Cellulose Acetate-g-Polycaprolactone Copolymerization Using Diisocyanate Intermediates and the Effect of Polymer Chain Length on Surface, Thermal, and Antibacterial Properties
Abstract
:1. Introduction
2. Results and Discussion
2.1. Synthesis of Polycaprolactone (PCL)
2.2. Determination of DP (for PCL) by Viscosity Measurements
2.3. Measurement of Hydroxyl Value
2.4. Synthesis of HEMA-PCL-NCO
2.5. Preparation of CA-g-PCL
2.6. Effect of Degree of Polymerization (for PCL) in Grafting Density
2.7. Thermal Properties of CA-g-PCL
2.8. Effect of DP of PCL on the Contact Angle
2.9. SEM Analysis of Surfaces
2.10. Impact of Chain Length on Antibacterial Activity
3. Materials and Methods
3.1. Materials
3.2. Synthesis of Polycaprolactone (PCL)
3.3. Synthesis of DI-Terminated PCL (PCL-NCO)
3.4. Synthesis of Cellulose Acetate-g-PCL
3.5. Instruments/Equipment
3.5.1. Fourier Transform Infrared Spectroscopy (FTIR)
3.5.2. Differential Scanning Calorimetry (DSC)
3.5.3. Standard Thermogravimetric Analysis (TGA)
3.5.4. Viscosity Measurements
3.5.5. Measurement of Hydroxyl Value
3.5.6. Water Contact Angle (WCA)Tests
3.5.7. Scanning Electron Microscopy (SEM)
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Zhang, M.Q.; Rong, M.Z.; Lu, X. Fully biodegradable natural fiber composites from renewable resources: All-plant fiber composites. Compos. Sci. Technol. 2005, 65, 2514–2525. [Google Scholar] [CrossRef]
- Schroeter, J.; Felix, F. Melting cellulose. Cellulose 2005, 12, 159–165. [Google Scholar] [CrossRef]
- Frisoni, G.; Baiardo, M.; Scandola, M.; Lednická, D.; Cnockaert, M.C.; Mergaert, J.; Swings, J. Natural cellulose fibers: Heterogeneous acetylation kinetics and biodegradation behavior. Biomacromolecules 2001, 2, 476–482. [Google Scholar] [CrossRef] [PubMed]
- Joly, N.; Granet, R.; Krausz, P. Olefin metathesis applied to cellulose derivatives? Synthesis, analysis, and properties of new crosslinked cellulose plastic films. J. Polym. Sci. Part A Polym. Chem. 2005, 43, 407–418. [Google Scholar] [CrossRef]
