5,2′-Dibromo-2,4′,5′-trihydroxydiphenylmethanone Inhibits LPS-Induced Vascular Inflammation by Targeting the Cav1 Protein
Abstract
:1. Introduction
2. Results
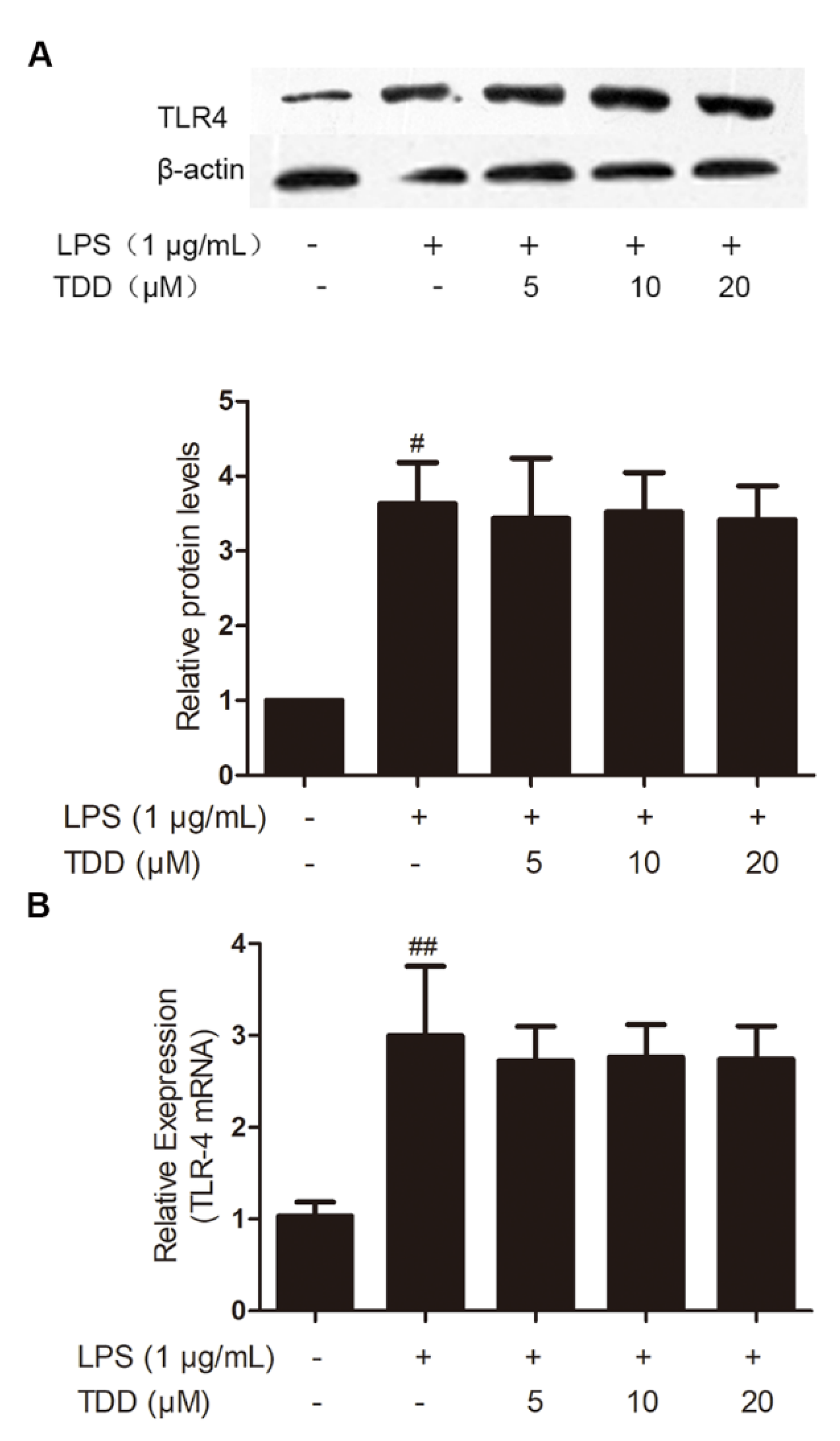
2.1. Effects of TDD on the Expression of TLR4 in LPS-Treated EA.hy926 Cells
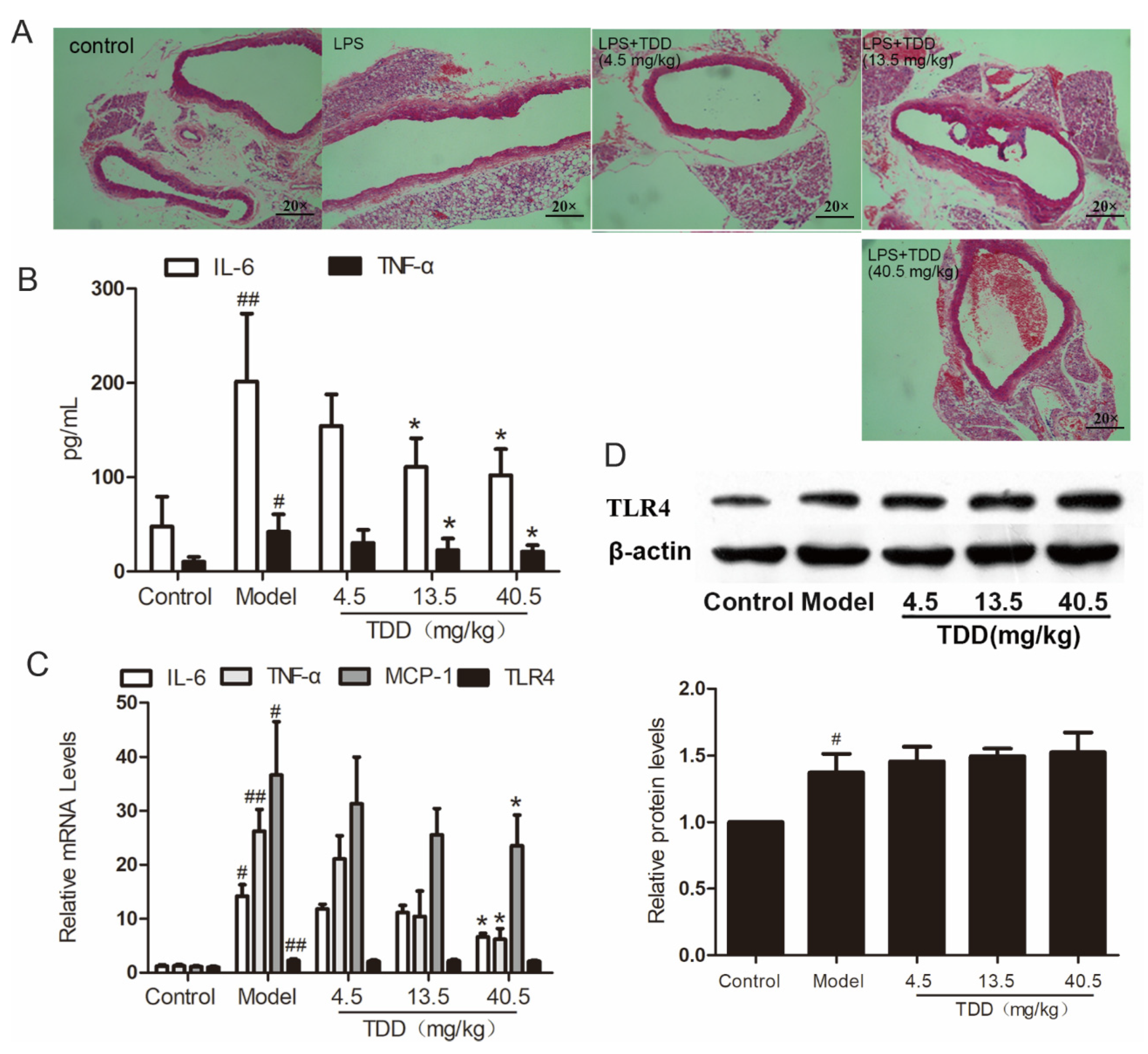
2.2. Effects of TDD on the Expression of TLR4 in the Aortae of LPS-Treated Mice
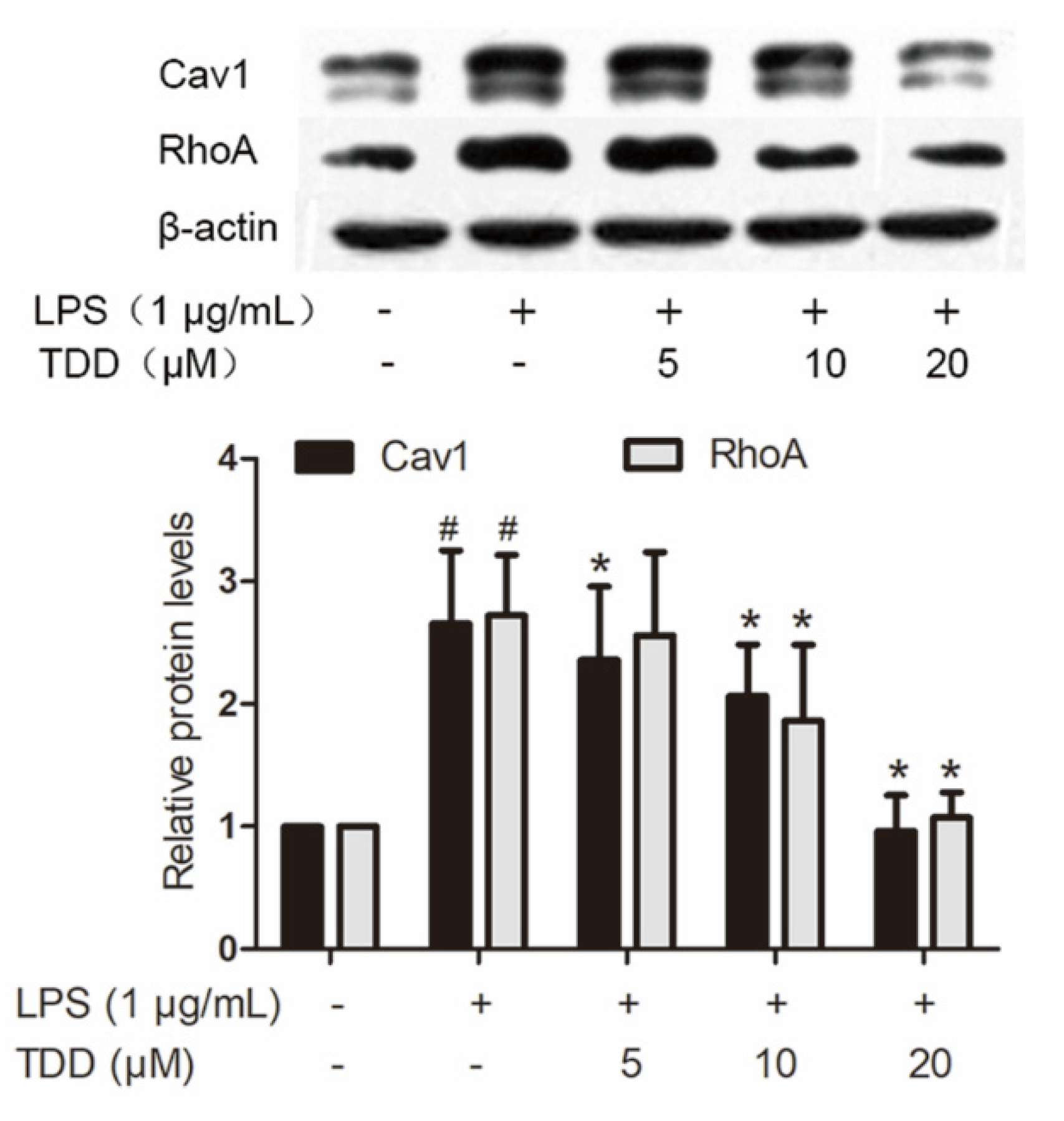
2.3. Effects of TDD on Protein Expression
2.4. Validation of the Activity of the NF-κB Signaling
2.5. Effects of TDD on Selected Proteins
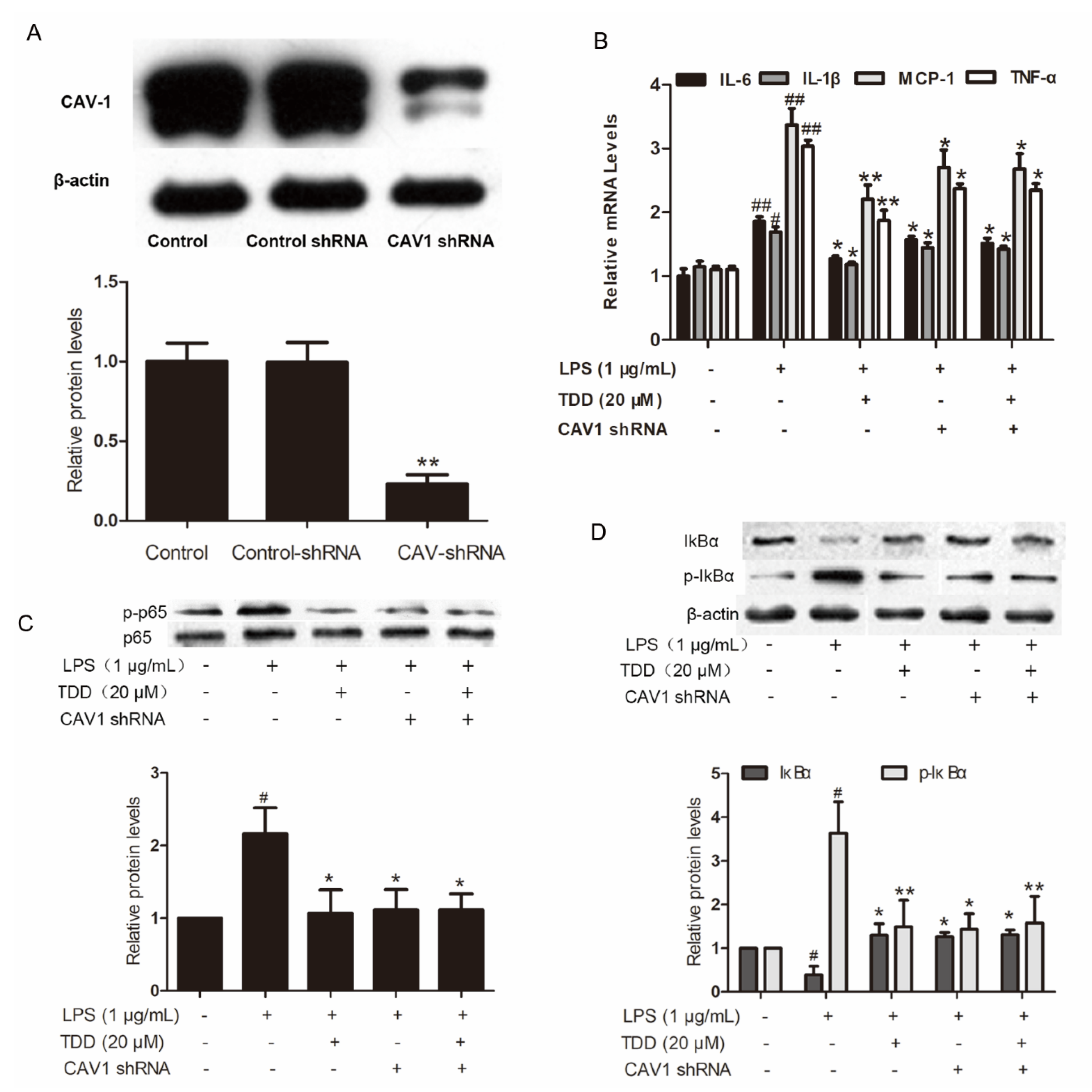
2.6. Effects of TDD on IL-6, IL-1β, MCP-1, and TNF-α Expression in LPS-Treated EA.hy926 Cells with Cav1 Knockdown
