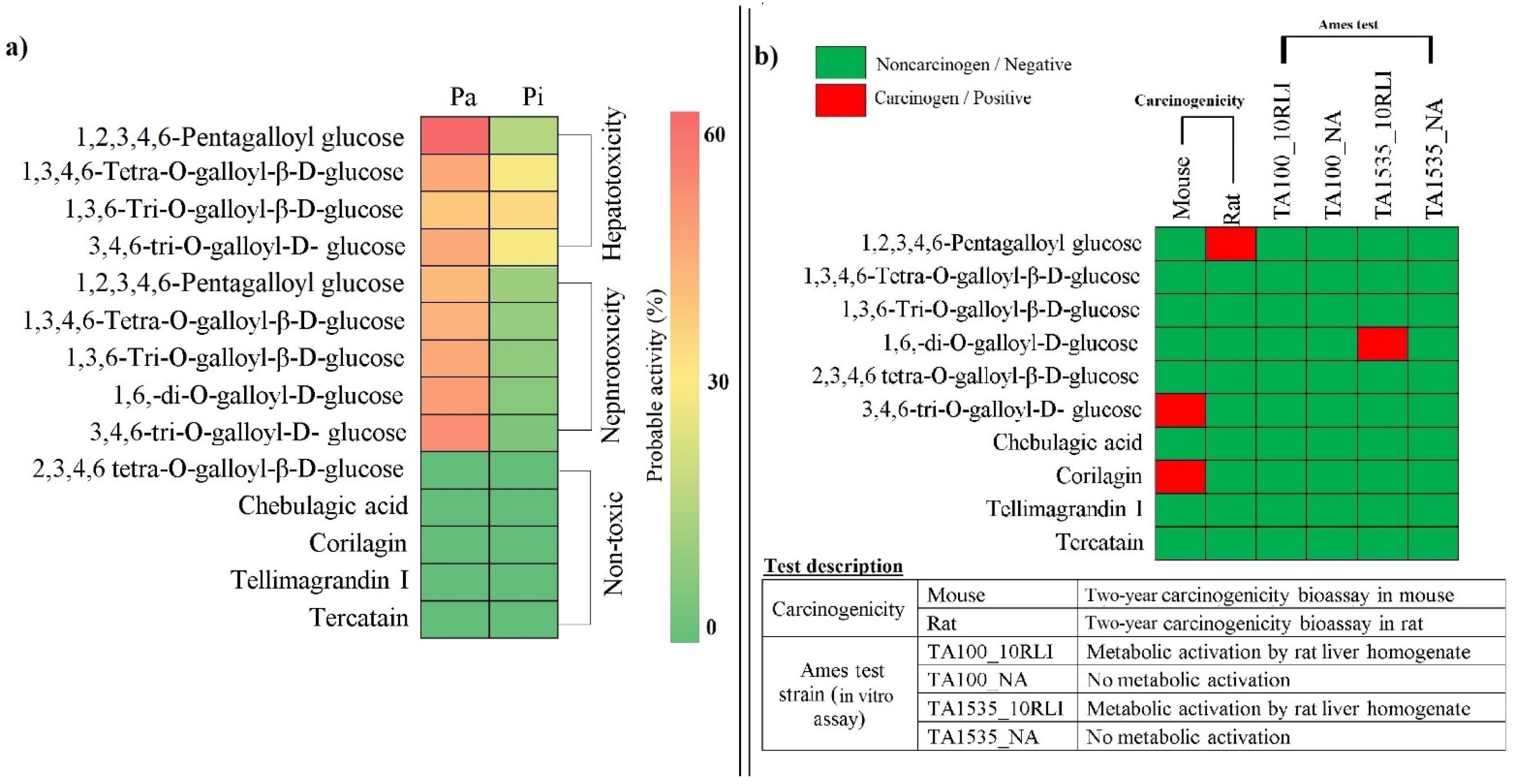
3.7. Druglikeness, Side Effects, and Toxicity of Phytocompounds
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mohamed, A.A.; Elbedewy, T.A.; El-Serafy, M.; El-Toukhy, N.; Ahmed, W.; El Din, Z.A. Hepatitis C virus: A global view. World J. Hepatol. 2015, 7, 2676. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Hepatitis C. Available online: https://www.who.int/news-room/fact-sheets/detail/hepatitis-c (accessed on 5 January 2021).