- Nishio, Y.; Dong, Y.; Paris, M.; O’Keefe, R.J.; Schwarz, E.M.; Drissi, H. Runx2-mediated regulation of the zinc finger Osterix/Sp7 gene. Gene 2006, 372, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Yu, J.; Wan, J. Urea and ethanolamine as a mixed plasticizer for thermoplastic starch. Carbohydr. Polym. 2006, 64, 267–273. [Google Scholar] [CrossRef]
- Mayumi, A.; Kitaoka, T.; Wariishi, H. Partial substitution of cellulose by ring-opening esterification of cyclic esters in a homogeneous system. J. Appl. Polym. Sci. 2006, 102, 4358–4364. [Google Scholar] [CrossRef]
- Guruprasad, K.H.; Shashidhara, G.M. Grafting, blending, and biodegradability of cellulose acetate. J. Appl. Polym. Sci. 2004, 91, 1716–1723. [Google Scholar] [CrossRef]
- Hatakeyama, H.; Yoshida, T.; Hatakeyama, T. The effect of side chain association on thermal and viscoelastic properties: Cellulose acetate based polycaprolactones. J. Therm. Anal. Calorim. 2000, 59, 157–168. [Google Scholar] [CrossRef]
- Lee, S.-H.; Shiraishi, N. Plasticization of cellulose diacetate by reaction with maleic anhydride, glycerol, and citrate esters during melt processing. J. Appl. Polym. Sci. 2001, 81, 243–250. [Google Scholar] [CrossRef]
- Teramoto, Y.; Ama, S.; Higeshiro, T.; Nishio, Y. Cellulose Acetate-graft-Poly(hydroxyalkanoate)s: Synthesis and dependence of the thermal properties on copolymer composition. Macromol. Chem. Phys. 2004, 205, 1904–1915. [Google Scholar] [CrossRef]
- Warth, H.; Mülhaupt, R.; Schätzle, J. Thermoplastic cellulose acetate and cellulose acetate compounds prepared by reactive processing. J. Appl. Polym. Sci. 1997, 64, 231–242. [Google Scholar] [CrossRef]
- Yoshioka, M.; Hagiwara, N.; Shiraishi, N. Thermoplasticization of cellulose acetates by grafting of cyclic esters. Cellulose 1999, 6, 193–212. [Google Scholar] [CrossRef]
