Fluoroquinolones’ Biological Activities against Laboratory Microbes and Cancer Cell Lines
Abstract
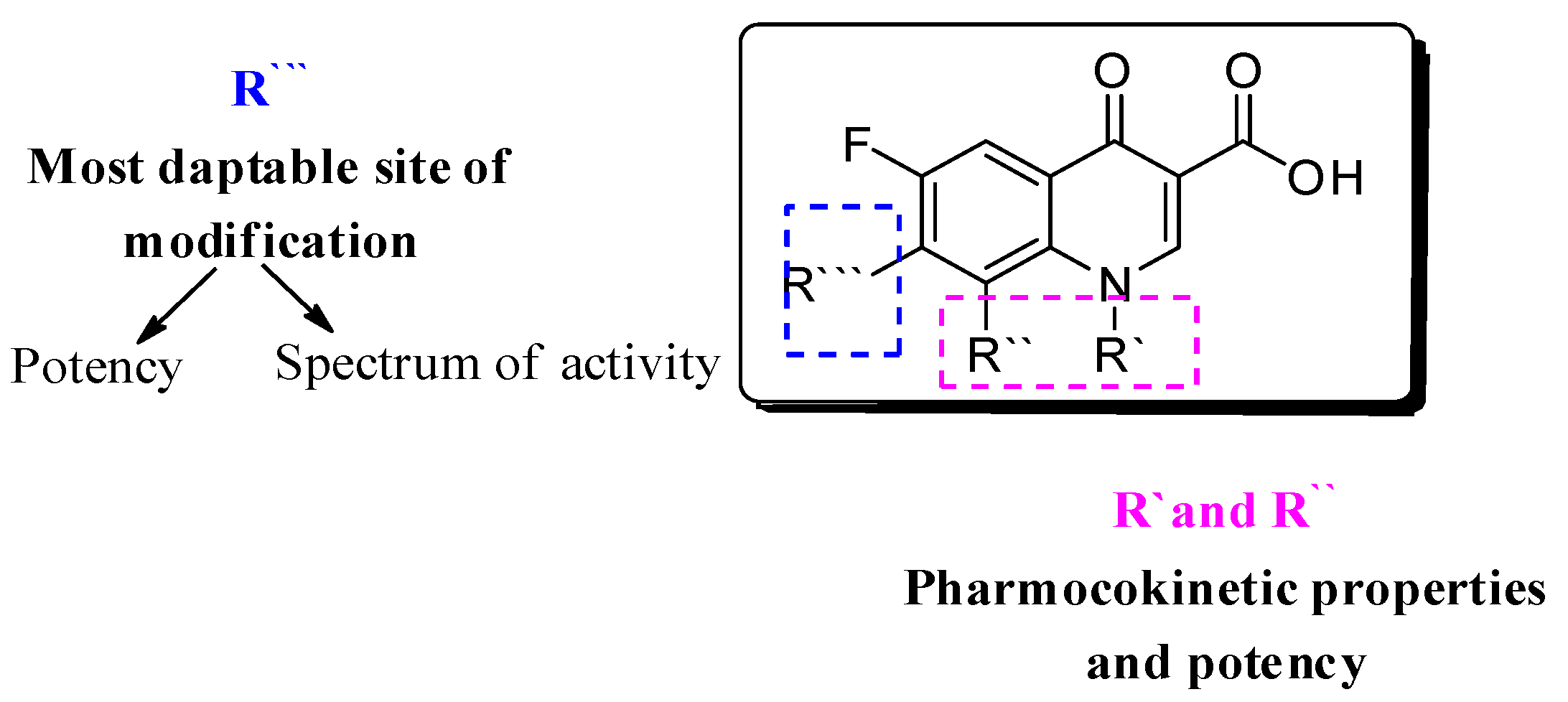
1. Introduction
2. Comparison of the In Vitro Antimicrobial Assays
3. FQ’s Antibacterial Biological Activity
3.1. FQ’s Antibacterial Activity against Gram-Positive Bacteria
3.2. FQs Antibacterial Activity against Gram-Negative Bacteria
3.3. FQs’ Antimycobacterial Activity
3.4. FQs’ Antifungal, Antiparasitic, and Anticancer Activity
3.5. FQs Inhibitory Effect as Anti-Viral Agaents against SARS-CoV-2 and HIV-1
4. Recommendations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Pintilie, L.; Stefaniu, A.; Nicu, A.I.; Maganu, M.; Caproiu, M.T. Design, synthesis and docking studies of some novel fluoroquinolone compounds with antibacterial activity. Synthesis 2018, 665, 636w. [Google Scholar] [CrossRef]
- World Health Organization. The World Health Report: 2004: Changing History; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- Andriole, V.T. The quinolones: Past, present, and future. Clin. Infect. Dis. 2005, 41 (Suppl. S2), S113–S119. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, A.A.; Suaifan, G.A.; Shehadeh, M.B.; Okechukwu, P.N. Design, synthesis, and biological evaluation of 1, 8-naphthyridine glucosamine conjugates as antimicrobial agents. Drug Dev. Res. 2019, 80, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Suaifan, G.A.; Mohammed, A.A.M.; Shehadeh, M.B. 1,8-naphthyridine Glucosamine Derivatives, Their Use in the Treatment of Microbial Infections, and a Method for Preparation 2020-WO202084648. Available online: https://patents.google.com/patent/WO2020084648A1/en?oq=PCT%2fJO2019%2f050010 (accessed on 13 October 2021).
- Suaifan, G.A.; Mohammed, A.A.M.; Shehadeh, M.B. Glycosylated 3-Substituted Fluoroquinolone Derivatives, Preparation Methods Thereof, and Their Use in the Treatment of Antimicrobial Infections 2020, WO2020/202239. Available online: https://patents.google.com/patent/WO2020202239A1/en?oq=WO2020202239A1 (accessed on 13 October 2021).
- Suaifan, G.A.; Mohammed, A.A.M.; Shehadeh, M.B. 1,8-naphthyridine Glucosamine Derivatives, and Uses Thereof for Treating Microbial Infections 2018. Reg. No. P/JO/2018/000097. Available online: https://patentscope.wipo.int/search/ar/detail.jsf?docId=JO320922571&_cid=P10-KZSKQB-93797-1 (accessed on 13 October 2021).
- Ezelarab, H.A.; Abbas, S.H.; Hassan, H.A.; Abuo-Rahma, G.E.D.A. Recent updates of fluoroquinolones as antibacterial agents. Arch. Pharm. 2018, 351, 1800141. [Google Scholar]
- Suaifan, G.A.; Mohammed, A.A. Fluoroquinolones structural and medicinal developments (2013–2018): Where are we now? Bioorg. Med. Chem. 2019, 27, 3005–3060. [Google Scholar] [CrossRef] [PubMed]
- Suaifan, G.A.; Mohammed, A.A. Erratum to “Fluoroquinolones structural and medicinal developments (2013–2018): Where are we now?”. Bioorganic Med. Chem. 2019, 27, 115072. [Google Scholar] [CrossRef]
- Markham, A. Delafloxacin: First global approval. Drugs 2017, 77, 1481–1486. [Google Scholar] [CrossRef]
- Patrick, G.L. An Introduction to Medicinal Chemistry; Oxford University Press: Oxford, UK, 2013. [Google Scholar]
- Idowu, T.; Schweizer, F. Ubiquitous nature of fluoroquinolones: The oscillation between antibacterial and anticancer activities. Antibiotics 2017, 6, 26. [Google Scholar] [CrossRef]
- Dalhoff, A. Antiviral, antifungal, and antiparasitic activities of fluoroquinolones optimized for treatment of bacterial infections: A puzzling paradox or a logical consequence of their mode of action? Eur. J. Clin. Microbiol. Infect. Dis. 2015, 34, 661–668. [Google Scholar] [CrossRef]
- Ozdek, S.C.; Miller, D.; Flynn, P.M.; Flynn, H.W., Jr. In vitro antifungal activity of the fourth generation fluoroquinolones against Candida isolates from human ocular infections. Ocul. Immunol. Inflamm. 2006, 14, 347–351. [Google Scholar] [CrossRef]
- Oliphant, C.M.; Green, G. Quinolones: A comprehensive review. Am. Fam. Physician 2002, 65, 455. [Google Scholar]
- Abidi, M.; Ledeboer, N.; Banerjee, A.; Hari, P. Morbidity and mortality attributable to Rothia bacteremia in neutropenic and nonneutropenic patients. Diagn. Microbiol. Infect. Dis. 2016, 85, 116–120. [Google Scholar] [CrossRef] [PubMed]
- Mitscher, L.A. Bacterial topoisomerase inhibitors: Quinolone and pyridone antibacterial agents. Chem. Rev. 2005, 105, 559–592. [Google Scholar] [CrossRef] [PubMed]
- Zhanel, G.G.; Walkty, A.; Vercaigne, L.; Karlowsky, J.A.; Embil, J.; Gin, A.S.; Hoban, D.J. The new fluoroquinolones: A critical review. Can. J. Infect. Dis. Med. Microbiol. 1999, 10, 207–238. [Google Scholar] [CrossRef] [PubMed]
- Zhanel, G.G.; Ennis, K.; Vercaigne, L.; Walkty, A.; Gin, A.S.; Embil, J.; Smith, H.; Hoban, D.J. A critical review of the fluoroquinolones. Drugs 2002, 62, 13–59. [Google Scholar] [CrossRef] [PubMed]
- Gorityala, B.K.; Guchhait, G.; Goswami, S.; Fernando, D.M.; Kumar, A.; Zhanel, G.G.; Schweizer, F. Hybrid antibiotic overcomes resistance in P. aeruginosa by enhancing outer membrane penetration and reducing efflux. J. Med. Chem. 2016, 59, 8441–8455. [Google Scholar] [CrossRef]
- Jorgensen, S.C.; Mercuro, N.J.; Davis, S.L.; Rybak, M.J. Delafloxacin: Place in therapy and review of microbiologic, clinical and pharmacologic properties. Infect. Dis. Ther. 2018, 7, 197–217. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.A.; Panda, S.S.; Birs, A.S.; Serrano, J.C.; Gonzalez, C.F.; Alamry, K.A.; Katritzky, A.R. Synthesis and antibacterial evaluation of amino acid–antibiotic conjugates. Bioorganic Med. Chem. Lett. 2014, 24, 1856–1861. [Google Scholar] [CrossRef]
- Pardeshi, K.A.; Kumar, T.A.; Ravikumar, G.; Shukla, M.; Kaul, G.; Chopra, S.; Chakrapani, H. Targeted Antibacterial Activity Guided by Bacteria-Specific Nitroreductase Catalytic Activation to Produce Ciprofloxacin. Bioconjugate Chem. 2019, 30, 751–759. [Google Scholar] [CrossRef]
- Abdel-Aziz, M.; Park, S.-E.; Abuo-Rahma, G.E.-D.A.; Sayed, M.A.; Kwon, Y. Novel N-4-piperazinyl-ciprofloxacin-chalcone hybrids: Synthesis, physicochemical properties, anticancer and topoisomerase I and II inhibitory activity. Eur. J. Med. Chem. 2013, 69, 427–438. [Google Scholar] [CrossRef]