2.7. Effects of TDD on Inhibitor of NF-κB (IκB) and p65 Phosphorylation in LPS-Treated EA.hy926 Cells with Cav1 Knockdown
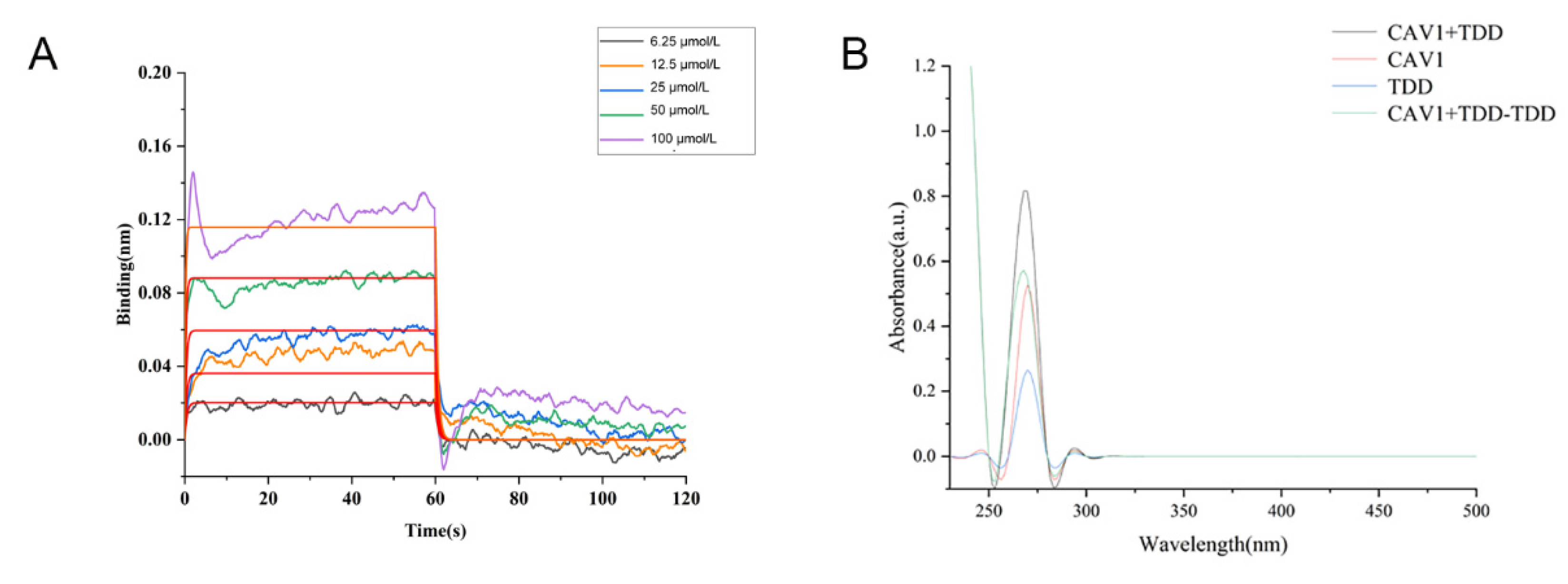
2.8. Biolayer Interference (BLI) Assay
2.9. UV-Vis Spectroscopy
2.10. Fluorescence Spectroscopy
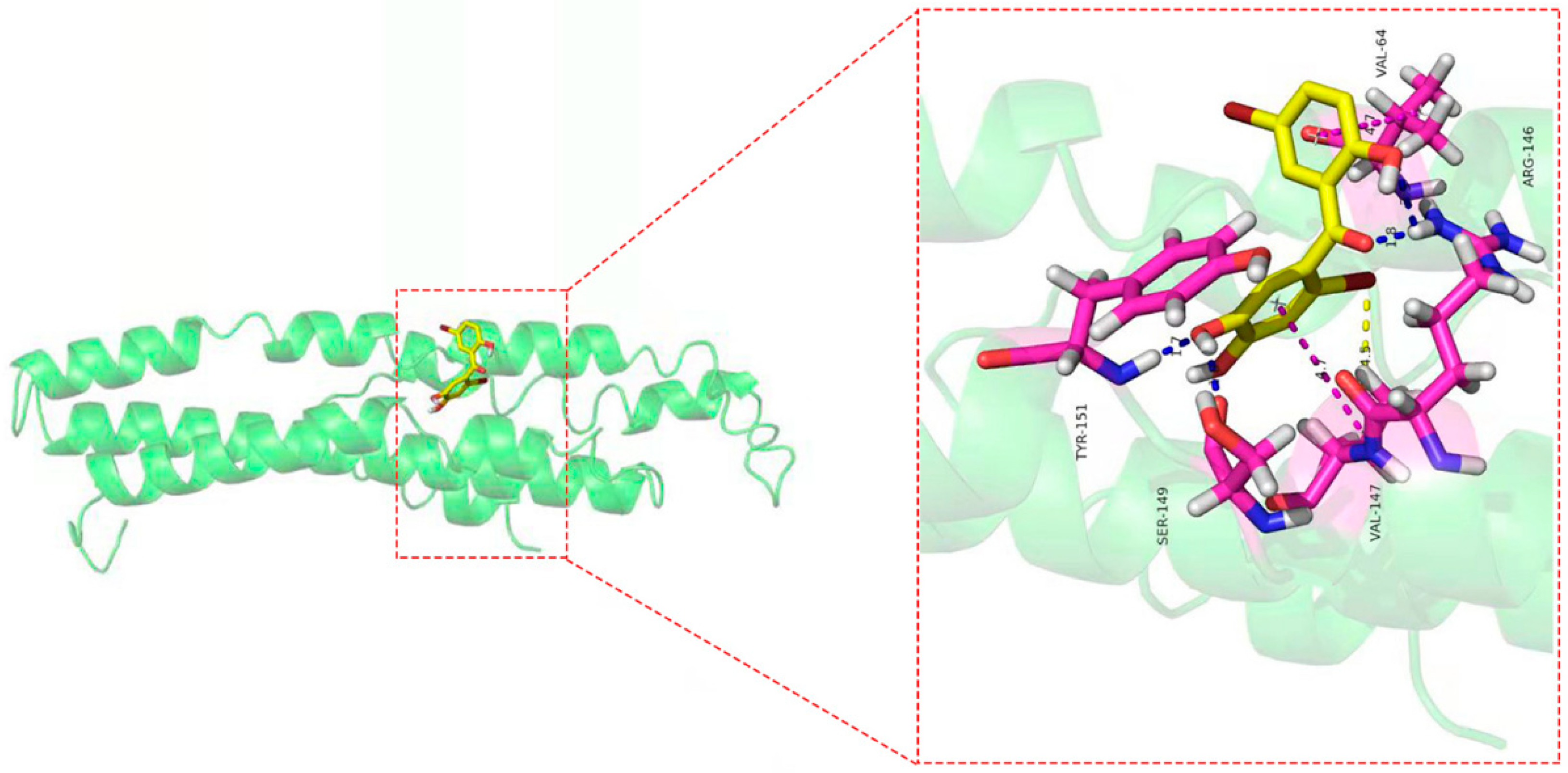
2.11. Molecular Docking
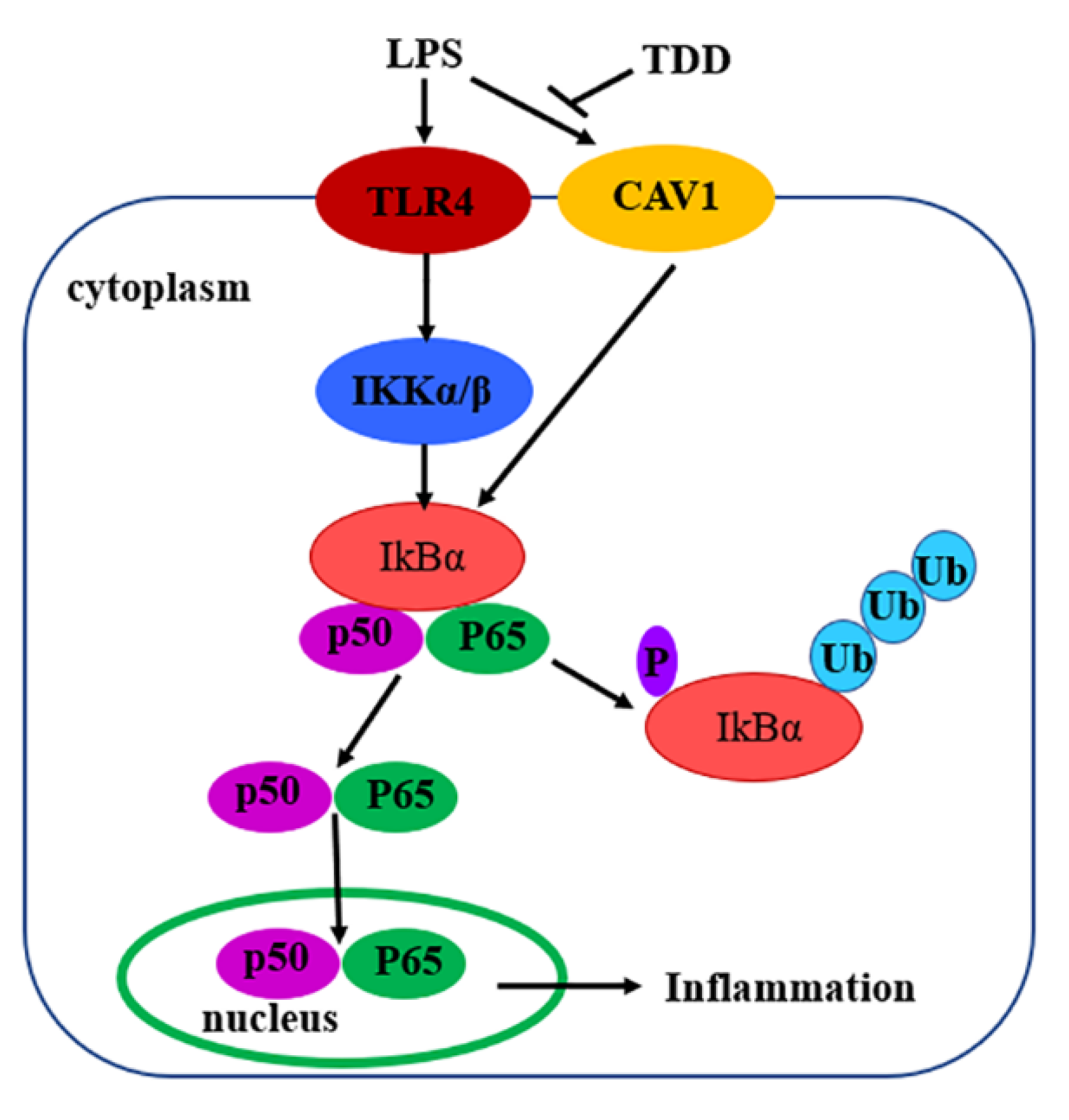
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Cell Culture
4.3. Animals and Treatments
4.4. Histopathological Evaluation
4.5. Enzyme-Linked Immunosorbent Assay (ELISA)
4.6. Quantitative Reverse Transcription-Polymerase Chain Reaction (qRT-PCR)
4.7. Western Blot Analysis
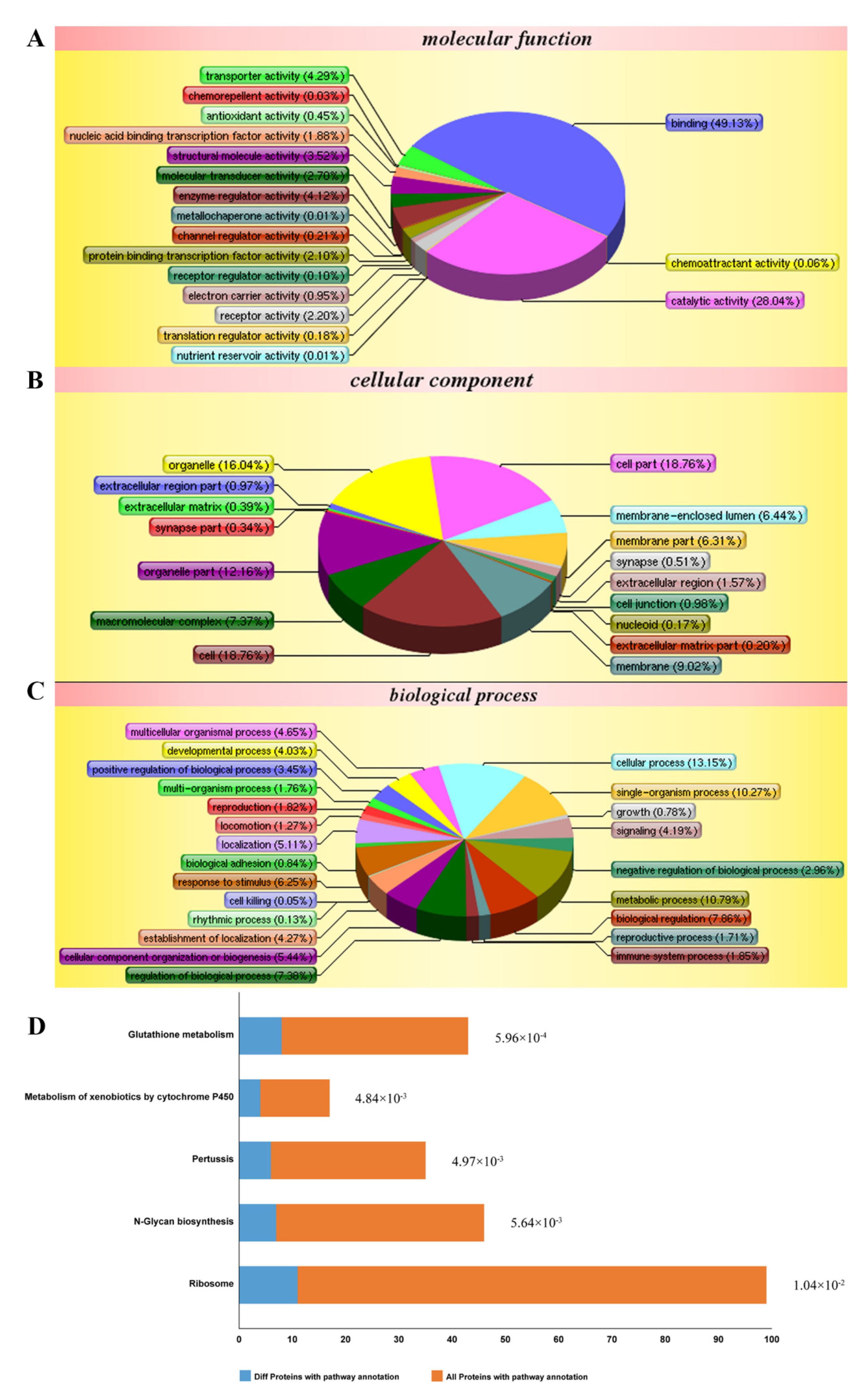
