In Vivo Anti-Inflammatory Effect, Antioxidant Activity, and Polyphenolic Content of Extracts from Capsicum chinense By-Products
Abstract
:1. Introduction
2. Results and Discussion
2.1. Anti-Inflammatory Effect of Extracts Obtained by Maceration
2.2. Antioxidant Activity of Extracts Obtained by Maceration
2.3. Polyphenolic Content of Extracts Obtained by Maceration
2.4. Correlation between the Anti-Inflammatory Effect or the Antioxidant Activity with the Polyphenolic Content of the Extracts Obtained by Maceration
2.5. Comparison of the Anti-Inflammatory Effect of Extracts Obtained by Maceration (ME), Soxhlet (SOX), and Supercritical Fluid Extraction (SFE)
2.6. Comparison of the Antioxidant Activity of Extracts Obtained by ME, SOX and SFE
2.7. Comparison of Polyphenolic Content of Extracts Obtained by ME, SOX and SFE
2.8. Correlation between the Anti-Inflammatory Effect or the Antioxidant Activity and Polyphenolic Content of Extracts of C. chinense By-Products Obtained by ME, SOX and SFE
2.9. Dose–Response Curves of the Extracts That Exhibited Anti-Inflammatory Activity
3. Materials and Methods
3.1. Obtaining Extracts
3.1.1. Plant Material
3.1.2. Drying of Habanero Pepper By-Products
3.1.3. Maceration Extraction (ME)
3.1.4. Soxhlet Extraction (SOX)
3.1.5. Supercritical Fluid Extraction (SFE)
3.1.6. Extraction of Polyphenols
3.2. Anti-Inflammatory Activity and Dose–Response Curve
3.2.1. Animals
3.2.2. TPA-Induced Ear Edema
3.3. Antioxidant Activity (ABTS)
3.4. Polyphenolic Determination by UPLC-DAD
Determination of Polyphenolic Content
3.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jahnavi, K.; Pavani-Reddy, P.; Vasudha, B.; Narender, B. Non-steroidal anti-inflammatory drugs: An overview. J. Drug Deliv. Ther. 2019, 9, 442–448. [Google Scholar] [CrossRef]
- Charlier, C.; Michaux, C. Dual inhibition of cyclooxygenase-2 (COX-2) and 5-lipoxygenase (5-LOX) as a new strategy to provide safer non-steroidal anti-inflammatory drugs. Eur. J. Med. Chem. 2003, 38, 645–659. [Google Scholar] [CrossRef]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and other phenolic compounds from medicinal plants for pharmaceutical and medical aspects: An overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef] [PubMed]
- Sowjanya, R.; Shankar, M.; Sireesha, B.; Naik, A.E.; Yudharaj, P.; Priyadarshini, R.R. An overview on inflammation and plant havinganti inflammatory activity. Int. J. Phytopharm. Res. 2017, 7, 25–32. [Google Scholar]
- Arulselvan, P.; Tangestani-Fard, M.; Sean-Tan, W.; Gothai, S.; Fakurazi, S.; Norhaizan, M.E.; Suresh-Kumar, S. Role of antioxidants and natural products in inflammation. Oxid. Med. Cell. Longev. 2016, 2016, 5276130. [Google Scholar] [CrossRef] [Green Version]
- Shaikh, R.U.; Pund, M.M.; Gacche, R.N. Evaluation of anti-inflammatory activity of selected medicinal plants used in Indian traditional medication system in vitro as well as in vivo. J. Tradit. Complement. Med. 2016, 6, 355–361. [Google Scholar] [CrossRef] [Green Version]
- Lourenço, S.C.; Moldão-Martins, M.; Alves, V.D. Antioxidants of natural plant origins: From sources to food industry applications. Molecules 2019, 24, 4132. [Google Scholar] [CrossRef] [Green Version]
- Huang, X.X.; Ming, B.; Zhou, L.; Lou, L.L.; Liu, Q.B.; Zhang, Y.; Li, L.Z.; Song, S.J. Food by-products as a new and cheap source of bioactive compounds: Lignans with antioxidant and anti-inflammatory properties from Crataegus pinnatifida seeds. J. Agric. Food Chem. 2015, 63, 7252–7260. [Google Scholar] [CrossRef]
- Rodríguez-Buenfil, I.M.; Ramírez-Sucre, M.O.; Ramírez-Rivera, E. Metabolómica y Cultivo del Chile Habanero (Capsicum Chinense Jacq) de la Península de Yucatán, 1st ed.; CIATEJ: Jalisco, Mexico, 2020; Chapters 6–9, 11; pp. 95–169, 185–216. ISBN 978-607-8734-09-2. [Google Scholar]
- Roy, A. Bhut Jolokia (Capsicum chinense Jacq): A Review. Int. J. Pharm. Sci. Res. 2016, 7, 882–889. [Google Scholar] [CrossRef]
- SAGARPA. Agenda Técnica Agrícola Yucatán, 2nd ed.; Secretaría de Agricultura, Ganadería, Desarrollo Rural, Pesca y Alimentación: Mexico City, Mexico, 2015; pp. 21–32. [Google Scholar]
- SIAP: Anuario Estadístico de la Producción Agrícola. Available online: https://nube.siap.gob.mx/cierreagricola/ (accessed on 31 August 2021).
