Molecular Distillation of Lavender Supercritical Extracts: Physicochemical and Antimicrobial Characterization of Feedstocks and Assessment of Distillates Enriched with Oxygenated Fragrance Components
Abstract
:1. Introduction
2. Results
2.1. Composition of Lavender scCO2 Extracts
2.2. Thermal Properties of scCO2 Extracts
2.3. Application of Molecular Distillation in Lavender scCO2 Extract Fractionation
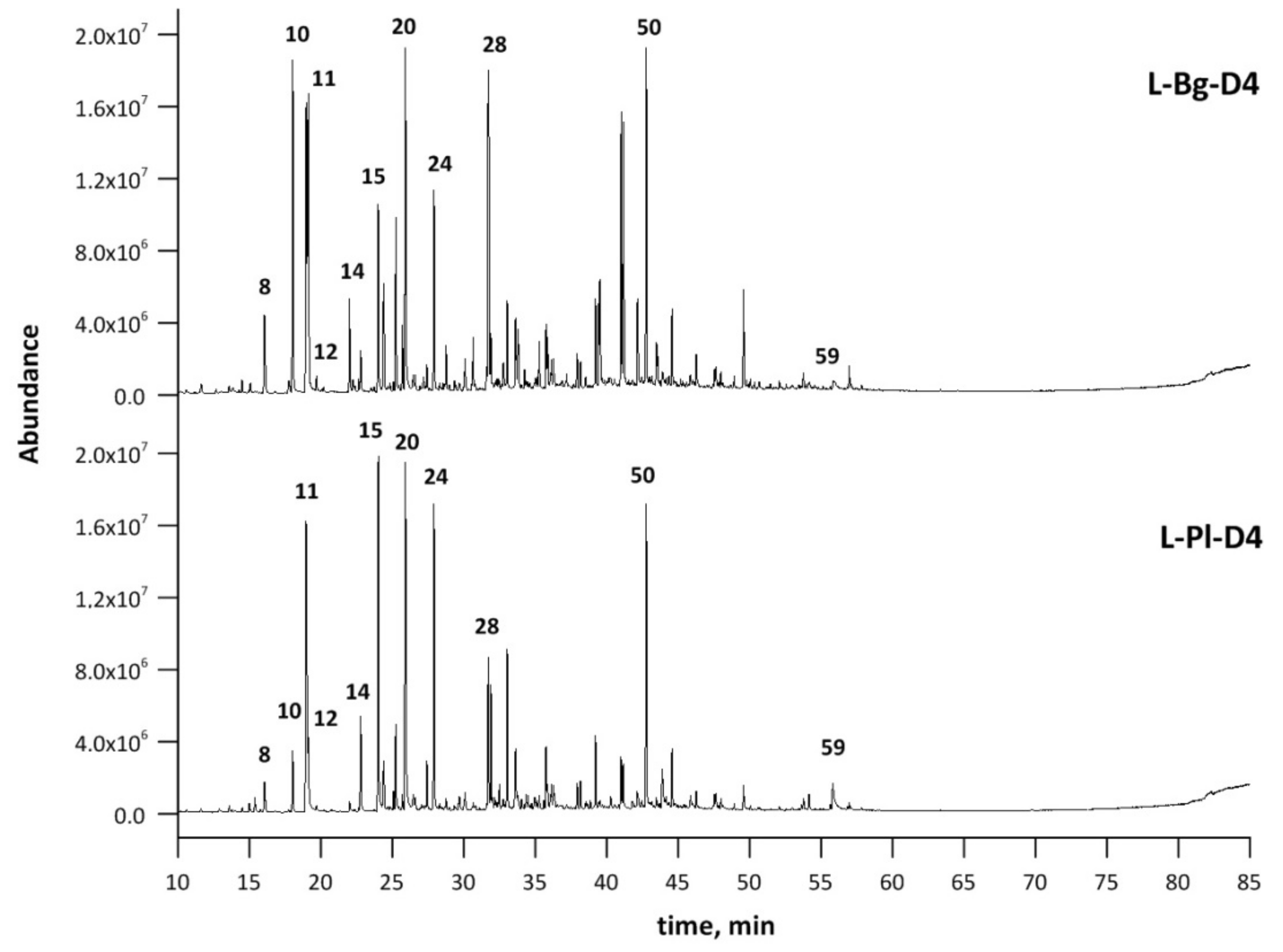
2.4. The Effect of EVT on Distillate Enrichment with Key Lavender Fragrance Molecules
2.5. Quality Evaluation of Distillates and Light Oil Fractions
2.6. Antimicrobial Activity
3. Discussion
4. Materials and Methods
4.1. Chemical and Reagents
4.2. Plants Materials
4.3. Supercritical Fluid Extraction
4.4. Molecular Distillation
4.5. Chemical Analysis
4.5.1. Gas Chromatography Equipped with Tandem Mass Spectrometry (GC–MS/MS)
4.5.2. Gas Chromatography Equipped with Flame Ionization Detection (GC–FID)
4.5.3. Supercritical Fluid Chromatography (SFC)
4.6. Differential Scanning Calorimetry
4.7. HATR-FTIR
4.8. Antimicrobial Tests
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Sample Availability
References
- Śmigielski, K.; Raj, A.; Krosowiak, K.; Gruska, R. Chemical composition of the essential oil of Lavandula angustifolia cultivated in Poland. J. Essent. Oil Bear. Plants 2009, 12, 338–347. [Google Scholar] [CrossRef]
- Prashar, A.; Locke, I.C.; Evans, C.S. Cytotoxicity of lavender oil and its major components to human skin cells. Cell. Proliferat. 2004, 37, 221–299. [Google Scholar] [CrossRef]
- Denner, S.S. Lavandula angustifolia Miller, English lavender. Holist. Nurs. Pract. 2009, 23, 57–64. [Google Scholar] [CrossRef]
- Orchard, A.; van Vuuren, S. Commercial Essential Oils as Potential Antimicrobials to Treat Skin Diseases. Review Article. Evid. Based Compl. Altern. Med. 2017, 2017, 1–92. [Google Scholar] [CrossRef] [Green Version]
- Malcolm, B.J.; Tallian, K. Essential oil of lavender in anxiety disorders: Ready for prime time? Ment. Health Clin. 2017, 7, 147–155. [Google Scholar] [CrossRef]
- Kędzia, A.; Kędzia, A.W. Effectiveness of lavender oil (Oleum Lavandulae) against yeastlike fungi from Candida species. Post. Fitoter. 2020, 21, 73–77. [Google Scholar]
- Ali, B.; Al-Wabel, N.A.; Shams, S.; Ahamad, A.; Khan, S.A.; Anwar, F. Essential oils used in aromatherapy: A systemic review. Asian Pac. J. Trop. Biomed. F. 2015, 5, 601–611. [Google Scholar] [CrossRef] [Green Version]
- Białoń, M.; Krzyśko-Łupicka, T.; Nowakowska-Bogdan, E.; Wieczorek, P.P. Chemical Composition of Two Different Lavender Essential Oils and Their Effect on Facial Skin Microbiota. Molecules 2019, 24, 3270–3287. [Google Scholar] [CrossRef] [Green Version]
- Andrei, F.; Ersilia, A.; Tulcan, C.; Dragomirescu, A. Chemical Composition and the Potential of Lavandula angustifolia L. Oil as a Skin Depigmentant. Rec. Nat. Prod. 2018, 12, 340–349. [Google Scholar] [CrossRef]
- Wesołowska, A.; Jadczak, D.; Grzeszczuk, M. Influence of distillation time on the content and composition of essential oil isolated from lavender (Lavandula angustifolia Mill.). Herba Polonica 2010, 56, 24–36. [Google Scholar]
- Filly, A.; Fabiano-Tixier, A.S.; Louis, C.; Fernandez, X.; Chemat, F. Water as a green solvent combined with different techniques for extraction of essential oil from lavender flowers. Comptes Rendus Chim. 2016, 19, 707–717. [Google Scholar] [CrossRef]
- Michalak, I.; Górka, B.; Wieczorek, P.P.; Rój, E.; Lipok, J.; Łęska, B.; Messyasz, B.; Wilk, R.; Schroeder, G.; Dobrzyńska-Inger, A.; et al. Supercritical fluid extraction of algae enhances levels of biologically active compounds promoting plant growth. Eur. J. Phycol. 2016, 51, 243–252. [Google Scholar] [CrossRef]
- Jerkovic, I.; Molnar, M.; Vidovic, S.; Vladic, J.; Jokic, S. Supercritical CO2 of Lavandula angustifolia Mill. Flowers, Coumarin and Herniarin Content. Phytochem. Anal. 2017, 28, 558–566. [Google Scholar] [CrossRef] [PubMed]
- Nadalin, V.; Lepojevic, Z.; Ristic, M.; Vladic, J.; Nikolovski, B.; Adamovic, D. Investigation of cultivated lavender (Lavandula officinalis L.) extraction and its extracts. Chem. Ind. Chem. Eng. Q. 2014, 20, 71–86. [Google Scholar] [CrossRef] [Green Version]