- Rehman, S.; Ashfaq, U.A.; Javed, T. Antiviral drugs against hepatitis C virus. Genet. Vaccines Ther. 2011, 9, 11. [Google Scholar] [CrossRef] [PubMed]
- Ringehan, M.; McKeating, J.A.; Protzer, U. Viral hepatitis and liver cancer. Philos. Trans. R. Soc. B Biol. Sci. 2017, 372, 20160274. [Google Scholar] [CrossRef] [PubMed]
- Palumbo, E. Pegylated interferon and ribavirin treatment for hepatitis C virus infection. Ther. Adv. Chronic Dis. 2011, 2, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.; Kumar, A.; Sharma, P.; Arora, A. Newer direct-acting antivirals for hepatitis C virus infection: Perspectives for India. Indian J. Med. Res. 2017, 146, 23. [Google Scholar]
- Hu, J.H.; Chang, M.L.; Huang, T.J.; Yeh, C.T.; Chiu, W.N.; Chiang, M.S.; Chen, M.Y. Comparison of Compliance and Efficacy of Pegylated Interferon α-2a and α-2b in Adults with Chronic Hepatitis C. J. Interferon Cytokine Res. 2019, 39, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Coppola, N.; Pisaturo, M.; Tonziello, G.; Sagnelli, C.; Sagnelli, E.; Angelillo, I.F. Efficacy of Pegylated interferon α-2a and α-2b in patients with genotype 1 chronic hepatitis C: A meta-analysis. BMC Infect. Dis. 2012, 12, 357. [Google Scholar] [CrossRef]
- Sulkowski, M.S.; Cooper, C.; Hunyady, B.; Jia, J.; Ogurtsov, P.; Peck-Radosavljevic, M.; Shiffman, M.L.; Yurdaydin, C.; Dalgard, O. Management of adverse effects of Peg-IFN and ribavirin therapy for hepatitis C. Nat. Rev. Gastroenterol. Hepatol. 2011, 8, 212–223. [Google Scholar] [CrossRef] [PubMed]
- Shiffman, M.L.; Suter, F.; Bacon, B.R.; Nelson, D.; Harley, H.; Solá, R.; Shafran, S.D.; Barange, K.; Lin, A.; Soman, A.; et al. Peginterferon alfa-2a and ribavirin for 16 or 24 weeks in HCV genotype 2 or 3. N. Engl. J. Med. 2007, 357, 124–134. [Google Scholar] [CrossRef]
- McHutchison, J.G.; Lawitz, E.J.; Shiffman, M.L.; Muir, A.J.; Galler, G.W.; McCone, J.; Nyberg, L.M.; Lee, W.M.; Ghalib, R.H.; Schiff, E.R.; et al. Peginterferon alfa-2b or alfa-2a with ribavirin for treatment of hepatitis C infection. N. Engl. J. Med. 2009, 361, 580–593. [Google Scholar] [CrossRef]
- Manns, M.P.; McHutchison, J.G.; Gordon, S.C.; Rustgi, V.K.; Shiffman, M.; Reindollar, R.; Goodman, Z.D.; Koury, K.; Ling, M.H.; Albrecht, J.K.; et al. Peginterferon alfa-2b plus ribavirin compared with interferon alfa-2b plus ribavirin for initial treatment of chronic hepatitis C: A randomised trial. Lancet 2001, 358, 958–965. [Google Scholar] [CrossRef]
- Fried, M.W.; Shiffman, M.L.; Reddy, K.R.; Smith, C.; Marinos, G.; Gonçales, F.L., Jr.; Häussinger, D.; Diago, M.; Carosi, G.; Dhumeaux, D.; et al. Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N. Engl. J. Med. 2002, 347, 975–982. [Google Scholar] [CrossRef] [PubMed]
- Hadziyannis, S.J.; Sette, H., Jr.; Morgan, T.R.; Balan, V.; Diago, M.; Marcellin, P.; Ramadori, G.; Bodenheimer, H., Jr.; Bernstein, D.; Rizzetto, M.; et al. Peginterferon-α2a and ribavirin combination therapy in chronic hepatitis C: A randomized study of treatment duration and ribavirin dose. Ann. Intern. Med. 2004, 140, 346–355. [Google Scholar] [CrossRef] [PubMed]
- Salam, K.A.; Akimitsu, N. Hepatitis C virus NS3 inhibitors: Current and future perspectives. BioMed Res. Int. 2013, 2013, 467869. [Google Scholar] [CrossRef]