- Rodríguez-Buenfil, I.M.; Ramírez-Sucre, M.O.; Echevarría-Machado, I. Soils of Yucatan: Effect on the growth of the habanero chili plant (Capsicum chinense) mini review. Agric. Res. Technol. Open Access J. 2017, 8, 1–2. [Google Scholar] [CrossRef]
- Borges-Gómez, L.; Moo-Kauil, C.; Ruíz-Novelo, J.; Osalde-Balam, M.; González-Valencia, C.; Yam-Chimal, C.; Can-Puc, F. Suelos destinados a la producción de chile habanero en Yucatán: Características físicas y químicas predominantes. Agrociencia 2014, 48, 347–359. [Google Scholar]
- Oney-Montalvo, J.; Uc-Varguez, A.; Ramírez-Rivera, E.; Ramírez-Sucre, M.; Rodríguez-Buenfil, I. Influence of soil composition on the profile and content of polyphenols in habanero peppers (Capsicum chinense Jacq). Agronomy 2020, 10, 1234. [Google Scholar] [CrossRef]
- Herrera-Pool, E.; Patrón-Vázquez, J.; Ramos-Díaz, A.; Ayora-Talavera, T.; Pacheco, N. Extraction and identification of phenolic compounds in roots and leaves of Capsicum chinense by UPLC–PDA/MS. J. Bioeng. 2019, 3, 17–27. [Google Scholar]
- Ukwuani, A.N.; Hassan, I.B. In vitro anti-inflammatory activity of hydromethanolic seed, fruit and leave extracts of Capsicum chinense (red pepper). Eur. J. Biomed. Pharm. 2015, 2, 57–65. [Google Scholar]
- Ferrentino, G.; Morozova, K.; Kongi-Mosibo, O.; Ramezani, M.; Scampicchio, M. Biorecovery of antioxidants from apple pomace by supercritical fluid extraction. J. Clean. Prod. 2018, 186, 253–261. [Google Scholar] [CrossRef]
- Chel-Guerrero, L.D.; Sauri-Duch, E.; Fragoso-Serrano, M.C.; Pérez-Flores, L.J.; Gómez-Olivares, J.L.; Salinas-Arreortua, N.; Sierra-Palacios, E.C.; Mendoza-Espinoza, J.A. Phytochemical profile, toxicity, and pharmacological potential of peels from four species of tropical fruits. J. Med. Food 2018, 21, 734–743. [Google Scholar] [CrossRef]
- Ameer, K.; Shahbaz, H.M.; Kwon, J.H. Green Extraction methods for polyphenols from plant matrices and their byproducts: A review. Compr. Rev. Food Sci. Food Saf. 2017, 16, 295–315. [Google Scholar] [CrossRef] [Green Version]
- Ghasemian, M.; Owlia, S.; Owlia, M.B. Review of anti-inflammatory herbal medicines. Adv. Pharmacol. Sci. 2016, 2016, 9130979. [Google Scholar] [CrossRef] [Green Version]
- Isah, T. Stress and defense responses in plant secondary metabolites production. Biol. Res. 2019, 52, 39. [Google Scholar] [CrossRef] [Green Version]
- Yang, W.; Chen, X.; Li, Y.; Guo, S.; Wang, Z.; Yu, X. Advances in pharmacological activities of terpenoids. Nat. Prod. Commun. 2020, 15, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Suwa, R.; Tajima, H.; Gima, S.; Uehara, N.; Watanabe, K.; Yabuta, S.; Tominaga, J.; Kawamitsu, Y. Polyphenol production in Peucedanum japonicum Thunb. varies with soil type and growth stage. Hort. J. 2018, 87, 382–388. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Tsao, R. Dietary polyphenols, oxidative stress and antioxidant and anti-inflammatory effects. Curr. Opin. Food Sci. 2016, 8, 33–42. [Google Scholar] [CrossRef]
- Ormeño, E.; Fernandez, C. Effect of soil nutrient on production and diversity of volatile terpenoids from plants. Curr. Bioact. Compds. 2012, 8, 71–79. [Google Scholar]