4.8. Isobaric Tag for the Relative and Absolute Quantification Technique (iTRAQ)
4.9. Cav1 Adenovirus Infection
4.10. BLI Assay
4.11. UV-vis Absorption Spectra
4.12. Fluorescence Spectrum
4.13. Molecular Docking
4.14. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Liu, Y.; Luo, X.; Jia, H.; Yu, B. The Effect of Blood Pressure Variability on Coronary Atherosclerosis Plaques. Front. Cardiovasc. Med. 2022, 9, 803810. [Google Scholar] [CrossRef]
- Jensen, J.K.; Binderup, T.; Grandjean, C.E.; Bentsen, S.; Ripa, R.S.; Kjaer, A. Semaglutide reduces vascular inflammation investigated by PET in a rabbit model of advanced atherosclerosis. Atherosclerosis, 2022; in press. [Google Scholar] [CrossRef]
- Marchio, P.; Guerra-Ojeda, S.; Vila, J.M.; Aldasoro, M.; Victor, V.M.; Mauricio, M.D. Targeting Early Atherosclerosis: A Focus on Oxidative Stress and Inflammation. Oxidative Med. Cell. Longev. 2019, 2019, 8563845. [Google Scholar] [CrossRef] [PubMed]
- Gencer, S.; Evans, B.R.; van der Vorst, E.P.C.; Doring, Y.; Weber, C. Inflammatory Chemokines in Atherosclerosis. Cells 2021, 10, 226. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela, C.A.; Baker, E.J.; De Souza, C.O.; Miles, E.A.; Calder, P.C. Differential Effects of Ruminant and Industrial 18-Carbon trans-Monounsaturated Fatty Acids (trans Vaccenic and Elaidic) on the Inflammatory Responses of an Endothelial Cell Line. Molecules 2021, 26, 5834. [Google Scholar] [CrossRef] [PubMed]