- Reddy, U.; Tandon, H.; Pradhan, M.K.; Adhikesavan, H.; Srinivasan, N.; Das, S.; Jayaraman, N. Potent HCV NS3 Protease Inhibition by a Water-Soluble Phyllanthin Congener. ACS Omega 2020, 5, 11553. [Google Scholar]
- Chatel-Chaix, L.; Baril, M.; Lamarre, D. Hepatitis C virus NS3/4A protease inhibitors: A light at the end of the tunnel. Viruses 2010, 2, 1752–1765. [Google Scholar] [CrossRef]
- Romano, K.P.; Laine, J.M.; Deveau, L.M.; Cao, H.; Massi, F.; Schiffer, C.A. Molecular mechanisms of viral and host cell substrate recognition by hepatitis C virus NS3/4A protease. J. Virol. 2011, 85, 6106–6116. [Google Scholar] [CrossRef]
- Ashfaq, U.A.; Javed, T.; Rehman, S.; Nawaz, Z.; Riazuddin, S. An overview of HCV molecular biology, replication and immune responses. Virol. J. 2011, 8, 161. [Google Scholar] [CrossRef]
- Naeem, A.; Waheed, Y. Sequence analysis of hepatitis C virus nonstructural protein 3-4A serine protease and prediction of conserved B and T cell epitopes. Biomed. Rep. 2017, 7, 563–566. [Google Scholar] [CrossRef][Green Version]
- Rimmert, B.; Sabet, S.; Ackad, E.; Yousef, M.S. A 3D structural model and dynamics of hepatitis C virus NS3/4A protease (genotype 4a, strain ED43) suggest conformational instability of the catalytic triad: Implications in catalysis and drug resistivity. J. Biomol. Struct. Dyn. 2014, 32, 950–958. [Google Scholar] [CrossRef]
- Meewan, I.; Zhang, X.; Roy, S.; Ballatore, C.; O’Donoghue, A.J.; Schooley, R.T.; Abagyan, R. Discovery of New Inhibitors of Hepatitis C Virus NS3/4A Protease and Its D168A Mutant. ACS Omega 2019, 4, 16999–17008. [Google Scholar] [CrossRef] [PubMed]
- Goto, K.; Roca Suarez, A.A.; Wrensch, F.; Baumert, T.F.; Lupberger, J. Hepatitis c virus and hepatocellular carcinoma: When the host loses its grip. Int. J. Mol. Sci. 2020, 21, 3057. [Google Scholar] [CrossRef] [PubMed]
- Gerold, G.; Pietschmann, T. Hepatitis C virus NS5B polymerase primes innate immune signaling. Hepatology 2013, 57, 1275–1277. [Google Scholar] [CrossRef] [PubMed]
- Szabo, G.; Dolganiuc, A. Hepatitis C and innate immunity: Recent advances. Clin. Liver Dis. 2008, 12, 675–692. [Google Scholar] [CrossRef] [PubMed]
- Deng, L. NS3 protein of Hepatitis C virus associates with the tumour suppressor p53 and inhibits its function in an NS3 sequence-dependent manner. J. Gen. Virol. 2006, 87, 1703–1713. [Google Scholar] [CrossRef] [PubMed]
- Li, X.D.; Sun, L.; Seth, R.B.; Pineda, G.; Chen, Z.J. Hepatitis C virus protease NS3/4A cleaves mitochondrial antiviral signaling protein off the mitochondria to evade innate immunity. Proc. Natl. Acad. Sci. USA 2005, 102, 17717–17722. [Google Scholar] [CrossRef] [PubMed]
- Karayiannis, P. The hepatitis C virus NS3/4A protease complex interferes with pathways of the innate immune response. J. Hepatol. 2005, 43, 743–745. [Google Scholar] [CrossRef]
- Ben-Shabat, S.; Yarmolinsky, L.; Porat, D.; Dahan, A. Antiviral effect of phytochemicals from medicinal plants: Applications and drug delivery strategies. Drug Deliv. Transl. Res. 2020, 10, 354–367. [Google Scholar] [CrossRef]