- Sinkovic, L.; Demšar, L.; Žnidarcic, D.; Vidrih, R.; Hribar, J.; Treutter, D. Phenolic profiles in leaves of chicory cultivars (Cicho-rium intybus L.) as influenced by organic and mineral fertilizers. Food Chem. 2015, 166, 507–513. [Google Scholar] [CrossRef]
- Herrero, M.; Plaza, M.; Cifuentes, A.; Ibañez, E. Extraction techniques for the determination of phenolic compounds in food. Compr. Sampl. Sample Prep. 2012, 4, 159–180. [Google Scholar] [CrossRef] [Green Version]
- Enogieru, A.B.; Haylett, W.; Hiss, D.C.; Bardien, S.; Ekpo, O.E. Rutin as a potent antioxidant: Implications for neurodegenerative disorders. Oxid. Med. Cell. Longev. 2018, 2018, 6241017. [Google Scholar] [CrossRef]
- Sova, M. Antioxidant and antimicrobial activities of cinnamic acid derivatives. Mini Rev. Med. Chem. 2012, 12, 749–767. [Google Scholar] [CrossRef]
- Kadioglu, O.; Nass, J.; Saeed, M.E.; Schuler, B.; Efferth, T. Kaempferol is an anti-inflammatory compound with activity towards NF-κB pathway proteins. Anticancer Res. 2015, 35, 2645–2650. [Google Scholar] [PubMed]
- Abubakar, A.R.; Haque, M. Preparation of Medicinal Plants: Basic Extraction and Fractionation Procedures for Experimental Purposes. J. Pharm. Bioallied. Sci. 2020, 12, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Eun-Yi, K.; Weon-Jong, Y.; Hae-Won, L.; Soo-Jin, H.; Young-Hwan, K.; Shanura, I.P.F.; Kichul, C.; Chi-Heon, L.; Sung-Pyo, H.; Su-Hyeon, C.; et al. Anti-inflammatory effect of supercritical extract and its constituents from Ishige okamurae. EXCLI J. 2016, 15, 434–445. [Google Scholar] [CrossRef]
- Herrera-Pool, E.; Ramos-Díaz, A.; Lizardi-Jiménez, M.A.; Pech-Cohuo, S.; Ayora-Talavera, T.; Cuevas-Bernardino, J.C.; García-Cruz, U.; Pacheco, N. Effect of solvent polarity on the Ultrasound Assisted extraction and antioxidant activity of phenolic compounds from habanero pepper leaves (Capsicum chinense) and its identification by UPLC-PDA-ESI-MS/MS. Ultrason. Sonochem. 2021, 76, 105658. [Google Scholar] [CrossRef]
- Gougoulias, N.; Wogiatzi, E.; Vagelas, L.; Giurgiulescu, L.; Gogou, I.; Ntalla, M.; Kalfountzos, D. Comparative study on polyphenols content, capsaicin and antioxidant activity of different hot peppers varieties (Capsicum annuum L.) under environmental conditions of thessaly region, Greece. Carpathian J. Food Sci. Technol. 2017, 9, 109–116. [Google Scholar]
- Fernández, S.; Carreras, T.; Castro, R.; Perelmuter, K.; Giorgi, V.; Vila, A.; Rosales, A.; Pazos, M.; Moyna, G.; Carrera, I.; et al. A comparative study of supercritical fluid and ethanol extracts of Cannabis inflorescences: Chemical profile and biological activity. J. Supercrit. Fluids 2021, 179, 105385. [Google Scholar] [CrossRef]
- Kőszegi, K.; Vatai1, G.; Békássy-Molnár, E. Comparison the soxhlet and supercritical fluid extraction of nettle root (Urtica dioica L.). Period. Polytech. Chem. Eng. 2015, 59, 168–173. [Google Scholar] [CrossRef] [Green Version]