- lidrissi, A.; El barkany, S.; Amhamdi, H.; Maaroufi, A.; Hammouti, B. New approach to predict the solubility of polymers Application: Cellulose Acetate at various DS, preparedfrom Alfa “Stipa -tenassicima” of Eastern Morocco. J. Mater. Environ. Sci. 2012, 3, 270–285. [Google Scholar]
- Azzaoui, K.; Lamhamdi, A.; Mejdoubi, E.M.; Berrabah, M.; Hammouti, B.; Elidrissi, A.; Fouda, M.M.; Al-Deyab, S.S. Synthesis and characterization of composite based on cellulose acetate and hydroxyapatite application to the absorption of harmful substances. Carbohydr. Polym. 2014, 111, 41–46. [Google Scholar] [CrossRef]
- Zhang, N.; Wardwell, P.R.; Bader, R.A. Polysaccharide-Based Micelles for Drug Delivery. Pharmaceutics 2013, 5, 329–352. [Google Scholar] [CrossRef] [Green Version]
- Miller, T.; Goude, M.C.; McDevitt, T.C.; Temenoff, J.S. Molecular engineering of glycosaminoglycan chemistry for biomolecule delivery. Acta Biomater. 2014, 10, 1705–1719. [Google Scholar] [CrossRef] [Green Version]
- Posocco, B.; Dreussi, E.; De Santa, J.; Toffoli, G.; Abrami, M.; Musiani, F.; Grassi, M.; Farra, R.; Tonon, F.; Grassi, G.; et al. Polysaccharides for the Delivery of Antitumor Drugs. Materials 2015, 8, 2569–2615. [Google Scholar] [CrossRef] [Green Version]
- Rodrigues, S.; Cardoso, L.; Da Costa, A.M.R.; Grenha, A. Biocompatibility and stability of polysaccharide polyelectrolyte complexes aimed at respiratory delivery. Materials 2015, 8, 5647–5670. [Google Scholar] [CrossRef] [Green Version]
- Raeisi, F.R.E. Mini review of polysaccharide nanoparticles and drugdelivery process. Adv. Appl. NanoBio-Technol. 2020, 1, 33–44. [Google Scholar]
- Dwivedi, R.; Kumar, S.; Pandey, R.; Mahajan, A.; Nandana, D.; Katti, D.S.; Mehrotra, D. Polycaprolactone as biomaterial for bone scaffolds: Review of literature. J. Oral Biol. CraniofacialRes. 2020, 10, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Cesur, S.; Köroğlu, C.; Yalçın, H.T. Antimicrobial and biodegradable food packaging applications of polycaprolactone/organo nanoclay/chitosan polymeric composite films. J. VinylAddit. Technol. 2018, 24, 376–387. [Google Scholar] [CrossRef]