- Lv, G.P.; Hu, D.J.; Zhou, Y.-Q.; Zhang, Q.W.; Zhao, J.; Li, S.-P. Preparation and application of standardized typical volatile components fraction from turmeric (Curcuma longa L.) by supercritical fluid extraction and step molecular distillation. Molecules 2018, 23, 1831. [Google Scholar] [CrossRef] [Green Version]
- Fang, T.; Goto, M.; Sasaki, M.; Hirose, T. Combination of supercritical CO2 and vacuum distillation for the fractionation of bergamot oil. J. Agric. Food Chem. 2004, 52, 5162–5167. [Google Scholar] [CrossRef]
- Olivares, A.; Martínez, I.; Illanes, A. Enzyme assisted fractionation of wood sterols mixture by short path distillation. Chem. Eng. J. 2012, 191, 557–562. [Google Scholar] [CrossRef]
- Lutisan, J.; Cvengros, J.; Micov, M. Heat and mass transfer in the evaporating film of a molecular evaporator. Chem. Eng. J. 2002, 85, 225–234. [Google Scholar] [CrossRef]
- Cvengros, J.; Lutisan, J.; Micov, M. Feed temperature influence on the efficiency of a molecular evaporation. Chem. Eng. J. 2000, 78, 61–67. [Google Scholar] [CrossRef]
- Tovar, L.P.; Pinto, G.M.F.; Wolf-Maciel, M.R.; Batistella, C.B.; Maciel-Filho, R. Short-Path-Distillation Process of Lemongrass Essential Oil: Physicochemical Characterization and Assessment Quality of the Distillate and the Residue Products. Ind. Eng. Chem. Res. 2011, 50, 8185–8194. [Google Scholar] [CrossRef]
- Rossi, P.C.; Willnecker, A.A.; Berti, J.; Borgarello, A.V.; Mezza, G.N.; Pramparo, M.C. D-Limonene and Geranial Fractionation from Lemon Essential oil by Molecular Distillation. Lat. Am. Appl. Res. 2011, 41, 81–85. [Google Scholar]
- Martinello, M.A.; Pagliero, C.L.; Allevi, C.A. Deterpenation of Orange Essential Oil by Molecular Distillation. Int. J. Eng. Trends Technol. 2015, 30, 161–165. [Google Scholar] [CrossRef]
- Laksmono, J.A.; Augustian, E.; Adilina, I.B. Patchouli Alcohol Enrichment from Patchouli Oil using Molecular Distillation Unit. J. Tek. Ind. Pert. 2007, 17, 74–79. [Google Scholar]
- Mezza, G.N.; Borgarello, A.V.; Daguero, J.D.; Pramparo, M.C. Obtention of Rosemary Essential Oil Concentrates by Molecular Distillation and Free Radical Scavenging Capacity Analysis. J. Food Eng. 2013, 9, 147–153. [Google Scholar] [CrossRef]
- Olmedo, R.H.; Nepote, V.; Grosso, N.R. Antioxidant activity of fractions from oregano essential oils obtained by molecular distillation. Food Chem. 2014, 156, 212–219. [Google Scholar] [CrossRef]
- Yia, F.; Suna, J.; Baoa, X.; Mab, B.; Suna, M. Influence of molecular distillation on antioxidant and antimicrobial activities of rose essential oils. LWT-Food Sci. Technol. 2018, 102, 310–316. [Google Scholar] [CrossRef]
- Li, Y.; Xia, L.; Vazquez, J.F.T.; Song, S. Optimization of supercritical CO2 extraction of essential oil from Artemisia annua L. by means of response surface methodology. J. Essent. Oil Bear. Plants 2017, 20, 314–327. [Google Scholar] [CrossRef]
- Zhong, J.-L.; Muhammad, N.; Chen, S.-Q.; Guo, L.-W.; Li, J.-S. Pilot-scale supercritical CO2 extraction coupled molecular distillation and hydrodistillation for the separation of essential oils from artemisia argyi Lévl. et Vant. Sep. Sci. Technol. 2021, 56, 3127–3135. [Google Scholar] [CrossRef]
- Chen, F.; Wang, Z.; Zhao, G.; Liao, X.; Cai, T.; Guo, L.; Hu, X. Purification process of octacosanol extracts from rice bran wax by molecular distillation. J. Food Eng. 2007, 79, 63–68. [Google Scholar] [CrossRef]
- Maziero, E.V.; Salles, R.B.; Tovar, L.P.; Tanabe, E.H.; Bertuol, D.A. Fractionation of polyethylene wax by pilot-scale molecular distillation: New insights on process development. Chem. Eng. Res. Design. 2019, 152, 201–215. [Google Scholar] [CrossRef]
- Tyśkiewicz, K.; Konkol, M.; Rój, E. Supercritical carbon dioxide (scCO2) extraction of phenolic compounds from lavender (Lavandula angustifolia) flowers: A box-behnken experimental optimization. Molecules. 2019, 24, 3354–3369. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sovova, H.; Stateva, R.P. New Approach to Modelling Supercritical CO2 Extraction of Cuticular Waxes: Interplay between Solubility and Kinetics. Ind. Eng. Chem. Res. 2015, 54, 4861–4870. [Google Scholar] [CrossRef]
- Reverchon, E. Supercritical fluid extraction and fractionation of essential oils and related products. J. Supercrit. Fluid. 1997, 10, 1–37. [Google Scholar] [CrossRef]
