Chloroaluminate Ionic Liquid Immobilized on Magnetic Nanoparticles as a Heterogeneous Lewis Acidic Catalyst for the Friedel–Crafts Sulfonylation of Aromatic Compounds
Abstract
:1. Introduction
2. Results and Discussion
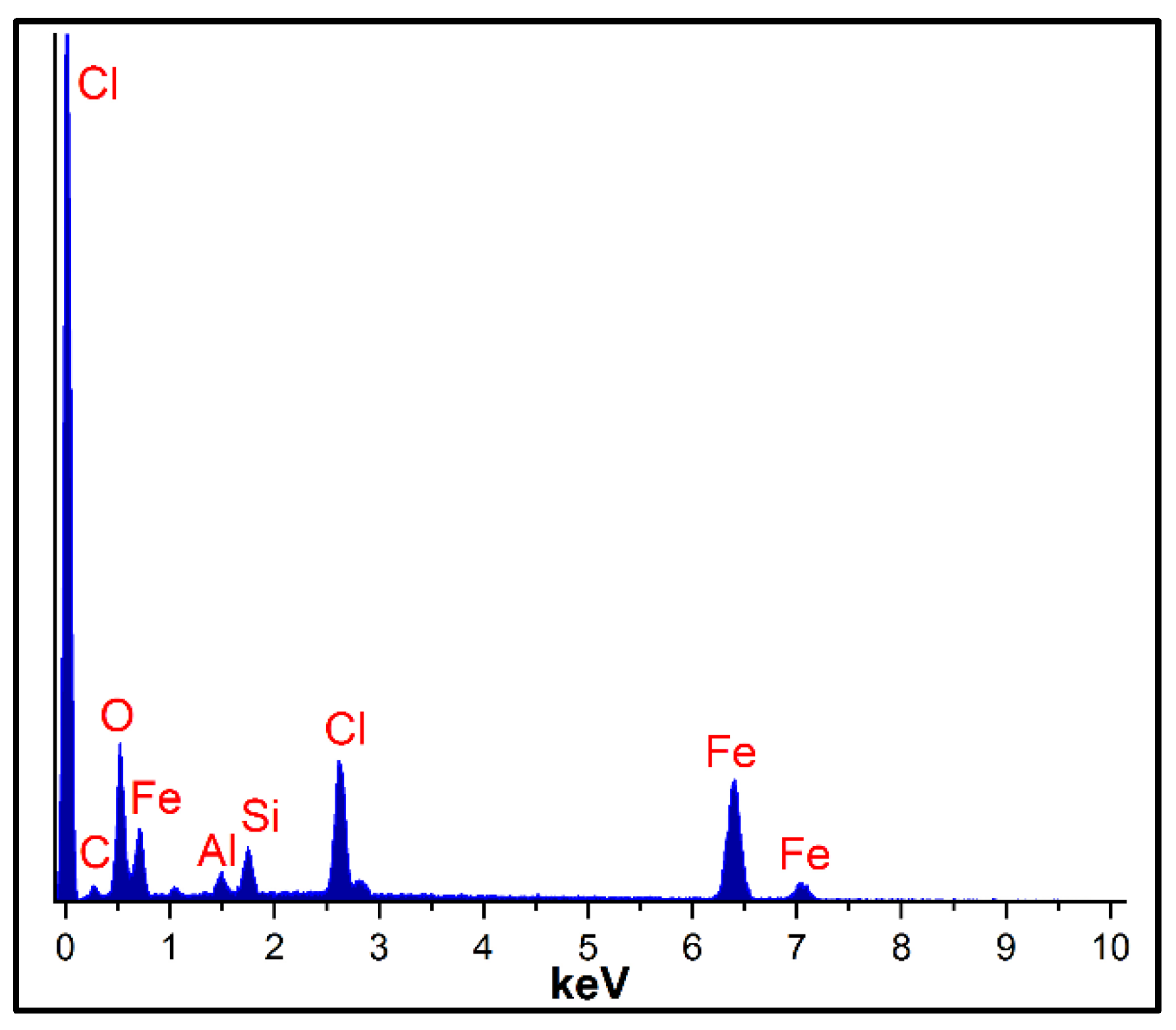
2.1. Catalyst Characterization
2.2. Friedel–Crafts Sulfonylation
3. Materials and Methods
3.1. General Procedure for Preparation of Heterogeneous Catalyst Fe3O4@O2Si[PrMIM]Cl·AlCl3
3.1.1. The Preparation of MNPs via the Modified Chemical Coprecipitation Method
3.1.2. The Preparation of 3-Methyl-1-(3-trimethoxysilylpropyl)-1H-imidazole-3-ium Chloride
3.1.3. Methyl-1-(3-trimethoxysilylpropyl)-1H-imidazole-3-ium Chloride
3.1.4. The Preparation of Fe3O4@O2Si[PrMIM]Cl
3.1.5. The Preparation of Fe3O4@O2Si[PrMIM]Cl·AlCl3
3.2. General Procedure for the Friedel–Crafts Sulfonylation
3.3. Spectroscopic Data
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Padwa, A.; Bullock, W.H.; Dyszlewski, A.D. Studies dealing with the alkylation-[1,3]-rearrangement reaction of some phenylthio-substituted allylic sulfones. J. Org. Chem. 1990, 55, 955–964. [Google Scholar] [CrossRef]
- Block, E. The organosulfur chemistry of the Genus Allium—Implications for the organic chemistry of sulfur. Angew. Chem. Int. Ed. 1992, 31, 1135–1178. [Google Scholar] [CrossRef]
- Shaaban, O.; Rizk, O.; Bayad, A.; El-Ashmawy, I. Synthesis of some 4,5-Dihydrothieno[3,2-e][1,2,4]triazolo[4,3-a] Pyrimi-dine-2-carboxamides as anti-inflammatory and analgesic agents. Open J. Med. Chem. 2013, 7, 49–65. [Google Scholar] [CrossRef] [Green Version]
- Hwang, S.H.; Wagner, K.M.; Morisseau, C.; Liu, J.Y.; Dong, H.; Wecksler, A.T.; Hammock, B.D. Synthesis and structure-activity relationship studies of urea-containing pyrazoles as dual inhibitors of cyclooxygenase-2 and soluble epoxide hydrolase. J. Med. Chem. 2011, 54, 3037–3050. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meadows, D.C.; Sanchez, T.; Neamati, N.; North, T.W.; Gervay-Hague, J. Ring substituent effects on biological activity of vinyl sulfones as inhibitors of HIV-1. Bioorg. Med. Chem. 2007, 15, 1127–1137. [Google Scholar] [CrossRef] [Green Version]
- Capela, R.; Oliveira, R.; Gonçalves, L.M.; Domingos, A.; Gut, J.; Rosenthal, P.J.; Lopes, F.; Moreira, R. Artemisinin-dipeptidyl vinyl sulfone hybrid molecules: Design, synthesis and preliminary SAR for antiplasmodial activity and falcipain-2 inhibition. Bioorg. Med. Chem. Lett. 2009, 19, 3229–3232. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosenthal, A.S.; Chen, X.; Liu, J.O.; West, D.C.; Hergenrother, P.J.; Shapiro, T.A.; Posner, G.H. Malaria-infected mice are cured by a single oral dose of new dimeric trioxane sulfones which are also selectively and powerfully cytotoxic to cancer cells. J. Med. Chem. 2009, 52, 1198–1203. [Google Scholar] [CrossRef] [Green Version]