- Liu, H.; Huang, J.; Wang, J.; Wang, M.; Liu, M.; Wang, B.; Guo, H.; Lu, Y. Synthesis, antimycobacterial and antibacterial evaluation of l-[(1R, 2S)-2-fluorocyclopropyl] fluoroquinolone derivatives containing an oxime functional moiety. Eur. J. Med. Chem. 2014, 86, 628–638. [Google Scholar] [CrossRef]
- Cilliers, P.; Seldon, R.; Smit, F.J.; Aucamp, J.; Jordaan, A.; Warner, D.F.; N’Da, D.D. Design, synthesis, and antimycobacterial activity of novel ciprofloxacin derivatives. Chem. Biol. Drug Des. 2019, 94, 1518–1536. [Google Scholar] [CrossRef] [PubMed]
- Türe, A.; Kulabaş, N.; Dingiş, S.İ.; Birgül, K.; Bozdeveci, A.; Karaoğlu, Ş.A.; Krishna, V.S.; Sriram, D.; Küçükgüzel, İ. Design, synthesis and molecular modeling studies on novel moxifloxacin derivatives as potential antibacterial and antituberculosis agents. Bioorganic Chem. 2019, 88, 102965. [Google Scholar] [CrossRef] [PubMed]
- Wayne, P. Clinical and Laboratory Standards Institute (CLSI) performance standards for antimicrobial disk diffusion susceptibility tests 19th ed. approved standard. CLSI Doc. M100-S19 2009, 29, M100-S21. [Google Scholar]
- Pfaller, M.; Sheehan, D.; Rex, J. Determination of fungicidal activities against yeasts and molds: Lessons learned from bactericidal testing and the need for standardization. Clin. Microbiol. Rev. 2004, 17, 268–280. [Google Scholar] [CrossRef]
- Balouiri, M.; Sadiki, M.; Ibnsouda, S.K. Methods for in vitro evaluating antimicrobial activity: A review. J. Pharm. Anal. 2016, 6, 71–79. [Google Scholar] [CrossRef]
- Khan, Z.A.; Siddiqui, M.F.; Park, S. Current and emerging methods of antibiotic susceptibility testing. Diagnostics 2019, 9, 49. [Google Scholar] [CrossRef]
- SLCI Performance Standards for Antimicrobial Disk Susceptibility Tests. Available online: https://clsi.org/standards/products/microbiology/documents/m02/ (accessed on 30 March 2020).
- Xiao, Z.-P.; Wang, X.-D.; Wang, P.-F.; Zhou, Y.; Zhang, J.-W.; Zhang, L.; Zhou, J.; Zhou, S.-S.; Ouyang, H.; Lin, X.-Y. Design, synthesis, and evaluation of novel fluoroquinolone–flavonoid hybrids as potent antibiotics against drug-resistant microorganisms. Eur. J. Med. Chem. 2014, 80, 92–100. [Google Scholar] [CrossRef]
- Towle, T.R.; Kulkarni, C.A.; Oppegard, L.M.; Williams, B.P.; Picha, T.A.; Hiasa, H.; Kerns, R.J. Design, synthesis, and evaluation of novel N-1 fluoroquinolone derivatives: Probing for binding contact with the active site tyrosine of gyrase. Bioorganic Med. Chem. Lett. 2018, 28, 1903–1910. [Google Scholar] [CrossRef]
- Jorgensen, J.H.; Turnidge, J.D. Susceptibility test methods: Dilution and disk diffusion methods. Man. Clin. Microbiol. 2015, 1253–1273. [Google Scholar]
- Korgenski, E.K.; Daly, J.A. Evaluation of the BIOMIC video reader system for determining interpretive categories of isolates on the basis of disk diffusion susceptibility results. J. Clin. Microbiol. 1998, 36, 302–304. [Google Scholar] [CrossRef]
- Benkova, M.; Soukup, O.; Marek, J. Antimicrobial susceptibility testing: Currently used methods and devices and the near future in clinical practice. J. Appl. Microbiol. 2020, 129, 806–822. [Google Scholar] [CrossRef] [PubMed]
- Le Page, S.; van Belkum, A.; Fulchiron, C.; Huguet, R.; Raoult, D.; Rolain, J.-M. Evaluation of the PREVI® Isola automated seeder system compared to reference manual inoculation for antibiotic susceptibility testing by the disk diffusion method. Eur. J. Clin. Microbiol. Infect. Dis. 2015, 34, 1859–1869. [Google Scholar] [CrossRef] [PubMed]
- Hada, D.; Chauhan, N.P.S. 11. Antimicrobial testing methods. In Biocidal Polymers; De Gruyter: Berlin, Boston, 2019; pp. 241–262. [Google Scholar]
- Bruin, J.P.; Diederen, B.M.; IJzerman, E.P.; Den Boer, J.W.; Mouton, J.W. Correlation of MIC value and disk inhibition zone diameters in clinical Legionella pneumophila serogroup 1 isolates. Diagn. Microbiol. Infect. Dis. 2013, 76, 339–342. [Google Scholar] [CrossRef]
- Sharma, P.C.; Kumar, R.; Chaudhary, M.; Sharma, A.; Rajak, H. Synthesis and biological evaluation of novel benzothiazole clubbed fluoroquinolone derivatives. J. Enzym. Inhib. Med. Chem. 2013, 28, 1–10. [Google Scholar] [CrossRef]
- Patel, M.M.; Patel, L.J. Design, synthesis, molecular docking, and antibacterial evaluation of some novel flouroquinolone derivatives as potent antibacterial agent. Sci. World J. 2014, 2014, 10. [Google Scholar] [CrossRef]
- Chugunova, E.; Akylbekov, N.; Bulatova, A.; Gavrilov, N.; Voloshina, A.; Kulik, N.; Zobov, V.; Dobrynin, A.; Syakaev, V.; Burilov, A. Synthesis and biological evaluation of novel structural hybrids of benzofuroxan derivatives and fluoroquinolones. Eur. J. Med. Chem. 2016, 116, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Leyva-Ramos, S.; de Loera, D.; Cardoso-Ortiz, J. In vitro antibacterial activity of 7-substituted-6-fluoroquinolone and 7-substituted-6, 8-difluoroquinolone derivatives. Chemotherapy 2017, 62, 194–198. [Google Scholar] [CrossRef]
- Mentese, M.; Demirbas, N.; Mermer, A.; Demirci, S.; Demirbas, A.; Ayaz, F.A. Novel azole-functionalited flouroquinolone hybrids: Design, conventional and microwave irradiated synthesis, evaluation as antibacterial and antioxidant agents. Lett. Drug Des. Discov. 2018, 15, 46–64. [Google Scholar] [CrossRef]
- Seliem, I.A.; Panda, S.S.; Girgis, A.S.; Nagy, Y.I.; George, R.F.; Fayad, W.; Fawzy, N.G.; Ibrahim, T.S.; Al-Mahmoudy, A.M.; Sakhuja, R. Design, synthesis, antimicrobial, and DNA gyrase inhibitory properties of fluoroquinolone–dichloroacetic acid hybrids. Chem. Biol. Drug Des. 2020, 95, 248–259. [Google Scholar] [CrossRef]
- Chrzanowska, A.; Roszkowski, P.; Bielenica, A.; Olejarz, W.; Stępień, K.; Struga, M. Anticancer and antimicrobial effects of novel ciprofloxacin fatty acids conjugates. Eur. J. Med. Chem. 2020, 185, 111810. [Google Scholar] [CrossRef]
- Mokaber-Esfahani, M.; Eshghi, H.; Akbarzadeh, M.; Gholizadeh, M.; Mirzaie, Y.; Hakimi, M.; Lari, J. Synthesis and Antibacterial Evaluation of New Pyrimidyl N-Ciprofloxacin Derivatives. ChemistrySelect 2019, 4, 8930–8933. [Google Scholar] [CrossRef]
