Anti-Tumor Active Isopropylated Fused Azaisocytosine-Containing Congeners Are Safe for Developing Danio rerio as Well as Red Blood Cells and Activate Apoptotic Caspases in Human Breast Carcinoma Cells
Abstract
:1. Introduction
2. Results and Discussion
2.1. The Effect of the Tested Isopropylated Fused Azaisocytosine-Containing Congeners (I–VI) on Developing Zebrafish
2.2. The Hemolytic Activities of the Investigated Isopropylated Fused Azaisocytosine-Containing Congeners (I–VI)
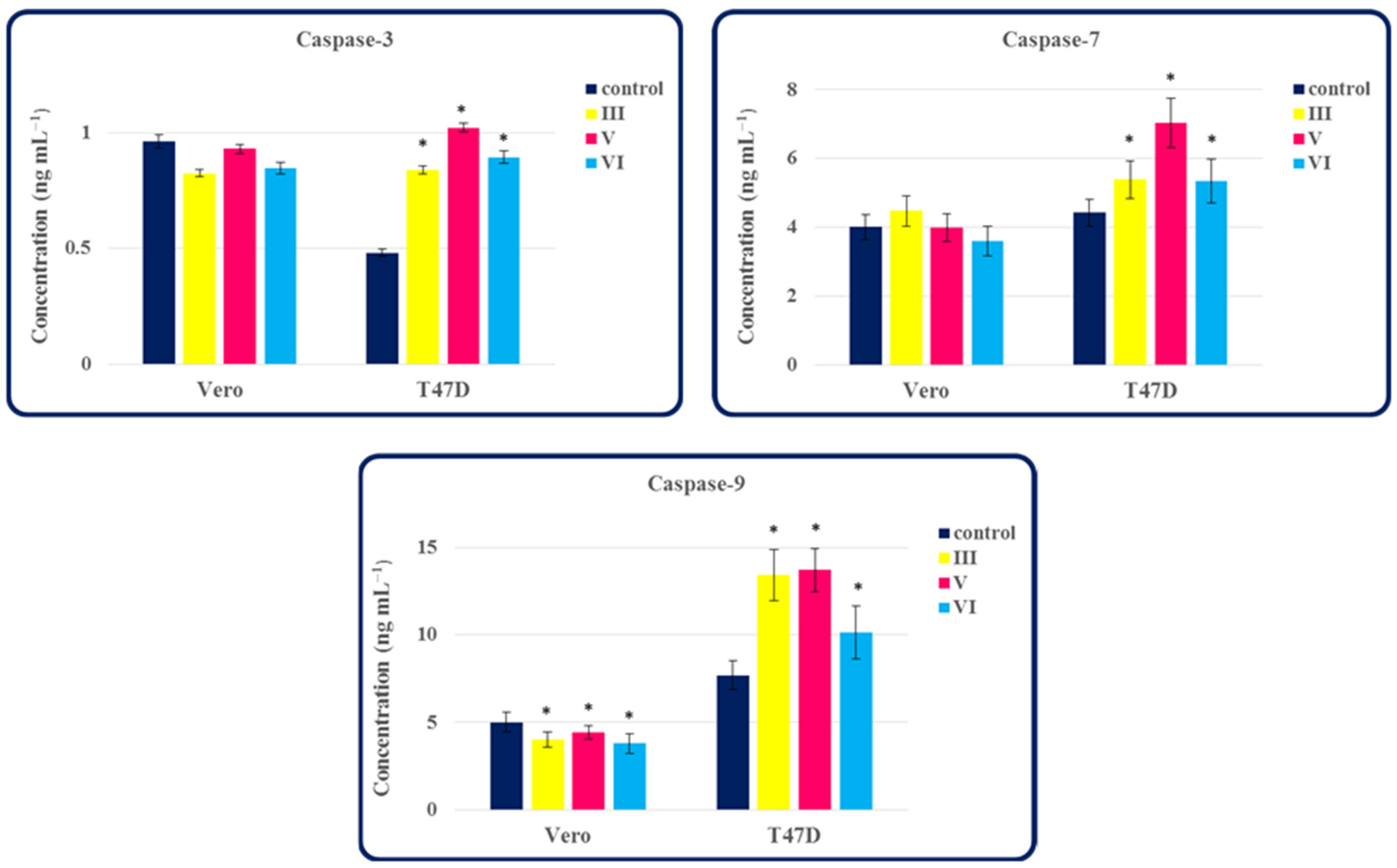
2.3. The Ability of Compounds III, V and VI to Activate Caspase-3, -7 and -9 in Normal Cells and Tumor Cells of the Breast
2.4. Predicting Molecular Targets for the Isopropylated Fused Azaisocytosine-Containing Congeners (I–VI)
3. Materials and Methods
3.1. The Investigated Compounds (I–VI)
3.2. In Vivo Studies
3.2.1. Preparation of Solutions of the Tested Compounds
3.2.2. Zebrafish Embryo Acute Toxicity Studies
3.3. Ex Vivo Studies
3.3.1. Red Blood Cells
3.3.2. Hemolysis Assay
3.4. In Vitro Studies
Measurement of Caspase-3, -7 and -9 Levels in Normal and Tumor Cells
3.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Sztanke, M.; Rzymowska, J.; Janicka, M.; Sztanke, K. Two novel classes of fused azaisocytosine-containing congeners as promising drug candidates: Design, synthesis as well as in vitro, ex vivo and in silico studies. Bioorg. Chem. 2020, 95, 103480. [Google Scholar] [CrossRef]
- Janicka, M.; Mycka, A.; Sztanke, M.; Sztanke, K. Predicting pharmacokinetic properties of potential anticancer agents via their chromatographic behavior on different reversed phase materials. Int. J. Mol. Sci. 2021, 22, 4257. [Google Scholar] [CrossRef]
