Cationic Axial Ligand Effects on Sulfur-Substituted Subphthalocyanines
Abstract
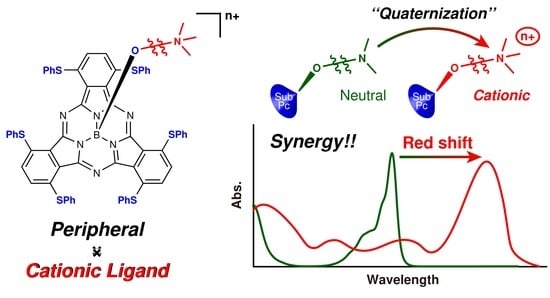
:1. Introduction
2. Results and Discussion
2.1. Synthesis of Subphthalocyanines
2.2. Cationic Axial Ligand Effect of Sulfur-Substituted Subphthalocyanines
2.3. Electrochemical Properties
2.4. Molecular Orbital Calculations
3. Materials and Methods
Synthesis of Subphthalocyanines
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Ooyama, Y.; Yagi, S. (Eds.) Progress in the Science of Functional Dyes; Springer: Singapore, 2021. [Google Scholar]
- Kanaparthi, R.K.; Kandhadi, J.; Giribabu, L. Metal-free organic dyes for dye-sensitized solar cells: Recent advances. Tetrahedron 2012, 68, 8383–8393. [Google Scholar] [CrossRef]
- Sharma, K.; Sharma, V.; Sharma, S.S. Dye-sensitized solar cells: Fundamentals and current status. Nanoscale Res. Lett. 2018, 13, 381. [Google Scholar] [CrossRef] [PubMed]
- Mei, J.; Diao, Y.; Appleton, A.L.; Fang, L.; Bao, Z. Integrated materials design of organic semiconductors for field-effect transistors. J. Am. Chem. Soc. 2013, 135, 6724–6746. [Google Scholar] [CrossRef] [PubMed]
- Yuvarja, S.; Nawaz, A.; Liu, Q.; Dubal, D.; Surya, S.G.; Salama, K.N.; Sonar, P. Organic field-effect transistor-based flexible sensors. Chem. Soc. Rev. 2020, 49, 3423–3460. [Google Scholar] [CrossRef] [PubMed]
- Inganäs, O. Organic photovoltaics over three decades. Adv. Mater. 2018, 30, 1800388. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Taima, T.; Miyadera, T.; Yamanari, T.; Yoshida, Y. Structural modifications of zinc phthalocyanine thin films for organic photovoltaic applications. J. Appl. Phys. 2012, 111, 103117. [Google Scholar] [CrossRef]
- Mako, T.L.; Racicot, J.M.; Levine, M. Supramolecular luminescent sensors. Chem. Rev. 2019, 119, 322–477. [Google Scholar] [CrossRef]
- Zhao, X.; Liu, J.; Fan, J.; Chao, H.; Peng, X. Recent progress in photosensitizers for overcoming the challenges of photodynamic therapy: From molecular design to application. Chem. Soc. Rev. 2021, 50, 4185–4219. [Google Scholar] [CrossRef]
- Moret, F.; Reddi, E. Strategies for optimizing the delivery to tumors of macrocyclic photosensitizers used in photocdynamic therapy (PDT). J. Porphyr. Phthalocyanines 2017, 21, 239–256. [Google Scholar] [CrossRef] [Green Version]
- Sekkat, N.; van den Bergh, H.; Nyokong, T.; Lange, N. Like a bolt from the blue: Phthalocyanines in biomedical optics. Molecules 2012, 17, 98–144. [Google Scholar] [CrossRef] [Green Version]
- Rio, Y.; Rodríguez-Morgade, M.S.; Torres, T. Modulating the electronic properties of porphyrinoids: A voyage from the violet to the infrared regions of the electromagnetic spectrum. Org. Biomol. Chem. 2008, 6, 1877–1894. [Google Scholar] [CrossRef] [PubMed]
- Furuyama, T.; Kobayashi, N. Azaporphyrin phosphorus(V) complexes: Synthesis, structure, and modification of optical properties. Phys. Chem. Chem. Phys. 2017, 19, 15596–15612. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Yutronkie, N.J.; Lessard, B.H.; Brusso, J.L. From chemical curiosity to versatile building blocks: Unmasking the hidden potential of main-group phthalocyanines in organic field-effect transistors. Mater. Adv. 2021, 2, 165–185. [Google Scholar] [CrossRef]
- Mitra, K.; Harman, M.C.T. Silicon phthalocyanines: Synthesis and resurgent applications. Org. Biomol. Chem. 2021, 19, 1168–1190. [Google Scholar] [CrossRef]
- Jiang, X.-J.; Lo, P.-C.; Yeung, S.-L.; Fong, W.-P.; Ng, D.K.P. A pH-responsive fluorescence probe and photosensitizer based on a tetraamino silicon(IV) phthalocyanine. Chem. Commun. 2010, 46, 3188–3190. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Furuyama, T.; Ishii, T.; Ieda, N.; Maeda, H.; Segi, M.; Uchiyama, M.; Nakagawa, H. Cationic axial ligands on sulfur substituted silicon(IV) phthalocyanines: Improved hydrophilicity and exceptionally red-shifted absorption into the NIR region. Chem. Commun. 2019, 55, 7311–7314. [Google Scholar] [CrossRef]