- Al-Said, M.S.; Ghorab, M.M.; Nissan, Y.M. Dapson in heterocyclic chemistry, part VIII: Synthesis, molecular docking and anticancer activity of some novel sulfonylbiscompounds carrying biologically active 1,3-dihydropyridine, chromene and chromenopyridine moieties. Chem. Cent. J. 2012, 6, 64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soni, S.; Seth, M.; Sah, P. Synthesis and in-vitro antimicrobial evaluation of some novel phthalyl substituted aryl sulphones and sulphonamides. Res. J. Pharm. Biol. Chem. Sci. 2012, 3, 898–907. [Google Scholar]
- Li, P.; Yin, J.; Xu, W.; Wu, J.; He, M.; Hu, D.; Yang, S.; Song, B. Synthesis, antibacterial activities, and 3D-QSAR of sulfone derivatives containing 1, 3, 4-oxadiazole moiety. Chem. Biol. Drug Chem. 2013, 82, 546–556. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.T.; Lira, R.; Hansell, E.; McKerrow, J.H.; Roush, W.R. Synthesis of macrocyclic trypanosomal cysteine protease inhibitors. Bioorg. Med. Chem. Lett. 2008, 18, 5860–5863. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jereb, M. Highly atom-economic, catalyst- and solvent-free oxidation of sulfides into sulfones using 30% aqueous H2O2. Green Chem. 2012, 14, 3047–3052. [Google Scholar] [CrossRef]
- Kirihara, M.; Itou, A.; Noguchi, T.; Yamamoto, J. Tantalum carbide or niobium carbide catalyzed oxidation of sulfides with hydrogen peroxide: Highly efficient and chemoselective syntheses of sulfoxides and sulfones. Synlett 2010, 2010, 1557–1561. [Google Scholar] [CrossRef]
- Pritzius, A.B.; Breit, B. Asymmetric rhodium-catalyzed addition of thiols to allenes: Synthesis of branched allylic thioethers and sulfones. Angew. Chem. Int. Ed. 2015, 54, 3121–3125. [Google Scholar] [CrossRef] [PubMed]
- Maloney, K.M.; Kuethe, J.T.; Linn, K. A practical, one-pot synthesis of sulfonylated pyridines. Org. Lett. 2011, 13, 102–105. [Google Scholar] [CrossRef] [PubMed]
- Umierski, N.; Manolikakes, G. Metal-free synthesis of diaryl sulfones from arylsulfinic acid salts and diaryliodonium salts. Org. Lett. 2013, 15, 188–191. [Google Scholar] [CrossRef] [PubMed]
- Pandya, V.G.; Mhaske, S.B. Transition-metal-free C–S bond formation: A facile access to aryl sulfones from sodium sulfinates via arynes. Org. Lett. 2014, 16, 3836–3839. [Google Scholar] [CrossRef]
- Taniguchi, N. Aerobic copper-catalyzed synthesis of (E)-alkenyl sulfones and (E)-β-halo-alkenyl sulfones via addition of sodium sulfinates to alkynes. Tetrahedron 2014, 70, 1984–1990. [Google Scholar] [CrossRef]
- Yang, W.; Yang, S.; Li, P.; Wang, L. Visible-light initiated oxidative cyclization of phenyl propiolates with sulfinic acids to coumarin derivatives under metal-free conditions. Chem. Commun. 2015, 51, 7520–7523. [Google Scholar] [CrossRef]