- Roy, D.; Semsarilar, M.; Guthrie, J.T.; Perrier, S. Cellulose modification by polymer grafting: A review. Chem. Soc. Rev. 2009, 38, 2046–2064. [Google Scholar] [CrossRef] [PubMed]
- Tizzotti, M.; Charlot, A.; Fleury, E.; Stenzel, M.; Bernard, J. Modification of polysaccharides through controlled/living radical polymerization grafting-towards the generation of high performance hybrids. Macromol. Rapid Commun. 2010, 31, 1751–1772. [Google Scholar] [CrossRef]
- Tabaght, F.E.; El Idrissi, A.; Achelhi, N.; Elyousfi, R.; Bellaouchi, R.; Asehraou, A.; Elbarkany, S. Grafting of fibrillated cellulose with acrylic compounds: Synthesis, properties and biodegradation. Mor. J. Chem. 2019, 3, 595–614. [Google Scholar]
- Barsbay, M.; Güven, O.; Davis, T.P.; Barner-Kowollik, C.; Barner, L. RAFT-mediated polymerization and grafting of sodium 4-styrenesulfonate from cellulose initiated via γ-radiation. Polymer 2009, 50, 973–982. [Google Scholar] [CrossRef]
- Carlmark, A.; Malmström, E.E. ATRP grafting from cellulose fibers to create block-copolymer grafts. Biomacromolecules 2003, 4, 1740–1745. [Google Scholar] [CrossRef]
- Malmström, E.; Carlmark, A. Controlled grafting of cellulose fibres—An outlook beyond paper and cardboard. Polym. Chem. 2011, 3, 1702–1713. [Google Scholar] [CrossRef]
- Roy, D.; Guthrie, A.J.T.; Perrier, S. Graft polymerization: Grafting poly(styrene) from cellulose via reversible addition−fragmentation chain transfer (raft) polymerization. Macromolecules 2005, 38, 10363–10372. [Google Scholar] [CrossRef]
- Hansson, S.; Trouillet, V.; Tischer, T.; Goldmann, A.S.; Carlmark, A.; Barner-Kowollik, C.; Malmström, E. Grafting efficiency of synthetic polymers onto biomaterials: A comparative study of grafting-from versus grafting-to. Biomacromolecules 2012, 14, 64–74. [Google Scholar] [CrossRef]
- Kang, H.; Liu, R.; Huang, Y. Graft modification of cellulose: Methods, properties and applications. Polymer 2015, 70, A1–A16. [Google Scholar] [CrossRef]