- Reverchon, E.; Della Porta, G.; Senatore, F. Supercritical CO2 extraction and fractionation of lavender essential Oil. J. Agric. Food Chem. 1995, 43, 1654–1658. [Google Scholar] [CrossRef]
- Da Porto, C.; Decorti, D.; Kikic, I. Flavour compounds of Lavandula angustifolia L. to use in food manufacturing: Comparison of three different extraction methods. Food Chem. 2009, 112, 1072–1078. [Google Scholar] [CrossRef]
- Mallavarapu, G.R.; Mehta, V.K.; Sastry, K.P.; Krishnan, R.; Ramesh, S.; Kumar, S. Composition of lavender oils produced in Kashmir and Kodaikanal. J. Med. Arom. Plant Sci. 2000, 22, 768–770. [Google Scholar]
- Guo, X.; Wang, P. Aroma characteristics of lavender extract and essential oil from Lavandula angustifolia Mill. Molecules 2020, 25, 5541–5555. [Google Scholar] [CrossRef]
- Duskova, E.; Dusek, K.; Indrak, P.; Smekalova, K. Postharvest changes in essential oil content and quality of lavender flowers. Ind. Crop. Prod. 2016, 79, 225–231. [Google Scholar] [CrossRef]
- Ozek, T.; Tabanca, N.; Demirci, F.; Wedge, D.E.; Husnu Can Baser, K. Enantiomeric Distribution of Some Linalool Containing Essential Oils and Their Biological Activities. Rec. Nat. Prod. 2010, 4, 180–192. [Google Scholar]
- Jerkovic, J.; Kuś, P.M. Terpenes in honey: Occurrence, origin and their role as chemical biomarkers. RSC Adv. 2014, 4, 31710–31728. [Google Scholar] [CrossRef]
- Alissandrakis, E.; Tarantilis, P.A.; Harizanis, P.C.; Polissiou, M. Aroma investigation of unifloral Greek citrus honey using solid-phase microextraction coupled to gas chromatographic—Mass spectrometric analysis. Food Chem. 2007, 100, 396–404. [Google Scholar] [CrossRef]
- Chen, X.; Yauk, Y.-K.; Nieuwenhuizen, N.J.; Matich, A.J.; Wang, M.Y.; Perez, R.L.; Atkinson, R.G.; Beuning, L.L. Characterisation of an (S)-linalool synthase from kiwifruit (Actinidia arguta) that catalyses the first committed step in the production of floral lilac compounds. Funct. Plant Biol. 2010, 37, 232–243. [Google Scholar] [CrossRef]
- Lesage-Meessen, L.; Bou, M.; Sigoillot, J.-C.; Faulds, C.B.; Lomascolo, A. Essential oils and distilled straws of lavender and lavandin: A review of current use and potential application in white biotechnology. Appl. Microbiol. Biotechnol. 2015, 99, 3375–3385. [Google Scholar] [CrossRef] [PubMed]
- Cavanagh, H.M.A.; Wilkinson, J.M. Biological activities of lavender essential oil. J. Phytochem. Res. 2002, 16, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Lis-Balchin, M.; Hart, S. Studies on the mode of action of the essential oil of lavender (Lavandula angustifolia P. Miller). Phytother. Res. 1999, 13, 540–542. [Google Scholar] [CrossRef]
- Fidyt, K.; Fiedorowicz, A.; Strządała, L.; Szumny, A. Review β-caryophyllene and β-caryophyllene oxide—Natural compounds of anticancer and analgesic properties. Cancer Med. 2016, 5, 3007–3017. [Google Scholar] [CrossRef]
- Ngamprasertish, S.; Menwa, J.; Sawangkeaw, R. Caryophyllene oxide extraction from lemon basil (Ocimum citriodorum Vis.) straw by hydrodistillation and supercritical CO2. J. Supercrit. Fluid. 2018, 138, 1–6. [Google Scholar] [CrossRef]
- Najmanova, I.; Dosedel, M.; Hrdina, R.; Anzenbacher, P.; Filipsý, T.; Říha, M.; Mladenka, P. Cardiovascular effects of coumarins besides their antioxidant activity. Curr. Top. Med. Chem. 2015, 15, 830–849. [Google Scholar] [CrossRef]
- Ruguo, Z.; Hua, Z.; Hong, Z.; Ying, F.; Kun, L.; Wenwen, Z. Thermal analysis of four insect waxes based on differential scanning calorimetry (DSC). Procedia Eng. 2011, 18, 101–106. [Google Scholar] [CrossRef]
- Donhowe, G.; Fennema, O. Water vapor and oxygen permeability of wax films. JAOCS 1993, 70, 867–873. [Google Scholar] [CrossRef]
- Barthlott, W.; Mail, M.; Bhushan, B.; Koch, K. Plant surfaces: Structures and functions for biomimetic innovations, Review. Nano-Micro Lett. 2017, 9, 1–40. [Google Scholar] [CrossRef] [Green Version]
- Charumanee, S.; Yotsawimonwat, S.; Sirisa-ard, P.; Pholsongkram, K. Characterization and chemical composition of epicuticular wax from banana leaves grown in Northern Thailand. Songklanakarin J. Sci. Technol. 2017, 39, 509–516. [Google Scholar]
- Freitas, C.A.S.; Vieira, I.G.P.; Sousa, P.H.M.; Muniz, C.R.; da Costa Gonzaga, M.L.; Guedes, M.I.F. Carnauba wax p-methoxycinnamic diesters: Characterization, antioxidant activity and simulated gastrointestinal digestion followed by in vitro bioaccessibility. Food Chem. 2016, 196, 1293–1300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Panteli, E.; Saratsioti, P.; Stamatis, H.; Voutsas, E. Solubilities of cinnamic acid esters in organic solvents. J. Chem. Eng. Data 2010, 55, 745–749. [Google Scholar] [CrossRef]