- Bag, A.; Bhattacharyya, S.K.; Chattopadhyay, R.R. The development of Terminalia chebula Retz. (Combretaceae) in clinical research. Asian Pac. J. Trop. Biomed. 2013, 3, 244–252. [Google Scholar] [CrossRef]
- Ratha, K.K.; Joshi, G.C. Haritaki (Chebulic myrobalan) and its varieties. Ayu 2013, 34, 331–334. [Google Scholar] [CrossRef]
- Chattopadhyay, R.R.; Bhattacharyya, S. Plant Review Terminalia chebula: An update. Pharmacogn. Rev. 2007, 1, 151–156. [Google Scholar]
- Chang, Z.; Zhang, Q.; Liang, W.; Zhou, K.; Jian, P.; She, G.; Zhang, L. A Comprehensive Review of the Structure Elucidation of Tannins from Terminalia Linn. Evid.-Based Complement. Altern. Med. 2019, 2019, 8623909. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Ye, T.; Liang, L.; Liang, W.; Jian, P.; Zhou, K.; Zhang, L. Anti-cancer activity of an ethyl-acetate extract of the fruits of Terminalia bellerica (Gaertn.) Roxb. through an apoptotic signaling pathway in vitro. J. Tradit. Chin. Med. Sci. 2018, 5, 370–379. [Google Scholar] [CrossRef]
- Murali, Y.K.; Anand, P.; Tandon, V.; Singh, R.; Chandra, R.; Murthy, P.S. Long-term effects of Terminalia chebula Retz. on hyperglycemia and associated hyperlipidemia, tissue glycogen content and in vitro release of insulin in streptozotocin induced diabetic rats. Exp. Clin. Endocrinol. Diabetes 2007, 115, 641–646. [Google Scholar] [CrossRef]
- Tasduq, S.A.; Singh, K.; Satti, N.K.; Gupta, D.K.; Suri, K.A.; Johri, R.K. Terminalia chebula (fruit) prevents liver toxicity caused by sub-chronic administration of rifampicin, isoniazid and pyrazinamide in combination. Hum. Exp. Toxicol. 2006, 25, 111–118. [Google Scholar] [CrossRef]
- Choi, M.K.; Kim, H.G.; Han, J.M.; Lee, J.S.; Lee, J.S.; Chung, S.H.; Son, C.G. Hepatoprotective effect of Terminalia chebula against t-BHP-induced acute liver injury in C57/BL6 mice. Evid.-Based Complement. Altern. Med. 2015, 2015, 517350. [Google Scholar] [CrossRef]
- Shivaprasad, H.N.; Kharya, M.D.; Rana, A.C.; Mohan, S. Preliminary Immunomodulatory Activities of the Aqueous Extract of Terminalia chebula. Pharm. Biol. 2006, 44, 32–34. [Google Scholar] [CrossRef]
- Suguna, L.; Singh, S.; Sivakumar, P.; Sampath, P.; Chandrakasan, G. Influence of Terminalia chebula on dermal wound healing in rats. Phytother. Res. 2002, 16, 227–231. [Google Scholar] [CrossRef]
- Li, K.; Diao, Y.; Zhang, H.; Wang, S.; Zhang, Z.; Yu, B.; Huang, S.; Yang, H. Tannin extracts from immature fruits of Terminalia chebula Fructus Retz. promote cutaneous wound healing in rats. BMC Complement. Altern. Med. 2011, 11, 86. [Google Scholar] [CrossRef]
- Suchalatha, S.; Devi, C.S. Protective effect of Terminalia chebula against experimental myocardial injury induced by isoproterenol. Indian J. Exp. Biol. 2004, 42, 174–178. [Google Scholar]
- Hedina, A.; Kotti, P.; Kausar, J.; Anand, V. Phytopharmacological overview of Terminalia chebula Retz. Pharmacogn. J. 2016, 8, 307–309. [Google Scholar] [CrossRef]
- Carounanidy, U.; Satyanarayanan, R.; Velmurugan, A. Use of an aqueous extract of Terminalia chebula as an anticaries agent: A clinical study. Indian J. Dent. Res. 2007, 18, 152. [Google Scholar] [PubMed]
- Lin, L.T.; Chen, T.Y.; Lin, S.C.; Chung, C.Y.; Lin, T.C.; Wang, G.H.; Anderson, R.; Lin, C.C.; Richardson, C.D. Broad-spectrum antiviral activity of chebulagic acid and punicalagin against viruses that use glycosaminoglycans for entry. BMC Microbiol. 2013, 13, 187. [Google Scholar] [CrossRef] [PubMed]