- Azzaoui, K.; Mejdoubi, E.; Lamhamdi, A.; Hammouti, B.; Akartasse, N.; Berrabah, M.; Elidrissi, A.; Jodeh, S.; Hamed, O.; Abidi, N. Novel tricomponenets composites films from polylactic Acid/Hydroxyapatite/Poly-Caprolactone suitable for Biomedical Applications. J. Mater. Environ. Sci. 2016, 7, 761–769. [Google Scholar]
- Paquet, O.; Krouit, M.; Bras, J.; Thielemans, W.; Belgacem, M.N. Surface modification of cellulose by PCL grafts. Acta Mater. 2010, 58, 792–801. [Google Scholar] [CrossRef]
- Tabaght, F.E.; El Idrissi, A.; Bellaouchi, R.; Asehraou, A.; Aqil, M.; El Barkany, S.; Benarbia, A.; Achalhi, N.; Tahani, A. Cellulose grafted aliphatic polyesters: Synthesis, characterization and biodegradation under controlled conditions in a laboratory test system. J. Mol. Struct. 2020, 1205, 127582. [Google Scholar] [CrossRef]
- El Idrissi, A.; El Barkany, S.; Amhamdi, H.; Maaroufi, A.-K. Physicochemical characterization of celluloses extracted from Esparto “Stipa tenacissima” of Eastern Morocco. J. Appl. Polym. Sci. 2013, 128, 537–548. [Google Scholar] [CrossRef]
- El Barkany, S.; Jilal, I.; El Idrissi, A.; Abou-Salama, M.; Zanagui, C.; Salhi, A.; Amhamdi, H. Homogenous synthesis of new c6 regioselective cellulose acetate Butyl hexane-1,6-Diyldicarbamate based on esparto cellulose of morocco oriental. Appl. J. Environ. Eng. Sci. 2017, 3, 3. [Google Scholar]
- Valipour, F.; Valipour, F.; Rahbarghazi, R.; Navali, A.M.; Rashidi, M.R.; Davaran, S. Novel hybrid polyester-polyacrylate hydrogels enriched with platelet-derived growth factor for chondrogenic differentiation of adipose-derived mesenchymal stem cells in vitro. J. Biol. Eng. 2021, 15, 6. [Google Scholar] [CrossRef]
- Gnanou, Y.; Fontanille, M. Organic and Physical Chemistry of Polymers; Wiley: Hoboken, NJ, USA, 2008. [Google Scholar]
- Huggins, M.L. Methods for the estimation of molecularweights of high polymersfrom data on properties of the irdilute solutions are critically discussed. Specificrecommendations are made regarding future procedure. J. Am. Chem. Soc. 1942, 64, 2716. [Google Scholar]