- Tyskiewicz, K.; Konkol, M.; Rój, E. the application of supercritical fluid extraction in phenolic compounds isolation from natural plant materials. Molecules 2018, 23, 2625. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chaves, J.O.; De Souza, M.C.; Da Silva, L.C.; Lachos-Perez, D.; Torres-Mayanga, P.C.; Machado, A.P.F.; Forster-Carneiro, T.; Vázquez-Espinosa, M.; González-de-Peredo, A.V.; Barbero, G.F.; et al. Extraction of flavonoids from natural sources using modern techniques. Front. Chem. 2020, 8, 507887. [Google Scholar] [CrossRef]
- Poletto, P.; Alvarez-Rivera, G.; Torres, T.M.S.; Mendiola, J.A.; Ibañez, E.; Cifuentes, A. Compressed fluids and phytochemical profiling tools to obtain and characterize antiviral and anti-inflammatory compounds from natural sources. Trends Anal. Chem. 2020, 129, 115942. [Google Scholar] [CrossRef]
- Bakasataea, N.; Kunworaratha, N.; Yupanqui, C.T.; Voravuthikunchai, S.P.; Joycharat, N. Bioactive components, antioxidant, and anti-inflammatory activities of the wood of Albizia myriophylla. Revista Brasileira de Farmacognosia 2018, 28, 444–450. [Google Scholar] [CrossRef]
- Grigore, A.; Colceru-Mihul, S.; Litescu, S.; Panteli, M.; Rasit, I. Correlation between polyphenol content and anti-inflammatory activity of Verbascum phlomoides (mullein). Pharm. Biol. 2013, 51, 925–929. [Google Scholar] [CrossRef] [Green Version]
- Villanueva-Bermejo, D.; Vázquez, E.; Villalva, M.; Santoyo, S.; Fornari, T.; Reglero, G.; García-Risco, M.R. Simultaneous supercritical fluid extraction of Heather (Calluna vulgaris L.) and Marigold (Calendula ocinalis L.) and anti-inflammatory activity of the extracts. Appl. Sci. 2019, 9, 2245. [Google Scholar] [CrossRef] [Green Version]
- Paes, J.; Dotta, R.; Barbero, G.F.; Martínez, J. Extraction of phenolic compounds and anthocyanins from blueberry (Vaccinium myrtillus L.) residues using supercritical CO2 and pressurized liquids. J. Supercrit. Fluids 2014, 95, 8–16. [Google Scholar] [CrossRef]
- Evaluacion.ssm.gob.mx. Available online: http://evaluacion.ssm.gob.mx/pdf/normateca/NOM-220-SSA1–2012.pdf (accessed on 21 September 2021).
- Ren, Z.; Zhang, R.; Li, Y.; Li, Y.; Yang, Z.; Yang, H. Ferulic acid exerts neuroprotective effects against cerebral ischemia/reperfusion-induced injury via antioxidant and anti-apoptotic mechanisms in vitro and in vivo. Int. J. Mol. Med. 2017, 40, 1444–1456. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gilman, S.C.; Carlson, R.P.; Chang, J.; Lewis, A.J. The antiinflammatory activity of the immunomodulator Wy-18,251 (3-(p-chlorophenyl)thiazolo[3,2-a]benzimidazole-2-acetic acid). Agents Actions 1985, 17, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Sigma Aldrich. Available online: https://www.sigmaaldrich.com/deepweb/assets/sigmaaldrich/product/documents/281/332/cs0790bul.pdf (accessed on 1 September 2020).
- Magnusson, B.; Örnemark, U. Eurachem Guide: The Fitness for Purpose of Analytical Methods—A Laboratory Guide to Method Validation and Related Topics, 2nd ed.; Eurachem Publisher: Teddington, UK, 2014; pp. 1–70. Available online: www.eurachem.org (accessed on 6 June 2021).