- Najibullah, S.N.M.; Ahamad, J.; Aldahish, A.A.; Sultana, S.; Sultana, S. Chemical Characterization and α-Glucosidase Inhibitory Activity of Essential Oil of Lavandula angustifolia Flowers. J. Essent. Oil Bear. Plants 2021, 24, 431–438. [Google Scholar] [CrossRef]
- Tankeu, S.Y.; Vermaak, I.; Kamatou, G.P.P.; Viljoen, A.M. Vibrational spectroscopy and chemimetric modelling: An economical and robust quality control method for lavender oil. Ind. Crops Prod. 2014, 59, 234–240. [Google Scholar] [CrossRef]
- Agatonovic-Kustrin, S.; Ristivojevic, P.; Gegechkori, V.; Litvinova, T.M.; Morton, D.W. Essential oil quality and purity evaluation via ft-ir spectroscopy and pattern recognition techniques. Appl. Sci. 2020, 10, 7294–7305. [Google Scholar] [CrossRef]
- Nelson, R.R.S. In vitro activities of five plant essential oils against methicillin-resistant Staphylococcus aureus and vancomycin-resistant Enterococcus faecium. J. Antimicrob. Chemother. 1997, 40, 305–306. [Google Scholar] [CrossRef] [Green Version]
- Lis-Balchin, M.; Deans, S.G.; Eaglesham, E. Relationship between bioactivity and chemical composition of commercial essential oils. Flavour Fragr. J. 1998, 13, 98–104. [Google Scholar] [CrossRef]
- Shellie, R.; Mondello, L.; Marriott, P.; Dugo, G. Characterization of lavender essential oils by using gas chromatography-mass spectrometry with correlation of linear retention indices and comparison with comprehensive two-dimensional gas chromatography. J. Chromatogr. A. 2002, 970, 225–234. [Google Scholar] [CrossRef]
- Predoi, D.; Iconaru, S.L.; Buton, N.; Badea, M.L.; Marutescu, L. Antimicrobial activity of new materials based on lavender and brasil essential oils and hydroxyapatite. Nanomaterials 2018, 8, 291. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pankey, G.A.; Sabath, L.D. Clinical relevance of bacteriostatic versus bactericidal mechanisms of action in the treatment of gram-positive bacterial infections. Clin. Infect. Dis. 2004, 38, 864–870. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moussi Imane, M.; Houda, F.; Said Amal, A.H.; Kaotar, N.; Mohammed, T.; Imane, R.; Farid, H. Phytochemical composition and antibacterial activity of Moroccan Lavandula angustifolia Mill. J. Essent. Oil Bear. Plants 2017, 20, 1074–1082. [Google Scholar] [CrossRef]
- Ciocarlan, A.; Lupascu, L.; Aricu, A.; Dragalin, I.; Popescu, V.; Geana, E.-I.; Ionete, R.E.; Vornicu, N.; Duliu, O.G.; Hristozova, G.; et al. Chemical composition and assessment of antimicrobial activity of lavender essential oil and some by-products. Plants 2021, 10, 1829. [Google Scholar] [CrossRef] [PubMed]
- Caprari, C.; Fantasma, F.; Divino, F.; Bucci, A.; Iorizzi, M.; Naclerio, G.; Ranalli, G.; Saviano, G. Chemical Profile, In vitro biological activity and comparisonof essential oils from fresh and dried flowers of Lavandula angustifolia L. Molecules. 2021, 26, 5317–5337. [Google Scholar] [CrossRef]
- Peana, A.T.; D’Aquila, P.S.; Panin, F.; Serra, G.; Pippia, P.; Moretti, M.D.L. Anti-inflammatory activity of linalool and linalyl acetate constituents of essential oils. Phytomedicine 2002, 9, 721–726. [Google Scholar] [CrossRef]
- Trombetta, D.; Castelli, F.; Sarpietro, M.G.; Venuti, V.; Cristani, M.; Daniele, C.; Saija, A.; Mazzani, G.; Bisignano, G. Mechanism of antibacterial action of three monoterpenes. Antimicrob. Agents Chemother. 2005, 49, 2474–2478. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Cai, J.; Chen, H.; Zhong, Q.; Hou, Y.; Chen, W.; Chen, W. Antibacetrial activity and mechanism of linalool against Pseudomonas aeruginosa. Microb. Pathog. 2020, 141, 103980. [Google Scholar] [CrossRef]
- D’Auria, F.D.; Tecca, M.; Strippoli, V.; Salvatore, G.; Battinelli, L.; Mazzanti, G. Antifungal activity of Lavandula angustifolia essential oil against Candida albicans and mycelial form. Med. Mycol. 2005, 43, 391–396. [Google Scholar] [CrossRef] [Green Version]
- Babu, G.D.K.; Sharma, A.; Singh, B. Volatile composition of Lavandula angustifolia produced by different extraction techniques. J. Ess. Oil Res. 2016, 28, 489–500. [Google Scholar] [CrossRef]