- Sztanke, M.; Sztanke, K. Medical University of Lublin (Assignee). 8-Substituted-3-(propan-2-yl)-7,8-dihydroimidazo[2,1-c][1,2,4]triazin-4(6H)-ones, Method of Their Preparation and Medical Use. Polish Patent 236364, 11 January 2021. Patent Application Publication No: PL 2019/429163 A1, 2019. [Google Scholar]
- Miyawaki, I. Application of zebrafish to safety evaluation in drug discovery. Toxicol. Pathol. 2020, 33, 197–210. [Google Scholar] [CrossRef]
- Aspatwar, A.; Becker, H.M.; Parvathaneni, N.K.; Hammaren, M.; Svorjova, A.; Barker, H.; Supuran, C.T.; Dubois, L.; Lambin, P.; Parikka, M.; et al. Nitroimidazole-based inhibitors DTP338 and DTP348 are safe for zebrafish embryos and efficiently inhibit the activity of human CA IX in Xenopus oocytes. J. Enzyme Inhib. Med. Chem. 2018, 33, 1064–1073. [Google Scholar] [CrossRef] [Green Version]
- Aspatwar, A.; Parvathaneni, N.K.; Barker, H.; Anduran, E.; Supuran, C.T.; Dubois, L.; Lambin, P.; Parkkila, S.; Winum, J.-Y. Design, synthesis, in vitro inhibition and toxicological evaluation of human carbonic anhydrases I, II and IX inhibitors in 5-nitroimidazole series. J. Enzyme Inhib. Med. Chem. 2020, 35, 109–117. [Google Scholar] [CrossRef] [Green Version]
- Bambino, K.; Chu, J. Zebrafish in toxicology and environmental health. Curr. Top. Dev. Biol. 2017, 124, 331–367. [Google Scholar] [CrossRef] [Green Version]
- Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the protection of animals used for scientific purposes. Off. J. Eur. Union 2010, L276, 33–79.