- Wiedermann, C.J.; Kiechl, S.; Dunzendorfer, S.; Schratzberger, P.; Egger, G.; Oberhollenzer, F.; Willeit, J. Association of endotoxemia with carotid atherosclerosis and cardiovascular disease. J. Am. Coll. Cardiol. 1999, 34, 1975–1981. [Google Scholar] [CrossRef] [Green Version]
- Losito, A.; Kalidas, K.; Santoni, S.; Errico, R.; Jeffery, S. Association of the -159C/T polymorphism of the endotoxin receptor (CD14) with carotid artery disease and cardiovascular mortality in dialysis patients. Blood Purif 2005, 23, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Videm, V.; Svennevig, J.L.; Fosse, E.; Mohr, B.; Aasen, A.O. Plasma endotoxin concentrations during cardiac surgery may be related to atherosclerosis. Perfusion 2000, 15, 421–426. [Google Scholar] [CrossRef]
- Reifenberg, K.; Lehr, H.A.; Fan, J.; Koike, T.; Wiese, E.; Kupper, I.; Sagban, T.A.; Schaefer, S.C.; Zahringer, U.; Torzewski, M.; et al. Endotoxin accelerates atherosclerosis independent of complement activation. Thromb. Res. 2009, 123, 653–658. [Google Scholar] [CrossRef]
- Gitlin, J.M.; Loftin, C.D. Cyclooxygenase-2 inhibition increases lipopolysaccharide-induced atherosclerosis in mice. Cardiovasc. Res. 2009, 81, 400–407. [Google Scholar] [CrossRef] [Green Version]