- Chen, J.; Mao, J.; Zheng, Y.; Liu, D.; Rong, G.; Yan, H.; Zhang, C.; Shi, D. Iodine-promoted decarboxylative C–S cross-coupling of cinnamic acids with sodium benzene sulfinates. Tetrahedron 2015, 71, 5059–5063. [Google Scholar] [CrossRef]
- Truce, W.E.; Milionis, J.P. Friedel—Crafts cyclization of ι-phenylalkanesulfonyl chlorides. J. Am. Chem. Soc. 1952, 74, 974–977. [Google Scholar] [CrossRef]
- Truce, W.E.; Vriesen, C.W. Friedel—Crafts reactions of methanesulfonyl chloride with benzene and certain substituted benzenes. J. Am. Chem. Soc. 1953, 75, 5032–5036. [Google Scholar] [CrossRef]
- Jensen, F.R.; Brown, H.C. Kinetics of the Friedel-Crafts sulfonylation of aromatics with aluminum chloride as catalyst and nitrobenzene as solvent. J. Am. Chem. Soc. 1958, 80, 4038–4041. [Google Scholar] [CrossRef]
- Olah, G.A.; Kobayashi, S.; Nishimura, J. Aromatic substitution. XXXI. Friedel-Crafts sulfonylation of benzene and toluene with alkyl- and arylsulfonyl halides and anhydrides. J. Am. Chem. Soc. 1973, 95, 564–569. [Google Scholar] [CrossRef]
- Marquié, J.; Laporterie, A.; Dubac, J.; Roques, N.; Desmurs, J.-R. Acylation and related reactions under microwaves. 4. Sulfonylation reactions of aromatics. J. Org. Chem. 2001, 66, 421–425. [Google Scholar] [CrossRef]
- Fleck, T.J.; Chen, J.J.; Lu, C.V.; Hanson, K.J. Isomerization-free sulfonylation and its application in the synthesis of PHA-565272A. Org. Process Res. Dev. 2006, 10, 334–338. [Google Scholar] [CrossRef]
- Bandgar, B.P.; Kasture, S.P. Zinc-Mediated fast sulfonylation of aromatics. Synth. Commun. 2001, 31, 1065–1068. [Google Scholar] [CrossRef]
- Jang, D.O.; Moon, K.S.; Cho, D.H.; Kim, J.-G. Highly selective catalytic Friedel-Crafts acylation and sulfonylation of activated aromatic compounds using indium metal. Tetrahedron Lett. 2006, 47, 6063–6066. [Google Scholar] [CrossRef]
- De Noronha, R.G.; Fernandes, A.C.; Romão, C.C. MoO2Cl2 as a novel catalyst for Friedel-Crafts acylation and sulfonylation. Tetrahedron Lett. 2009, 50, 1407–1410. [Google Scholar] [CrossRef]
- Singh, R.P.; Kamble, R.M.; Chandra, K.L.; Saravanan, P.; Singh, V.K. An efficient method for aromatic Friedel-Crafts alkylation, acylation, benzoylation, and sulfonylation reactions. Tetrahedron 2001, 57, 241–247. [Google Scholar] [CrossRef]
- Nguyen, V.T.A.; Duus, F.; Le, T.N. Upward trend in catalytic efficiency of rare-earth triflate catalysts in Friedel-Crafts aromatic sulfonylation reactions. Asian J. Org. Chem. 2014, 3, 963–968. [Google Scholar] [CrossRef]
- Jin, T.; Zhao, Y.; Ma, Y.; Li, T. A practical and efficient method for the preparation of aromatic sulfones by the reaction of aryl sulfonyl chlorides with arenes catalyzed by Fe(OH)3. Indian J. Chem. 2005, 44B, 2183–2185. [Google Scholar] [CrossRef]