- Reimers, M.J.; Flockton, A.R.; Tanguay, R.L. Ethanol- and acetaldehyde-mediated developmental toxicity in zebrafish. Neurotoxicol. Teratol. 2004, 26, 769–781. [Google Scholar] [CrossRef]
- Anduran, E.; Aspatwar, A.; Parvathaneni, N.K.; Suylen, D.; Bua, S.; Nocentini, A.; Parkkila, S.; Supuran, C.T.; Dubois, L.; Lambin, P.; et al. Hypoxia-activated prodrug derivatives of carbonic anhydrase inhibitors in benzenesulfonamide series: Synthesis and biological evaluation. Molecules 2020, 25, 2347. [Google Scholar] [CrossRef]
- Jarque, S.; Rubio-Brotons, M.; Ibarra, J.; Ordoñez, V.; Dyballa, S.; Miñana, R.; Terriente, J. Morphometric analysis of developing zebrafish embryos allows predicting teratogenicity modes of action in higher vertebrates. Reprod. Toxicol. 2020, 96, 337–348. [Google Scholar] [CrossRef]
- Kundishora, A.; Sithole, S.; Mukanganyama, S. Determination of the cytotoxic effect of different leaf extracts from Parinari curatellifolia (Chrysobalanaceae). J. Toxicol. 2020, 2020, 8831545. [Google Scholar] [CrossRef]
- Kesavardhana, S.; Malireddi, R.K.S.; Kanneganti, T.D. Caspases in cell death, inflammation, and pyroptosis. Annu. Rev. Immunol. 2020, 38, 567–595. [Google Scholar] [CrossRef] [Green Version]
- Boice, A.; Bouchier-Hayes, L. Targeting apoptotic caspases in cancer. Biochim. Biophys. Acta Mol. Cell Res. 2020, 1867, 118688. [Google Scholar] [CrossRef]
- Sztanke, M.; Rzymowska, J.; Sztanke, K. Anticancer active trifluoromethylated fused triazinones are safe for early-life stages of zebrafish (Danio rerio) and reveal a proapoptotic action. J. Enzyme Inhib. Med. Chem. 2021, 6, 335–343. [Google Scholar] [CrossRef]
- de Koning, C.; Beekhuijzen, M.; Tobor-Kapłon, M.; de Vries-Buitenweg, S.; Schoutsen, D.; Leeijen, N.; van de Waart, B.; Emmen, H. Visualizing compound distribution during zebrafish embryo development: The effects of lipophilicity and DMSO. Birth Defects Res. B Dev. Reprod. Toxicol. 2015, 104, 253–272. [Google Scholar] [CrossRef]
- Sittaramane, V.; Padgett, J.; Salter, P.; Williams, A.; Luke, S.; McCall, R.; Arambula, J.F.; Graves, V.B.; Blocker, M.; Van Leuven, D.; et al. Discovery of quinoline-derived trifluoromethyl alcohols, determination of their in vivo toxicity and anticancer activity in a zebrafish embryo model. ChemMedChem 2015, 10, 1802–1807. [Google Scholar] [CrossRef]
- Yumnamcha, T.; Devi, M.D.; Roy, D.; Nongthomba, U. Evaluation of developmental toxicity and genotoxicity of aqueous seed extract of Croton tiglium L. using zebrafish. Drug Chem. Toxicol. 2022, 45, 398–406. [Google Scholar] [CrossRef]
- Chahardehi, A.M.; Arsad, H.; Lim, V. Zebrafish as a successful animal model for screening toxicity of medicinal plants. Plants 2020, 9, 1345. [Google Scholar] [CrossRef]