- Ajala, O.S.; Jukov, A.; Ma, C.M. Hepatitis C virus inhibitory hydrolysable tannins from the fruits of Terminalia chebula. Fitoterapia 2014, 99, 117–123. [Google Scholar] [CrossRef]
- Duan, D.; Li, Z.; Luo, H.; Zhang, W.; Chen, L.; Xu, X. Antiviral compounds from traditional Chinese medicines Galla Chinese as inhibitors of HCV NS3 protease. Bioorgan. Med. Chem. Lett. 2004, 14, 6041–6044. [Google Scholar] [CrossRef]
- Lin, L.T.; Chen, T.Y.; Chung, C.Y.; Noyce, R.S.; Grindley, T.B.; McCormick, C.; Lin, T.C.; Wang, G.H.; Lin, C.C.; Richardson, C.D. Hydrolyzable tannins (chebulagic acid and punicalagin) target viral glycoprotein-glycosaminoglycan interactions to inhibit herpes simplex virus 1 entry and cell-to-cell spread. J. Virol. 2011, 85, 4386–4398. [Google Scholar] [CrossRef]
- Kesharwani, A.; Polachira, S.K.; Nair, R.; Agarwal, A.; Mishra, N.N.; Gupta, S.K. Anti-HSV-2 activity of Terminalia chebula Retz extract and its constituents, chebulagic and chebulinic acids. BMC Complement. Altern. Med. 2017, 17, 110. [Google Scholar] [CrossRef]
- Vinayak, U.; Hegde, H.V.; Divakar, M.; Shankar, B.; Veeresh, H.; Kholkute, S.D. Digital database on ethno-medicinal plants of Western Ghats. Curr. Sci. 2010, 99, 1658–1659. [Google Scholar]
- Liu, T.; Lin, Y.; Wen, X.; Jorissen, R.N.; Gilson, M.K. BindingDB: A web-accessible database of experimentally determined protein–ligand binding affinities. Nucleic Acids Res. 2007, 35, D198–D201. [Google Scholar] [CrossRef]
- Wassermann, A.M.; Bajorath, J. BindingDB and ChEMBL: Online compound databases for drug discovery. Expert Opin. Drug Discov. 2011, 6, 683–687. [Google Scholar] [CrossRef]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef] [PubMed]
- Patil, V.S.; Khatib, N.A. Triterpene saponins from Barringtonia acutangula (L.) Gaertn as a potent inhibitor of 11β-HSD1 for type 2 diabetes mellitus, obesity, and metabolic syndrome. Clin. Phytosci. 2020, 6, 61. [Google Scholar] [CrossRef]
- Khanal, P.; Patil, B.M.; Chand, J.; Naaz, Y. Anthraquinone derivatives as an immune booster and their therapeutic option against COVID-19. Nat. Prod. Bioprospect. 2020, 10, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Patil, V.S.; Deshpande, S.H.; Harish, D.R.; Patil, A.S.; Virge, R.; Nandy, S.; Roy, S. Gene set enrichment analysis, network pharmacology and in silico docking approach to understand the molecular mechanism of traditional medicines for the treatment of diabetes mellitus. J. Proteins Proteom. 2020, 11, 297–310. [Google Scholar] [CrossRef]
- Samdani, A.; Vetrivel, U. POAP: A GNU parallel based multithreaded pipeline of open babel and AutoDock suite for boosted high throughput virtual screening. Comput. Biol. Chem. 2018, 74, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Bowers, K.J.; Chow, D.E.; Xu, H.; Dror, R.O.; Eastwood, M.P.; Gregersen, B.A.; Klepeis, J.L.; Kolossvary, I.; Moraes, M.A.; Sacerdoti, F.D.; et al. Scalable Algorithms for Molecular Dynamics Simulations on Commodity Clusters. In Proceedings of the ACM/IEEE Conference on Supercomputing (SC06), Tampa, FL, USA, 11–17 November 2006. [Google Scholar]