- Olah, G.A.; Mathew, T.; Surya Prakash, G.K. Nafion-H catalysed sulfonylation of aromatics with arene/alkenesulfonic acids for the preparation of sulfones. Chem. Commun. 2001, 17, 1696–1697. [Google Scholar] [CrossRef] [PubMed]
- Choudary, B.M.; Chowdari, N.S.; Kantam, M.L.; Kannan, R. Fe(III) exchanged montmorillonite: A mild and ecofriendly catalyst for sulfonylation of aromatics. Tetrahedron Lett. 1999, 40, 2859–2862. [Google Scholar] [CrossRef]
- Borujeni, K.P.; Tamami, B. Polystyrene and silica gel supported AlCl3 as highly chemoselective heterogeneous Lewis acid catalysts for Friedel-Crafts sulfonylation of aromatic compounds. Catal. Commun. 2007, 8, 1191–1196. [Google Scholar] [CrossRef]
- Boroujeni, K.P. Sulfonylation of aromatic compounds with sulfonic acids using silica gel-supported AlCl3 as a heterogeneous Lewis acid catalyst. J. Sulphur. Chem. 2010, 31, 197–203. [Google Scholar] [CrossRef]
- Nara, S.J.; Harjani, J.R.; Salunkhe, M.M. Friedel-Crafts sulfonylation in 1-Butyl-3-methylimidazolium chloroaluminate ionic liquids. J. Org. Chem. 2001, 66, 8616–8620. [Google Scholar] [CrossRef]
- Bahrami, K.; Khodei, M.M.; Shahbazi, F. Highly selective catalytic Friedel-Crafts sulfonylation of aromatic compounds using a FeCl3-based ionic liquid. Tetrahedron Lett. 2008, 49, 3931–3934. [Google Scholar] [CrossRef]
- Hajipour, A.R.; Zarei, A.; Khazdooz, L.; Pourmousavi, S.A.; Mirjalili, B.B.F.; Ruoho, A.E. Direct sulfonylation of aromatic rings with aryl or alkyl sulfonic acid using supported P2O5/Al2O3. Phosphorus Sulfur Silicon Relat. Elem. 2006, 180, 2029–2034. [Google Scholar] [CrossRef]
- Mirjalili, F.; Zolfigol, M.A.; Bamoniri, A.; Khazdooz, L. An efficient method for the sulfonylation of aromatic rings with arene/alkane sulfonic acid by using P2O5/SiO2 under heterogeneous conditions. Bull. Korean Chem. Soc. 2003, 24, 1009–1010. [Google Scholar] [CrossRef]
- Zhao, D.; Wu, M.; Kou, Y.; Min, E. Ionic liquids: Applications in catalysis. Catal. Today 2002, 74, 157–189. [Google Scholar] [CrossRef]
- Boroujeni, K.P.; Jafarinasab, M. Polystyrene-supported chloroaluminate ionic liquid as a new heterogeneous Lewis acid catalyst for Knoevenagel condensation. Chin. Chem. Lett. 2012, 23, 1067–1070. [Google Scholar] [CrossRef]
- Mouradzadegun, A.; Elahi, S.; Abadast, F. Synthesis of a 3D-network polymer supported Bronsted acid ionic liquid based on calix[4]resorcinarene via two post-functionalization steps: A highly efficient and recyclable acid catalyst for the preparation of symmetrical bisamides. RSC Adv. 2014, 4, 31239–31248. [Google Scholar] [CrossRef]
- Wei-Li, D.; Bi, J.; Sheng-Lian, L.; Xu-Biao, L.; Xin-Man, T.; Chak-Tong, A. Polymers anchored with carboxyl-functionalized di-cation ionic liquids as efficient catalysts for the fixation of CO2 into cyclic carbonates. Catal. Sci. Technol. 2014, 4, 556–562. [Google Scholar] [CrossRef]