- Oliveira, R.; Domingues, I.; Grisolia, C.K.; Soares, A.M.V.M. Effects of triclosan on zebrafish early-life stages and adults. Environ. Sci. Pollut. Res. 2009, 16, 679–688. [Google Scholar] [CrossRef]
- Mu, X.; Pang, S.; Sun, X.; Gao, J.; Chen, J.; Chen, X.; Li, X.; Wang, C. Evaluation of acute and developmental effects of difenoconazole via multiple stage zebrafish assays. Environ. Pollut. 2013, 175, 147–157. [Google Scholar] [CrossRef]
- Mu, X.; Chai, T.; Wang, K.; Zhu, L.; Huang, Y.; Shen, G.; Li, Y.; Li, X.; Wang, C. The developmental effect of difenoconazole on zebrafish embryos: A mechanism research. Environ. Pollut. 2016, 212, 18–26. [Google Scholar] [CrossRef]
- Luo, X.; Chen, L.; Zhang, Y.; Liu, J.; Xie, H. Developmental and cardiac toxicities of propofol in zebrafish larvae. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2020, 237, 108838. [Google Scholar] [CrossRef]
- Yang, X.; Sun, Z.; Wang, W.; Zhou, Q.; Shi, G.; Wei, F.; Jiang, G. Developmental toxicity of synthetic phenolic antioxidants to the early life stage of zebrafish. Sci. Total Environ. 2018, 643, 559–568. [Google Scholar] [CrossRef]
- Cedron, V.P.; Weiner, A.M.J.; Vera, M.; Sanchez, L. Acetaminophen affects the survivor, pigmentation and development of craniofacial structures in zebrafish (Danio rerio) embryos. Biochem. Pharmacol. 2020, 174, 113816. [Google Scholar] [CrossRef]
- Qian, L.; Qi, S.; Cao, F.; Zhang, J.; Li, C.; Song, M.; Wang, C. Effects of penthiopyrad on the development and behaviour of zebrafish in early-life stages. Chemosphere 2019, 214, 184–194. [Google Scholar] [CrossRef]
- Patrick, G.L. An Introduction to Medicinal Chemistry; Oxford University Press Inc.: New York, NY, USA, 2009. [Google Scholar]
- Teran, R.; Guevara, R.; Mora, J.; Dobronski, L.; Barreiro-Costa, O.; Beske, T.; Pérez-Barrera, J.; Araya-Maturana, R.; Rojas-Silva, P.; Poveda, A.; et al. Characterization of antimicrobial, antioxidant, and leishmanicidal activities of Schiff base derivatives of 4-aminoantipyrine. Molecules 2019, 24, 2696. [Google Scholar] [CrossRef] [Green Version]
- Szuster-Ciesielska, A.; Sztanke, K.; Kandefer-Szerszeń, M. A novel fused 1,2,4-triazine aryl derivative as antioxidant and nonselective antagonist of adenosine A2A receptors in ethanol-activated liver stellate cells. Chem. Biol. Interact. 2012, 195, 18–24. [Google Scholar] [CrossRef]
- Taliani, S.; Pugliesi, I.; Barresi, E.; Simorini, F.; Salerno, S.; La Motta, C.; Marini, A.M.; Cosimelli, B.; Cosconati, S.; Di Maro, S.; et al. 3-Aryl-[1,2,4]triazino[4,3-a]benzimidazol-4(10H)-one: A novel template for the design of highly selective A2B adenosine receptor antagonists. J. Med. Chem. 2012, 55, 1490–1499. [Google Scholar] [CrossRef]
- OECD Guidelines for the Testing of Chemicals, Test No. 236: Fish Embryo Acute Toxicity (FET) Test; OECD: Paris, France, 2013.