- Chel-Guerrero, D.L.; Oney-Montalvo, J.E.; Rodriguez-Buenfil, I.M. Phytochemical Characterization of By-Products of Habanero Pepper Grown in Two Different Types of Soils from Yucatán, Mexico. Plants 2021, 10, 779. [Google Scholar] [CrossRef] [PubMed]
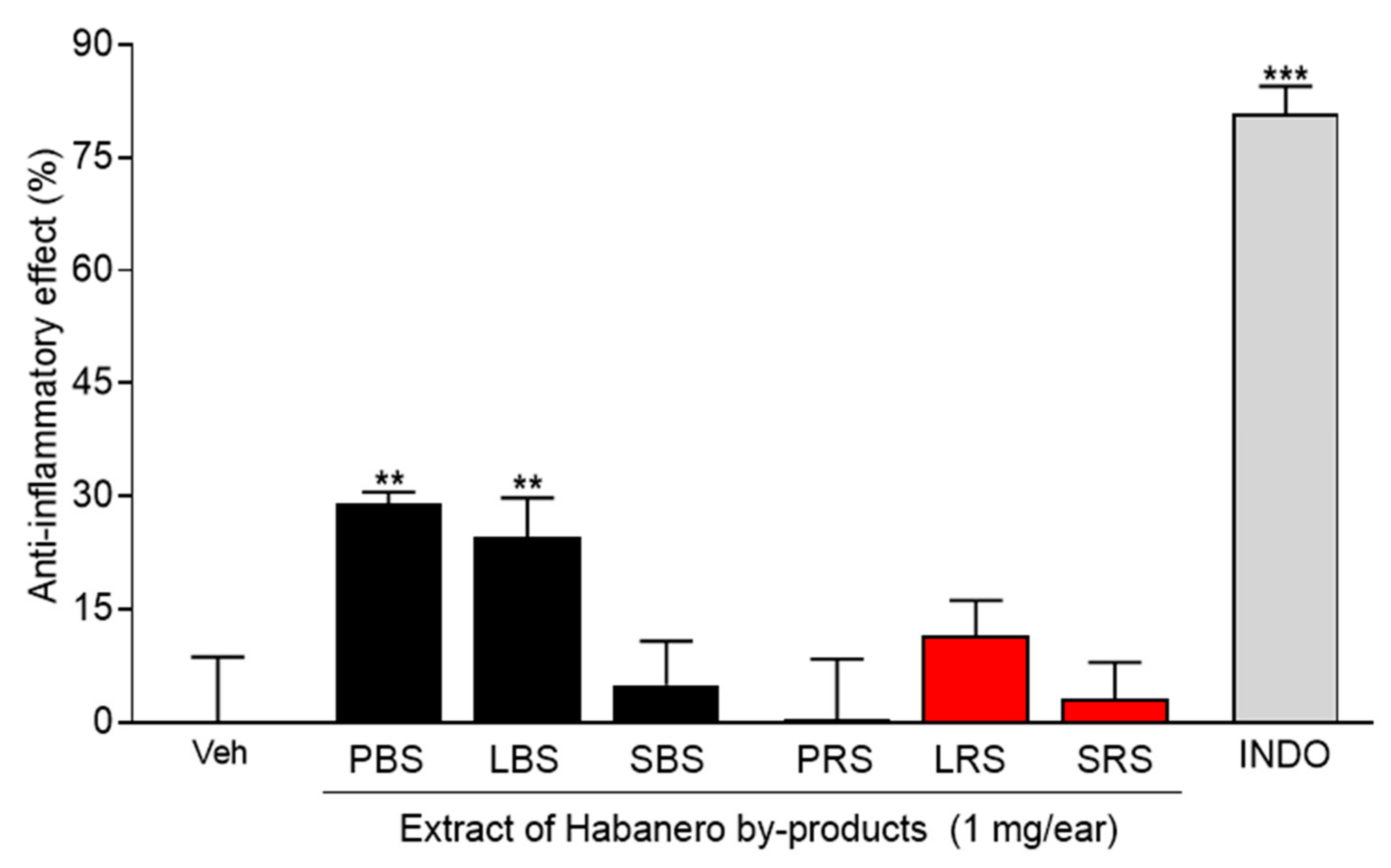


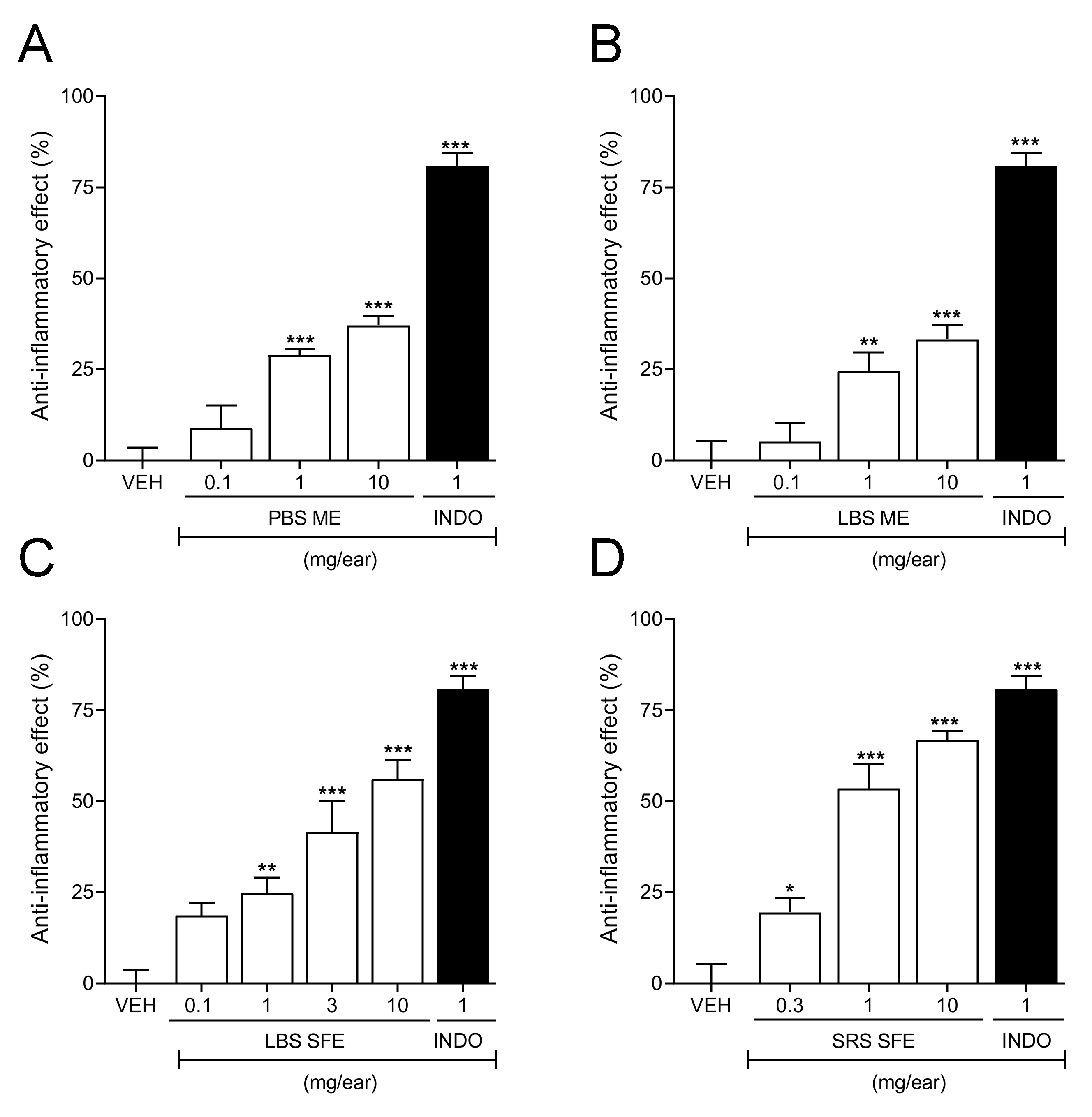
| Sample | Type of Soil | Antioxidant Activity (ABTS) Trolox Equivalent (mM) A |
|---|---|---|
| Peduncles | Black soil | 2.29 ± 0.0039 d |
| Leaves | Black soil | 0.74 ± 0.0007 f |
| Stems | Black soil | 2.56 ± 0.0026 c |
| Peduncles | Red soil | 2.57 ± 0.0065 b |
| Leaves | Red soil | 0.83 ± 0.0007 e |
| Stems | Red soil | 2.80 ± 0.0052 a |
| Polyphenols | Content (mg 100 g−1 DB) A | |||||
|---|---|---|---|---|---|---|
| Black Soil | Red Soil | |||||
| Peduncles | Leaves | Stems | Peduncles | Leaves | Stems | |
| Chlorogenic acid | 292.70 ± 0.75 d | 308.53 ± 1.53 a | 302.50 ± 3.41 b | 293.93 ± 1.10 d | 311.13 ± 0.50 a | 297.80 ± 0.87 c |
| Coumaric acid | 137.20 ± 1.73 b | 123.90 ± 0.46 c | 113.07 ± 0.23 d | 141.67 ± 0.06 a | 137.77 ± 0.25 b | 112.10 ± 0.56 d |
| p-coumaric acid | 99.60 ± 0.30 b | 94.17 ± 0.06 c | 86.47 ± 0.06 d | 105.30 ± 0.44 a | 95.27 ± 2.29 c | 86.47 ± 0.06 d |
| Cinnamic acid | 132.03 ± 1.63 a | 125.93 ± 1.68 c | 131.70 ± 0.20 a | 133.23 ± 0.21 a | 127.90 ± 0.44 b | 133.07 ± 0.67 a |
| Vanillin | 64.43 ± 0.21 b | 46.20 ± 0.72 d | 29.27 ±0.35 e | 71.63 ± 0.31 a | 61.47 ± 1.98 c | 26.53 ± 0.64 f |
| Catechin | 0.00 ± 0.00 b | 0.00 ± 0.00 b | 0.00 ± 0.00 b | 142.90 ± 0.99 a,* | 0.00 ± 0.00 b | 0.00 ± 0.00 b |