- Kraemer, E.O. Molecular weights of celluloses. Ind. Eng. Chem. 1938, 30, 1200. [Google Scholar] [CrossRef]
- Narr, R.Z.; Zabusky, H.H.; Heitmiller, R.F. Single point determination of intrinsic viscosity. J. Appl. Polym. Sci. 1963, 7, S30. [Google Scholar] [CrossRef]
- Wagner, H.L. The Mark–Houwink–Sakurada equation for the viscosity of linear polyethylene. J. Phys. Chem. Ref. Data 1985, 14, 611–617. [Google Scholar] [CrossRef] [Green Version]
- Koleske, J.V.; Lundberg, R.D. Lactone polymers. II. Hydrodynamic properties and unperturbed dimensions of poly-ε-caprolactone. J. Polym. Sci. A-2 Polym. Phys. 1969, 7, 897–907. [Google Scholar] [CrossRef]
- AOAC Official Method 965.32, Hydroxyl Value of Oils and Fats Acetylation Method. First Action 1965-Final Action 1969. Available online: https://www.coursehero.com/file/31058218/AOAC-96532-Hydroxyl-Valuespdf/ (accessed on 7 February 2022).
- Grumezescu, V.; Grumezescu, A.M. (Eds.) Materials for Biomedical Engineering Thermoset and Thermoplastic Polymers 2019; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Mathew, A.P.; Dufresne, A. Morphological investigation of nanocomposites from sorbitol plasticized starch and tunicin whiskers. Biomacromolecules 2002, 3, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Marcovich, N.; Auad, M.; Bellesi, N.; Nutt, S.; Aranguren, M.I. Cellulose micro/nanocrystals reinforced polyurethane. J. Mater. Res. 2006, 21, 870–881. [Google Scholar] [CrossRef]
- Grunert, M.; Winter, W.T. Nanocomposites of cellulose acetate butyrate reinforced with cellulose nanocrystals. J. Polym. Environ. 2002, 10, 27–30. [Google Scholar] [CrossRef]
- Hassan, M.; Bras, J.; Hassan, E.A.; Fadel, S.M.; Dufresne, A. Polycaprolactone/modified bagasse whisker nanocomposites with improved moisture-barrier and biodegradability properties. J. Appl. Polym. Sci. 2012, 125, E10–E19. [Google Scholar] [CrossRef]