- Tardugno, R.; Serio, A.; Pellati, F.; D’Amato, S.; Chaves López, C.; Bellardi, M.G.; Di Vito, M.; Savini, V.; Paparella, A.; Benvenuti, S. Lavandula x intermedia and Lavandula angustifolia essential oils: Phytochemical composition and antimicrobial activity against foodborne pathogens. Nat. Prod. Res. 2019, 33, 3330–3335. [Google Scholar] [CrossRef] [PubMed]
- Śmigielski, K.B.; Prusinowska, R.; Krosowiak, K.; Sikora, M. Comparison of qualitative and quantitative chemical composition of hydrolate and essential oils of lavender (Lavandula angustifolia). J. Essent. Oil Res. 2013, 25, 291–299. [Google Scholar] [CrossRef]
- Aćimović, M.G.; Tešević, V.V.; Smiljanić, K.T.; Cvetković, M.T.; Stanković, J.M.; Kiprovski, B.M.; Sikora, V.S. Hydrolates-by-products of essential oil distillation: Chemical composition, biological activity and potential uses. Adv. Mater. Technol. 2020, 9, 54–70. [Google Scholar] [CrossRef]
- Garzoli, S.; Laghezza Masci, V.; Franceschi, S.; Tiezzi, A.; Giacomello, P.; Ovidi, E. Headspace/GC–MS Analysis and investigation of antibacterial, antioxidant and cytotoxic activity of essential oils and hydrolates from Rosmarinus officinalis L. and Lavandula angustifolia Miller. Foods 2021, 10, 1768. [Google Scholar] [CrossRef] [PubMed]
- European Committee for Antimicrobial Susceptibility Testing (EUCAST) of the European Society of Clinical Microbiology and Infectious Diseases (ESCMID). Determination of minimum inhibitory concentrations (MICs) of antibacterial agents by broth dilution. EUCAST Discussion Document E.Dis 5.1; The European Committee on Antimicrobial Susceptibility Testing. Clin. Microbiol. Inf. Dis. 2003, 9, 1–7. [Google Scholar]

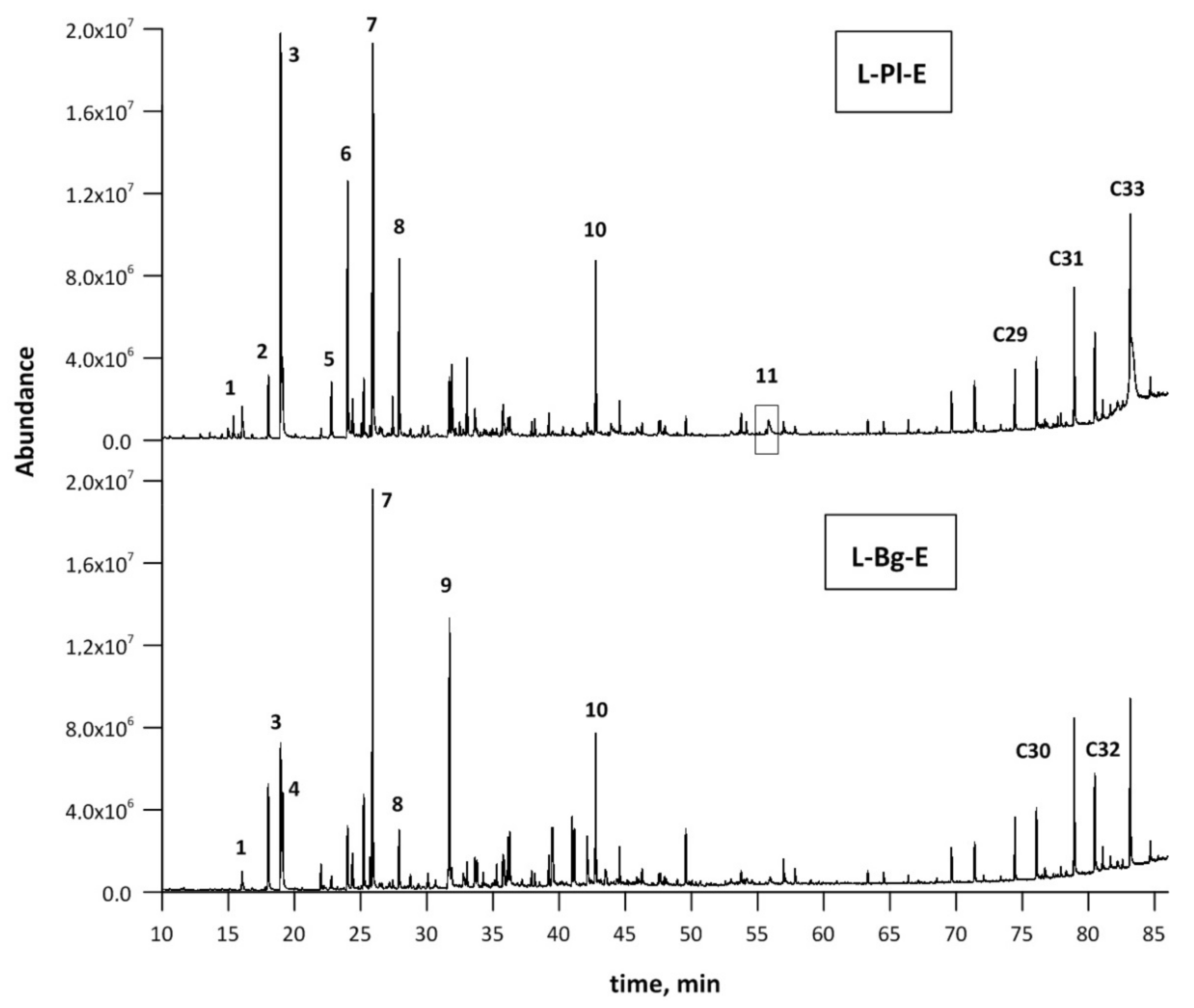
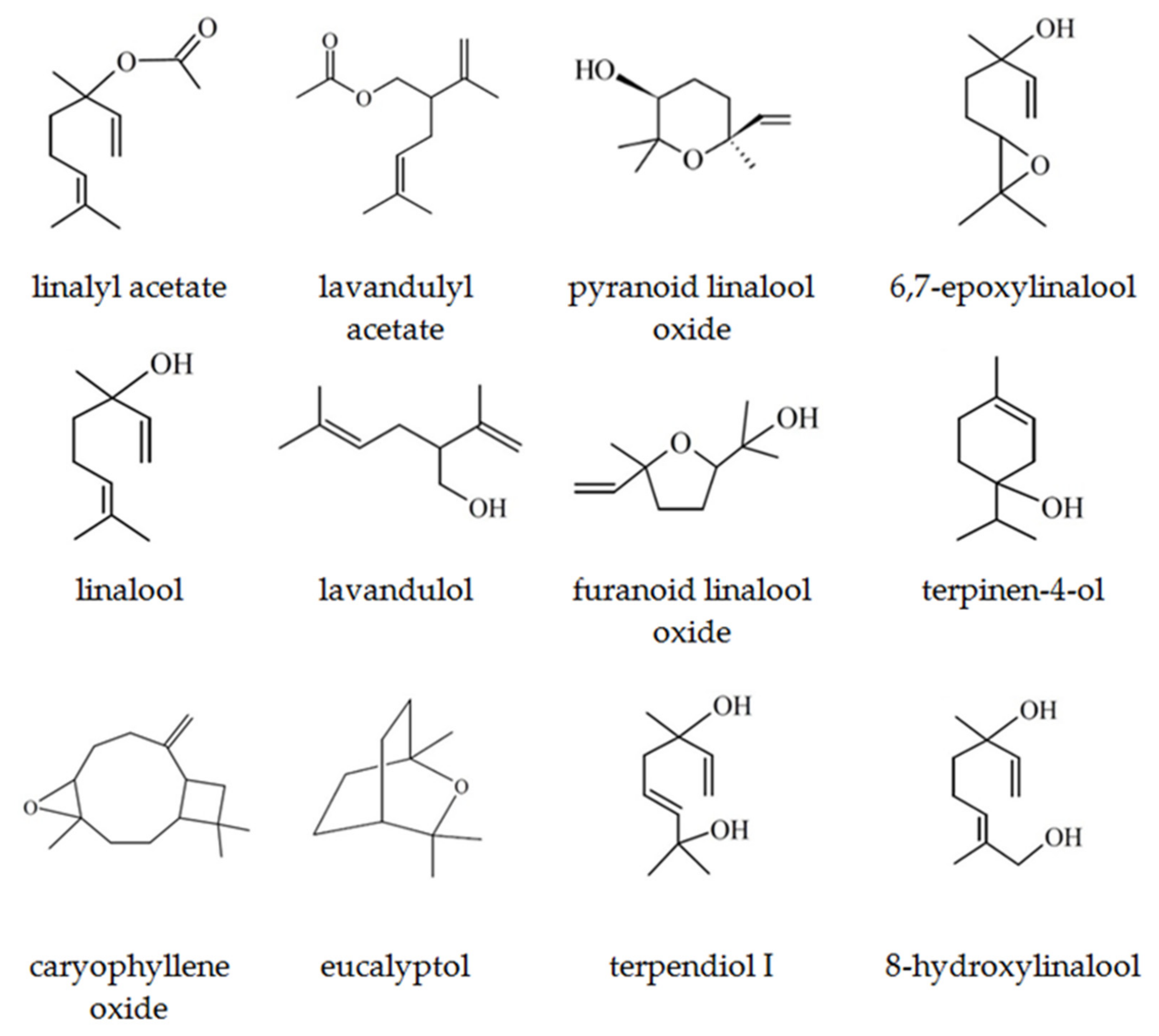

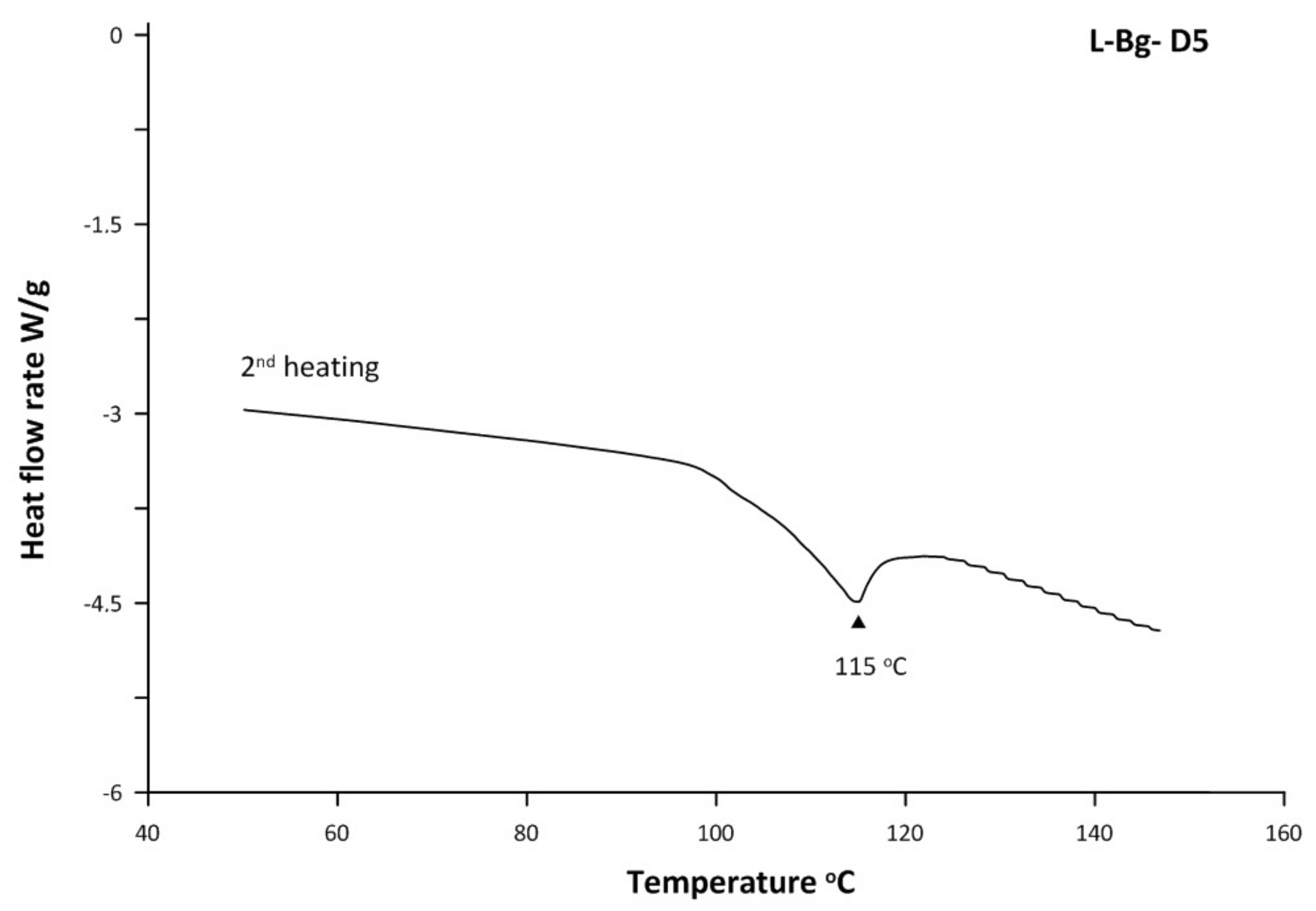

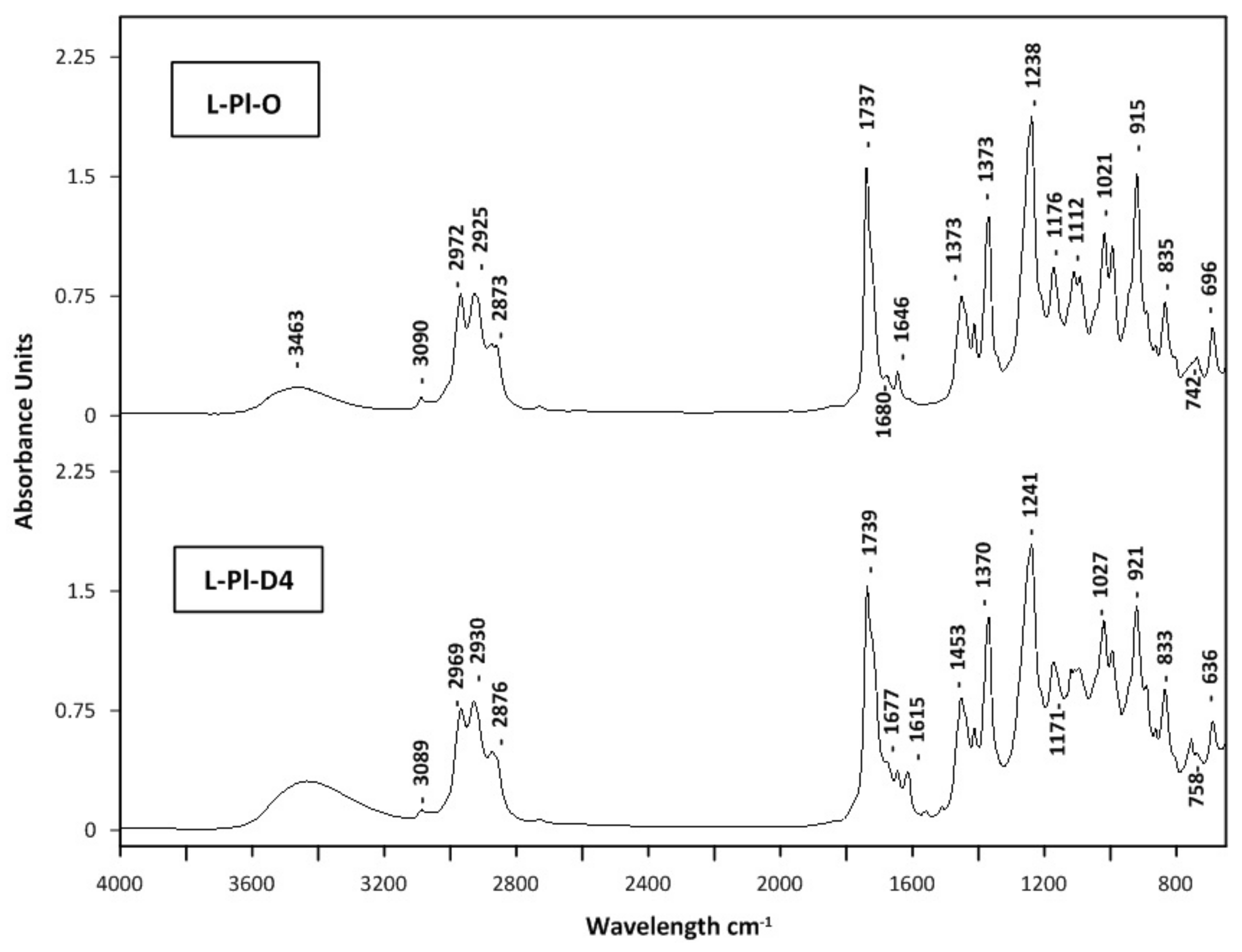

| No. | Compounds | RI | RT | Composition, % | |
|---|---|---|---|---|---|
| L-Pl-E | L-Bg-E | ||||
| 1 | (-)-Limonene | 1114 | 14.99 | 0.26 | - |
| 2 | trans-β-Ocimene | 1123 | 15.39 | 0.55 | - |
| 3 | Eucalyptol | 1139 | 16.05 | 1.05 | 0.87 |
| 4 | cis-Linalool oxide (furanoid) | 1186 | 18.03 | 1.51 | 3.59 |
| 5 | Linalool (3,7-dimethyl-1,6-octadien-3-ol) | 1209 | 18.98 | 17.02 | 4.56 |
| 6 | trans-Linalool oxide (furanoid) | 1212 | 19.12 | 1.42 | 3.63 |
| 7 | Lavender lactone | 1278 | 22.01 | 0.26 | 0.79 |
| 8 | Lavandulol | 1295 | 22.79 | 1.40 | 0.43 |
| 9 | Terpinen-4-ol | 1323 | 24.02 | 6.02 | 2.15 |
| 10 | Linalool oxide (pyranoid) | 1332 | 24.40 | 0.95 | 1.55 |
| 11 | α-Terpineol | 1348 | 25.08 | 0.28 | - |
| 12 | 2,6-Dimethyl-3,7-octadiene-2,6-diol | 1351 | 25.24 | 1.34 | 2.81 |
| 13 | 3,7-Dimethyl-1,5- octadiene-3,7-diol (terpendiol I) | 1362 | 25.70 | 0.26 | 0.85 |
| 14 | Linalool acetate | 1367 | 25.93 | 25.68 | 12.78 |
| 15 | Crypton | 1402 | 27.42 | 0.87 | 0.28 |
| 16 | (±)-Lavandulyl acetate | 1413 | 27.91 | 3.96 | 1.86 |
| 17 | Bornyl acetate | 1433 | 28.76 | - | 0.40 |
| 18 | p-Cumic aldehyde | 1456 | 29.71 | 0.35 | - |
| 19 | 2,6-Dimethyl-1,7-octadiene-3,6-diol | 1465 | 30.10 | 0.33 | 0.59 |
| 20 | p-Cymen-7-ol | 1501 | 31.59 | - | 0.54 |
| 21 | 3,7-Dimethyl-1,7-octadiene-3,6-diol, (terpendiol II) | 1504 | 31.71 | 1.41 | 8.16 |
| 22 | Longicyclene | 1508 | 31.90 | 1.61 | 0.7 |
| 23 | α-Bergamotene | 1523 | 32.50 | 0.24 | - |
| 24 | β-Caryophyllene | 1537 | 33.04 | 1.76 | 0.67 |
| 25 | β-Farnesene | 1551 | 33.64 | 0.89 | 1.13 |
| 26 | 2,6-Dimethyl-2,7-octadiene-1,6-diol (8-hydroxylinalool) | 1555 | 33.79 | 0.21 | 1.02 |
| 27 | Nerolidol | 1592 | 35.29 | - | 0.69 |
| 28 | cis-2-Methyl-3-oxo-cyclohexanebutanal | 1604 | 35.76 | 0.86 | 0.92 |
| 29 | cis-9-Tetradecen-1-ol | 1607 | 35.86 | 0.23 | 0.65 |
| 30 | Epoxylinalool | 1614 | 36.14 | 0.49 | 1.55 |
| 31 | Farnesyl formate | 1618 | 36.28 | 0.5 | 1.76 |
| 32 | Nerolidyl acetate | 1661 | 37.93 | 0.32 | 0.48 |
| 33 | α-Santalol | 1667 | 38.17 | 0.38 | 0.4 |
| 34 | α-Bisabolol | 1694 | 39.22 | 0.44 | 0.84 |
| 35 | NI a | 1701 | 39.46 | - | 1.51 |
| 36 | NI a | 1703 | 39.54 | - | 1.67 |
| 37 | NI a | 1743 | 40.99 | - | 2.06 |
| 38 | NI a | 1747 | 41.16 | - | 1.38 |
| 39 | (1S,2S,4S)-Trihydroxy-p-menthane | 1774 | 42.14 | 0.25 | 1.88 |
| 40 | β-Caryophyllene oxide | 1791 | 42.77 | 3.8 | 4.54 |
| 41 | Coumarin | 1821 | 43.92 | 0.18 | - |