- Ivanov, S.M.; Lagunin, A.A.; Rudik, A.V.; Filimonov, D.A.; Poroikov, V.V. ADVERPred–web service for prediction of adverse effects of drugs. J. Chem. Inf. Modeling 2018, 58, 8–11. [Google Scholar] [CrossRef]
- Hegde, V.R.; Pu, H.; Patel, M.; Das, P.R.; Butkiewicz, N.; Arreaza, G.; Gullo, V.P.; Chan, T.M. Two antiviral compounds from the plant Stylogne cauliflora as inhibitors of HCV NS3 protease. Bioorgan. Med. Chem. Lett. 2003, 13, 2925–2928. [Google Scholar] [CrossRef]
- Tamura, S.; Yang, G.M.; Yasueda, N.; Matsuura, Y.; Komoda, Y.; Murakami, N. Tellimagrandin I, HCV invasion inhibitor from Rosae rugosae Flos. Bioorgan. Med. Chem. Lett. 2010, 20, 1598–1600. [Google Scholar] [CrossRef]
- Lin, L.T.; Hsu, W.C.; Lin, C.C. Antiviral natural products and herbal medicines. J. Tradit. Complement. Med. 2014, 4, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Xiu, J.; Liu, J.; Zhang, L.; Li, X.; Xu, Y.; Qin, C.; Zhang, L. Chebulagic acid, a hydrolyzable tannin, exhibited antiviral activity in vitro and in vivo against human enterovirus 71. Int. J. Mol. Sci. 2013, 14, 9618–9627. [Google Scholar] [CrossRef]
- Behrendt, P.; Perin, P.; Menzel, N.; Banda, D.; Pfaender, S.; Alves, M.P.; Thiel, V.; Meuleman, P.; Colpitts, C.C.; Schang, L.M.; et al. Pentagalloylglucose, a highly bioavailable polyphenolic compound present in Cortex moutan, efficiently blocks hepatitis C virus entry. Antivir. Res. 2017, 147, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Tu, Z.; Gong, W.; Zhang, Y.; Feng, Y.; Liu, Y.; Tu, C. Inhibition of rabies virus by 1,2,3,4,6-Penta-O-galloyl-β-D-Glucose involves mTOR-dependent autophagy. Viruses 2018, 10, 201. [Google Scholar] [CrossRef] [PubMed]
- Korcsmáros, T.; Szalay, M.S.; Böde, C.; Kovács, I.A.; Csermely, P. How to design multi-target drugs: Target search options in cellular networks. Expert Opin. Drug Discov. 2007, 2, 799–808. [Google Scholar] [CrossRef] [PubMed]






| Phytocompounds | PubChem ID | Structural Similarity (p ≥ 0.7–1) | Experimental Assay | Reference Compound PubChem CID | Experimental IC50 of Reference Compound (µM) | References |
|---|---|---|---|---|---|---|
| 1,2,3,4,6-Pentagalloyl glucose | 374874 | 1 | Inhibitory activity against HCV NS3 protease by ELISA | 15945060 | 0.75 | [46] |
| 1,3,4,6-Tetra-O-galloyl-β-D-glucose | 471531 | 0.98 | ||||
| 1,3,6-Tri-O-galloyl-β-D-glucose | 452707 | 0.98 | ||||
| 1,6,-di-O-galloyl-D-glucose | 91227631 | 0.94 | ||||
| 2,3,4,6 tetra-O-galloyl-β-D-glucose | 101011018 | 0.7 | ||||
| 3,4,6-tri-O-galloyl-D-glucose | 14188641 | 0.87 | ||||
| Chebulagic acid | 250397 | 0.7 | HCV NS3 protease binding assay | 511658 | 0.3 | [59] |
| 511659 | 0.8 | [59] | ||||
| Corilagin | 73568 | 0.74 | HCV NS3 protease binding assay | 511658 | 0.3 | [59] |
| 511659 | 0.8 | [59] | ||||
| Inhibitory activity against HCV NS3 protease by ELISA | 15945060 | 0.75 | [46] | |||
| Tellimagrandin I | 442690 | 0.7 | HCV NS3 protease binding assay | 511658 | 0.3 | [59] |
| 511659 | 0.8 | [59] | ||||
| Inhibitory activity against HCV NS3 protease by ELISA | 15945060 | 0.75 | [46] | |||
| Tercatain | 14411426 | 0.75 | HCV NS3 protease binding assay | 511658 | 0.3 | [59] |