- Garza, I.; Wallace, M.J.; Fernando, D.; Singh, A.; Lee, R.E.; Gerding, J.S.; Franklin, C.; Yendapally, R. Synthesis and Evaluation of Thiazolidine Amide and N-Thiazolyl Amide Fluoroquinolone Derivatives. Arch. Der. Pharm. 2017, 350, e201700029. [Google Scholar] [CrossRef]
- Gao, Y.; Na, L.X.; Xu, Z.; Zhang, S.; Wang, A.P.; Lü, K.; Guo, H.Y.; Liu, M.L. Design, Synthesis and Antibacterial Evaluation of 1-[(1R, 2S)-2-Fluorocyclopropyl] ciprofloxacin-1, 2, 4-triazole-5 (4H)-thione Hybrids. Chem. Biodivers. 2018, 15, e1800261. [Google Scholar] [CrossRef]
- Gao, L.-Z.; Xie, Y.-S.; Li, T.; Huang, W.-L.; Hu, G.-Q. Synthesis and antibacterial activity of novel [1, 2, 4] triazolo [3, 4-h][1, 8] naphthyridine-7-carboxylic acid derivatives. Chin. Chem. Lett. 2015, 26, 149–151. [Google Scholar] [CrossRef]
- Bykowska, A.; Starosta, R.; Jezierska, J.; Jeżowska-Bojczuk, M. Coordination versatility of phosphine derivatives of fluoroquinolones. New Cu I and Cu II complexes and their interactions with DNA. RSC Adv. 2015, 5, 80804–80815. [Google Scholar] [CrossRef]
- Zhang, T.; Shen, W.; Liu, M.; Zhang, R.; Wang, M.; Li, L.; Wang, B.; Guo, H.; Lu, Y. Synthesis, antimycobacterial and antibacterial activity of fluoroquinolone derivatives containing an 3-alkoxyimino-4-(cyclopropylanimo) methylpyrrolidine moiety. Eur. J. Med. Chem. 2015, 104, 73–85. [Google Scholar] [CrossRef]
- Xu, Z.; Zhang, S.; Feng, L.-S.; Li, X.-N.; Huang, G.-C.; Chai, Y.; Lv, Z.-S.; Guo, H.-Y.; Liu, M.-L. Synthesis and in vitro antimycobacterial and antibacterial activity of 8-OMe ciprofloxacin-hydrozone/azole hybrids. Molecules 2017, 22, 1171. [Google Scholar] [CrossRef]
- Gorityala, B.K.; Guchhait, G.; Fernando, D.M.; Deo, S.; McKenna, S.A.; Zhanel, G.G.; Kumar, A.; Schweizer, F. Adjuvants based on hybrid antibiotics overcome resistance in Pseudomonas aeruginosa and enhance fluoroquinolone efficacy. Angew. Chem. Int. Ed. 2016, 55, 555–559. [Google Scholar] [CrossRef]
- Plech, T.; Wujec, M.; Kosikowska, U.; Malm, A.; Rajtar, B.; Polz-Dacewicz, M. Synthesis and in vitro activity of 1, 2, 4-triazole-ciprofloxacin hybrids against drug-susceptible and drug-resistant bacteria. Eur. J. Med. Chem. 2013, 60, 128–134. [Google Scholar] [CrossRef]
- Ji, C.; Miller, P.A.; Miller, M.J. Syntheses and antibacterial activity of N-acylated ciprofloxacin derivatives based on the trimethyl lock. ACS Med. Chem. Lett. 2015, 6, 707–710. [Google Scholar] [CrossRef]
- Kassab, A.E.; Gedawy, E.M. Novel ciprofloxacin hybrids using biology oriented drug synthesis (BIODS) approach: Anticancer activity, effects on cell cycle profile, caspase-3 mediated apoptosis, topoisomerase II inhibition, and antibacterial activity. Eur. J. Med. Chem. 2018, 150, 403–418. [Google Scholar] [CrossRef]
- Rajulu, G.G.; Naik, H.S.B.; Abhilash, V.; Thiruvengadam, J.; Rajesh, K.; Ganesh, S.; Jagadheshan, H.; Kesavan, P.K. New hydroxamic acid derivatives of fluoroquinolones: Synthesis and evaluation of antibacterial and anticancer properties. Chem. Pharm. Bull. 2014, 62, 168–175. [Google Scholar] [CrossRef][Green Version]
- Rajulu, G.G.; Naik, H.S.B.; Kumar, G.C.; Ramaraj, S.; Sambasivam, G.; Koppolu, K.P. New azetidine-3-carbonyl-N-methyl-hydrazino derivatives of fluoroquinolones: Synthesis and evaluation of antibacterial and anticancer properties. Med. Chem. Res. 2014, 23, 2856–2868. [Google Scholar] [CrossRef]
- Mermer, A.; Faiz, O.; Demirbas, A.; Demirbas, N.; Alagumuthu, M.; Arumugam, S. Piperazine-azole-fluoroquinolone hybrids: Conventional and microwave irradiated synthesis, biological activity screening and molecular docking studies. Bioorganic Chem. 2019, 85, 308–318. [Google Scholar] [CrossRef] [PubMed]
- Vu, T.H.; Ha-Duong, N.-T.; Aubry, A.; Capton, E.; Fechter, P.; Plesiat, P.; Verbeke, P.; Serradji, N. In vitro activities of a new fluoroquinolone derivative highly active against Chlamydia trachomatis. Bioorganic Chem. 2019, 83, 180–185. [Google Scholar] [CrossRef]
- Marquez, B.; Pourcelle, V.; Vallet, C.M.; Mingeot-Leclercq, M.-P.; Tulkens, P.M.; Marchand-Bruynaert, J.; Van Bambeke, F. Pharmacological characterization of 7-(4-(piperazin-1-yl)) ciprofloxacin derivatives: Antibacterial activity, cellular accumulation, susceptibility to efflux transporters, and intracellular activity. Pharm. Res. 2014, 31, 1290–1301. [Google Scholar] [CrossRef]
- Mohammed, H.H.; Abbas, S.H.; Abdelhafez, E.-S.M.; Berger, J.M.; Mitarai, S.; Arai, M.; Abuo-Rahma, G.E.-D.A. Synthesis, molecular docking, antimicrobial evaluation, and DNA cleavage assay of new thiadiazole/oxadiazole ciprofloxacin derivatives. Mon. Für Chem.-Chem. Mon. 2019, 150, 1809–1824. [Google Scholar] [CrossRef]
- Szczupak, Ł.; Kowalczyk, A.; Trzybiński, D.; Woźniak, K.; Mendoza, G.; Arruebo, M.; Steverding, D.; Stączek, P.; Kowalski, K. Organometallic ciprofloxacin conjugates with dual action: Synthesis, characterization, and antimicrobial and cytotoxicity studies. Dalton Trans. 2020, 49, 1403–1415. [Google Scholar] [CrossRef]
- Li, X.; Zhang, Y.-K.; Plattner, J.J.; Mao, W.; Alley, M.; Xia, Y.; Hernandez, V.; Zhou, Y.; Ding, C.Z.; Li, J. Synthesis and antibacterial evaluation of a novel tricyclic oxaborole-fused fluoroquinolone. Bioorganic Med. Chem. Lett. 2013, 23, 963–966. [Google Scholar] [CrossRef]
- Huang, J.; Wang, M.; Wang, B.; Wu, Z.; Liu, M.; Feng, L.; Zhang, J.; Li, X.; Yang, Y.; Lu, Y. Synthesis, antimycobacterial and antibacterial activity of 1-(6-amino-3, 5-difluoropyridin-2-yl) fluoroquinolone derivatives containing an oxime functional moiety. Bioorganic Med. Chem. Lett. 2016, 26, 2262–2267. [Google Scholar] [CrossRef]
- Zhang, T.; Wu, J.; Chen, S.; Liu, K.; Lin, Y.; Guo, H.; Liu, M. Synthesis and antibacterial activity of amino acid and dipeptide prodrugs of IMB-070593, a fluoroquinolone candidate. Molecules 2014, 19, 6822–6837. [Google Scholar] [CrossRef] [PubMed]