| Myricetin | 465.13 ± 3.27 b | 461.47 ± 16.66 b | 454.93 ± 17.12 b | 457.70 ± 1.35 b | 489.33 ± 16.57 a | 450.10 ± 1.55 b |
| Diosmetin | 0.00 ± 0.00 b | 0.00 ± 0.00 b | 0.00 ± 0.00 b | 0.00 ± 0.00 b | 123.10 ± 0.00 a,* | 0.00 ± 0.00 b |
| Rutin | 239.30 ± 8.88 b | 99.43 ± 2.25 e | 192.23 ± 2.34 c | 265.13 ± 1.63 a | 116.15 ± 2.76 d | 232.00 ± 9.85 b |
| Kaempferol | 208.33 ± 4.05 a | 204.07 ± 1.46 b | 0.00 ± 0.00 c | 203.67 ± 2.34 b | 204.70 ± 2.48 a,b | 0.00 ± 0.00 c |
| Quercetin + Luteolin ** | 531.37 ± 0.61 b | 506.77 ± 9.47 d | 500.77 ± 1.87 d | 519.00 ± 0.82 c | 505.30 ± 0.79 d | 562.10 ± 1.47 a |
| Hesperidin + Diosmin ** | 187.97 ± 0.49 b | 122.57 ± 4.16 e | 126.40 ± 0.95 e | 204.77 ± 0.81 a | 143.33 ± 6.07 c | 133.80 ± 1.93 d |
| Neohesperidin | 36.63 ± 1.15 a | 28.53 ± 2.40 b | 31.57 ± 5.35 a,b | 33.10 ± 5.11 a,b | 36.23 ± 1.26 a | 27.47 ± 0.31 b |
| Naringenin | 67.13 ± 0.15 a | 0.00 ± 0.00 d | 0.00 ± 0.00 d | 65.97 ± 0.15 b | 63.23 ± 0.32 c | 0.00 ± 0.00 d |
| Total Content B | 2462.83 ± 10.98 b | 2121.57 ± 23.97 d | 1968.90 ± 14.48 e | 2638.00 ± 87.44 a | 2414.92 ± 86.65 c | 2061.43 ± 10.43 d |
| Compounds or Bioactivity | Anti-Inflammatory Effect (% Inhibition) | Antioxidant Activity (ABTS) 1 |
|---|---|---|
| Chlorogenic acid | 0.11 | −0.83 |
| Coumaric acid | 0.24 | −0.28 |
| p-Coumaric acid | 0.19 | −0.11 |
| Cinnamic acid | −0.52 | 0.97 |
| Vanillin | 0.26 | − 0.26 |
| Catechin | −0.49 | 0.32 |
| Myricetin | 0.29 | −0.72 |
| Rutin | −0.38 | 0.92 |
| Kaempferol | 0.54 | −0.59 |
| Quercetin + Luteolin | −0.14 | 0.56 |