| 511659 | 0.8 | [59] | ||||
| Inhibitory activity against HCV NS3 protease by ELISA | 15945060 | 0.75 | [46] |
| Reactome ID | Pathways | Gene Count | Background Gene Count | FDR | Genes within Pathway |
|---|---|---|---|---|---|
| HSA-109582 | Hemostasis | 8 | 601 | 1.52E-05 | F10, F11, F2, LCK, PLAT, PLAU, PTPN1, SERPINE1 |
| HSA-140837 | Intrinsic pathway of fibrin clot formation | 3 | 22 | 0.00014 | F10, F11, F2 |
| HSA-140877 | Formation of fibrin clot (clotting cascade) | 3 | 39 | 0.00051 | F10, F11, F2 |
| HSA-162582 | Signal transduction | 11 | 2605 | 0.00051 | F2, HSP90AA1, LCK, PDK1, PLAT, PTPN1, PTPN2, RGS4, RGS7, RGS8, SERPINE1 |
| HSA-9006934 | Signaling by receptor tyrosine kinases | 5 | 437 | 0.0019 | HSP90AA1, LCK, PLAT, PTPN1, PTPN2 |
| HSA-76002 | Platelet activation, signaling, and aggregation | 4 | 256 | 0.0023 | F2, LCK, PTPN1, SERPINE1 |
| HSA-6807004 | Negative regulation of MET activity | 2 | 20 | 0.0038 | PTPN1, PTPN2 |
| HSA-140875 | Common pathway of fibrin clot formation | 2 | 22 | 0.0042 | F10, F2 |
| HSA-1280215 | Cytokine signaling in immune system | 5 | 654 | 0.0062 | GSTO1, HSP90AA1, LCK, PTPN1, PTPN2 |
| HSA-76009 | Platelet aggregation (plug formation) | 2 | 37 | 0.0096 | F2, PTPN1 |
| HSA-449147 | Signaling by interleukins | 4 | 439 | 0.0111 | GSTO1, HSP90AA1, LCK, PTPN2 |
| HSA-168256 | Immune system | 7 | 1925 | 0.0245 | F2, GSTO1, HSP90AA1, LCK, PLAU, PTPN1, PTPN2 |
| Phytocompounds | PubChem CID | BE (kcal/mol) | Total No. of Interactions | No. of Interactions with Active Site Residues | HBI (Amino Acid…Ligand) | Van der Waals, Pi–Alkyl, CH, Pi–Cation, Pi–Sigma, Pi–Pi Stacked, Pi–Pi T-Shaped Interactions |
|---|---|---|---|---|---|---|
| 1,2,3,4,6-Pentagalloyl glucose | 374874 | −7.7 | 20 | 20 | Gln1041…OH, His1057…OH (2), Asp1081…OH, Ile1132…OH, Ser1139…O-, Ser1139…OH (2), Arg1155…O- (2) | Val1055, His1057 (2), Gly1058, Ile1132, Lys1136, Ala1156 (3), Ala1157 |
| 1,3,4,6-Tetra-O-galloyl-β-D-glucose | 471531 | −7.6 | 14 | 14 | Gln1041…OH, Asp1081…OH, Ile1132…OH, Ser1139…OH (2), Ser1139…O- (2), Arg1155…OH | Ile1132, Lys1136 (2), Gly1137, Ala1156, Ala1157 |
| 1,3,6-Tri-O-galloyl-β-D-glucose | 452707 | −7.0 | 8 | 8 | Gly1058…O-, Gly1137…O-, Ser1139….O- (2), Ser1139…OH | His1057, Lys1136, Ala1156 |
| 1,6,-di-O-galloyl-D-glucose | 91227631 | −6.6 | 6 | 5 | Thr1042…OH, Ile1132…OH, Ser1139…O- | His1057 (2), Lys1136 |
| 2,3,4,6 tetra-O-galloyl-β-D-glucose | 101011018 | −6.5 | 11 | 11 | Gln1041…OH, Ser1139…OH (3) | Ile1132, Lys1136, Gly1137, Ser1139, Ala1156 (2), Ala1157 |
| 3,4,6-tri-O-galloyl-D-glucose | 14188641 | −6.6 | 11 | 10 | Gln1041…OH, Thr1042…OH, Ile1132…OH, Ser1139…OH, Ser1139…O- | His1057, Ile1132, Lys1136, Gly1137, Ala1156, Ala1157 |
| Chebulagic acid | 250397 | −8.4 | 13 | 11 | Gly1137…O-, Arg1155…=O, Arg1155…=O, Gly1058…O-, Ile1132…OH (2), Ser1159…OH (2) | Lys1136 (2), His1057, Ile1132, Ser1139 |
| Corilagin | 73568 | −7.3 | 3 | 3 | Gln1041…OH, His1057…O- | Lys1136 |
| Tellimagrandin I | 442690 | −7.7 | 10 | 9 | Gln1041…OH, Thr1042…OH, Gly1058…O-, Ser1139…=O, Ala1157…O- | His1057, Ile1132 (2), Lys1136, Ala1157 |
| Tercatain | 14411426 | −7.5 | 8 | 6 | Leu1135…OH, Ser1139…O-, Ser1159…OH, Ser1159…O- | His1057 (2), Lys1136, Ala1156 |