- Khoshnevis, M.; Davoodnia, A.; Zare-Bidaki, A.; Tavakoli-Hoseini, N. Alumina supported acidic ionic liquid: Preparation, characterization, and its application as catalyst in the synthesis of 1,8-dioxo-octahydroxanthenes. Synth. React. Inorg. Met. Org. Nano-Met. Chem. 2013, 43, 1154–1161. [Google Scholar] [CrossRef]
- Tamboli, A.H.; Chaugule, A.A.; Sheikh, F.A.; Chung, W.-J.; Kim, H. Synthesis, characterization, and application of silica supported ionic liquid as catalyst for reductive amination of cyclohexanone with formic acid and triethyl amine as hydrogen source. Chin. J. Catal. 2015, 36, 1365–1371. [Google Scholar] [CrossRef]
- Hu, Y.L.; Fang, D. Preparation of silica supported ionic liquids for highly selective hydroxylation of aromatics with hydrogen peroxide under solvent-free conditions. J. Mex. Chem. Soc. 2016, 60, 207–217. [Google Scholar] [CrossRef]
- Qian, C.; Yao, C.; Yang, L.; Yang, B.; Liu, S.; Liu, Z. Preparation and application of silica films supported imidazolium-based ionic liquid as efficient and recyclable catalysts for benzoin condensations. Catal. Lett. 2020, 150, 1389–1396. [Google Scholar] [CrossRef]
- Shojaei, R.; Zahedifar, M.; Mohammadi, P.; Saidi, K.; Sheibani, H. Novel magnetic nanoparticle supported ionic liquid as an efficient catalyst for the synthesis of spiro [pyrazole-pyrazolo[3,4-b]pyridine]-dione derivatives under solvent free conditions. J. Mol. Struct. 2019, 1178, 401–407. [Google Scholar] [CrossRef]
- Safari, J.; Zarnegar, Z. Brønsted acidic ionic liquid based magnetic nanoparticles: A new promoter for the Biginelli synthesis of 3,4-dihydropyrimidin-2(1H)-ones/thiones. New J. Chem. 2014, 38, 358–365. [Google Scholar] [CrossRef]
- Li, P.-H.; Li, B.-L.; Hu, H.-C.; Zhao, X.-N.; Zhang, Z.-H. Ionic liquid supported on magnetic nanoparticles as highly efficient and recyclable catalyst for the synthesis of β-keto enol ethers. Catal. Commun. 2014, 46, 118–122. [Google Scholar] [CrossRef]
- Naeimi, H.; Aghaseyedkarimi, D. Fe3O4@SiO2·HM·SO3H as a recyclable heterogeneous nanocatalyst for the microwave-promoted synthesis of 2,4,5-trisubstituted imidazoles under solvent free conditions. New J. Chem. 2015, 39, 9415–9421. [Google Scholar] [CrossRef]
- Ghorbani-Choghamarani, A.; Norouzi, M. Synthesis and characterization of ionic liquid immobilized on magnetic nanoparticles: A recyclable heterogeneous organocatalyst for the acetylation of alcohols. J. Magn. Magn. Mater. 2016, 401, 832–840. [Google Scholar] [CrossRef]
- Ghorbani-Choghamarani, A.; Taherinia, Z.; Nikoorazm, M. Ionic liquid supported on magnetic nanoparticles as a novel reusable nanocatalyst for the efficient synthesis of tetracyclic quinazoline compounds. Res. Chem. Intermed. 2018, 44, 6591–6604. [Google Scholar] [CrossRef]