- Salunke, R.A.; Shukla, M.; Kaul, G.; Bansal, B.R.; Chopra, S.; Chhibber, M. New fluoroquinolone compounds with endo-nortropine derivatives at C-7 position show antibacterial activity against fluoroquinolone-resistant strains of Staphylococcus aureus. Chem. Biol. Drug Des. 2019, 94, 1626–1633. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Xing, J.; Cheng, H.; Wang, H.; Wang, J.; Wang, S.; Zhou, J.; Zhang, H. Design, Synthesis, Antibacterial Evaluation and Docking Study of Novel 2-Hydroxy-3-(nitroimidazolyl)-propyl-derived Quinolone. Chem. Biol. Drug Des. 2015, 85, 79–90. [Google Scholar] [CrossRef]
- Abu-Sini, M.; Mayyas, A.; Al-Karablieh, N.; Darwish, R.; Al-Hiari, Y.; Aburjai, T.; Arabiyat, S.; Abu-Qatouseh, L. Synthesis of 1, 2, 3-triazolo [4, 5-h] quinolone derivatives with novel anti-microbial properties against Metronidazole resistant Helicobacter pylori. Molecules 2017, 22, 841. [Google Scholar] [CrossRef]
- El-Megharbel, S.; Adam, A.; Megahed, A.; Refat, M. Synthesis and molecular structure of moxifloxacin drug with metal ions as a model drug against some kinds of bacteria and fungi. Russ. J. Gen. Chem. 2015, 85, 2366–2373. [Google Scholar] [CrossRef]
- Long, T.E.; Keding, L.C.; Lewis, D.D.; Anstead, M.I.; Withers, T.R.; Hongwei, D.Y. Anionic fluoroquinolones as antibacterials against biofilm-producing Pseudomonas aeruginosa. Bioorganic Med. Chem. Lett. 2016, 26, 1305–1309. [Google Scholar] [CrossRef]
- Charushin, V.N.; Mochulskaya, N.N.; Antipin, F.V.; Kotovskaya, S.K.; Nosova, E.V.; Ezhikova, M.A.; Kodess, M.I.; Kravchenko, M.A. Synthesis and antimycobacterial evaluation of new (2-oxo-2H-chromen-3-yl) substituted fluoroquinolones. J. Fluor. Chem. 2018, 208, 15–23. [Google Scholar] [CrossRef]
- Ross, A.G.; Benton, B.M.; Chin, D.; De Pascale, G.; Fuller, J.; Leeds, J.A.; Reck, F.; Richie, D.L.; Vo, J.; LaMarche, M.J. Synthesis of ciprofloxacin dimers for evaluation of bacterial permeability in atypical chemical space. Bioorganic Med. Chem. Lett. 2015, 25, 3468–3475. [Google Scholar] [CrossRef]
- Bartzatt, R.; Cirillo, S.L.; Cirillo, J.D. Antibacterial derivatives of ciprofloxacin to inhibit growth of necrotizing fasciitis associated penicillin resistant Escherichia coli. J. Pharm. 2013, 2013, 517638. [Google Scholar] [CrossRef]
- AA Abdel-Aal, M.; Abdel-Aziz, S.A.; Shaykoon, M.S.A.; Mohamed, M.F.; Abuo-Rahma, G.E.-D.A. Antibacterial and Urease Inhibitory activity of New Piperazinyl N-4 Functionalized Ciprofloxacin-oxadiazoles. J. Mod. Res. 2019, 1, 1–7. [Google Scholar] [CrossRef][Green Version]
- Fardeau, S.; Dassonville-Klimpt, A.; Audic, N.; Sasaki, A.; Pillon, M.; Baudrin, E.; Mullié, C.; Sonnet, P. Synthesis and antibacterial activity of catecholate–ciprofloxacin conjugates. Bioorganic Med. Chem. 2014, 22, 4049–4060. [Google Scholar] [CrossRef]
- Riahifard, N.; Tavakoli, K.; Yamaki, J.; Parang, K.; Tiwari, R. Synthesis and evaluation of antimicrobial activity of [r4w4k]-levofloxacin and [r4w4k]-levofloxacin-q conjugates. Molecules 2017, 22, 957. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Zhang, H.; Ma, T.; Xue, H.; Miao, Z.; Chen, L.; Shi, X. Ciprofloxacin-1, 2, 3-triazole-isatin hybrids tethered via amide: Design, synthesis, and in vitro anti-mycobacterial activity evaluation. Bioorg. Med. Chem. Lett. 2019, 29, 2635–2637. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Lv, Z.; Wen, J.; Zhao, S.; Xu, Z. Synthesis and in vitro evaluation of novel substituted isatin-propylene-1H-1, 2, 3-triazole-4-methylene-moxifloxacin hybrids for their anti-mycobacterial activities. Eur. J. Med. Chem. 2018, 143, 899–904. [Google Scholar] [CrossRef]
- Scroggs, S.L.; Offerdahl, D.K.; Flather, D.P.; Morris, C.N.; Kendall, B.L.; Broeckel, R.M.; Beare, P.A.; Bloom, M.E. Fluoroquinolone antibiotics exhibit low antiviral activity against SARS-CoV-2 and MERS-CoV. Viruses 2021, 13, 8. [Google Scholar] [CrossRef]
- Sarma, P. Norfloxacin: A new drug in the treatment of falciparum malaria. Ann. Intern. Med. 1989, 111, 336–337. [Google Scholar] [CrossRef]
- Hiltensperger, G.; Hecht, N.; Kaiser, M.; Rybak, J.-C.; Hoerst, A.; Dannenbauer, N.; Müller-Buschbaum, K.; Bruhn, H.; Esch, H.; Lehmann, L. Quinolone amides as antitrypanosomal lead compounds with in vivo activity. Antimicrob. Agents Chemother. 2016, 60, 4442–4452. [Google Scholar] [CrossRef]
- Allaka, T.R.; Polkam, N.; Rayam, P.; Sridhara, J.; Garikapati, N.S.; Kotapalli, S.S.; Ummanni, R.; Anireddy, J.S. Design, synthesis and biological activity evaluation of novel pefloxacin derivatives as potential antibacterial agents. Med. Chem. Res. 2016, 25, 977–993. [Google Scholar] [CrossRef]
- Wang, X.; Jiang, X.; Sun, S.; Liu, Y. Synthesis and biological evaluation of novel quinolone derivatives dual targeting histone deacetylase and tubulin polymerization as antiproliferative agents. RSC Adv. 2018, 8, 16494–16502. [Google Scholar] [CrossRef]

| Fluoroquinolone | Structure | Generation | Physical Properties | References |
|---|---|---|---|---|
| Norfloxacin | 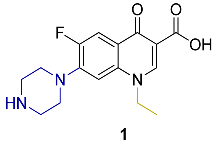 | 2nd | ClogP 1.81 | [23,24] |
| Ciprofloxacin | 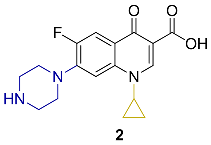 | LogPexp–0.1432 | [23,25,26,27] | |
| ClogP–0.725 | ||||
| ClogP 1.32 | ||||
| ClogP 1.55 | ||||
| Levofloxacin | 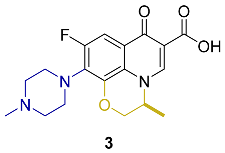 | 3rd | ClogP 1.35 ClogP–0.51 | [24,26] |
| Moxifloxacin |  | 4th | ClogP 2.53 LogP 1.60 | [24,28] |
| Testing Technique | Advantages | Disadvantages | Reference |
|---|---|---|---|
| Disk-diffusion |
|
| [36,37] |
| Dilution methods |
| - Broth macrodilution has higher risk of error- Broth microdilution may not detect contamination, inoculum viability and the inhibitory effect of cosolvents used (e.g., dimethyl sulphoxide)- Agar dilution method requires intense labor and high cost unless it is automated | [31,38] |
| Fluoroquinolone | G +ve Bacteria | Strain | MIC (µM) | Reference | |
|---|---|---|---|---|---|
| Generation | Name | ||||
| Second Generation | Norfloxacin | B. subtilis | NCDC 71 | 15.658 | [42] |
| B. cereus | 8035 | 4.697 | [44] | ||
| Roma 702 | <0.128 | [46] | |||
| Roma 709 | 8.267 | [28] | |||
| B. polymyxa | NCDC 64 | 78.289 | [42] | ||
| E. faecalis | ATCC 29212 | <0.128 | [46] | ||