| 42 | tau-Cadinol | 1842 | 44.58 | 0.66 | 1.01 |
| 43 | Ledene oxide-(II) | 1891 | 46.27 | 0.25 | 0.43 |
| 44 | α-Santalol | 1929 | 47.55 | 0.27 | 0.3 |
| 45 | β-Santalol | 1932 | 47.65 | 0.31 | |
| 46 | Hexahydrofarnesyl acetone | 1942 | 47.98 | 0.2 | - |
| 47 | Isolongifolol | 1990 | 49.58 | 0.39 | 1.55 |
| 48 | Neryl acetate | 2121 | 53.76 | 0.51 | 0.43 |
| 49 | Gerany-p-cymene | 2134 | 54.15 | 0.27 | 0.28 |
| 50 | Herniarin | 2189 | 55.83 | 1.13 | 0.38 |
| 51 | Neoisolongifolene, 8-oxo | 2228 | 56.98 | 0.21 | 0.7 |
| 52 | 7-Hydroxyfarnesene | 2257 | 57.83 | - | 0.46 |
| 53 | 11-Methyltricosane | 2453 | 63.33 | 0.27 | 0.31 |
| 54 | Pentacosane, C25 | 2497 | 64.54 | 0.22 | 0.31 |
| 55 | 2-Methylhexacosane | 2568 | 66.38 | 0.26 | - |
| 56 | Heptacosane, C27 | 2697 | 69.66 | 0.81 | 0.95 |
| 57 | 1-Iodo-docosane | 2769 | 71.40 | 1.43 | 1.21 |
| 58 | Nonacosane, C29 | 2897 | 74.45 | 1.28 | 1.78 |
| 59 | Tetracosane 1-iodo | 2969 | 76.08 | 1.62 | 1.97 |
| 60 | Triacontane, C30 | 2996 | 76.72 | - | 0.3 |
| 61 | 1-Heptacosanol | 3051 | 77.92 | 0.3 | 0.29 |
| 62 | Hentriacontane, C31 | 3097 | 78.93 | 3.14 | 4.68 |
| 63 | Methyltriacontane | 3169 | 80.48 | 2.23 | 3.22 |
| 64 | Dotriacontane, C32 | 3197 | 81.08 | 0.46 | 0.73 |
| 65 | Tritriacontane, C33 | 3297 | 83.16 | 4.57 | 5.43 |
| Compounds | Formula | Molecular Weight, g/mol | Boiling Point, °C | Melting Point, °C |
|---|---|---|---|---|
| Coumarin | C9H6O2 | 146.14 | 301.7 | 68–70 |
| 1,8-Cineole | C10H18O | 154.24 | 176.5 | 2.9 |
| Linalool | C10H18O | 154.25 | 198 | <−20 |
| Terpinen-4-ol | C10H18O | 154.25 | 211–213 | - |
| Lavandulol | C10H18O | 154.26 | 229–230 | - |
| Herniarin | C10H8O3 | 176.16 | 335.3 | 117–121 |
| Linalyl acetate | C12H20O2 | 196.29 | 220 | <25 |
| Lavandulyl acetate | C12H20O2 | 196.29 | 228–229 | −17.12 |
| Caryophyllene oxide | C15H24O | 220.35 | 279.7 | 60–62 |
| Parameters | |||||||
|---|---|---|---|---|---|---|---|
| Exp. | FT, °C | RdT, °C | EVT, °C | CT, °C | FF, mL/min | p, Pa | rpm |
| Bulgarian lavender | |||||||
| L-Bg D1 | 50 | 55 | 55 | −5 | 0.833 | ~1 Pa (10−2 mbar) | 350 |
| L-Bg D2 | 58 | 65 | −5 | ||||
| L-Bg D3 | 63 | 75 | 0 | ||||
| L-Bg D4 | 68 | 85 | 6 | ||||
| L-Bg D5 | 70 | 95 | 6 | ||||
| Polish lavender | |||||||
| L-Pl D1 | 45 | 55 | 55 | 10 | 0.833 | ~1 Pa (10−2 mbar) | 200 |
| L-Pl D2 | 65 | ||||||
| L-Pl D3 | 75 | ||||||
| L-Pl D4 | 85 | ||||||
| L-Pl D5 | 95 | ||||||
| EVT, °C | CT, °C | Fraction | %wtD | %wtR | D/R | %wtCT | %wt | Rec. | |
|---|---|---|---|---|---|---|---|---|---|
| Oil | Water | ||||||||
| Bulgarian lavender | |||||||||
| 55 | −5 | L-Bg-D1 | 15.23 | 75.73 | 0.20 | 1.73 | 3.46 | 3.85 | 20.2 |
| 65 | −5 | L-Bg-D2 | 20.12 | 70.54 | 0.29 | 1.88 | 3.38 | 4.08 | 30.3 |
| 75 | 0 | L-Bg-D3 | 34.87 | 55.51 | 0.63 | 2.32 | 3.54 | 3.76 | 64.0 |
| 85 | 6 | L-Bg-D4 | 39.28 | 51.12 | 0.77 | 2.20 | 3.50 | 3.90 | 81.4 |
| 95 | 6 | L-Bg-D5 | 44.65 | 44.86 | 1.00 | 3.12 | 3.25 | 4.12 | 82.8 |
| Polish lavender | |||||||||
| 55 | 10 | L-Pl-D1 | 19.13 | 70.31 | 0.27 | 3.32 | 3.12 | 4.12 | 23.9 |
| 65 | L-Pl-D2 | 24.86 | 64.43 | 0.39 | 3.15 | 3.31 | 4.25 | 37.2 | |
| 75 | L-Pl-D3 | 35.33 | 53.81 | 0.66 | 3.23 | 3.25 | 4.38 | 61.2 | |
| 85 | L-Pl-D4 | 44.44 | 44.06 | 1.01 | 3.70 | 3.30 | 4.50 | 86.6 | |
| 95 | L-Pl-D5 | 45.28 | 42.05 | 1.08 | 4.53 | 3.42 | 4.72 | 88.1 | |
| Stream | EVT, °C | Compounds | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 * | 2 * | 3 ** | 4 ** | 5 ** | 6 ** | 7 ** | 8 ** | 9 ** | ||
| Content, mg/g | ||||||||||
| L-Bg-E | - | 7.00 | 5.30 | 1.85 | 19.60 | 51.54 | 2.32 | 20.38 | 7.46 | 6.02 |
| D1 | 55 | 8.48 | 3.79 | 3.03 | 25.10 | 75.80 | 3.22 | 25.85 | 9.53 | 8.22 |
| D2 | 65 | 6.52 | 4.11 | 3.55 | 31.34 | 82.24 | 3.54 | 32.12 | 11.51 | 9.64 |
| D3 | 75 | 5.46 | 4.25 | 4.02 | 37.52 | 107.45 | 4.23 | 36.76 | 14.38 | 11.61 |
| D4 | 85 | 5.66 | 4.43 | 4.20 | 39.24 | 118.41 | 4.80 | 45.55 | 18.14 | 13.80 |
| T-C-D4 | 0.80 | 0.80 | 2.30 | 2.00 | 2.30 | 2.10 | 2.20 | 2.40 | 2.30 | |
| D5 | 95 | 5.59 | 4.85 | 3.74 | 34.94 | 105.40 | 4.27 | 40.55 | 16.14 | 12.20 |
| L-Pl-E | - | 14.89 | 11.07 | 1.65 | 59.16 | 89.53 | 8.96 | 43.61 | 14.40 | 15.88 |
| D1 | 55 | 14.11 | 7.58 | 2.48 | 75.34 | 127.54 | 10.32 | 50.45 | 15.81 | 19.65 |
| D2 | 65 | 12.84 | 6.53 | 2.73 | 95.39 | 146.76 | 12.87 | 65.39 | 19.25 | 26.84 |