- Naikwade, A.; Jagadale, M.; Kale, D.; Rashinkar, G. Magnetic nanoparticle supported ionic liquid phase catalyst for oxidation of alcohols. Aust. J. Chem. 2020, 73, 1088–1097. [Google Scholar] [CrossRef]
- Safari, J.; Zarnegar, Z. Immobilized ionic liquid on superparamagnetic nanoparticles as an effective catalyst for the synthesis of tetrasubstituted imidazoles under solvent-free conditions and microwave irradiation. Comptes Rendus Chim. 2013, 16, 920–928. [Google Scholar] [CrossRef]
- Zhang, Q.; Su, H.; Luo, J.; Wei, Y. A magnetic nanoparticle supported dual acidic ionic liquid: A “quasi-homogeneous” catalyst for the one-pot synthesis of benzoxanthenes. Green Chem. 2012, 14, 201–208. [Google Scholar] [CrossRef]
- Azgomi, N.; Mokhtary, M. Nano-Fe3O4@SiO2 supported ionic liquid as an efficient catalyst for the synthesis of 1,3-thiazolidin-4-ones under solvent-free conditions. J. Mol. Catal. A Chem. 2015, 398, 58–64. [Google Scholar] [CrossRef]
- Ngo, H.N.T.; Nguyen, N.L.T.; Luu, X.T.T. The Friedel-Crafts sulfonylation catalyzed by chloroaluminate ionic liquids. Sci. Tech. Dev. J. Nat. Sci. 2021, 5, 1581–1592. [Google Scholar]
- Lopez, J.A.; González, F.; Bonilla, F.A.; Zambrano, G.; Gómez, M.E. Synthesis and characterization of Fe3O4 magnetic nanofluid. Rev. Latinoam. Metal. Mater. 2010, 30, 60–66. [Google Scholar]
- Nazari, S.; Saadat, S.; Fard, P.K.; Gorjizadeh, M.; Nezhad, E.R.; Afshari, M. Imidazole functionalized magnetic Fe3O4 nanoparticles as a novel heterogeneous and efficient catalyst for synthesis of dihydropyrimidinones by Biginelli reaction. Monatsh. Chem. 2013, 144, 1877–1882. [Google Scholar] [CrossRef]
- Choudary, B.M.; Chowdari, N.S.; Kantam, M.L. Friedel–Crafts sulfonylation of aromatics catalysed by solid acids: An eco-friendly route for sulfone synthesis. J. Chem. Soc. Perkin Trans. I 2000, 16, 2689–2693. [Google Scholar] [CrossRef]
- Yang, M.; Shen, H.; Li, Y.; Shen, C.; Zhang, P. d-Glucosamine as a green ligand for copper catalyzed synthesis of aryl sulfones from aryl halides and sodium sulfinates. RSC Adv. 2014, 4, 26295–26300. [Google Scholar] [CrossRef]
- Liang, X.; Li, Y.; Xia, Q.; Cheng, L.; Guo, J.; Zhang, P.; Zhang, W.; Wang, Q. Visible-light-driven electron donor–acceptor complex induced sulfonylation of diazonium salts with sulfinates. Green Chem. 2021, 23, 8865–8870. [Google Scholar] [CrossRef]
- Bandgar, B.P.; Bettigeri, S.V.; Phopase, J. Unsymmetrical diaryl sulfones through palladium-catalyzed coupling of aryl boronic acids and arylsulfonyl chlorides. Org. Lett. 2004, 6, 2105–2108. [Google Scholar] [CrossRef] [PubMed]
- Bian, M.; Ma, C.; Xu, F. Anion-functionalized ionic liquids enhance the CuI-catalyzed cross-coupling reaction of sulfinic acid salts with aryl halides and vinyl bromides. Synthesis 2007, 2007, 2951–2956. [Google Scholar] [CrossRef]