| Diosmetin | −0.03 | −0.60 |
| Hesperidin + Diosmin | −0.01 | 0.37 |
| Neohesperidin | 0.28 | −0.14 |
| Naringenin | 0.13 | −0.06 |
| Antioxidant activity (ABTS) 1 | −0.51 | 1.00 |
| % Anti-inflammatory effect | 1.00 |
| Sample | Type of Soil | Type of Extraction | Trolox Equivalent (mM) A |
|---|---|---|---|
| Leaves | Black soil | ME | 0.74 ± 0.0007 g |
| Leaves | Black soil | SOX | 0.80 ± 0.002 d |
| Leaves | Black soil | SFE | 0.75 ± 0.001 f |
| Stems | Black soil | ME | 2.56 ± 0.0026 b |
| Stems | Black soil | SOX | 0.83 ± 0.0013 c |
| Stems | Black soil | SFE | 0.58 ± 0.0023 i |
| Stems | Red soil | ME | 2.80 ± 0.0052 a |
| Stems | Red soil | SOX | 0.79 ± 0.0016 e |
| Stems | Red soil | SFE | 0.73 ± 0.0016 h |
| Compound | Anti-Inflammatory Effect (% Inhibition) | Antioxidant Activity (ABTS) 1 |
|---|---|---|
| Gallic Acid | −0.32 | −0.17 |
| Protocatechuic Acid | −0.37 | −0.29 |
| Chlorogenic Acid | −0.63 | 0.48 |
| Coumaric Acid | −0.13 | 0.15 |
| p-Coumaric Acid | −0.69 | 0.40 |
| Cinnamic Acid | −0.22 | 0.23 |
| Vanillin | −0.14 | 0.13 |
| Catechin | −0.33 | −0.22 |
| Myricetin | −0.70 | 0.34 |
| Rutin | −0.31 | 0.80 |
| Kaempferol | −0.01 | −0.34 |
| Quercetin + Luteolin | −0.40 | 0.60 |
| Apigenin | −0.03 | −0.17 |
| Diosmetin | −0.03 | −0.17 |
| Hesperidin + Diosmin | −0.67 | 0.52 |
| Neohesperidin | −0.65 | 0.28 |
| Naringenin | −0.03 | −0.17 |
| Antioxidant activity (ABTS) 1 | −0.57 | 1.00 |