- Kumar, G.S.; Rajendran, S.; Karthi, S.; Govindan, R.; Girija, E.K.; Karunakaran, G.; Kuznetsov, D. Green synthesis and antibacterial activity of hydroxyapatite nanorods for orthopedic applications. MRS Commun. 2017, 7, 183–188. [Google Scholar] [CrossRef] [Green Version]
- Lin, J.; Ding, J.; Dai, Y.; Wang, X.; Wei, J.; Chen, Y. Antibacterial zinc oxide hybrid with gelatin coating. Mater. Sci. Eng. C 2017, 81, 321–326. [Google Scholar] [CrossRef]





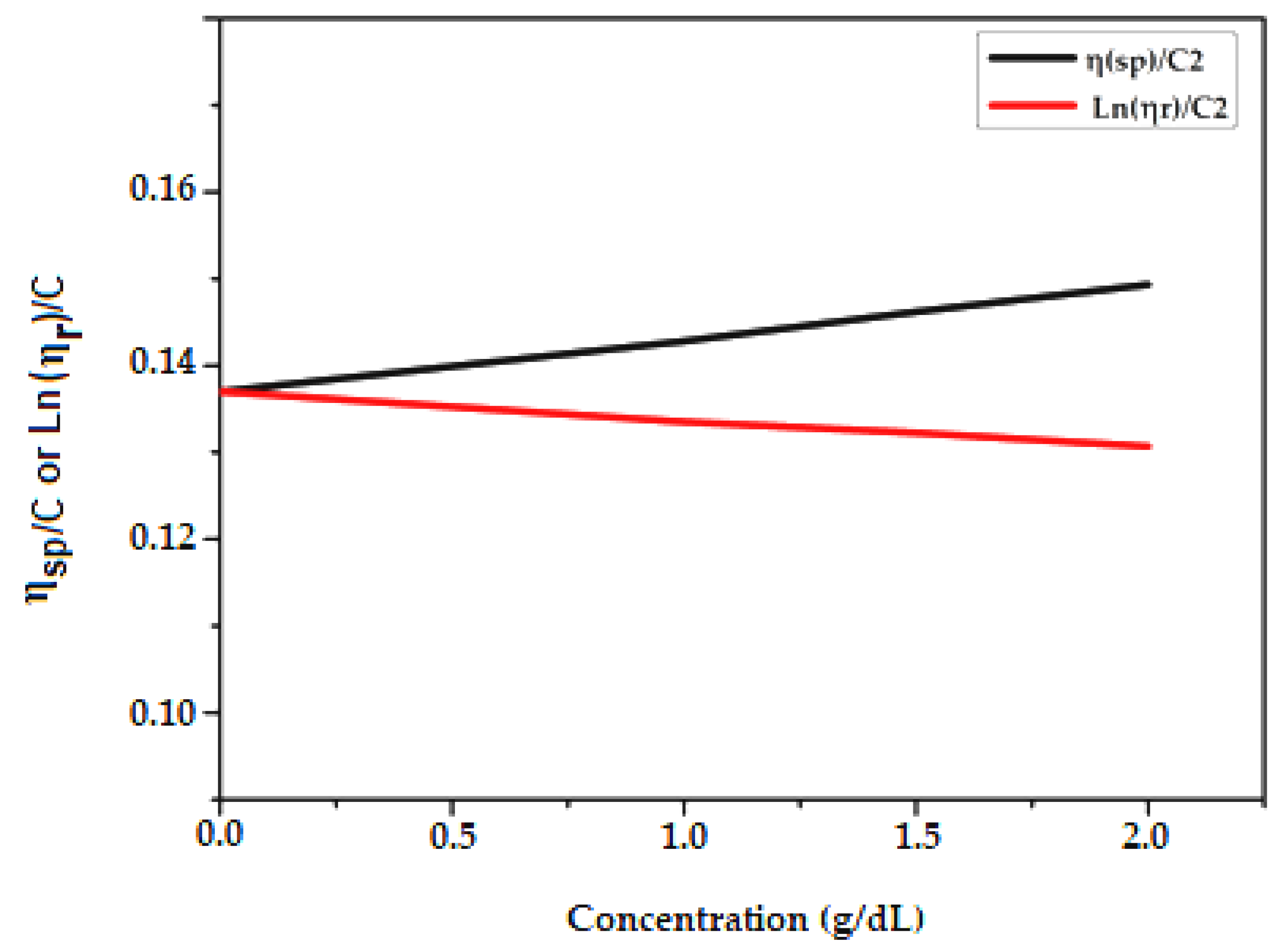



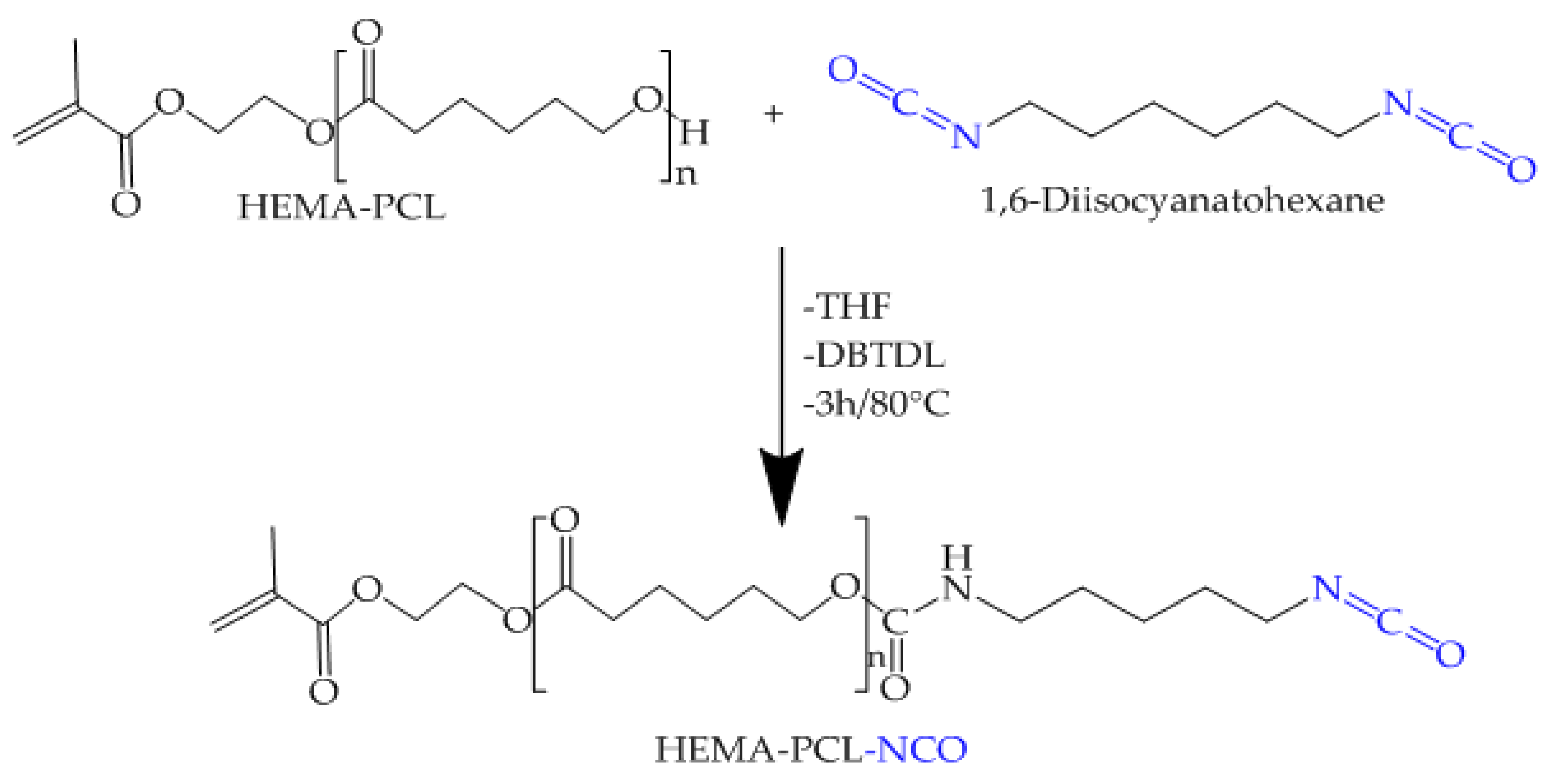

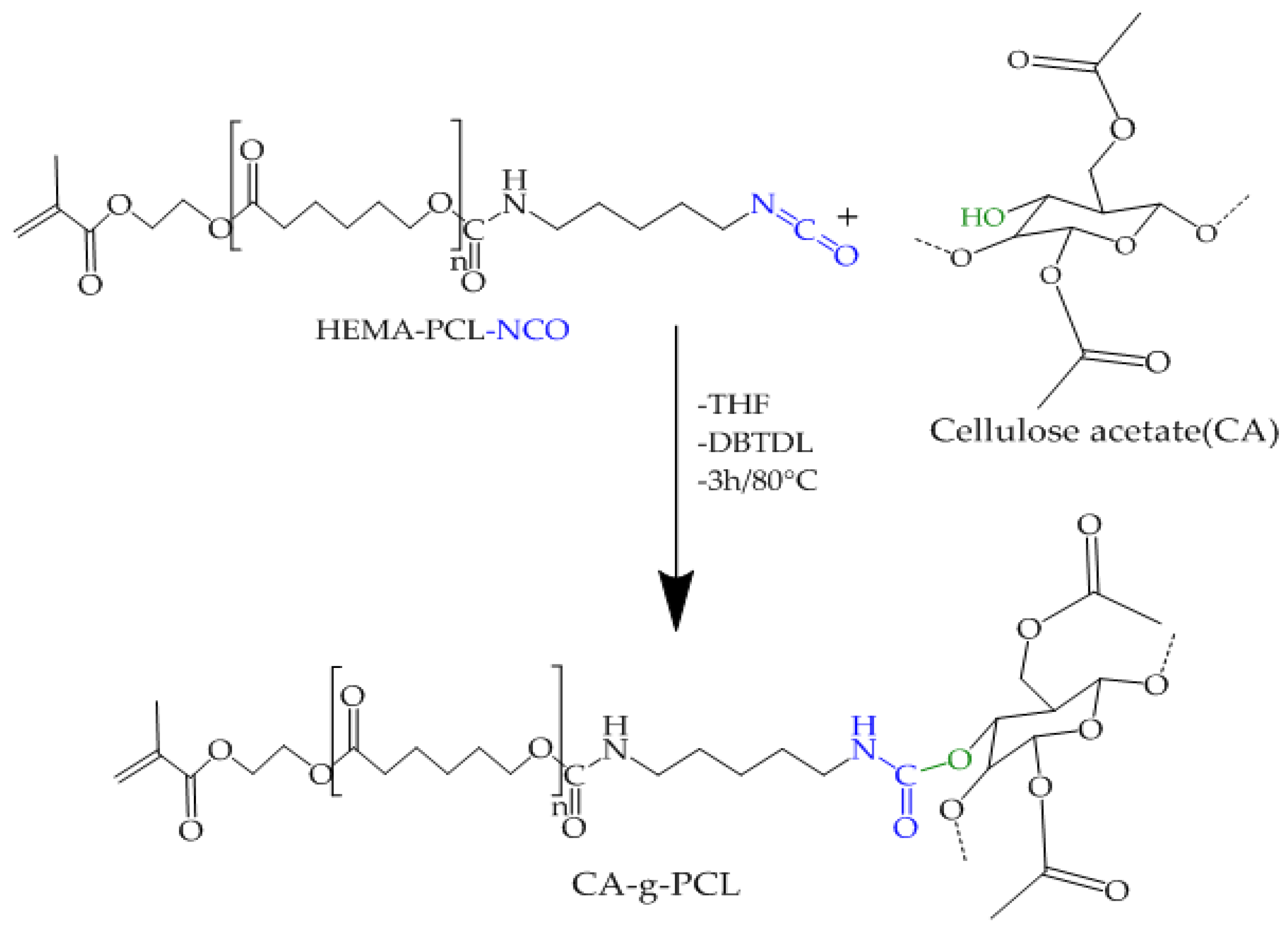







| Sample 1 | 1 g/dL | 1.5 g/dL | 2 g/dL |
|---|---|---|---|
| T (s) | 295.35 | 315.35 | 335.86 |
| 1.1428 | 1.2194 | 1.2987 | |
| 0.1428 | 0.2194 | 0.2987 | |
| 0.1428 | 0.1462 | 0.14935 | |
| 0.1335 | 0.13226 | 0.1307 |
| Sample 1 | 1 g/dL | 1.5 g/dL | 2 g/Dl |
|---|---|---|---|
| t (s) | 308.42 | 335.44 | 365.2 |
| 1.1926 | 1.297 | 1.412 | |
| 0.1926 | 0.297 | 0.412 | |
| 0.1926 | 0.198 | 0.206 | |
| 0.17618 | 0.1733 | 0.1725 |
| Sample 1 | 1 g/dL | 1.5 g/dL | 2 g/dL |
|---|---|---|---|
| t (s) | 334.65 | 374.32 | 415.52 |
| 1.294 | 1.4474 | 1.606 | |
| 0.294 | 0.4474 | 0.606 | |
| 0.294 | 0.2982 | 0.303 | |
| 0.2578 | 0.2465 | 0.236 |
| Sample | [η] (dL/g) (±0.01 dL/g) (Mark–Houwink–Sakurada) | [η] (dL/g) (±0.01 dL/g) (Naar et al.) |
|---|---|---|
| HEMA-PCL50 | 0.137 | 0.1366 |
| HEMA-PCL100 | 0.181 | 0.182 |
| HEMA-PCL200 | 0.2785 | 0.267 |
| Sample | Conv(%) 1 | [ε-CL]/[HEMA] | (DPn)Th 2 | (DP)v | (DP)n | Mn(th) 3 (g/mol) | Mv 4 (g/mol) | Mn 5 (g/mol) |
|---|---|---|---|---|---|---|---|---|
| HEMA-PCL50 | 85.7 | 16.4 | 14.1 | 70 | 48.7 | 1734 | 8163 | 5574 |
| HEMA-PCL100 | 87.14 | 43.6 | 38 | 103.6 | 72.1 | 4466.5 | 11956 | 8365 |
| HEMA-PCL200 | 90 | 82 | 73.8 | 187 | 130 | 8553.5 | 21,575.5 | 15,057 |
| Sample | Blank (mL) | Sample (mL) | I(OH) (mg/gof Sample) | Mn(g/mol) | (DP)n |
|---|---|---|---|---|---|
| HEMA-PCL50 | 47.2 | 45.8 | 13.09 | 4285.7 | 37.5 |
| HEMA-PCL100 | 47.2 | 46.3 | 8.415 | 6666.7 | 58.5 |