| 8.267 | [28] | ||||
| 100.207 | [47] | ||||
| L. acidophilus | RSKK 06029 | 2113.794 | [28] | ||
| L. monocytogenes | ATCC 43251 | 8.267 | [28] | ||
| S. aureus | NCDC 110 | 31.315 | [42] | ||
| ATCC 29213 | 3.132 | [43] | |||
| ATCC 25923 | 156.170 | [45] | |||
| 4.134 | [28] | ||||
| <0.128 | [46] | ||||
| S. aureus 209p | 1.221 | [44] | |||
| MRSA | 1.879 | [28] | |||
| S. pneumonia | ATCC 49619 | 19.572 | [43] | ||
| Lomefloxacin | B. cereus | 8035 | 17.931 | [44] | |
| S. aureus | 209p | 2.220 | |||
| Ciprofloxacin | A. baumannii | 24.144 | [24] | ||
| ATCC 19606 | 2.354 | [50] | |||
| B. cereus | ATCC 10876 | 0.360 | [57] | ||
| Roma 702 | 0.181 | [46] | |||
| Roma 709 | 3.954 | [28] | |||
| B. polymyxa | NCDC 64 | 30.180 | [42] | ||
| B. subtilis | ATCC 6633 | 0.090 | [57] | ||
| 0.030 | [58] | ||||
| 8.149 | [34] | ||||
| NCDC 71 | 60.361 | [42] | |||
| 72.433 | [59] | ||||
| E. Faecalis | ATCC 29212 | 3.018 | [56,60,61] | ||
| 1.360 | [51] | ||||
| 0.368 | [46] | ||||
| 1.509 | [55,62] | ||||
| 7.878 | [28] | ||||
| ATCC 33186 | 2.384 | [50] | |||
| ATCC 51575 | 1.360 | [51] | |||
| ATCC 51299 | 1.509 | [55] | |||
| JH2-2 | 6.036 | [63] | |||
| UCN41 | 3.018 | [63] | |||
| E. faecalis | 24.144 | [47] | |||
| 14-1 | 96.577 | [53,54] | |||
| 14-2 | 3.018 | [53,54] | |||
| E. faecium | ATCC-19434T | 3.018 | [63] | ||
| BM-4147 | 12.072 | [63] | |||
| ATCC 27270 | 2.651 | [56] | |||
| ATCC 700221 | >386.308 | [55] | |||
| 13-7 | >386.308 | [55] | |||
| 14-2 | 96.577 | [53,54] | |||
| 14-5 | 386.308 | [53,54] | |||
| 14-6 | >386.308 | [53,54] | |||
| E. hirae | ATCC 10541 | 24.144 | [48] | ||
| K. pneumonia | 193.154 | [24] | |||
| L. acidophilus | RSKK 06029 | 252.277 | [28] | ||
| L. monocytogenes | ATCC 43251 | 3.954 | [28] | ||
| EGD | 12.072 | [64] | |||
| CLIP21369 | 48.288 | [64] | |||
| S. aureus | ATCC 6538 | 26.015 | [65] | ||
| 0.800 | [66] | ||||
| 146.978 | [49] | ||||
| 1.509 | [48] | ||||
| ATCC 29213 | 0.400 | [66] | |||
| 1.509 | [48] | ||||
| 0.082 | [67] | ||||
| 1.509 | [60,61] | ||||
| 0.296 | [50] | ||||
| 0.680 | [51] | ||||
| 0.755 | [55] | ||||
| ATCC 25923 | 0.755 | [64] | |||
| 2.960 | [57] | ||||
| 0.010 | [52] | ||||
| 0.755 | [26] | ||||
| 0.368 | [46] | ||||
| 3.954 | [28] | ||||
| 3.018 | [62] | ||||
| S. aureus ATCC 25923 (clinical isolate) | 0.755 | [63] | |||
| SAI | 24.144 | [64] | |||
| SAI24 | 48.289 | [64] | |||
| SA036 | 96.577 | [64] | |||
| N41120032 | 193.154 | [64] | |||
| SG511 | 0.470 | [58] | |||
| Microbank 14001 (MRSA) | 1.480 | [57] | |||
| S. aureus D15 MRSA | 3.100 | [66] | |||
| S. aureus D17 MRSA | 3.100 | [66] | |||
| S. aureus CIPR | 50.000 | [66] | |||
| S. aureus NCTC 4163 | 0.755 | [48] | |||
| S. aureus HG001 (laboratory strain) | 0.377 | [63] | |||
| MSSA 12-1 | 0.755 | [26] | |||
| MSSA 12-2 | 0.755 | [26] | |||
| MSSA 12-4 | 0.755 | [26] | |||
| MSSA 12-5 | 0.755 | [26] | |||
| MSSA 14-1 | 96.577 | [53,54] | |||
| MSSA14-3 | 0.377 | [53,54] | |||
| MSSA 14-4 | 1.509 | [53,54] | |||
| MRSA | 3.954 | [28] | |||
| MRSA 14-4 | >386.308 | [53,54] | |||
| MRSA 14-5 | 48.288 | [53,54] | |||
| MRSA 12-2 | 193.154 | [26] | |||
| MRSA 12-4 | 193.154 | [26] | |||
| MRSA 12-5 | 96.577 | [26] | |||
| CMCC 26003 | 1.509 | [53,54] | |||
| S. aureus ATCC 700699 (resistant isolate) | >24.144 | [63] | |||
| Healthcare-acquired MRSA NRS70 | 0.604 | [50] | |||
| Community-acquired MRSAUSA300 | 19.014 | [50] | |||
| (MRSA) ATCC 33591 | 1.509 | [60,61] | |||
| 0.755 | [55] | ||||
| 0.680 | [51] | ||||
| MRSA ATCC 33592 | ≤0.083 | [56] | |||
| NCDC 110 | 150.901 | [42] | |||
| 12.072 | [47] | ||||
| 0.589 | [49] | ||||
| 0.377 | [24] | ||||
| S. epidermidis | ATCC 12228 | 0.400 | [66] | ||
| 1.480 | [57] | ||||
| 0.755 | [48] | ||||
| ATCC 14990 | 0.377 | [63] | |||
| ATCC 35984 | ≤0.181 | [63] | |||
| - | 0.589 | [49] | |||
| MSSE CANWARD-2008 81388 | ≤0.083 | [56] | |||
| MSSE ATCC 12228 | 0.377 | [55] | |||
| 0.340 | [51] | ||||
| MSSE 12-1 | 0.755 | [26] | |||
| MSSE12-3 | 6.036 | [26] | |||
| MSSE12-6 | 0.755 | [26] | |||
| MSSE12-8 | 12.072 | [26] | |||
| MSSE14-2 | >386.308 | [53,54] | |||
| MRSE CAN-ICU 61589 (CAZ > 32) | 42.411 | [56] | |||
| MRSE12-1 | 24.144 | [26] | |||
| MRSE12-6 | 48.288 | [26] | |||
| MRSE 13-3 | 193.154 | [55] | |||
| MRSE14-21 | 193.154 | [54] | |||
| MRSE14-22 | 386.308 | [53,54] | |||
| MRSE14-37 | 386.308 | [53,54] | |||
| MRSE14-39 | 386.308 | [53,54] | |||
| MRSE 16-3 | 32.897 | [54] | |||
| S. pneumoniae | ATCC 19615 | 6.036 | [54] | ||
| ATCC 49619 | 0.331 | [56] | |||
| R6 | 1.177 | [50] | |||
| Cipro HCl | B. cereus | Roma 709 | 1.636 | [28] | |
| E. faecalis | ATCC 29212 | 3.435 | [28] | ||
| L. acidophilus | RSKK 06029 | 219.385 | [28] | ||
| L. monocytogenes | ATCC 43251 | 3.435 | [28] | ||
| S. aureus | ATCC 25923 | 6.843 | [28] | ||
| MRSA | 3.435 | [28] | |||
| Third Generation | Levofloxacin | E. faecalis | ATCC 29212 | 2.770 | [51] |
| 2.767 | [55] | ||||
| ATCC 51575 | 1.380 | [51] | |||
| 1.384 | [55] | ||||
| ATCC 700221 | 177.220 | [51] | |||
| 14-1 | 44.276 | [68] | |||
| 354.210 | [53,54] | ||||
| 14-2 | 88.552 | [68] | |||
| 2.767 | [53,54] | ||||
| 14-3 | 177.104 | [68] | |||
| E. faecium | ATCC 700221 | 88.552 | [55] | ||
| 13-7 | 88.552 | [55] | |||
| 14-1 | 354.210 | [68] | |||
| 14-2 | 88.552 | [53,54] | |||
| 14-2 | 2.767 | [68] | |||
| 14-5 | 177.105 | [53,54] | |||
| 14-6 | 177.105 | [53,54] | |||
| 16-4 | 44.300 | [51] | |||
| S. aureus | ATCC 25923 | <0.022 | [26] | ||
| 0.166 | [69] | ||||
| ATCC 29213 | 0.350 | [55] | |||
| 0.350 | [51] | ||||
| CMCC 26003 | 0.346 | [68] | |||
| 0.346 | [53,54] | ||||
| MSSA12-2 | 0.346 | [26] | |||
| MSSA 12-4 | 0.166 | [69] | |||
| 0.344 | [26] | ||||
| MSSA12-5 | 0.346 | [26] | |||
| MSSA14-1 | 22.138 | [53,54] | |||
| MSSA 14-2 | 0.692 | [68] | |||
| MSSA14-3 | 0.346 | [53,54,68] | |||
| MSSA14-4 | 1.384 | [53,54,68] | |||
| MRSA 12-1 | 177.105 | [69] | |||
| MRSA12-2 | 88.552 | [26] | |||
| MRSA12-4 | 88.552 | [26] | |||
| MRSA12-5 | 88.552 | [26] | |||
| MRSA14-4 | 177.105 | [53,54,68] | |||
| MRSA14-5 | 22.138 | [26,53,54] | |||