| Anti-inflammatory effect (% Inhibition) | 1.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chel-Guerrero, L.D.; Castañeda-Corral, G.; López-Castillo, M.; Scampicchio, M.; Morozova, K.; Oney-Montalvo, J.E.; Ferrentino, G.; Acevedo-Fernández, J.J.; Rodríguez-Buenfil, I.M. In Vivo Anti-Inflammatory Effect, Antioxidant Activity, and Polyphenolic Content of Extracts from Capsicum chinense By-Products. Molecules 2022, 27, 1323. https://doi.org/10.3390/molecules27041323
Chel-Guerrero LD, Castañeda-Corral G, López-Castillo M, Scampicchio M, Morozova K, Oney-Montalvo JE, Ferrentino G, Acevedo-Fernández JJ, Rodríguez-Buenfil IM. In Vivo Anti-Inflammatory Effect, Antioxidant Activity, and Polyphenolic Content of Extracts from Capsicum chinense By-Products. Molecules. 2022; 27(4):1323. https://doi.org/10.3390/molecules27041323
Chicago/Turabian StyleChel-Guerrero, Lilian Dolores, Gabriela Castañeda-Corral, Misael López-Castillo, Matteo Scampicchio, Ksenia Morozova, Julio Enrique Oney-Montalvo, Giovanna Ferrentino, Juan José Acevedo-Fernández, and Ingrid Mayanín Rodríguez-Buenfil. 2022. "In Vivo Anti-Inflammatory Effect, Antioxidant Activity, and Polyphenolic Content of Extracts from Capsicum chinense By-Products" Molecules 27, no. 4: 1323. https://doi.org/10.3390/molecules27041323
APA StyleChel-Guerrero, L. D., Castañeda-Corral, G., López-Castillo, M., Scampicchio, M., Morozova, K., Oney-Montalvo, J. E., Ferrentino, G., Acevedo-Fernández, J. J., & Rodríguez-Buenfil, I. M. (2022). In Vivo Anti-Inflammatory Effect, Antioxidant Activity, and Polyphenolic Content of Extracts from Capsicum chinense By-Products. Molecules, 27(4), 1323. https://doi.org/10.3390/molecules27041323