- Cooke, M.; Clark, J.; Breeden, S. Lewis acid catalysed microwave-assisted synthesis of diaryl sulfones and comparison of associated carbon dioxide emissions. J. Mol. Catal. A Chem. 2009, 303, 132–136. [Google Scholar] [CrossRef]
- Deeming, A.S.; Russell, C.J.; Hennessy, A.J.; Willis, M.C. DABSO-based, three-component, one-pot sulfone synthesis. Org. Lett. 2014, 16, 150–153. [Google Scholar] [CrossRef]
- Chandrasekaran, R.; Perumal, S.; Wilson, D.A. NMR study of substituent effects in 4-substituted and 4,4′-disubstituted diphenyl sulphoxides and sulphones. Magn. Reson. Chem. 1989, 27, 360–367. [Google Scholar] [CrossRef]
- Srinivas, B.T.V.; Rawat, V.S.; Konda, K.; Sreedhar, B. Magnetically separable copper ferrite nanoparticles-catalyzed synthesis of diaryl, alkyl/aryl sulfones from arylsulfinic acid salts and organohalides/boronic acids. Adv. Synth. Catal. 2014, 356, 805–817. [Google Scholar] [CrossRef]
- Sharghi, H.; Shahsavari-Fard, Z. Al2O3/MeSO3H (AMA) a useful system for direct sulfonylation of phenols with p-toluenesulfonic acid. J. Iran. Chem. Soc. 2005, 2, 47–53. [Google Scholar] [CrossRef]









 | |||
|---|---|---|---|
| Entry | Acidic Catalyst | Ratio of 3a:3a′:3a″ | Yield (%) b |
| 1 | Fe3O4@O2Si[PrMIM]HSO4 (0.2 g) | 56:41:3 | 63 |
| 2 | MgFe2O4@O2Si[PrMIM]Cl·AlCl3 (0.2 g) | 63:31:6 | 57 |
| 3 | Fe3O4@O2Si[PrMIM]Cl·AlCl3 (0.1 g) | 49:44:7 | 78 |
| 4 | Fe3O4@O2Si[PrMIM]Cl·AlCl3 (0.2 g) | 55:39:6 | 85 |
| 5 | Fe3O4@O2Si[PrMIM]Cl·AlCl3 (0.3 g) | 47:46:7 | 86 |
 | ||||
|---|---|---|---|---|
| Entry | Arene | Sulfonyl Chloride | Product | Isolated Yield (%) (Time) d |
| 1 | 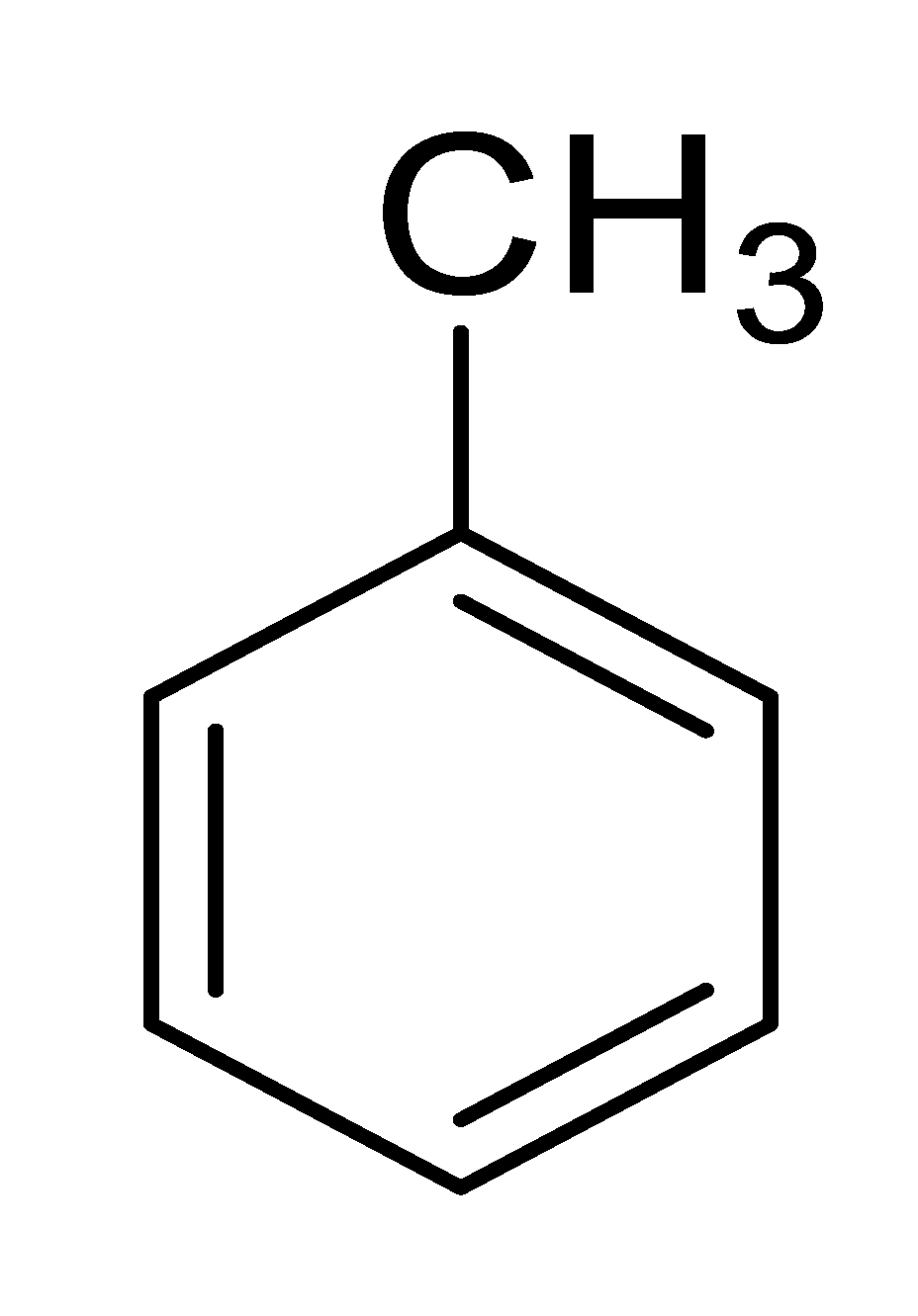 |  |  | 92 (4.0) |
| 2 | 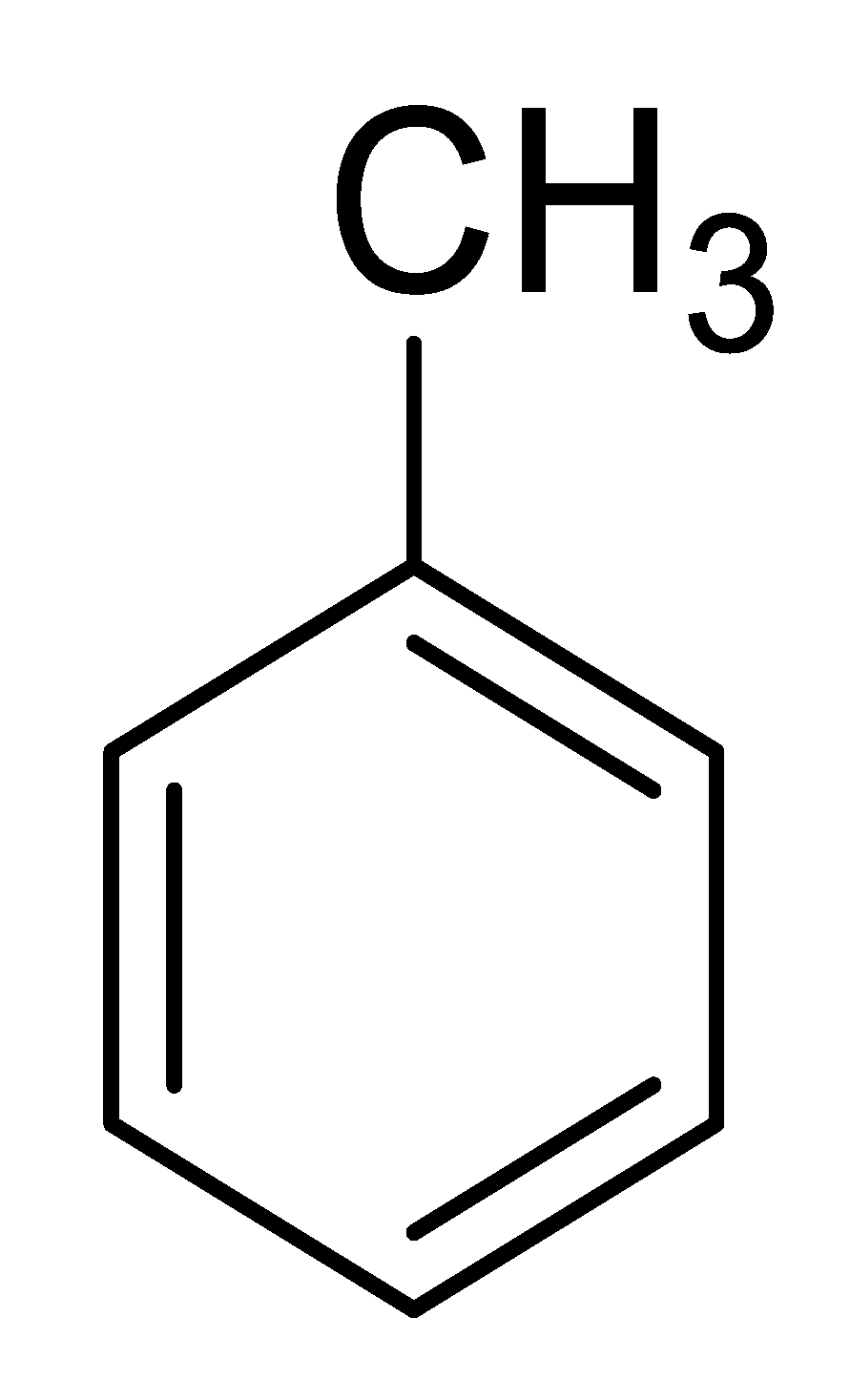 |  |  | 85 (4.5) |
| 3 |  |  | 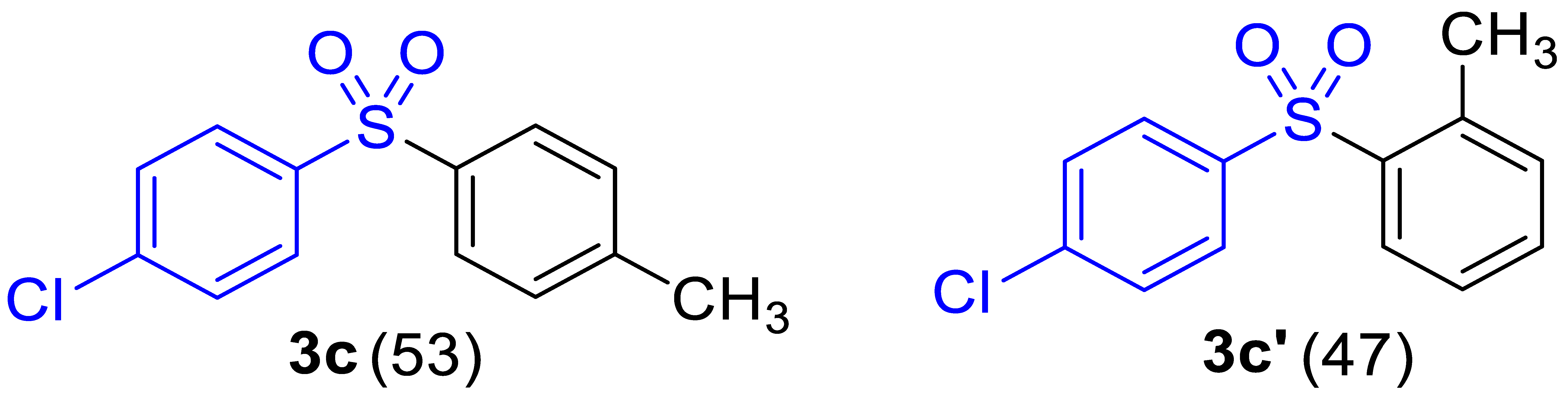 | 67 (4.0) |
| 4 |  | 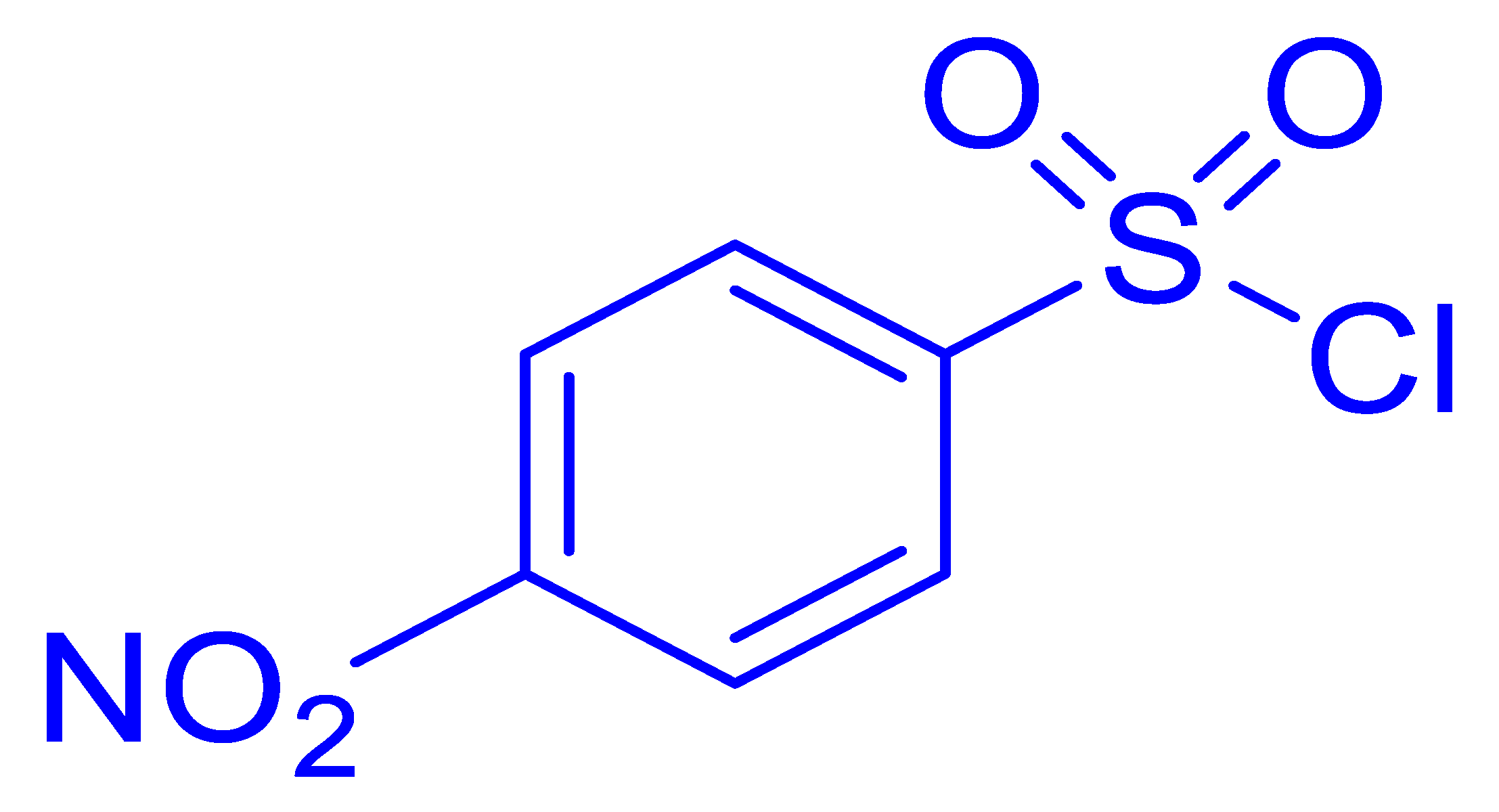 |  | 30 (5.0) |
| 5 |  |  |  | 85 (4.0) |
| 6 | 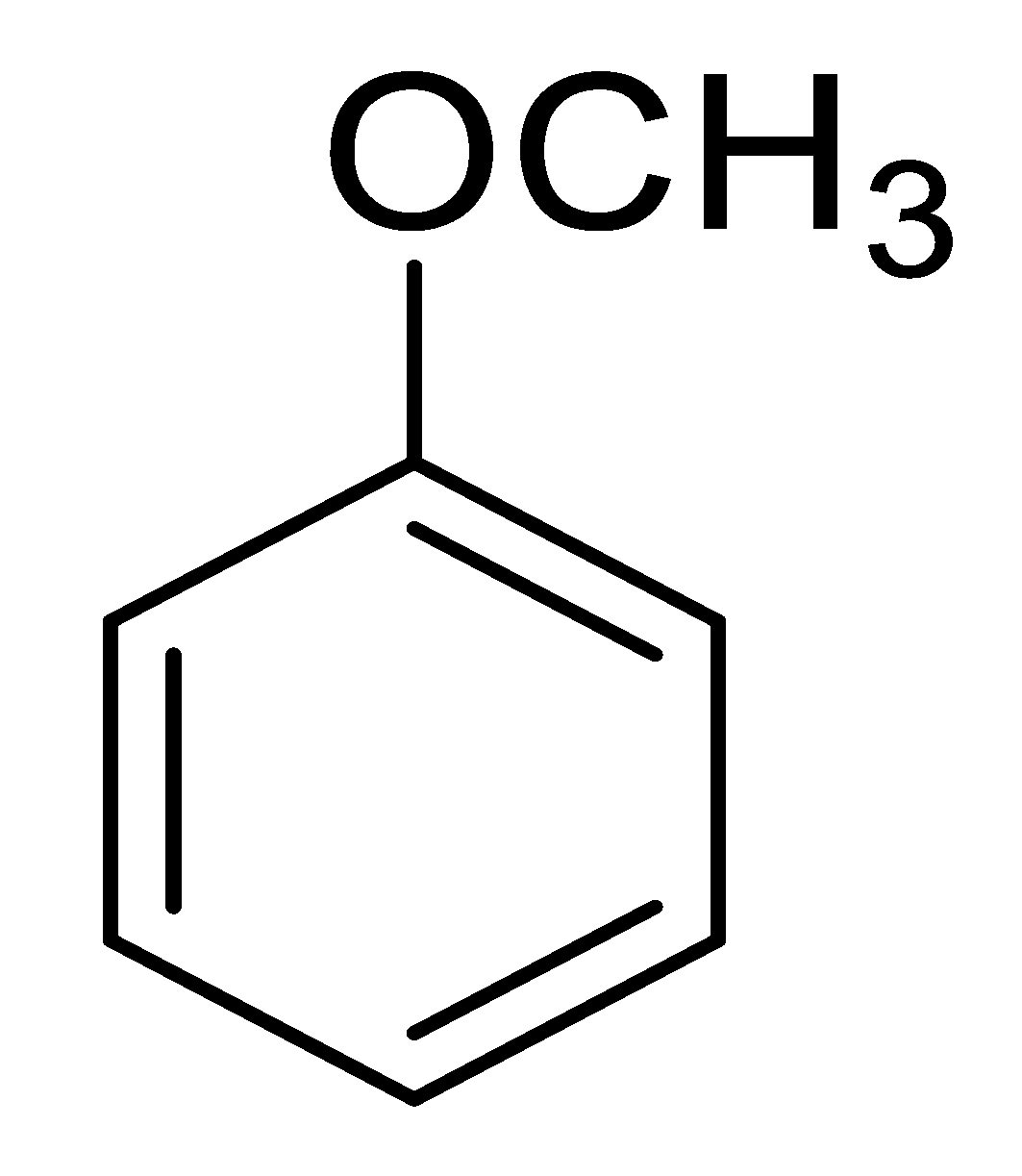 |  |  | 91 (1.0) |
| 7 | 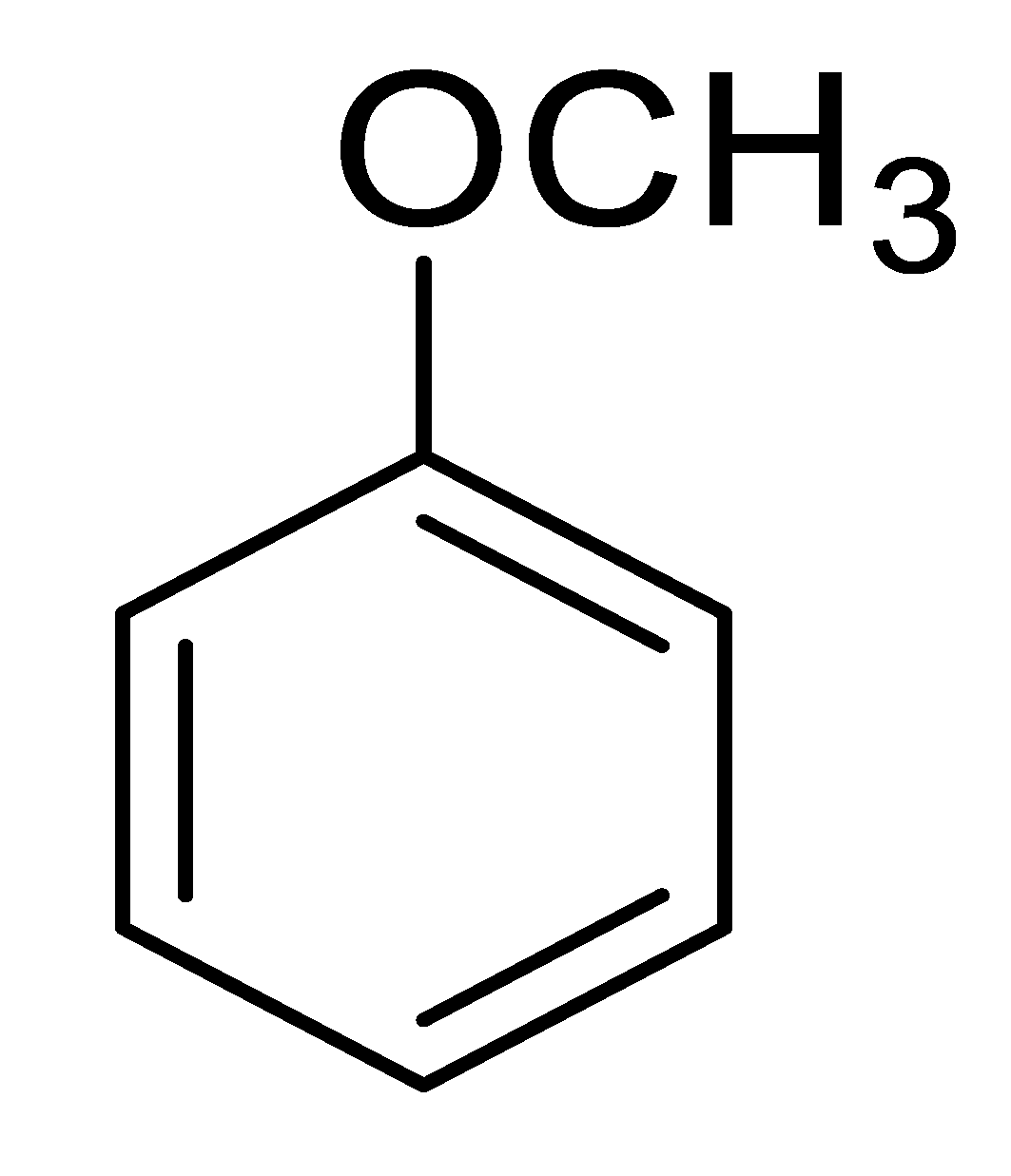 |  |  | 89 (4.0) |
| 8 |  |  |  | 54 (1.0) |
| 9 |  |  |  | 39 (3.5) |
| 10 | 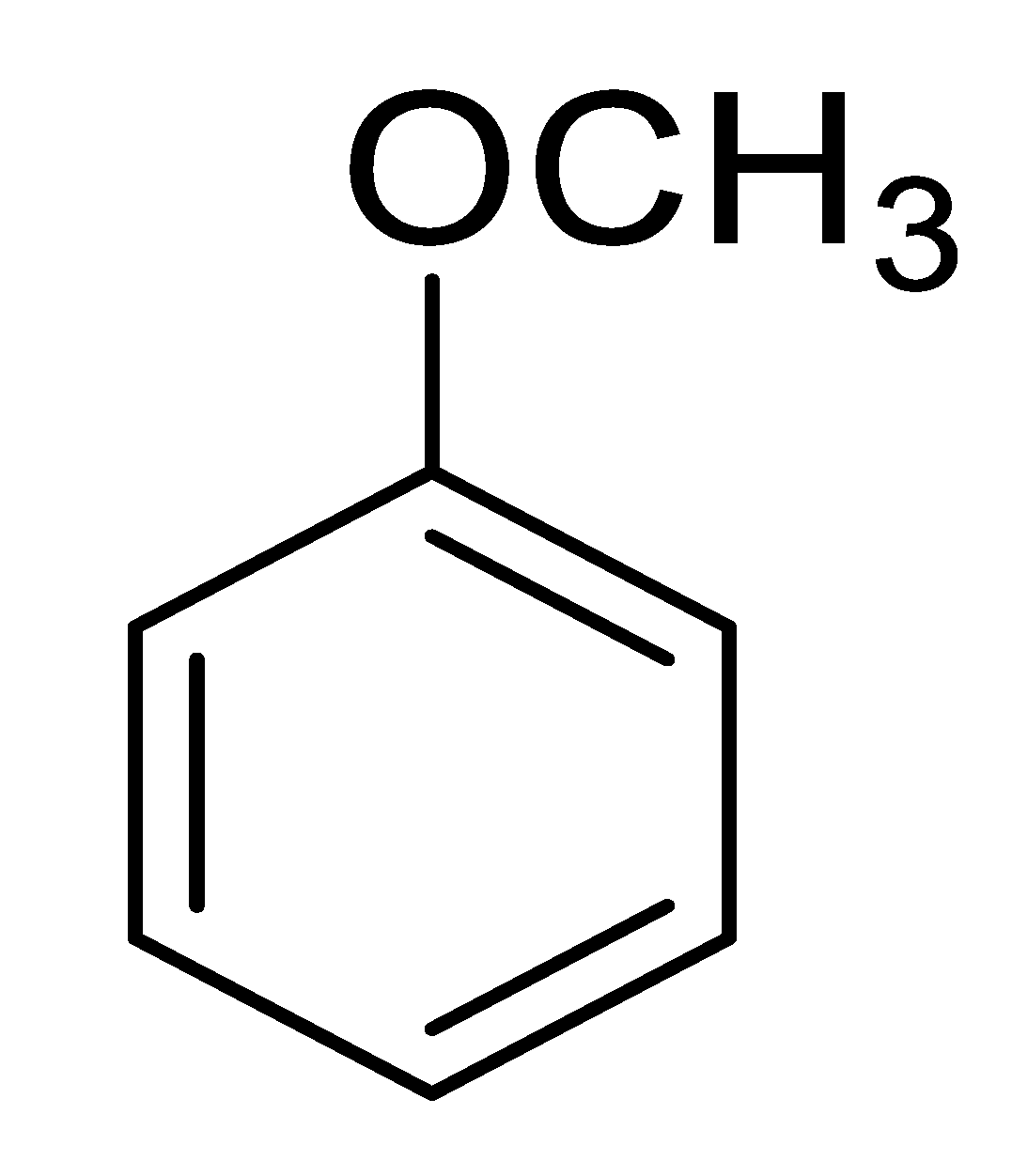 | 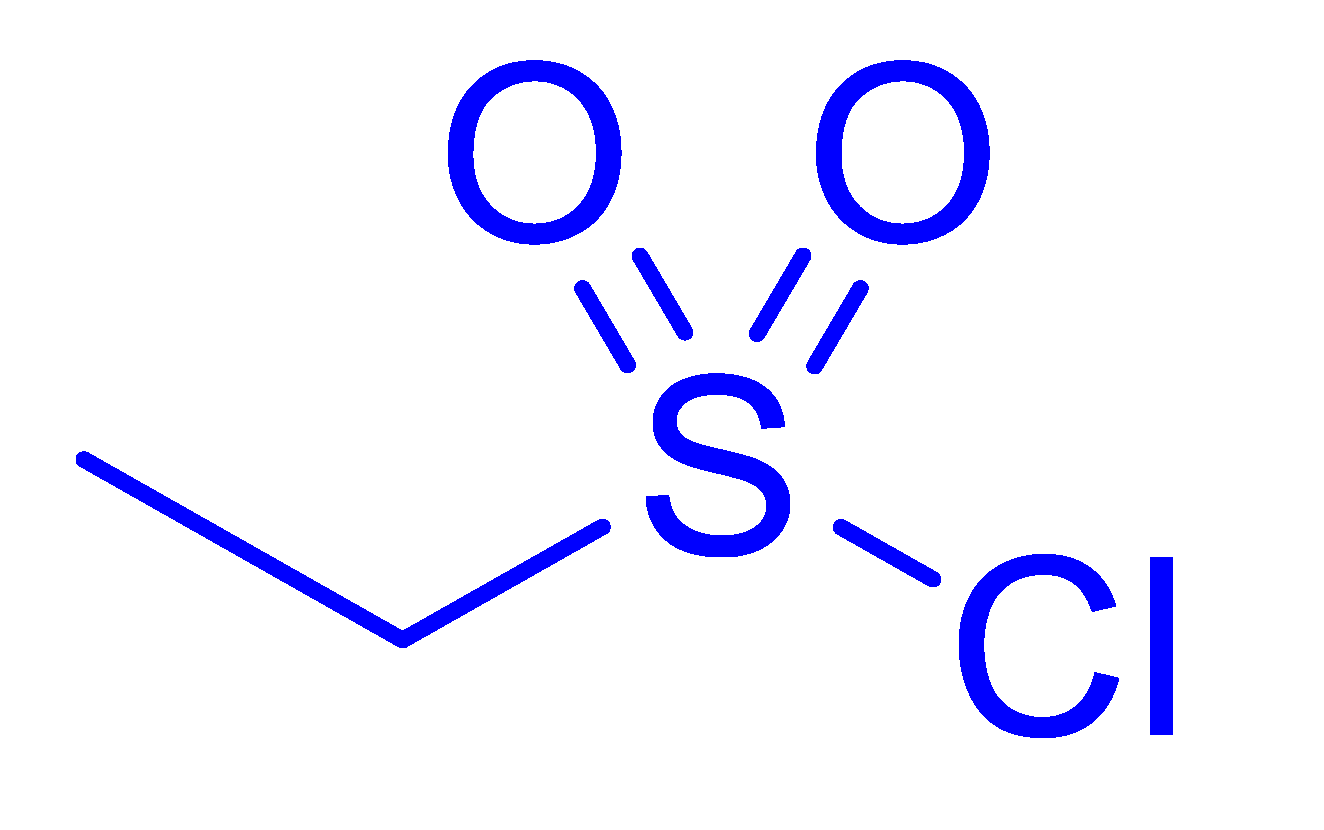 |  | 28 [e] (3.0) |
| 11 | 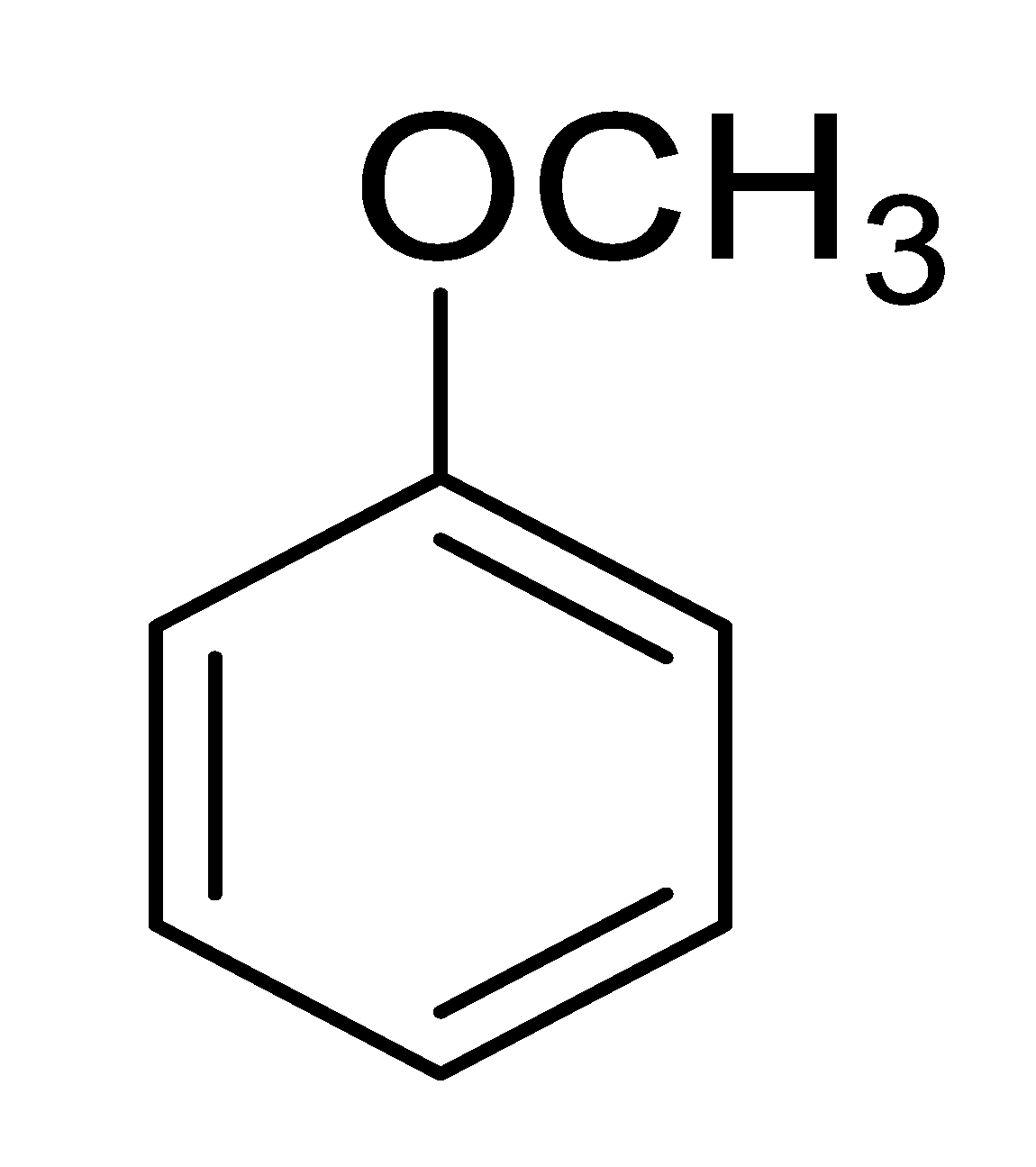 |  |  | 23 [e] (4.0) |
| 12 | 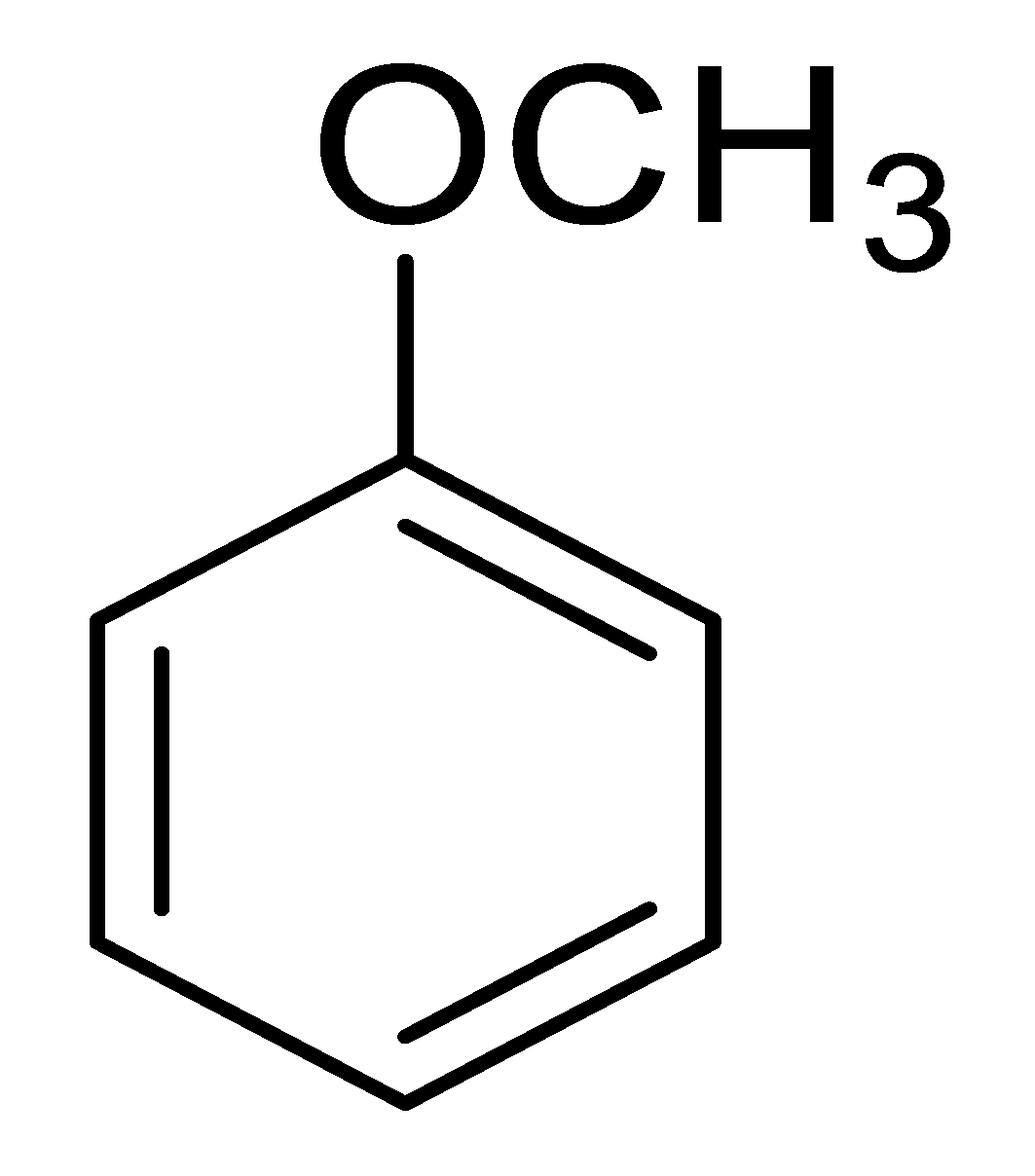 | 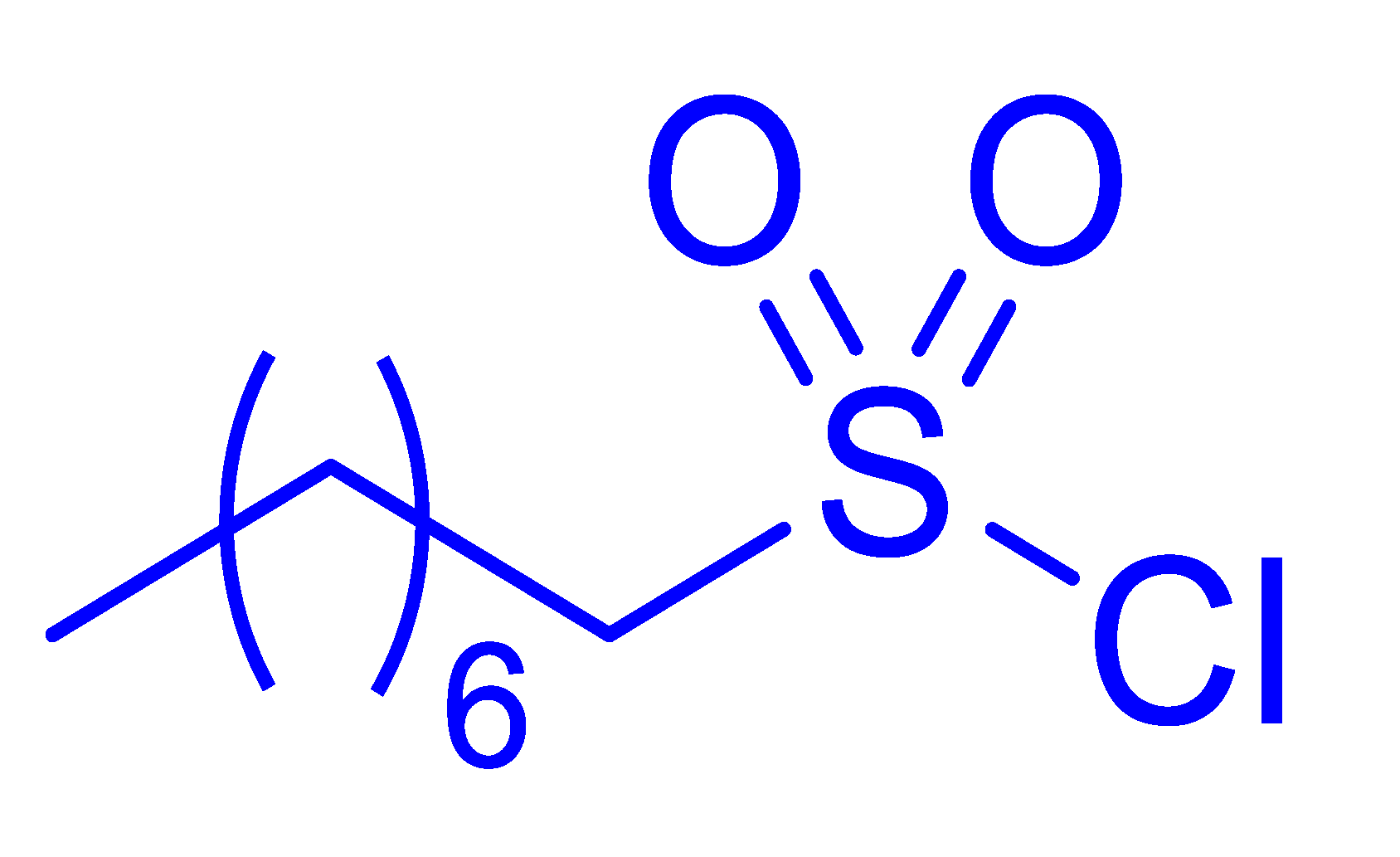 |  | 18 [e] (4.0) |
| 13 |  | 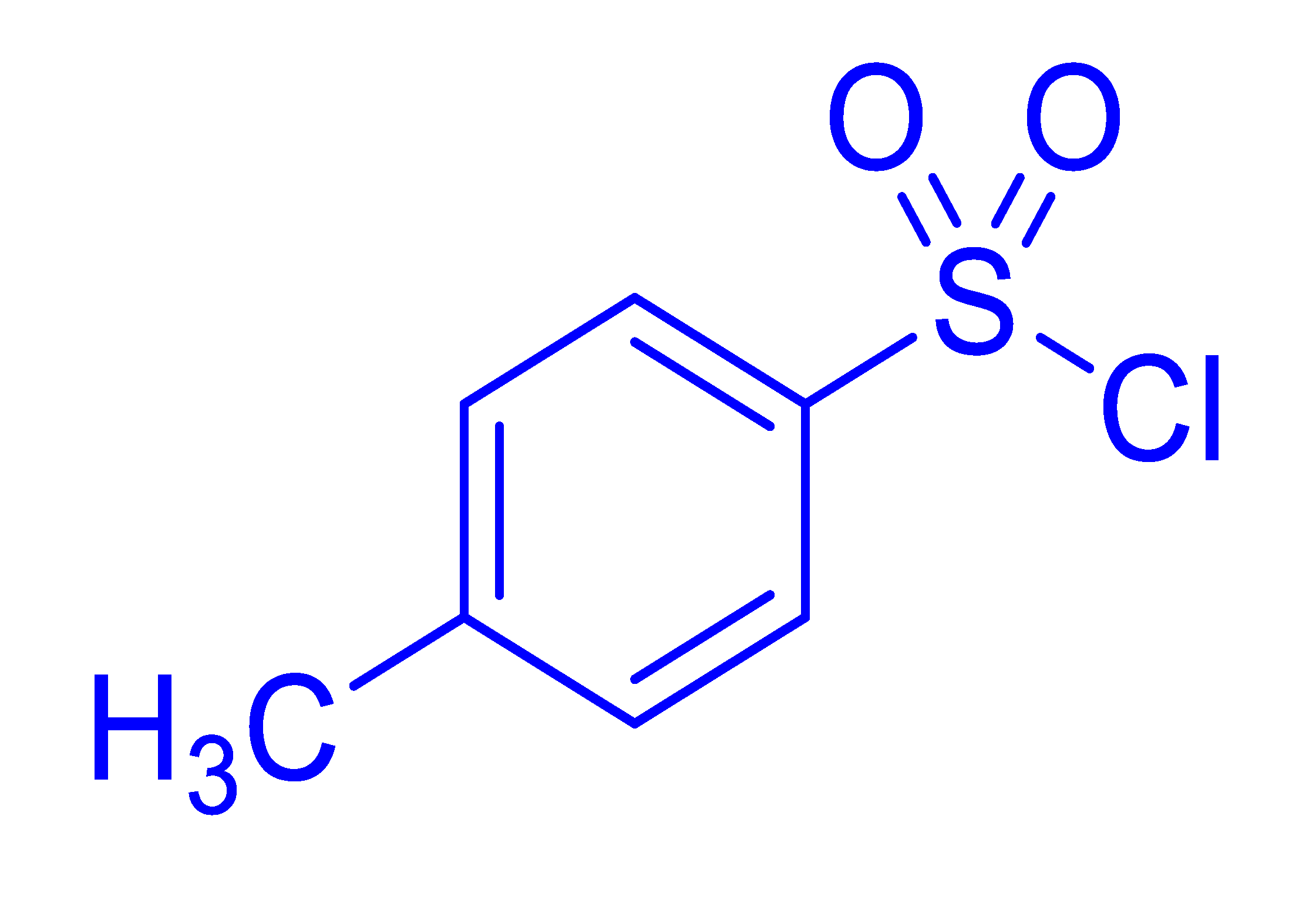 | 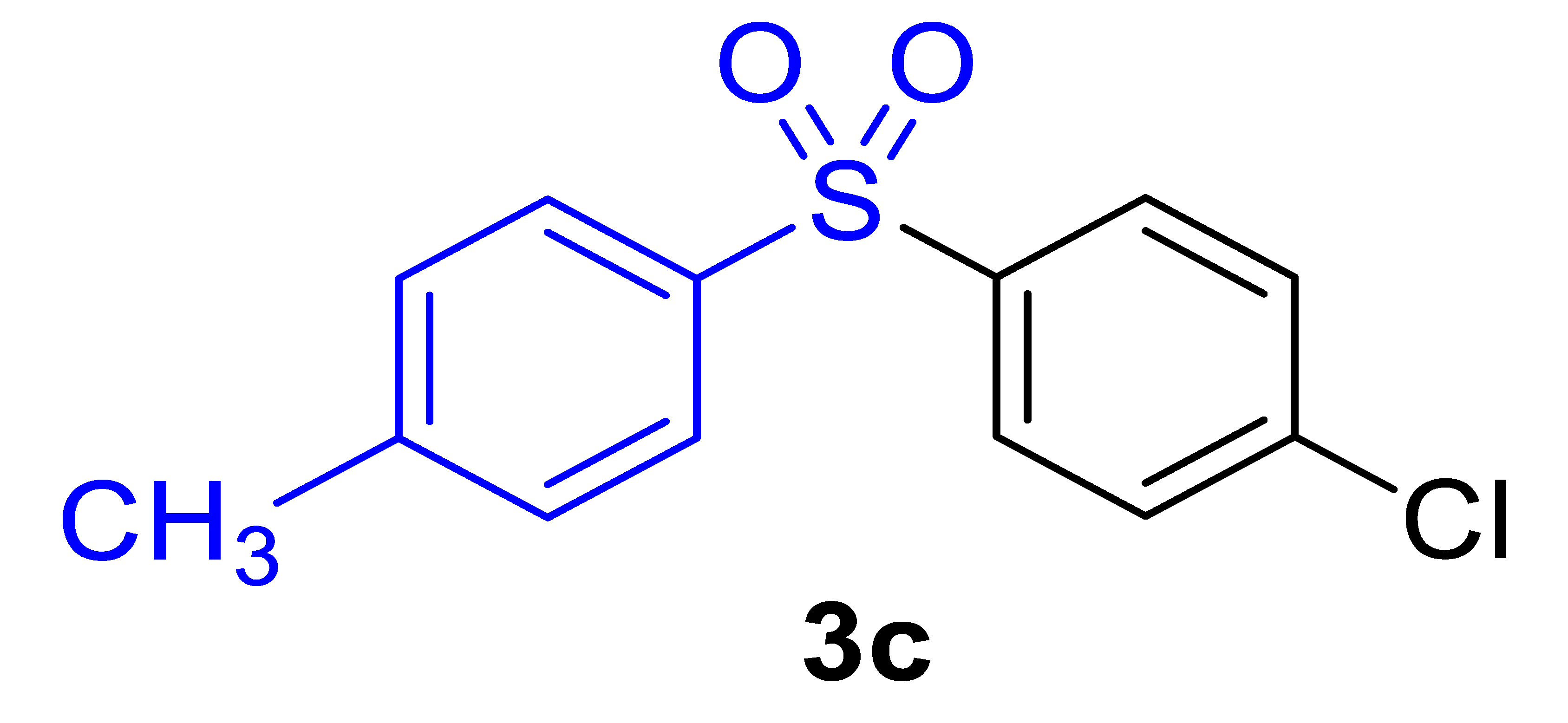 | 29 [e] (4.5) |
| 14 |  |  |  | 61 (2.5) |
| 15 |  | 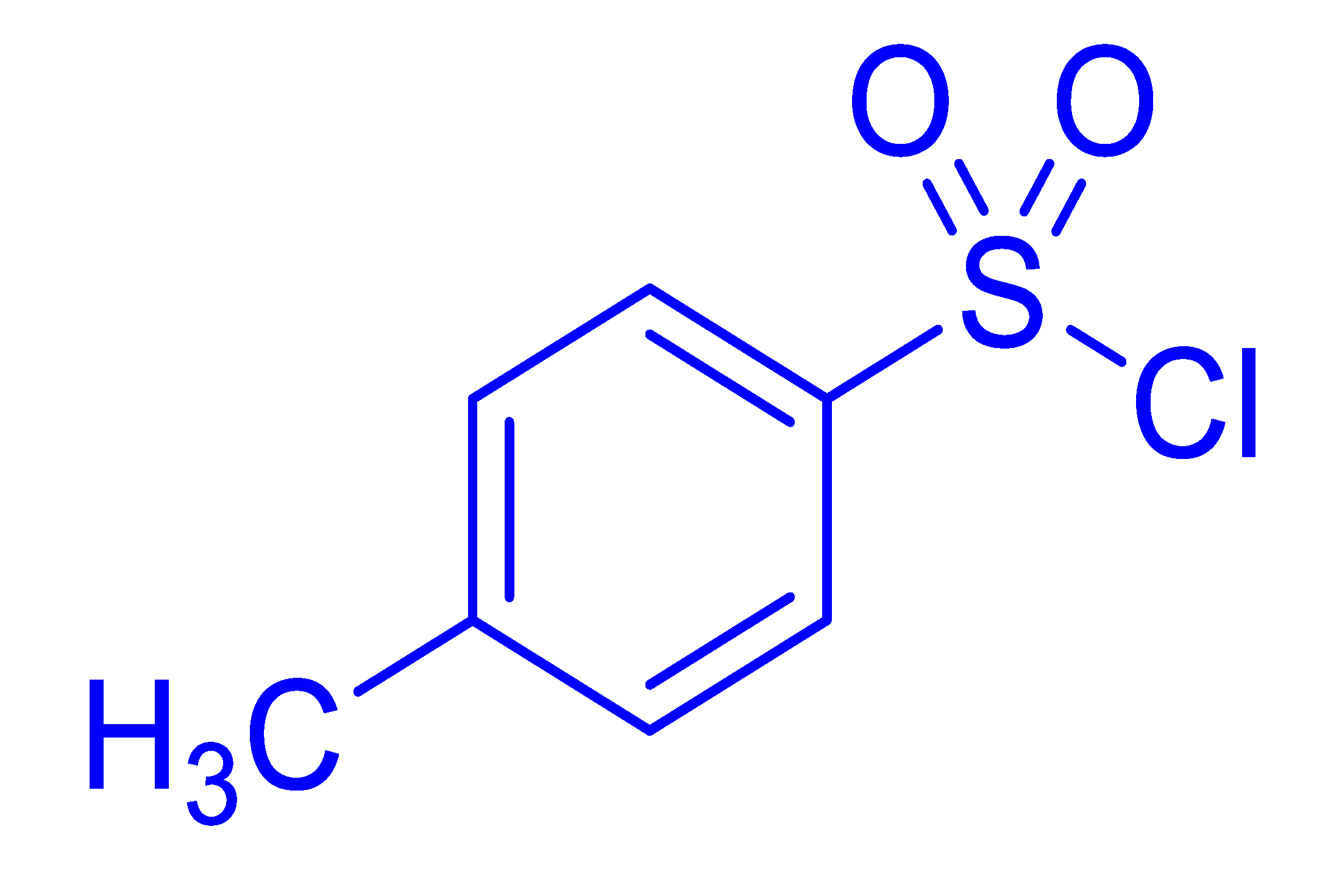 |  | 79 (4.0) |
| 16 |  |  |  | 85 (4.0) |
| 17 b |  |  |  | 90 (3.0) |
| 18 b |  |  |  | 83 (4.0) |
| 19 | 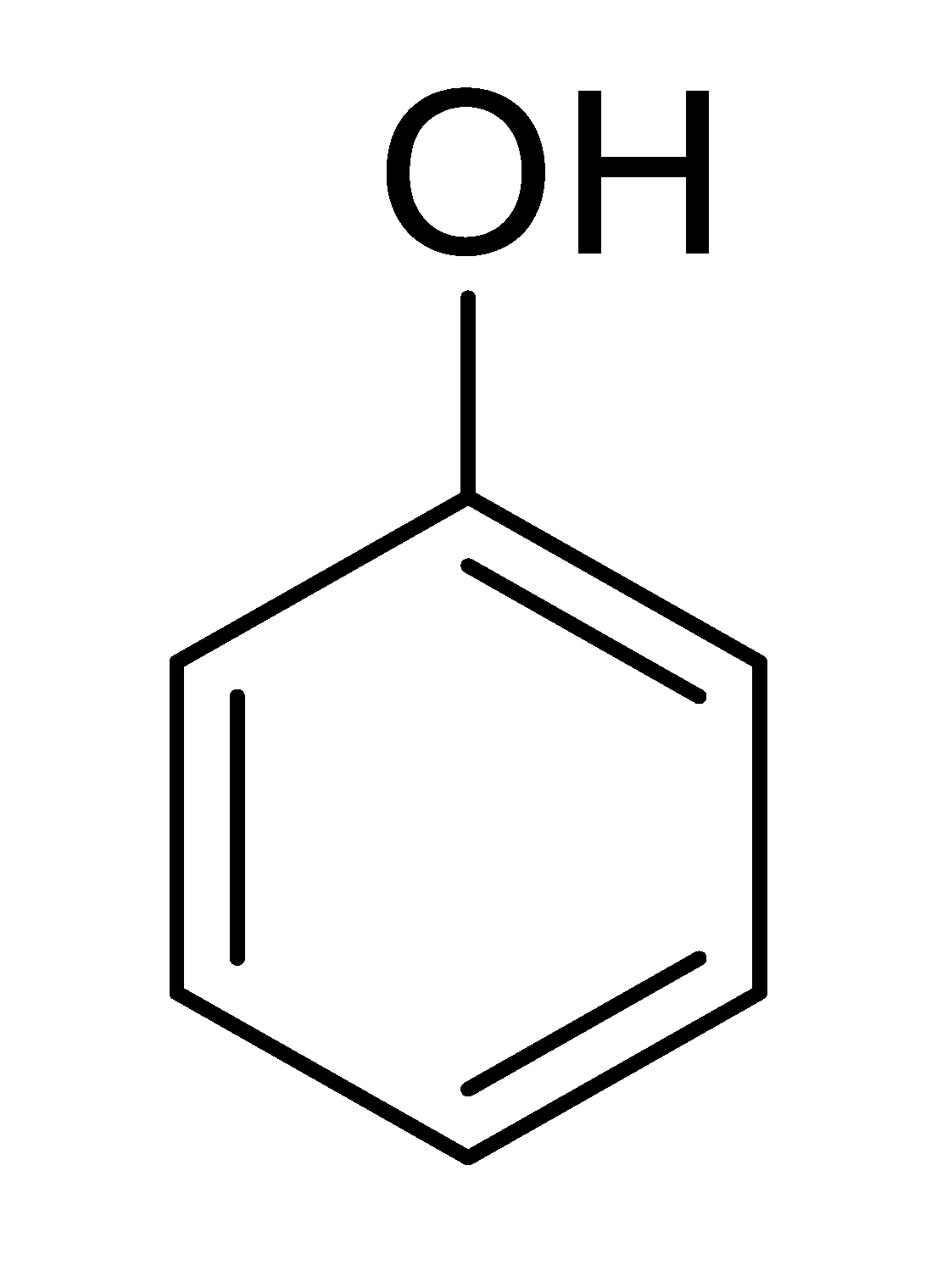 | 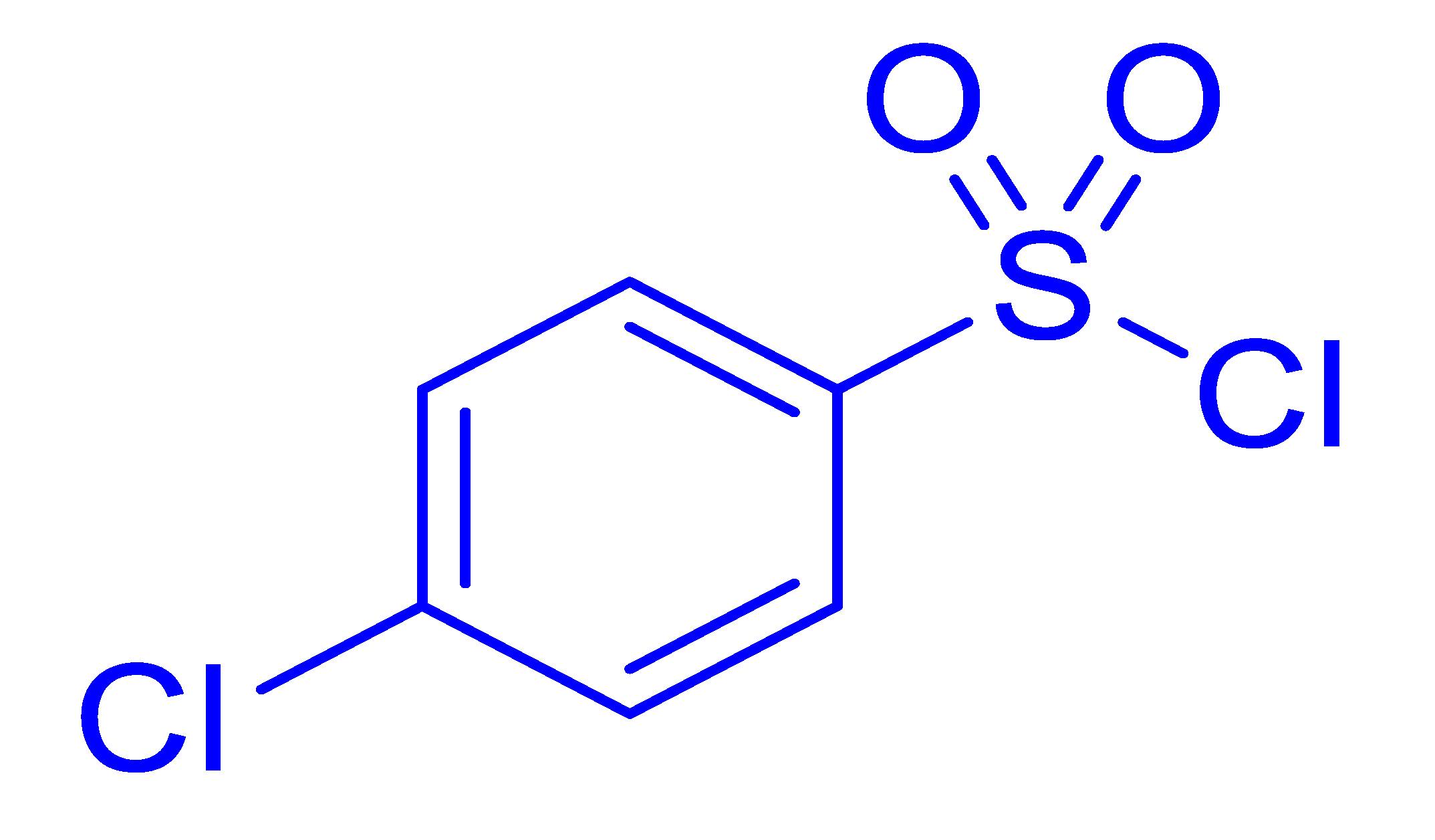 |  | 35 (5.0) |
| 20 c | 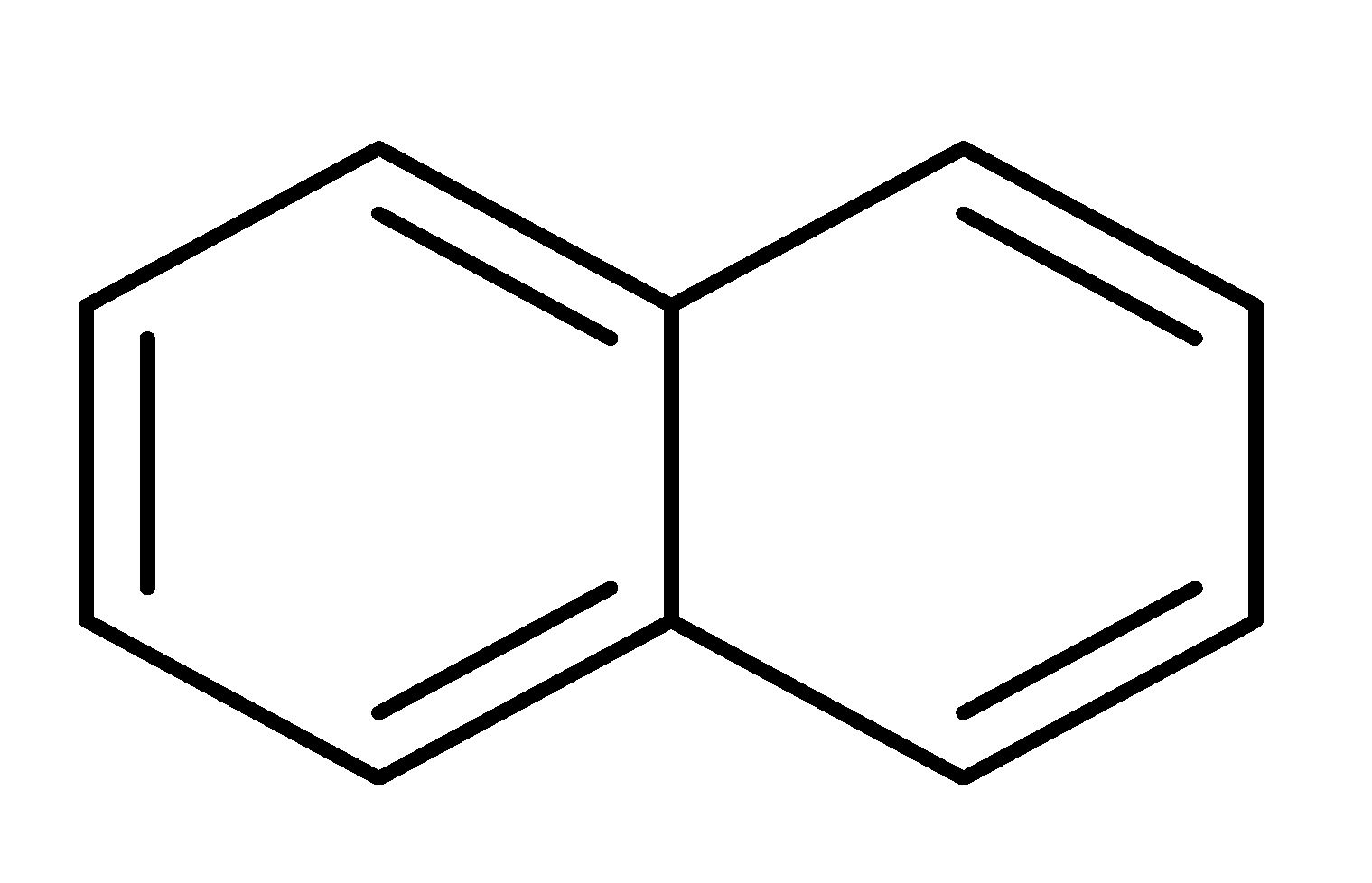 |  | 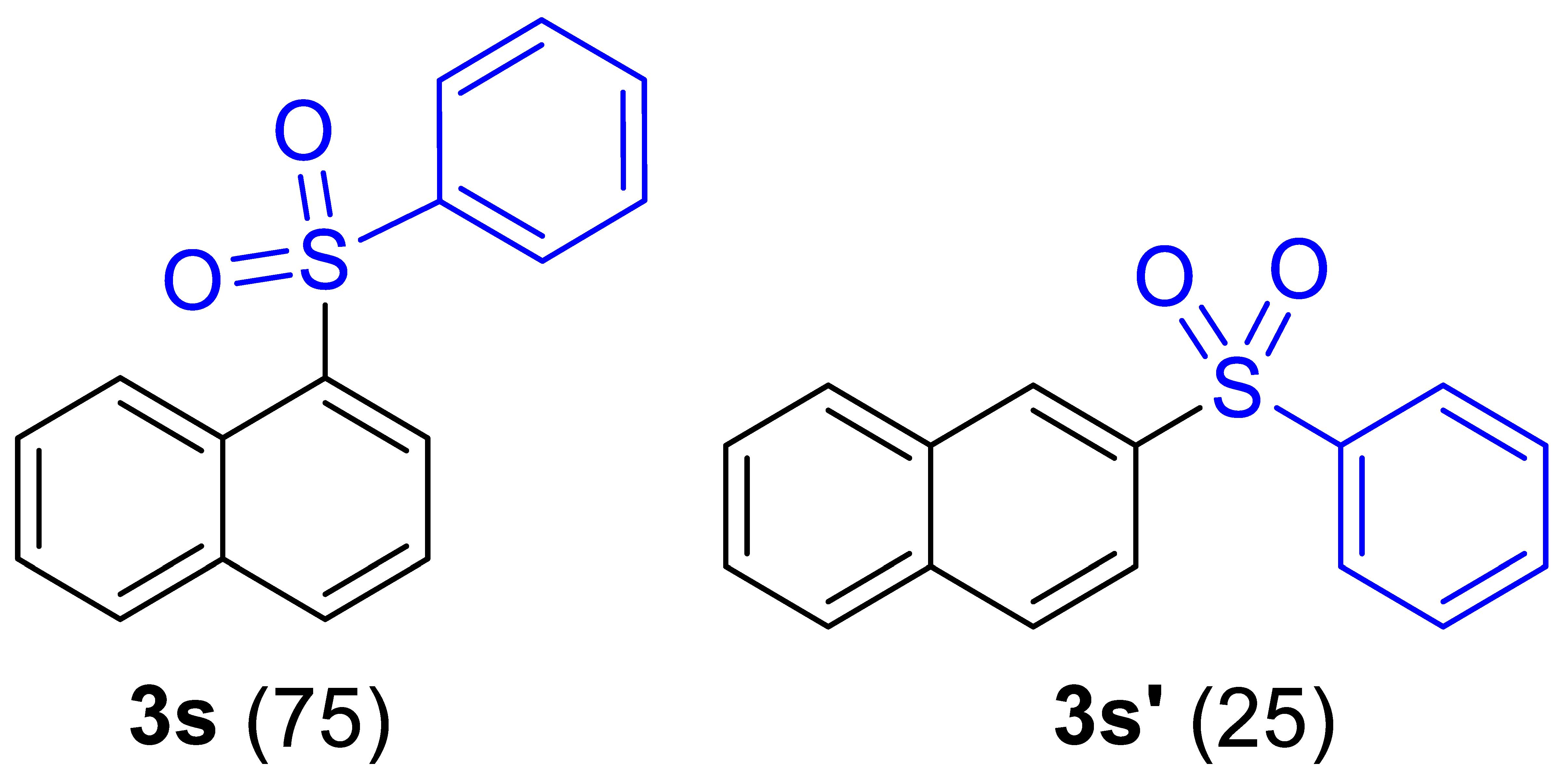 | 71 (6.0) |