| NARSA 10198 | 88.552 | [70] | |||
| NARSA 10193 | 88.552 | [70] | |||
| ATCC 29213 | 1.384 | [70] | |||
| S. epidermidis | MSSE ATCC 12228 | 0.350 | [51] | ||
| 0.346 | [55] | ||||
| 12-1 | 0.346 | [26] | |||
| 12-3 | 1.384 | [26] | |||
| 12-6 | 0.346 | [26] | |||
| 12-8 | 11.069 | [26] | |||
| 12-1 | 11.069 | [26] | |||
| 12-6 | 88.552 | [26] | |||
| MRSE12-1 | 0.083 | [69] | |||
| MSSE14-2 | >354.210 | [53,54] | |||
| 354.210 | [68] | ||||
| MSSE12-3 | 1.384 | [69] | |||
| MSSE14-4 | 2.767 | [68] | |||
| MSSE14-6 | 5.534 | [68] | |||
| MRSE 13-3 | 88.552 | [55] | |||
| MRSE14-21 | 177.105 | [53,54] | |||
| MRSE14-22 | 88.552 | [53,54,68] | |||
| MRSE14-37 | 177.105 | [53,54,68] | |||
| MRSE14-39 | 177.105 | [53,54,68] | |||
| MRSE 16-3 | 5.540 | [51] | |||
| S. pneumoniae | ATCC 49619 | 0.346 | [69] | ||
| ATCC 19615 | 1.384 | [53,54,68] | |||
| Sparifloxacin | B. cereus | 8035 | 0.484 | [44] | |
| S. aureus | 209p | 0.484 | |||
| Gatifloxacin | B. subtilis | NCDC 71 | 213.109 | [42] | |
| B. polymyxa | NCDC 64 | 26.639 | [42] | ||
| S. aureus | NCDC 110 | 13.319 | [42] | ||
| ATCC 29213 | 0.333 | [71] | |||
| MSSA clinical isolates | 0.333 | [71] | |||
| MRSA clinical isolates | 42.622 | [71] | |||
| S. epidermidis | ATCC 12228 | 0.160 | [71] | ||
| MSSE clinical isolates | 0.160 | [71] | |||
| MRSE clinical isolates | 0.160 | [71] | |||
| Moxifloxacin HCl | B. cereus | Roma 709 | <1.370 | [28] | |
| E. faecalis | ATCC 33186 | 0.891 | [50] | ||
| 14-1 | 18.296 | [68] | |||
| 14-2 | 36.539 | [68] | |||
| 14-3 | 18.296 | [68] | |||
| E. faecium | ATCC 29212 | <1.370 | [28] | ||
| 14-1 | 73.077 | [68] | |||
| 14-2 | 1.142 | [68] | |||
| MSSE 12-3 | 0.284 | [26,69] | |||
| MSSE 12-6 | 0.069 | [26] | |||
| MSSE 12-8 | 2.284 | [26] | |||
| MSSE 14-4 | 4.567 | [68] | |||
| MSSE 14-6 | 4.567 | [68] | |||
| MRSE 12-1 | 0.571 | [26,69] | |||
| MRSE 12-6 | 16.539 | [26] | |||
| MRSE 14-22 | 18.269 | [68] | |||
| MRSE 14-37 | 18.269 | [68] | |||
| MRSE 14-39 | 18.269 | [68] | |||
| L. acidophilus | RSKK 06029 | 92.785 | [28] | ||
| L. monocytogenes | ATCC 43251 | <1.370 | [28] | ||
| S. aureus | ATCC 25923 | 2.900 | [28] | ||
| <0.018 | [26,69] | ||||
| CMCC 26003 | 0.137 | [68] | |||
| MSSA ATCC 29213 | 0.057 | [50] | |||
| MSSA12-1 | 0.034 | [26] | |||
| MSSA12-2 | 0.018 | [26] | |||
| MSSA12-4 | <0.018 | [26,69] | |||
| MSSA12-5 | 0.034 | [26] | |||
| MSSA14-3 | <0.018 | [68] | |||
| MSSA14-4 | <0.018 | [68] | |||
| community-acquired MRSAUSA300 | 3.654 | [50] | |||
| healthcare-acquired MRSA NRS70 | 0.057 | [50] | |||
| MRSA 12-1 | 18.269 | [69] | |||
| MRSA12-2 | 18.269 | [26] | |||
| MRSA12-4 | 18.269 | [26] | |||
| MRSA12-5 | 18.269 | [26] | |||
| MRSA 14-4 | 27.404 | [68] | |||
| MRSA14-5 | 18.269 | [68] | |||
| MRSA | <1.370 | [28] | |||
| S. pneumoniae | ATCC 19615 | 0.034 | [68] | ||
| ATCC 49619 | 0.137 | [69] | |||
| R6 | 0.365 | [50] | |||
| Fluoroquinolone | G −ve Bacteria | Strain | MIC (µM) | Reference | |
|---|---|---|---|---|---|
| Generation | Name | ||||
| Second Generation | Norfloxacin | E. coli | ATCC 8739 | <0.251 | [1] |
| ATCC 25922 | 3.132 | [43] | |||
| <0.094 | [24] | ||||
| <1.879 | [28] | ||||
| 117.433 | [45] | ||||
| 0.128 | [46] | ||||
| ATCC 35218 | 6.263 | [46] | |||
| F-50 | 0.595 | [44] | |||
| NCDC 134 | 125.262 | [42] | |||
| K. pneumoniae | ATCC13883 | 4.134 | [28] | ||
| P. aeruginosa | ATCC 9027 | 9.708 | [44] | ||
| 1.002 | [1] | ||||
| ATCC 27853 | >1565.773 | [45] | |||
| 19.572 | [43] | ||||
| ATCC 43288 | 16.503 | [28] | |||
| NCDC 105 | 46.973 | [42] | |||
| PAO1 | 12.526 | [47] | |||
| Y. pseudotuberculosis | ATCC 911 | 1.879 | [28] | ||
| 0.128 | [46] | ||||
| Lomefloxacin | E. coli | F-50 | 8.823 | [44] | |
| P. aeruginosa | 9027 | 17.931 | [44] | ||
| Ciprofloxacin | A. haemolyticus | ATCC 19002 | 0.755 | [62] | |
| A. baumannii | ATCC17961 | 0.240 | [58] | ||
| CIP 7010 | 0.377 | [62] | |||
| CAN-ICU 63169 | 6.036 | [21] | |||
| A. coacetius | ATCC 19606 | 1.509 | [55] | ||
| 1.360 | [51] | ||||
| C. freundii | ATCC 43864 | ≤0.091 | [55] | ||
| 1.380 | [51] | ||||
| E. aerogenes | ATCC 13048 | ≤0.080 | [51] | ||
| ≤0.091 | [55] | ||||
| E. cloacae | ATCC 43560 | ≤0.091 | [55] | ||
| ≤0.080 | [51] | ||||
| E. coli | ESBLs(+)14-11 | 24.144 | [54] | ||
| 48.289 | [55] | ||||
| ESBL+ 14-2 | 96.577 | [54] | |||
| 14-1 | 24.144 | [54] | |||
| 14-2 | 24.144 | [54] | |||
| ATCC-29213 | ≤0.755 | [21,52] | |||
| ATCC 25922 | <1.811 | [28,63] | |||
| 0.024 | [54,57] | ||||
| 0.031 | [48] | ||||
| 0.010 | [66] | ||||
| 61.869 | [49] | ||||
| 0.091 | [62] | ||||
| 0.002 | [24] | ||||
| NR 17663 | 0.002 | [24] | |||
| NR 17666 | 0.045 | [24] | |||
| NR 17661 | 96.577 | [24] | |||
| ATCC 25922 ESBLs(-); | ≤0.091 | [55] | |||
| ≤0.080 | [51] | ||||
| ATCC 25922 (wild type) | ≤ 0.091 | [76] | |||
| ATCC 35218 | 0.045 | [60,61] | |||
| 16.961 | [34] | ||||
| ≤0.080 | [51] | ||||
| BW5328/pAH69 (wild type) | ≤ 0.091 | [76] | |||
| CAN-ICU 61714 (GEN-R) | ≤0.755 | [21] | |||
| CAN-ICU 63074 (AMK 32) | ≤0.755 | [21] | |||
| CANWARD-2011 97615 | 772.616 | [21] | |||
| gyrA S83LD87N, parC S80I E84G, AcrA+ | >96.577 | [76] | |||
| DC0 | 0.470 | [58] | |||
| DC2 | 0.240 | [58] | |||
| F-50 | 0.573 | [44] | |||
| K12 | 0.604 | [50] | |||
| K12 ΔlacU169 | 0.005 | [67] | |||
| K12 ΔlacU169 tolC::Tn10 | 0.001 | ||||
| K12 ΔlacU169 tolC::Tn10 gyrA S83L | 0.019 | ||||
| K12 ΔlacU169 tolC::Tn10 gyrA D87Y | 0.009 | ||||
| imp-4213 (permeable outer membrane) | ≤0.091 | [76] | |||
| JW5503-1 (ΔtoIC) | ≤0.0.091 | [76] | |||
| MC4100 (wild type) | ≤0.091 | [76] | |||
| NB27005-CDY0039 (ΔtolC, gyrA S83L D83G, parC S80I) | 6.036 | [76] | |||
| NCDC 134 | 75.451 | [42] | |||
| NCTC 8196 | 0.031 | [48] | |||
| 0.040 | [66] | ||||
| ATCC 8739 | 28.007 | [65] | |||
| Penicillin Resistant E. coli | 0.377 μM (68.9% survival of bacteria | [77] | |||
| H. pylori | NCTC 11916 | 1.811 | [72] | ||
| Clinical isolate | 0.905 | [72] | |||
| K. pneumoniae | ATCC 13883 | ≤0.755 | [21] | ||
| 1.811 | [28] | ||||
| 0.755 | [62] | ||||
| 0.050 | [66] | ||||