| HEMA-PCL200 | 47.2 | 46.7 | 4.675 | 12,000 | 105 |
| Chain Length | Grafting (%) | Yield (%) |
|---|---|---|
| CA-g-PCL50 | 45 | 88 |
| CA-g-PCL100 | 39 | 88 |
| CA-g-PCL200 | 32 | 85 |
| Sample | Diameter of Inhibition Zone (mm) | ||
|---|---|---|---|
| E. coli | S. aureus | P. aeruginosa | |
| CA-g-PCL200 | 12 ± 0.21mm | 21 ± 0.35mm | 7 ± 0.22mm |
| CA-g-PCL100 | 13 ± 0.39mm | 22 ± 0.25mm | 8 ± 0.33mm |
| CA-g-PCL50 | 15 ± 0.29mm | 25 ± 0.4mm | 8.5 ± 0.29mm |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benahmed, A.; Azzaoui, K.; El Idrissi, A.; Belkheir, H.; Said Hassane, S.O.; Touzani, R.; Rhazi, L. Cellulose Acetate-g-Polycaprolactone Copolymerization Using Diisocyanate Intermediates and the Effect of Polymer Chain Length on Surface, Thermal, and Antibacterial Properties. Molecules 2022, 27, 1408. https://doi.org/10.3390/molecules27041408
Benahmed A, Azzaoui K, El Idrissi A, Belkheir H, Said Hassane SO, Touzani R, Rhazi L. Cellulose Acetate-g-Polycaprolactone Copolymerization Using Diisocyanate Intermediates and the Effect of Polymer Chain Length on Surface, Thermal, and Antibacterial Properties. Molecules. 2022; 27(4):1408. https://doi.org/10.3390/molecules27041408
Chicago/Turabian StyleBenahmed, Abdessamade, Khalil Azzaoui, Abderahmane El Idrissi, Hammouti Belkheir, Said Omar Said Hassane, Rachid Touzani, and Larbi Rhazi. 2022. "Cellulose Acetate-g-Polycaprolactone Copolymerization Using Diisocyanate Intermediates and the Effect of Polymer Chain Length on Surface, Thermal, and Antibacterial Properties" Molecules 27, no. 4: 1408. https://doi.org/10.3390/molecules27041408
APA StyleBenahmed, A., Azzaoui, K., El Idrissi, A., Belkheir, H., Said Hassane, S. O., Touzani, R., & Rhazi, L. (2022). Cellulose Acetate-g-Polycaprolactone Copolymerization Using Diisocyanate Intermediates and the Effect of Polymer Chain Length on Surface, Thermal, and Antibacterial Properties. Molecules, 27(4), 1408. https://doi.org/10.3390/molecules27041408