- Meller, A.; Ossko, A. Phthalocyaninartige bor-komplexe. Monatsh. Chem. 1972, 103, 150–155. [Google Scholar] [CrossRef]
- Mayoral, M.J.; Guilleme, J.; Calbo, J.; Aragó, J.; Aparicio, F.; Ortí, E.; Torres, T.; González-Rodríguez, D. Dual-mode chiral self-assembly of cone-shaped subphthalocyanine aromatics. J. Am. Chem. Soc. 2020, 142, 21017–21031. [Google Scholar] [CrossRef]
- Sánchez-Molina, I.; Grimm, B.; Calderon, R.M.K.; Claessens, C.G.; Guldi, D.M.; Torres, T. Self-assembly, host-guest chemistry, and photophysical properties of subphthalocyanine-based metallosupramolecular capsules. J. Am. Chem. Soc. 2013, 135, 10503–10511. [Google Scholar] [CrossRef]
- Beaumont, N.; Cho, S.W.; Sullivan, P.; Newby, D.; Smith, K.E.; Jones, T.S. Boron subphthalocyanine chloride as an electron acceptor for high-voltage fullerene-free organic photovoltaics. Adv. Funct. Mater. 2012, 22, 561–566. [Google Scholar] [CrossRef]
- Takao, Y.; Masuoka, T.; Yamamoto, K.; Mizutani, T.; Matsumoto, F.; Moriwaki, K.; Hida, K.; Iwai, T.; Ito, T.; Mizuno, T.; et al. Synthesis and properties of novel fluorinated subnaphthalocyanines for organic photovoltaic cells. Tetrahedron Lett. 2014, 55, 4564–4567. [Google Scholar] [CrossRef] [Green Version]
- Claessens, C.G.; González-Rodríguez, D.; Rodríguez-Morgade, M.S.; Medina, A.; Torres, T. Subphthalocyanines, subporphyrazines, and subporphyrins: Singular nonplanar aromatic systems. Chem. Rev. 2014, 114, 2192–2277. [Google Scholar] [CrossRef]
- Shimizu, S.; Kobayashi, N. Structurally-modified subphthalocyanines: Molecular design towards realization of expected properties from the electronic structure and structural features of subphthalocyanine. Chem. Commun. 2014, 50, 6949–6966. [Google Scholar] [CrossRef]
- Roy, P.; Bressan, G.; Gretton, J.; Cammidge, A.N.; Meech, S.R. Ultrafast excimer formation and solvent controlled symmetry breaking charge separation in the excitonically coupled subphthalocyanine dimer. Angew. Chem. Int. Ed. 2021, 60, 10568–10572. [Google Scholar] [CrossRef]
- Caballero, E.; Guzmán, D.; Torres, T.; Rodriíguez-Morgade, M.S. Expanding the subprophyrazine chromophore by conjugation of phenylene and vinylene substituents: Rainbow SubPzs. J. Org. Chem. 2020, 85, 1948–1960. [Google Scholar] [CrossRef] [PubMed]
- Rhoda, H.M.; Kayser, M.P.; Wang, Y.; Nazarenko, A.Y.; Belosludov, R.V.; Kiprof, P.; Blank, D.A.; Nemykin, V.N. Tuning up an electronic structure of the subphthalocyanine derivatives toward electron-transfer process in noncovalent complexes with C60 and C70 fullerenes: Experimental and theoretical studies. Inorg. Chem. 2016, 55, 9549–9563. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, K.; Yoshino, S.; Takemoto, M.; Sugaya, K.; Kubo, H.; Komoriya, T.; Kamei, S.; Furukawa, S. Synthesis of arylsulfanyl-subphthalocyanines and their ring expansion reaction. J. Porphyrins Phthalocyanines 2015, 19, 688–694. [Google Scholar] [CrossRef]
- Fukuda, T.; Makarova, E.A.; Luk’yanets, E.A.; Kobayashi, N. Synthesis and spectroscopic and electrochemical studies of novel benzo- or 2,3-naphtho-fused tetraazachlorins, bacteriochlorins, and isobacteriochlorins. Chem. Eur. J. 2004, 10, 117–133. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, S.; Yamazaki, Y.; Kobayashi, N. Tetrathiafulvalene-annulated subphthalocyanines. Chem. Eur. J. 2013, 19, 7324–7327. [Google Scholar] [CrossRef]
- Furuyama, T.; Satoh, K.; Kushiya, T.; Kobayashi, N. Design, synthesis, and properties of phthalocyanine complexes with main-group elements showing main absorption and fluorescence beyond 1000 nm. J. Am. Chem. Soc. 2014, 136, 765–776. [Google Scholar] [CrossRef]