| ATCC 35657 | 0.021 | [60,61] | |||
| ATCC 700603 ESBLs (+) | 1.509 | [55] | |||
| 1.360 | [51] | ||||
| 0.755 | [63] | ||||
| 7 ESBLs(-) | ≤0.091 | [55] | |||
| 7 ESBLs (-) | ≤0.080 | [51] | |||
| ESBL+ 14–17 | 1.509 | [54] | |||
| ESBL+ 14–18 | 1.509 | [54] | |||
| ESBL+ 14–19 | 193.154 | [54] | |||
| 14-1 | 96.577 | [54] | |||
| 14-2 | 48.288 | [54] | |||
| 14-3 | >386.308 | [54] | |||
| 14-4 | 96.577 | [54] | |||
| K. pneumonia | 40.160 | [78] | |||
| M. catarrhalis | ATCC 25238 | 0.091 | [60,61] | ||
| M. morganii | ATCC 25830 | ≤0.091 | [55] | ||
| ≤0.080 | [51] | ||||
| P. aeruginosa | ATCC 9027 | 0.720 | [57] | ||
| 1.177 | [44] | ||||
| ATCC 15442 | 0.755 | [48] | |||
| ATCC 43288 | <0.091 | [62] | |||
| 3.954 | [28] | ||||
| ATCC 27853 | 1.509 | [48] | |||
| 1.509 | [54] | ||||
| 0.680 | [51] | ||||
| 0.755 | [55] | ||||
| 0.755 | [62,63] | ||||
| 3.018 | [21] | ||||
| CAN-ICU 62308 (GEN-R) | 6.036 | [21] | |||
| CANWARD-2011 96846 | 12.072 | [21] | |||
| DSM 1117 Mueller–Hinton | 0.755 | [79] | |||
| DSM 1117 Succinate minimum medium | 0.755 | ||||
| DSM 1117 Succinate minimum medium + FeCl3 (1 lM) | 0.755 | ||||
| AM 85 Mueller–Hinton | 48.288 | ||||
| AM 85 Succinate minimum medium | 48.288 | ||||
| AM 85 Succinate minimum medium + FeCl3 (1 lM) | 96.577 | ||||
| K799/wt | 0.470 | [58] | |||
| K799/61 | 0.240 | [58] | |||
| K1542 (ΔmexX, DmexB) | 0.181 | [76] | |||
| NCDC 105 | 150.901 | [42] | |||
| NB52023-CDK005 (ΔmexX, DmexB, gyrA T83I) | 1.509 | [76] | |||
| NB52023-CDK006 (ΔmexX, ΔmexB, gyrA T83I, parC S87L) | 12.072 | [76] | |||
| PAO1 | 1.177 | [50] | |||
| PA01 (Wild type) | 0.377 | [76] | |||
| - | 5.030 | [47] | |||
| - | 0.589 | [49] | |||
| 14-9 | 1.509 | [54] | |||
| 14-14 | 3.018 | [54] | |||
| 14-15 | 3.018 | [54] | |||
| 14-16 | 3.018 | [54] | |||
| P. mirabilis | ATCC 12453 | 0.045 | [57] | ||
| ATCC 49565 | ≤0.080 | [51] | |||
| 13-1 | ≤0.091 | [55] | |||
| P. rettgeri | ATCC 31052 | ≤0.091 | [55] | ||
| ≤0.080 | [51] | ||||
| P. vulgaris | ATCC 29905 | ≤0.091 | [55] | ||
| ≤0.080 | [51] | ||||
| S. marcescens | ATCC 21074 | 0.160 | [51] | ||
| 0.181 | [55] | ||||
| S. maltophilia | ATCC 13636 | 5.450 | [51] | ||
| 12.072 | [55] | ||||
| CAN-ICU 62584 | 1.325 | [56] | |||
| S. pneumoniae | ATCC 49619 | 0.755 | [26] | ||
| 12-18 | 3.018 | [26] | |||
| Y. pseudotuberculosis | ATCC 911 | 1.812 | [28] | ||
| Ciprofloxacin HCl | E. aerogenes | ATCC 13048 | 0.086–0.172 | [64] | |
| CM64 | 01.363 | [64] | |||
| E. coli | ATCC 25922 | <1.636 | [28] | ||
| 0.022 (pH 7.4) | [64] | ||||
| K. pneumoniae | ATCC13883 | <1.636 | [28] | ||
| P. aeruginosa | ATCC 43288 | 3.435 | [28] | ||
| Y. pseudotuberculosis | ATCC 911 | <1.636 | [28] | ||
| Third Generation | Levofloxacin | A. coacetious | ATCC 19606 | 0.346 | [55] |
| 0.350 | [51] | ||||
| C. freundii | ATCC 43864 | ≤.0.083 | [55] | ||
| ≤0.080 | [51] | ||||
| E. aerogenes | ATCC 13048 | 0.166 | [55] | ||
| 0.170 | [51] | ||||
| E. cloacae | ATCC 43560 | ≤.0.083 | [55] | ||
| ≤0.080 | [51] | ||||
| E. coli | ATCC 25922 | 0.346 | [68] | ||
| 0.0412 | [24] | ||||
| <0.022 | [69] | ||||
| ATCC 25922 ESBLs− | ≤0.083 | [55] | |||
| 88.610 | [51] | ||||
| ATCC 35218 ESBLs+ | ≤0.080 | [51] | |||
| NR 17663 | 0.083 | [24] | |||
| NR 17666 | 0.083 | [24] | |||
| NR 17661 | 88.552 | [24] | |||
| 12-6 | 0.692 | [69] | |||
| 12-11 | 11.069 | [69] | |||
| ESBL+ 14-1 | 11.069 | [54] | |||
| 44.276 | [69] | ||||
| 5.534 | [68] | ||||
| ESBL+ 14-2 | 21.810 | [54] | |||
| 21.810 | [68] | ||||
| 14-1 | 21.810 | [54] | |||
| 10.905 | [68] | ||||
| 14-2 | 21.810 | [54] | |||
| 10.905 | [68] | ||||
| K. pneumoniae | ESBL+ 14-17 | 1.363 | [54] | ||
| 10.905 | [68] | ||||
| ESBL+ 14-18 | 1.363 | [54] | |||
| 2.276 | [68] | ||||
| ESBL+ 14-19 | 174.482 | [54,68] | |||
| - | 11.069 | [80] | |||
| 14-1 | 43.621 | [54,68] | |||
| 14-2 | 21.810 | [54] | |||
| 14-3 | 87.241 | [54] | |||
| 43.621 | [68] | ||||
| 14-4 | 43.621 | [54] | |||
| 21.810 | [68] | ||||
| ATCC 700603 ESBLs+ | 1.364 | [55] | |||
| 1.380 | [51] | ||||
| ESBLs- | ≤0.082 | [55] | |||
| ESBLs- | 0.170 | [51] | |||
| 12-4 | 0.082 | [69] | |||
| 12-7 | 1.363 | [69] | |||
| P. aeruginosa | ATCC 27853 | 2.726 | [54,55,68] | ||
| 5.540 | [51] | ||||
| 14-9 | 1.363 | [54] | |||
| 2.726 | [68] | ||||
| 14-11 | 5.453 | [68] | |||
| 14-14 | 5.453 | [54] | |||
| 14-15 | 5.453 | [54,68] | |||
| 14-16 | 5.453 | [54] | |||
| 14-19 | 5.453 | [68] | |||
| 12-12 | 1.363 | [69] | |||
| 12-14 | 87.241 | [69] | |||
| 12-20 | 21.810 | [69] | |||
| M. morganii | ATCC 25830 | ≤0.083 | [55] | ||
| ≤0.080 | [51] | ||||
| P. mirabilis | 13-1 | 0.166 | [55] | ||
| ATCC 49565 | ≤0.080 | [51] | |||
| P. rettgeri | ATCC 31052 | ≤0.080 | [51] | ||
| ≤0.83 | [55] | ||||
| P. vulgaris | ATCC 29905 | ≤0.080 | [51] | ||
| ≤0.083 | [55] | ||||
| S. maltophilia | ATCC 13636 | 2.767 | [55] | ||
| 1.380 | [51] | ||||
| S. marcescens | ATCC 21074 | 0.350 | [51] | ||
| 0.356 | [55] | ||||
| S. pneumoniae | ATCC 49619 | 0.345 | [26] | ||
| 12-18 | 2.535 | [26] | |||
| Sparifloxacin | E. coli | F-50 | 0.484 | [44] | |
| P. aeruginosa | ATCC 9027 | 0.484 | [44] | ||
| Gatifloxacin | E. coli | ATCC 700603 | 0.160 | [71] | |
| NCDC 134 | 266.387 | [42] | |||
| K. pneumoniae | ATCC 25922 | 2.664 | [71] | ||
| P. aeruginosa | NCDC 105 | 106.555 | [42] | ||
| Moxifloxacin HCl | A. baumannii | ATCC 19606 | 0.972 | [50] | |
| E. coli | ATCC 25922 | 0.137 | [68] | ||
| <0.018 | [69] | ||||
| 0.037 | [24] | ||||
| <1.370 | [28] | ||||
| NR 17663 | 0.037 | [24] | |||
| NR 17666 | 0.075 | [24] | |||
| NR 17661 | 79.715 | [24] | |||
| 12-6 | 1.142 | [69] | |||
| 12-11 | 36.539 | [69] | |||
| ESBL+ 12-14 | 36.539 | [69] | |||
| ESBL+ 14-1 | 4.567 | [68] | |||
| ESBL+ 14-2 | 36.539 | [68] | |||
| 14-1 | 18.269 | [68] | |||
| 14-2 | 36.539 | [68] | |||
| K. pneumoniae | ATCC 13883 | <1.370 | [28] | ||
| ESBL+ 14-17 | 18.269 | [68] | |||
| ESBL+ 14-18 | 2.284 | [68] | |||
| ESBL+ 14-19 | 146.155 | [68] | |||
| 14-1 | 18.269 | [68] | |||
| 14-2 | 18.269 | [68] | |||
| 14-3 | 73.077 | [68] | |||
| 14-4 | 18.269 | [68] | |||