| * Asunaprevir | 16076883 | −7.4 | 20 | 20 | Gly1137…=O (2), Ala1157…NH, Ala1157…=O | Arg1155 (2), Asp1081 (2), His1057 (2), Ala1156 (4), Ile1132 (3), Val1158 (2), Lys1136 (2) |
| Compound/Ligand Name | Amino Acid Residue | Ligand Atom | Ligand Atom Position | Ligand Atom Interactions with the Protein Residues (%) |
|---|---|---|---|---|
| Asunaprevir | Gly1137 | =O | 16 | 97 |
| Ala1157 | =O | 7 | 99 | |
| Ala1157 | NH | 44 | 82 | |
| Ser1139 | =O | 9 | 56 | |
| Ser1159 | -O | 47 | 46 | |
| Gln1041 | +O | 18 | 39 | |
| Thr1042 | -O | 18 | 38 | |
| Thr1042 | -O | 18 | 28 | |
| Chebulagic acid | Ala1157 | =O | 41 | 61 |
| Asp1081 | OH | 68 | 51 | |
| Asp1081 | =O | 67 | 38 | |
| Asp1168 | =O | 20 | 31 | |
| Ile1132 | OH | 46 | 45 | |
| Arg1155 | OH | 50 | 31 | |
| His1057 | π-π | 25–30 | 46 | |
| 1,2,3,4,6-Pentagalloyl glucose | Asp1081 | OH | 41 | 99 |
| Asp1081 | OH | 40 | 99 | |
| Arg1155 | OH | 42 | 37 | |
| Arg1155 | OH | 42 | 22 | |
| Gly1137 | OH | 65 | 41 | |
| Ser1139 | OH | 65 | 61 | |
| Ser1139 | OH | 66 | 72 | |
| Leu1135 | OH | 67 | 47 | |
| Leu1157 | OH | 67 | 32 | |
| Tyr1056 | OH | 18 | 23 | |
| His1057 | OH | 18 | 23 |
| Phytocompounds | MW (g/mol) | MF | HBA | HBD | LogP | DLS |
|---|---|---|---|---|---|---|
| 1,2,3,4,6-Pentagalloyl glucose | 940.12 | C41 H32 O26 | 26 | 15 | 1.47 | 0.19 |
| 1,3,4,6-Tetra-O-galloyl-β-D-glucose | 788.11 | C34 H28 O22 | 22 | 13 | 0.62 | 0.92 |
| 1,3,6-Tri-O-galloyl-β-D-glucose | 636.1 | C27 H24 O18 | 18 | 11 | −0.08 | 0.92 |
| 1,6,-di-O-galloyl-D-glucose | 484.09 | C20 H20 O14 | 14 | 9 | 0.8 | 0.9 |
| 2,3,4,6 tetra-O-galloyl-β-D-glucose | 788.11 | C34 H28 O22 | 22 | 13 | 0.1 | 0.44 |
| 3,4,6-tri-O-galloyl-D-glucose | 636.1 | C27 H24 O18 | 18 | 11 | −0.2 | 0.87 |
| Chebulagic acid | 954.1 | C41 H30 O27 | 27 | 13 | 0.22 | 0.58 |
| Corilagin | 634.08 | C27 H22 O18 | 18 | 11 | 0.51 | 0.64 |
| Tellimagrandin I | 786.09 | C34 H26 O22 | 22 | 13 | 1.15 | 0.3 |
| Tercatain | 786.09 | C34 H26 O22 | 22 | 13 | 1.21 | 0.65 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Patil, V.S.; Harish, D.R.; Vetrivel, U.; Roy, S.; Deshpande, S.H.; Hegde, H.V. Hepatitis C Virus NS3/4A Inhibition and Host Immunomodulation by Tannins from Terminalia chebula: A Structural Perspective. Molecules 2022, 27, 1076. https://doi.org/10.3390/molecules27031076
Patil VS, Harish DR, Vetrivel U, Roy S, Deshpande SH, Hegde HV. Hepatitis C Virus NS3/4A Inhibition and Host Immunomodulation by Tannins from Terminalia chebula: A Structural Perspective. Molecules. 2022; 27(3):1076. https://doi.org/10.3390/molecules27031076
Chicago/Turabian StylePatil, Vishal S., Darasaguppe R. Harish, Umashankar Vetrivel, Subarna Roy, Sanjay H. Deshpande, and Harsha V. Hegde. 2022. "Hepatitis C Virus NS3/4A Inhibition and Host Immunomodulation by Tannins from Terminalia chebula: A Structural Perspective" Molecules 27, no. 3: 1076. https://doi.org/10.3390/molecules27031076
APA StylePatil, V. S., Harish, D. R., Vetrivel, U., Roy, S., Deshpande, S. H., & Hegde, H. V. (2022). Hepatitis C Virus NS3/4A Inhibition and Host Immunomodulation by Tannins from Terminalia chebula: A Structural Perspective. Molecules, 27(3), 1076. https://doi.org/10.3390/molecules27031076