- Maligaspe, E.; Hauwiller, M.R.; Zatsikha, Y.V.; Hinke, J.A.; Solntsev, P.V.; Blank, D.A.; Nemykin, V.N. Redox and photoinduced electron-transfer properties in short distance organoboryl ferrocene-subphthalocyanine dyads. Inorg. Chem. 2014, 53, 9336–9347. [Google Scholar] [CrossRef] [PubMed]
- Thorat, K.G.; Kamble, P.; Ray, A.K.; Sekar, N. Novel pyrromethene dyes with N-ethyl carbazole at the meso position: A comprehensive photophysical, lasing, photostability and TD-DFT strudy. Phys. Chem. Chem. Phys. 2015, 17, 17221–17236. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, N.; Ogata, H.; Nonaka, N.; Luk’yanets, E.A. Effect of peripheral substitution on the electronic absorption and fluorescence spectra of metal-free and zinc phthalocyanines. Chem. Eur. J. 2003, 9, 5123–5134. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Ng, D.K.P. Preparation, spectroscopic properties, and stability of water-soluble subphthalocyanines. Chem. Asian J. 2009, 4, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Becke, A.D. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. 1988, A38, 3098–3100. [Google Scholar] [CrossRef]
- Becke, A.D. A new mixing of Hartree–Fock and local density-functional theories. J. Chem. Phys. 1993, 98, 1372–1377. [Google Scholar] [CrossRef]
- Becke, A.D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef] [Green Version]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 1988, 37, 785–789. [Google Scholar] [CrossRef] [Green Version]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 16; Revision B.01; Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Bauernschmitt, R.d.; Ahlrichs, R. Treatment of electronic excitations within the adiabatic approximation of time dependent density functional theory. Chem. Phys. Lett. 1996, 256, 454–464. [Google Scholar] [CrossRef]
- Dreuw, A.; Head-Gordon, M. Single-reference ab initio methods for the calculation of excited states of large molecules. Chem. Rev. 2005, 105, 4009–4037. [Google Scholar] [CrossRef]
- Chai, J.-D.; Head-Gordon, M. Long-range corrected hybrid density functionals with damped atom–atom dispersion corrections. Phys. Chem. Chem. Phys. 2008, 10, 6615–6620. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tomasi, J.; Mennucci, B.; Cammi, R. Quantum mechanical continuum solvation models. Chem. Rev. 2005, 105, 2999–3094. [Google Scholar] [CrossRef] [PubMed]








| Compound | λabs/nm | λex/nm | λfl/nm | ΦPL | τ/ns | kr/×107 s−1 | knr/×108 s−1 |
|---|---|---|---|---|---|---|---|
| 1 | 561 | 500 | 575 | 0.07 | 0.90 | 7.7 | 10 |
| 1Q | 563 | 500 | 574 | 0.11 | 1.5 | 7.5 | 5.9 |
| 2 | 650 | 600 | 681 | 0.11 | 1.4 | 7.8 | 6.3 |
| 3 | 637 | 600 | 666 | 0.06 | 1.0 | 6.4 | 9.4 |
| 3Q | 650 | 600 | 681 | 0.06 | 0.99 | 5.6 | 9.5 |
| 4 | 638 | 600 | 668 | 0.07 | 0.92 | 7.9 | 10 |
| 4Q | 650 | 600 | 682 | 0.05 | 0.80 | 6.6 | 12 |
| λ/nm | f | Composition | |
|---|---|---|---|
| 1 | 506 | 0.41 | HOMO–4→LUMO+1 (2.4%), HOMO→LUMO (96.8%) |
| 505 | 0.41 | HOMO–4→LUMO (2.5%), HOMO→LUMO+1 (96.7%) | |
| 1Q | 509 | 0.41 | HOMO–3→LUMO+1 (2.5%), HOMO→LUMO (96.8%) |
| 507 | 0.41 | HOMO–3→LUMO (2.5%), HOMO→LUMO+1 (96.7%) | |
| 3′ | 614 | 0.43 | HOMO→LUMO (98.2%) |
| 612 | 0.43 | HOMO→LUMO+1 (98.2%) | |
| 3Q′ | 628 | 0.42 | HOMO→LUMO (98.4%) |
| 624 | 0.42 | HOMO→LUMO+1 (98.4%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ogura, Y.; Nakano, M.; Maeda, H.; Segi, M.; Furuyama, T. Cationic Axial Ligand Effects on Sulfur-Substituted Subphthalocyanines. Molecules 2022, 27, 2766. https://doi.org/10.3390/molecules27092766
Ogura Y, Nakano M, Maeda H, Segi M, Furuyama T. Cationic Axial Ligand Effects on Sulfur-Substituted Subphthalocyanines. Molecules. 2022; 27(9):2766. https://doi.org/10.3390/molecules27092766
Chicago/Turabian StyleOgura, Yusaku, Masahiro Nakano, Hajime Maeda, Masahito Segi, and Taniyuki Furuyama. 2022. "Cationic Axial Ligand Effects on Sulfur-Substituted Subphthalocyanines" Molecules 27, no. 9: 2766. https://doi.org/10.3390/molecules27092766