- Sztanke, M.; Sztanke, K.; Rajtar, B.; Świątek, Ł.; Boguszewska, A.; Polz-Dacewicz, M. The influence of some promising fused azaisocytosine-containing congeners on zebrafish (Danio rerio) embryos/larvae and their antihaemolytic, antitumour and antiviral activities. Eur. J. Pharm. Sci. 2019, 132, 34–43. [Google Scholar] [CrossRef]
- Aleström, P.; D’Angelo, L.; Midtlyng, P.J.; Schorderet, D.F.; Schulte-Merker, S.; Sohm, F.; Warner, S. Zebrafish: Housing and husbandry recommendations. Lab. Anim. 2020, 54, 213–224. [Google Scholar] [CrossRef] [Green Version]
- Finney, D.J. Probit analysis. J. Inst. Actuar. 1952, 78, 388–390. [Google Scholar]
- Sztanke, M.; Kandefer-Szerszeń, M.; Sztanke, K. Biologically and chemically important hydrazino-containing imidazolines as antioxidant agents. Free Rad. Res. 2018, 52, 685–697. [Google Scholar] [CrossRef] [PubMed]



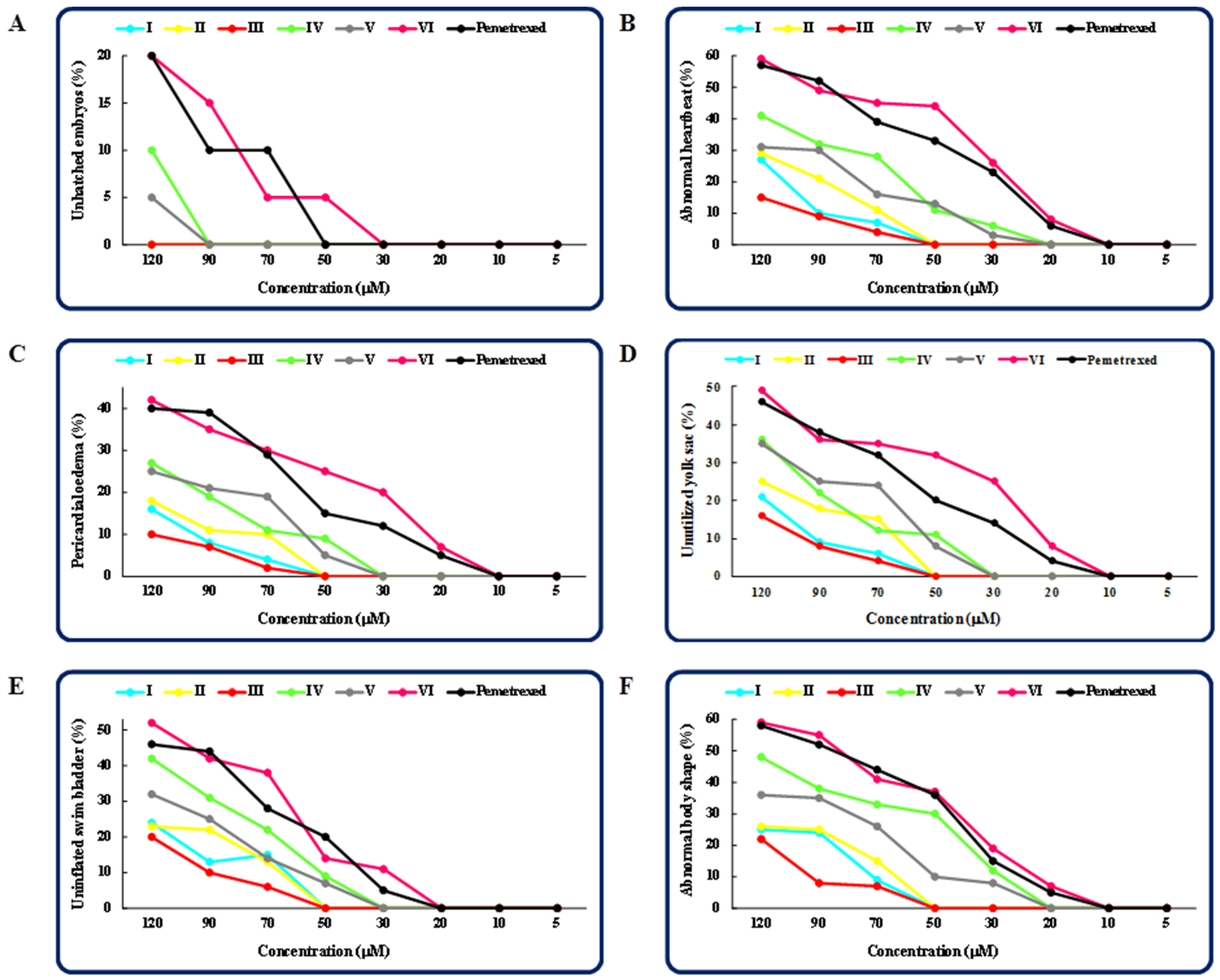


| Compound | MNTC (µM) 1 | LC50 (95% CL 2, µM) | NOAEC (µM) |
|---|---|---|---|
| I | 63.3 ± 11.5 | 117 (107–129) | 50 |
| II | 50.0 ± 0.0 | 105 (96–118) | 50 |
| III | 76.7 ± 11.5 | 121 (115–130) | 50 |
| IV | 30.0 ± 0.0 | 74 (65–86) | 20 |
| V | 43.3 ± 11.5 | 90 (79–100) | 20 |
| VI | 16.7 ± 5.8 | 54 (44–70) | 10 |
| Pemetrexed | 20.0 ± 0.0 | 68 (59–78) | 10 |
| Compound/Control | Hemolytic Activity (in %) | |
|---|---|---|
| Compound | I | 0 |
| II | 0 | |
| III | 0 | |
| IV | 0 | |
| V | 0 | |
| VI | 0 | |
| A positive control | 10% Triton X-100 solution | 100 |
| A negative control | Phosphate Buffered Saline | 0 |
| Compound/Standard Drug | Drug Score | Bioactivity Score | |||||
|---|---|---|---|---|---|---|---|
| GPCR Ligand | Enzyme Inhibitor | Kinase Inhibitor | Ion Channel Modulator | Nuclear Receptor Ligand | Protease Inhibitor | ||