| 21 c |  |  |  | 59 (4.0) |
 | |||
|---|---|---|---|
| Entry | Arene | Product | Isolated Yield (%) (Time) c |
| 1 |  |  | 76 (3.0) |
| 2 b |  |  | 73 (1.5) |
| 3 |  |  | 77 (2.0) |
| 4 | 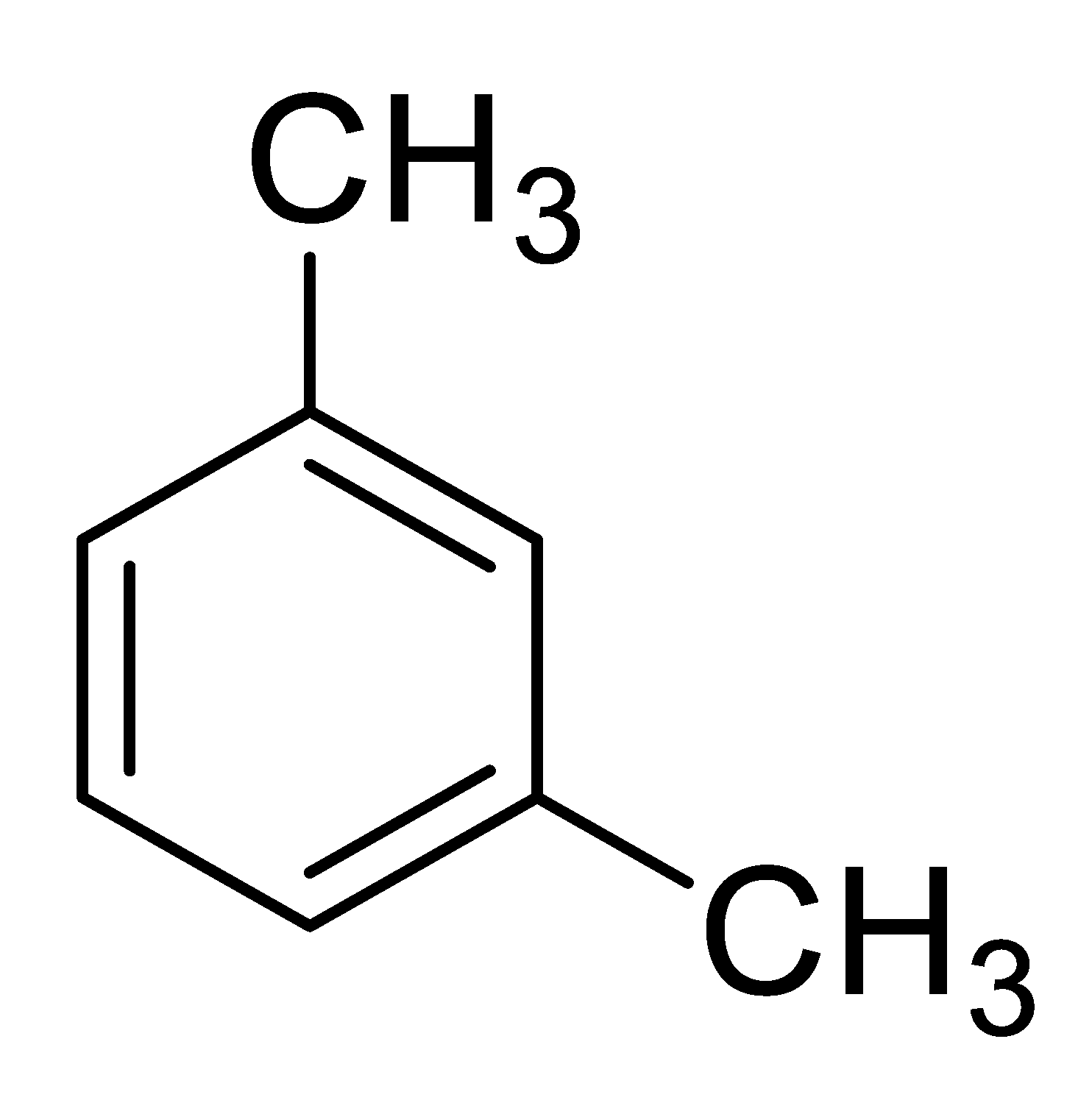 |  | 72 (2.0) |
| 5 |  |  | 77 (3.0) |
| 6 b |  | 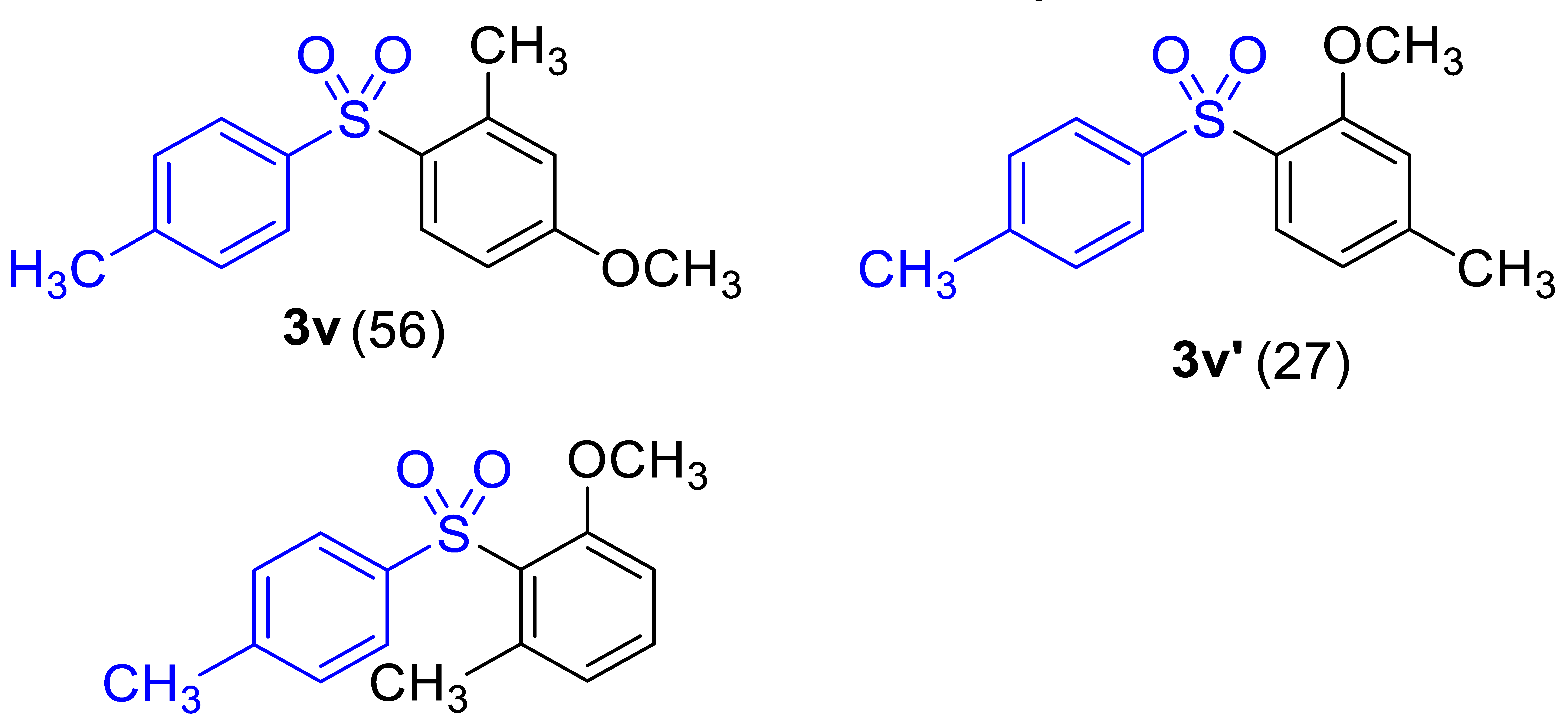 | 82 (2.0) |
| 7 b | 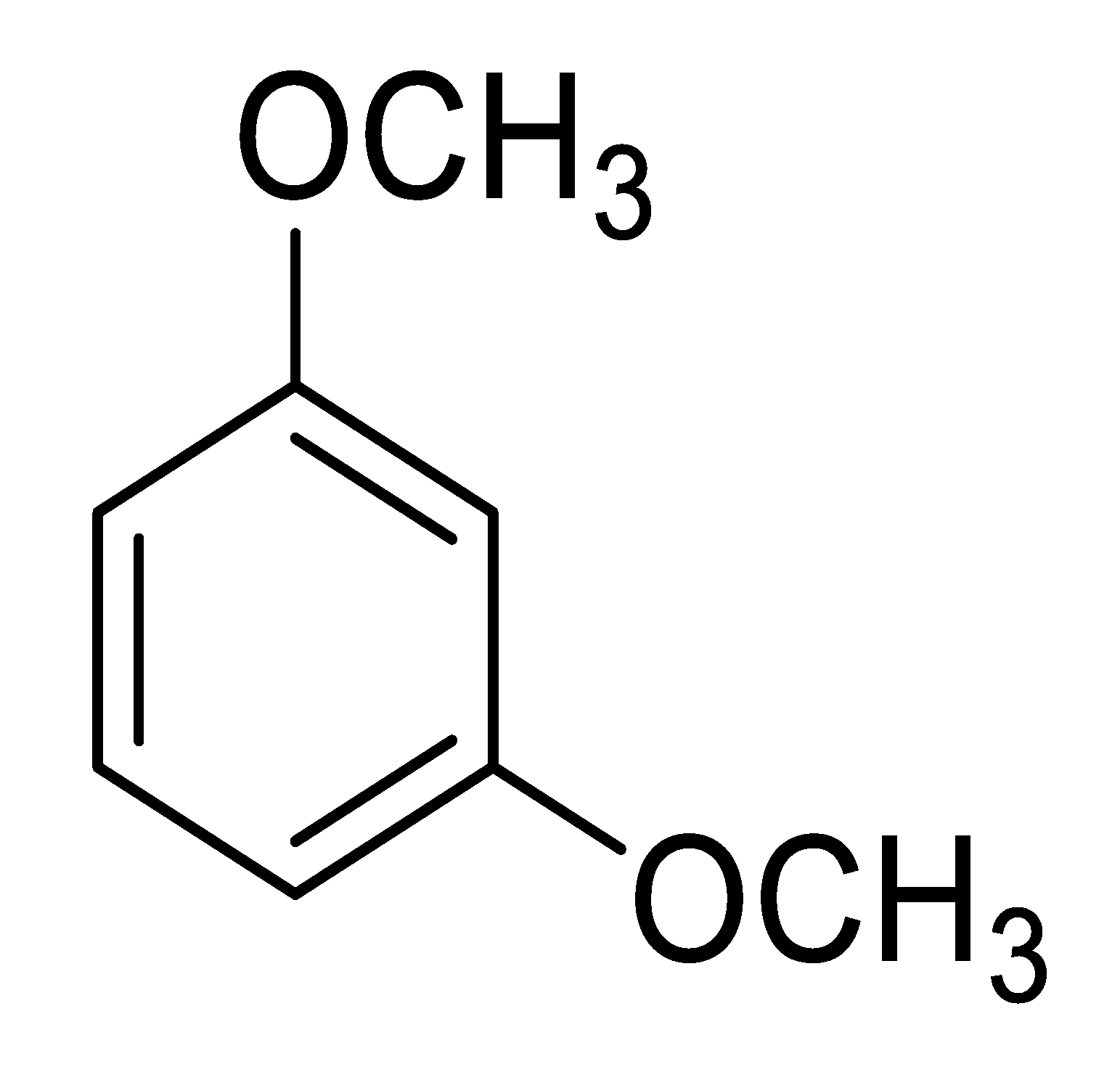 |  | 83 (2.0) |
| 8 b |  |  | 61 (2.5) |
| 9 b |  |  | 81 (2.0) |
 | ||||||||
|---|---|---|---|---|---|---|---|---|
| Catalyst | X | Method | Solvent | Temp a (°C) | Time (h) | Recyclable Times | Yield b (%) | Ref. |
| Indium (0.2 eq) | Cl | Stirring | Dioxane | 100 | 1.5–3.0 | None | 76–84 | [28] |
| Ps-AlCl3 (0.15 eq) | Cl | Stirring | Arene | 85 | 1.1–2.3 | 4 | 89–93 | [35] |
| SiO2-AlCl3 (0.1 eq) | Cl | Stirring | Arene | 85 | 1.0–2.0 | 4 | 91–95 | [35] |
| SiO2-AlCl3 (0.1 eq) | OH | Stirring | None | 80 | 1.3–1.9 | 4 | 88–94 | [36] |
| [BTBA]FeCl3 (1 eq) | Cl | Stirring | None | 60 | 0.02–0.08 | None | 90–97 | [38] |
| MoO2Cl2 (20 mol%) | Cl | Reflux | Arene | 20 | None | 44–89 | [29] | |
| Cu(OTf)2, Sn(OTf)2 (5–10% mol) | Cl | Heating | Arene | 120 | 8–12 | None | 37–98 | [30] |
| Fe(III)-exchanged montmorillonite (0.2 g) | OH | Reflux | Arene | 6–24 | None | 60–63 | [62] | |
| OTs | Reflux | Arene | 6.0 | None | 82–94 | |||
| Nafion-H (50 wt %) | OH | Reflux | Arene | 8–20 | None | 40–82 | [33] | |
| Fe(OH)3 (0.1 g) | Cl | Stirring | Arene | 130–160 | 0.5–3.0 | None | 74–88 | [32] |
| P2O5/Al2O3 (0.67 g) | OH | Reflux | Arene | 1.0 | None | 55–90 | [39] | |
| P2O5/SiO2 (1.2 g) | OH | Reflux | Arene | 0.5–1.8 | None | 50–90 | [40] | |
| Fe3O4@O2Si[PrMIM]Cl·AlCl3 (0.2 g) | Cl | Heating | None | 110 | 1.0–5.0 | 4 | 30–92 | [Our work] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, N.-L.T.; Nguyen, Q.-A.; Le, T.K.; Luu, T.X.T.; Tran, K.-N.T.; Pham, P.-B. Chloroaluminate Ionic Liquid Immobilized on Magnetic Nanoparticles as a Heterogeneous Lewis Acidic Catalyst for the Friedel–Crafts Sulfonylation of Aromatic Compounds. Molecules 2022, 27, 1644. https://doi.org/10.3390/molecules27051644
Nguyen N-LT, Nguyen Q-A, Le TK, Luu TXT, Tran K-NT, Pham P-B. Chloroaluminate Ionic Liquid Immobilized on Magnetic Nanoparticles as a Heterogeneous Lewis Acidic Catalyst for the Friedel–Crafts Sulfonylation of Aromatic Compounds. Molecules. 2022; 27(5):1644. https://doi.org/10.3390/molecules27051644
Chicago/Turabian StyleNguyen, Ngoc-Lan Thi, Quoc-Anh Nguyen, Tien Khoa Le, Thi Xuan Thi Luu, Kim-Ngan Thi Tran, and Phuoc-Bao Pham. 2022. "Chloroaluminate Ionic Liquid Immobilized on Magnetic Nanoparticles as a Heterogeneous Lewis Acidic Catalyst for the Friedel–Crafts Sulfonylation of Aromatic Compounds" Molecules 27, no. 5: 1644. https://doi.org/10.3390/molecules27051644