| 12-4 | 0.069 | [69] | |||
| ESBL+ 12-7 | 1.142 | [69] | |||
| S. pneumoniae | ATCC 49619 | 0.137 | [26] | ||
| 12-18 | 1.142 | [26] | |||
| P. aeruginosa | ATCC 27853 | 4.567 | [68] | ||
| ATCC 43288 | 11.601 | [28] | |||
| 14-9 | 9.135 | [68] | |||
| 14-11 | 36.539 | [68] | |||
| 14-15 | 36.539 | [68] | |||
| 14-16 | 18.269 | [68] | |||
| 14-19 | 2.284 | [68] | |||
| PA01 | 7.722 | [50] | |||
| 12-12 | 4.567 | [69] | |||
| 12-14 | 36.539 | [69] | |||
| 12-20 | 18.269 | [69] | |||
| Y. pseudotuberculosis | ATCC 911 | <1.495 | [28] | ||
| Fluoroquinolone | Mycobacterium Bacteria | Strain | MIC (mM) | Reference | |
|---|---|---|---|---|---|
| Generation | Name | ||||
| Second Generation | Norfloxacin | M. smegmatis | ATCC 607 | 16.503 | [28] |
| No activity | [46] | ||||
| Ciprofloxacin | M. tuberculosis | 36.216–51.307 | [63] | ||
| MTB H37Rv | MIC90 1.780 | [27] | |||
| 3.018 | [81] | ||||
| MTB H37Rv ATCC 27294 | 0.755 | [26,68] | |||
| MDR-TB | 6.036 | [81] | |||
| MDR-MTB 6133 resistant to INH and RFP | 0.377 | [26] | |||
| MDR-MTB 11277 resistant to INH and RFP | 0.377 | [26] | |||
| M. vaccae IMET10670 | 0.470 | [58] | |||
| M. smegmatis | ATCC607 | >120.721 | [28] | ||
| Cipro HCl | M. smegmatis | ATCC607 | >109.052 | [28] | |
| Third Generation | Levofloxacin | M. tuberculosis | H37RV 76? | 1.384 | [65] |
| MTB H37Rv ATCC 27294 | 0.692 | [26,68] | |||
| MDR-MTB 6133 resistant to INH and RFP | 0.377 | [26] | |||
| MDR-MTB 11277 resistant to INH and RFP | 0.692 | [26] | |||
| R2012-123 (pan-sensitive) | 0.692 | [65] | |||
| MDR-TB | ND | [75] | |||
| M. abscessus | 5.535 | [24] | |||
| M. chelonae | 5.535 | [24] | |||
| M. fortuitum | 0.346 | [24] | |||
| M. avium | ND | [75] | |||
| M. terrae | ND | [75] | |||
| R-2012-59 (MDR) | 0.692 | [65] | |||
| R-2012-97 (XDR) | 22.138 | [65] | |||
| M. abscessus | ATCC19977 | >88.552 | [65] | ||
| M. chelonae | ATCC35752 | 1.384 | [65] | ||
| M. fortuitum | ATCC06841 | 0.346 | [65] | ||
| Moxifloxacin | M. tuberculosis | H37Rv ATCC27294 | 0.311 | [65] | |
| MTB H37Rv | 0.228 | [82] | |||
| MDR-TB | 0.274 | [82] | |||
| R2012-123 (pan-sensitive) | 0.137 | [65] | |||
| M. smegmatis (MXF HCl) | ATCC607 | >91.347 | [28] | ||
| Antituberculosis | 0.440 | [28] | |||
| R-2012-59 (MDR) | ≤0.069 | [65] | |||
| R-2012-97 (XDR) | 4.567 | [65] | |||
| M. abscessus | ATCC19977 | >73.077 | [65] | ||
| M. chelonae | ATCC35752 | 0.571 | [65] | ||
| M. fortuitum | ATCC06841 | 0.137 | [65] | ||
| Fluoroquinolone | Fungi and Cancer | Strain | Inhibitory Effect | Reference | |
|---|---|---|---|---|---|
| Generation | Name | ||||
| Second Generation | Norfloxacin | C. albicans | ATCC 60193 | No zone of inhibition | [28] |
| S. cerevisiae | RSKK 251 | No zone of inhibition | |||
| Ciprofloxacin | A. clavatus | No zone of inhibition | [86] | ||
| C. albicans | ATCC 90873 amphotericin B-resistant | MIC 97.784 μM | [34] | ||
| C. albicans | ATCC 60193 | No zone of inhibition | [86] | ||
| T. brucei | 427/421 | MIC 100 μM GI50 30.9 ± 3.3 μM | [66] | ||
| Lung adenocarcinoma | A549 | MIC 50 μM | [61] | ||
| Colon cancer | HCT-116 | MIC 50 μM | [61] | ||
| Breast cancer | MCF-7 | MIC 50 μM | [61] | ||
| HEPG2, liver hepatocellular carcinoma cells | ATCC HB-8065 | IC50 ≥ 1207.211 μM | [50] | ||
| Vero, kidney epithelial cells | ATCC CCL-81. | IC50 ≥ 1207.211 μM | [50] | ||
| Human primary colon cancer | (SW480) | IC50 160.4 ± 6.7 μM | [48] | ||
| Human metastatic colon cancer | (SW620) | IC50 200.4 ± 4.9 μM | [48] | ||
| Human metastatic prostate cancer | (PC3) | IC50 101.4 ± 3.6 μM | [48] | ||
| Human immortal keratinocyte cell line from adult human skin | (HaCaT) | IC50 222.1 ± 5.2 μM | [48] | ||
| LDH release | HaCaT | LDH release % 4.6% at 60 μM 4.2% 40 μM 3.9% 20 μM 3.2% 10 μM | [48] | ||
| LDH release | SW480 | LDH release % 15% at 60 μM 14.5% at 40 μM 14.2% at 20 μM 12% at 10 μM | [48] | ||
| LDH release | SW620 | LDH release % 9.3% at 60 μM 9.1% at 40 μM 8.9% at 20 μM 8.1% at 10 μM | [48] | ||
| LDH release | PC3 | LDH release % 18% at 60 μM 17.5% at 40 μM 16.5% at 20 μM 14% at 10 μM | [48] | ||
| Urease inhibitory activity | 94.32 μM | [78] | |||
| HL-60 | MIC > 100 μM GI50 > 100 μM | [66] | |||
| Selectivity | MIC > 1 μM ratio GI50 > 3.2 μM ratio | [66] | |||
| L929 | GI50 >100 ± n.d. μM | [66] | |||
| HeLa | GI50 560 ± 22.6 μM | [66] | |||
| DNA gyrase | IC50 0.15 μM | [66] | |||
| Topoisomerase IV | 4.00 μM | [66] | |||
| Cytotoxicity | >100 μM | [27] | |||
| Cipro HCl | C. albicans | ATCC 60193 | No inhibition | [28] | |
| S. cerevisiae | RSKK 251 | No inhibition | [28] | ||
| Third Generation | Levofloxacin | Vero Cells | CC50 > 276.73 μM | [70] | |
| A549 | 76.3 ± 6.51 μM | [87] | |||
| HepG2 | >100 μM | ||||
| MCF-7 | 64.2 ± 5.67 μM | ||||
| PC-3 | >100 μM | ||||
| HeLa | 71.1 ± 4.98 μM | ||||
| MCF-10A (Human breast epithelial cell line) | >100 μM | ||||
| Moxifloxacin | S. cerevisiae | RSKK 251 | No inhibition | [28] | |
| HEPG2, liver hepatocellular carcinoma cells | ATCC HB-8065 | ≥ 996.435 μM | [50] | ||
| Vero, kidney epithelial cells | ATCC CCL-81 | ≥ 996.435 μM | [50] | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suaifan, G.A.R.Y.; Mohammed, A.A.M.; Alkhawaja, B.A. Fluoroquinolones’ Biological Activities against Laboratory Microbes and Cancer Cell Lines. Molecules 2022, 27, 1658. https://doi.org/10.3390/molecules27051658
Suaifan GARY, Mohammed AAM, Alkhawaja BA. Fluoroquinolones’ Biological Activities against Laboratory Microbes and Cancer Cell Lines. Molecules. 2022; 27(5):1658. https://doi.org/10.3390/molecules27051658
Chicago/Turabian StyleSuaifan, Ghadeer A. R. Y., Aya A. M. Mohammed, and Bayan A. Alkhawaja. 2022. "Fluoroquinolones’ Biological Activities against Laboratory Microbes and Cancer Cell Lines" Molecules 27, no. 5: 1658. https://doi.org/10.3390/molecules27051658
APA StyleSuaifan, G. A. R. Y., Mohammed, A. A. M., & Alkhawaja, B. A. (2022). Fluoroquinolones’ Biological Activities against Laboratory Microbes and Cancer Cell Lines. Molecules, 27(5), 1658. https://doi.org/10.3390/molecules27051658