| I | 0.84 | −0.41 | −0.37 | −0.61 | −0.77 | −0.95 | −1.00 |
| II | 0.79 | −0.40 | −0.41 | −0.59 | −0.82 | −0.89 | −0.98 |
| III | 0.73 | −0.28 | −0.41 | −0.40 | −0.74 | −0.93 | −0.98 |
| IV | 0.73 | −0.36 | −0.41 | −0.55 | −0.74 | −0.89 | −1.00 |
| V | 0.73 | −0.35 | −0.39 | −0.56 | −0.74 | −0.88 | −0.96 |
| VI | 0.60 | −0.30 | −0.39 | −0.48 | −0.70 | −0.80 | −0.91 |
| PMX | 0.58 | 0.27 | 0.39 | 0.45 | −0.02 | −0.51 | 0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sztanke, M.; Rzymowska, J.; Sztanke, K. Anti-Tumor Active Isopropylated Fused Azaisocytosine-Containing Congeners Are Safe for Developing Danio rerio as Well as Red Blood Cells and Activate Apoptotic Caspases in Human Breast Carcinoma Cells. Molecules 2022, 27, 1211. https://doi.org/10.3390/molecules27041211
Sztanke M, Rzymowska J, Sztanke K. Anti-Tumor Active Isopropylated Fused Azaisocytosine-Containing Congeners Are Safe for Developing Danio rerio as Well as Red Blood Cells and Activate Apoptotic Caspases in Human Breast Carcinoma Cells. Molecules. 2022; 27(4):1211. https://doi.org/10.3390/molecules27041211
Chicago/Turabian StyleSztanke, Małgorzata, Jolanta Rzymowska, and Krzysztof Sztanke. 2022. "Anti-Tumor Active Isopropylated Fused Azaisocytosine-Containing Congeners Are Safe for Developing Danio rerio as Well as Red Blood Cells and Activate Apoptotic Caspases in Human Breast Carcinoma Cells" Molecules 27, no. 4: 1211. https://doi.org/10.3390/molecules27041211
APA StyleSztanke, M., Rzymowska, J., & Sztanke, K. (2022). Anti-Tumor Active Isopropylated Fused Azaisocytosine-Containing Congeners Are Safe for Developing Danio rerio as Well as Red Blood Cells and Activate Apoptotic Caspases in Human Breast Carcinoma Cells. Molecules, 27(4), 1211. https://doi.org/10.3390/molecules27041211