| D3 | 75 | 10.49 | 5.12 | 3.07 | 112.80 | 165.87 | 15.89 | 88.22 | 24.32 | 29.32 |
| D4 | 85 | 8.95 | 4.30 | 3.22 | 131.79 | 185.73 | 18.95 | 92.53 | 29.67 | 34.75 |
| T-C-D4 | 0.60 | 0.40 | 2.00 | 2.20 | 2.10 | 2.10 | 2.10 | 2.20 | 2.10 | |
| D5 | 95 | 9.12 | 4.24 | 3.28 | 130.23 | 184.42 | 19.03 | 93.15 | 30.20 | 34.87 |
| No. | Compound | RI | RT | Composition, % | |||
|---|---|---|---|---|---|---|---|
| L-Pl-D4 | L-Bg-D4 | L-Pl-O | L-Bg-O | ||||
| 1 | α-Pinene | 1022 | 11.59 | - | - | 0.21 | 0.48 |
| 2 | Camphene | 1075 | 13.52 | - | - | 0.30 | 0.22 |
| 3 | (-)-β-Pinene | 1082 | 13.81 | - | - | 0.36 | 0.17 |
| 4 | 1-Octen-3-ol | 1095 | 14.43 | - | 0.20 | 0.38 | 0.53 |
| 5 | 3-Octanone | 1101 | 15.02 | - | - | 0.39 | 0.42 |
| 6 | (-)-Limonene | 1113 | 14.98 | - | - | 0.73 | - |
| 7 | trans-β-Ocimene | 1123 | 15.38 | 0.26 | - | 1.06 | - |
| 8 | Eucalyptol | 1139 | 16.05 | 0.84 | 1.80 | 3.27 | 4.23 |
| 9 | Sabinene hydrate | 1179 | 17.69 | - | - | - | 0.27 |
| 10 | cis-Linalool oxide (furanoid) | 1186 | 18.03 | 1.19 | 5.54 | 2.61 | 10.72 |
| 11 | Linalool (3,7-dimethyl-1,6-octadien-3-ol) | 1209 | 18.98 | 19.21 | 8.38 | 28.34 | 17.44 |
| 12 | trans-Linalool oxide (furanoid) | 1212 | 19.13 | 1.20 | 4.89 | 2.57 | 9.21 |
| 13 | Lavender lactone | 1278 | 22.01 | - | 1.32 | 0.41 | 2.49 |
| 14 | Lavandulol | 1295 | 22.78 | 1.84 | 0.71 | 1.52 | 0.87 |
| 15 | Terpinen-4-ol | 1323 | 24.01 | 7.17 | 3.06 | 7.56 | 4.20 |
| 16 | Linalool oxide (pyranoid) | 1332 | 24.40 | 1.05 | 2.33 | 1.42 | 2.87 |
| 17 | α-Terpineol | 1348 | 25.09 | 0.31 | - | 0.19 | - |
| 18 | 2,6-Dimethyl-3,7-octadiene-2,6-diol | 1352 | 25.24 | 1.41 | 2.64 | - | - |
| 19 | 3,7-Dimethyl-1,5- octadiene-3,7-diol (terpendiol I) | 1362 | 25.70 | 0.26 | 0.92 | - | 0.87 |
| 20 | Linalool acetate | 1368 | 25.94 | 30.51 | 20.84 | 36.42 | 32.34 |
| 21 | NI | 1380 | 26.46 | 0.31 | - | - | 0.21 |
| 22 | Isobornyl formate | 1392 | 26.52 | - | - | 0.29 | - |
| 23 | Crypton | 1402 | 27.42 | 0.80 | - | 1.07 | 0.55 |
| 24 | (±)-Lavandulyl acetate | 1413 | 27.91 | 5.17 | 3.02 | 4.50 | 3.99 |
| 25 | Bornyl acetate | 1433 | 28.76 | - | 0.66 | 0.22 | 1.03 |
| 26 | p-Cumic aldehyde | 1456 | 29.69 | 0.29 | - | 0.36 | 0.21 |
| 27 | 2,6-Dimethyl-1,7-octadiene-3,6-diol | 1465 | 30.08 | 0.48 | 0.65 | - | - |
| 28 | 3,7-Dimethyl-1,7-octadien-3,6-diol, (terpendiol II) | 1504 | 31.71 | 2.63 | 8.87 | - | 0.69 |
| 29 | Longicyclene | 1508 | 31.90 | 2.05 | 1.00 | 1.66 | 1.02 |
| 30 | α-Bergamotene | 1523 | 32.49 | 0.33 | - | 0.25 | - |
| 31 | β-Caryophyllene | 1537 | 33.04 | 2.68 | 1.33 | 2.73 | 1.69 |
| 32 | β-Farnesene | 1551 | 33.62 | 1.49 | 1.41 | 0.19 | 0.59 |
| 33 | 2,6-Dimethyl-2,7-octadiene-1,6-diol (8-hydroxylinalool) | 1555 | 33.78 | 0.29 | 1.17 | - | - |
| 34 | Limonene-1,2-diol | 1569 | 34.36 | 0.22 | - | - | - |
| 35 | Car-3-en-5-one | 1573 | 34.49 | 0.25 | - | - | - |
| 36 | Nerolidol | 1592 | 35.28 | 0.25 | 0.60 | - | - |
| 37 | cis-2-Methyl-3-oxo- cyclohexanebutanal | 1604 | 35.75 | 1.18 | 0.99 | - | - |
| 38 | cis-9-Tetradecen-1-ol | 1607 | 35.86 | 0.39 | 0.81 | - | - |
| 39 | Epoxylinalool | 1614 | 36.13 | 0.35 | - | - | - |
| 40 | Farnesyl formate | 1618 | 36.28 | 0.55 | 0.50 | - | - |
| 41 | Nerolidyl acetate | 1661 | 37.93 | 0.43 | 0.54 | - | - |
| 42 | α-Santalol | 1667 | 38.17 | 0.49 | - | - | - |
| 43 | α-Bisabolol | 1694 | 39.21 | 1.18 | 1.24 | - | - |
| 44 | NI | 1705 | 39.46 | - | 1.46 | - | - |
| 45 | NI | 1716 | 39.54 | - | 1.51 | - | - |
| 46 | Ascaridole | 1723 | 40.28 | 0.24 | 0.24 | - | - |
| 47 | NI | 1743 | 41.00 | 0.95 | 4.11 | - | - |
| 48 | NI | 1747 | 41.15 | 0.54 | 3.75 | - | - |
| 49 | (1S,2S,4S)-Trihydroxy-p-menthane | 1774 | 42.13 | 0.45 | 1.53 | - | - |
| 50 | β-Caryophyllene oxide | 1791 | 42.77 | 5.63 | 5.30 | 0.98 | 1.27 |
| 51 | NI | 1811 | 43.47 | - | 0.82 | - | - |
| 52 | Coumarin | 1823 | 43.88 | 1.10 | - | - | - |
| 53 | tau-Cadinol | 1842 | 44.57 | 0.94 | 1.07 | - | - |
| 54 | Longifolene aldehyde | 1880 | 45.87 | 0.30 | - | - | - |
| 55 | Ledene oxide-(II) | 1891 | 46.27 | 0.31 | 0.60 | - | - |
| 56 | Isolongifolol | 1990 | 49.57 | 0.46 | 1.41 | - | - |
| 57 | Neryl acetate | 2121 | 53.76 | 0.25 | 0.24 | - | - |
| 58 | Gerany-p-cymene | 2134 | 54.16 | 0.29 | - | - | - |
| 59 | Herniarin | 2189 | 55.82 | 1.38 | 0.55 | - | - |
| Microorganisms | L-Pl-D4 | L-Pl-O | L-Bg-D4 | L-Bg-O | L-Pl-E | L-Bg-E | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gram-positive bacteria | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC |
| Staphylococcus aureus ATCC 25923 | 1 | 2 | 2 | 4 | 2 | 2 | 4 | 8 | 2 | 4 | 2 | 4 |
| Staphylococcus aureus ATCC 29213 | 2 | 4 | 4 | 8 | 4 | 4 | 4 | 8 | 4 | 8 | 8 | 8 |
| Staphylococcus aureus ATCC BAA1707 | 2 | 4 | 4 | 8 | 4 | 8 | 4 | 8 | 2 | 8 | 4 | 8 |
| Bacillus subtilis ATCC 6633 | 2 | 2 | 1 | 4 | 1 | 4 | 2 | 4 | 0.25 | 4 | 0.06 | 4 |
| Bacillus cereus ATCC 10876 | 2 | 2 | 4 | 4 | 2 | 2 | 4 | 4 | 1 | 16 | 2 | 16 |
| Micrococcus luteus ATCC 10240 | 0.5 | 2 | 1 | 4 | 1 | 2 | 1 | 4 | 0.5 | 4 | 0.5 | 4 |
| Micrococcus faecalis ATCC 29212 | 1 | 4 | 1 | 4 | 2 | 8 | 2 | 8 | 1 | 16 | 4 | 16 |
| Gram-negative bacteria | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC |
| Escherichia coli ATCC 25922 | 8 | 8 | 8 | 8 | 8 | 16 | 8 | 16 | 8 | 16 | 16 | 16 |
| Yeast strains | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC |
| Candida albicans ATCC 10231 | 0.5 | 1 | 0.5 | 1 | 0.5 | 1 | 0.5 | 1 | 2 | 4 | 4 | 4 |
| Candida glabrata ATCC 90030 | 1 | 2 | 1 | 2 | 1 | 2 | 1 | 2 | 2 | 4 | 4 | 8 |
| Candida krusei ATCC 14243 | 1 | 1 | 1 | 1 | 1 | 2 | 1 | 2 | 2 | 4 | 4 | 8 |
| Microorganisms | A | B | C | D | E | F | G | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gram-positive bacteria | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC |
| Staphylococcus aureus ATCC 25923 | 1 | 2 | 1 | 1 | 2 | 4 | 2 | 2 | 1 | 4 | 0.5 | 1 | 0.5 | 1 |
| Staphylococcus aureus ATCC 29213 | 0.25 | 4 | 2 | 4 | 2 | 4 | 2 | 2 | 1 | 2 | 0.5 | 1 | 1 | 2 |
| Staphylococcus aureus ATCC BAA1707 | 0.25 | 4 | 2 | 4 | 2 | 4 | 2 | 4 | 1 | 4 | 0.5 | 1 | 1 | 2 |
| Bacillus subtilis ATCC 6633 | 1 | 4 | 0.125 | 2 | 0.5 | 2 | 2 | 2 | 0.5 | 4 | 0.06 | 0.5 | 0.06 | 2 |
| Bacillus cereus ATCC 10876 | 1 | 4 | 0.25 | 4 | 2 | 4 | 2 | 4 | 1 | 4 | 0.5 | 4 | 1 | 4 |
| Micrococcus luteus ATCC 10240 | 0.5 | 2 | 0.5 | 4 | 0.25 | 1 | 0.5 | 2 | 0.03 | 4 | 0.125 | 1 | 0.125 | 1 |
| Micrococcus faecalis ATCC 29212 | 1 | 4 | 2 | 4 | 1 | 4 | 0.5 | 2 | 0.5 | 4 | 0.5 | 2 | 2 | 8 |
| Gram-negative bacteria | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC |
| Escherichia coli ATCC 25922 | 2 | 4 | 0.5 | 4 | 1 | 4 | 2 | 4 | 2 | 4 | 1 | 1 | 1 | 2 |
| Yeast strains | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC |
| Candida albicans ATCC 10231 | 1 | 2 | 0.5 | 1 | 1 | 1 | 0.5 | 1 | 0.5 | 2 | 0.5 | 0.5 | 0.5 | 1 |
| Candida glabrata ATCC 90030 | 1 | 2 | 0.5 | 1 | 1 | 1 | 1 | 2 | 0.5 | 2 | 0.5 | 0.5 | 0.5 | 1 |
| Candida krusei ATCC 14243 | 1 | 4 | 1 | 2 | 1 | 1 | 1 | 2 | 1 | 2 | 0.5 | 0.5 | 0.25 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dębczak, A.; Tyśkiewicz, K.; Fekner, Z.; Kamiński, P.; Florkowski, G.; Konkol, M.; Rój, E.; Grzegorczyk, A.; Malm, A. Molecular Distillation of Lavender Supercritical Extracts: Physicochemical and Antimicrobial Characterization of Feedstocks and Assessment of Distillates Enriched with Oxygenated Fragrance Components. Molecules 2022, 27, 1470. https://doi.org/10.3390/molecules27051470
Dębczak A, Tyśkiewicz K, Fekner Z, Kamiński P, Florkowski G, Konkol M, Rój E, Grzegorczyk A, Malm A. Molecular Distillation of Lavender Supercritical Extracts: Physicochemical and Antimicrobial Characterization of Feedstocks and Assessment of Distillates Enriched with Oxygenated Fragrance Components. Molecules. 2022; 27(5):1470. https://doi.org/10.3390/molecules27051470
Chicago/Turabian StyleDębczak, Agnieszka, Katarzyna Tyśkiewicz, Zygmunt Fekner, Piotr Kamiński, Grzegorz Florkowski, Marcin Konkol, Edward Rój, Agnieszka Grzegorczyk, and Anna Malm. 2022. "Molecular Distillation of Lavender Supercritical Extracts: Physicochemical and Antimicrobial Characterization of Feedstocks and Assessment of Distillates Enriched with Oxygenated Fragrance Components" Molecules 27, no. 5: 1470. https://doi.org/10.3390/molecules27051470
APA StyleDębczak, A., Tyśkiewicz, K., Fekner, Z., Kamiński, P., Florkowski, G., Konkol, M., Rój, E., Grzegorczyk, A., & Malm, A. (2022). Molecular Distillation of Lavender Supercritical Extracts: Physicochemical and Antimicrobial Characterization of Feedstocks and Assessment of Distillates Enriched with Oxygenated Fragrance Components. Molecules, 27(5), 1470. https://doi.org/10.3390/molecules27051470