- Engelmann, M.G.; Redl, C.V.; Nikol, S. Recurrent perivascular inflammation induced by lipopolysaccharide (endotoxin) results in the formation of atheromatous lesions in vivo. Lab. Investig. 2004, 84, 425–432. [Google Scholar] [CrossRef]
- Suh, J.S.; Kim, S.; Boström, K.I.; Wang, C.-Y.; Kim, R.H.; Park, N.-H. Periodontitis-induced systemic inflammation exacerbates atherosclerosis partly via endothelial–mesenchymal transition in mice. Int. J. Oral Sci. 2019, 11, 201–212. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.; Wu, G.; Li, W.; Yang, J.; Yan, J.; Wang, Y.; Yao, W.; Zhou, X.; He, Z.; Wu, L.; et al. Preparation and protective effects of 1,8-cineole-loaded self-microemulsifying drug delivery system on lipopolysaccharide-induced endothelial injury in mice. Eur. J. Pharm. Sci. 2019, 127, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Han, S.; Zhou, Y.; Qi, B.; Wang, X. Therapeutic effects of mangiferin on sepsis-associated acute lung and kidney injuries via the downregulation of vascular permeability and protection of inflammatory and oxidative damages. Eur. J. Pharm. Sci. 2020, 152, 105400. [Google Scholar] [CrossRef] [PubMed]
- Pussinen, P.J.; Tuomisto, K.; Jousilahti, P.; Havulinna, A.S.; Sundvall, J.; Salomaa, V. Endotoxemia, immune response to periodontal pathogens, and systemic inflammation associate with incident cardiovascular disease events. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 1433–1439. [Google Scholar] [CrossRef] [Green Version]
- Yin, K.; Tang, S.L.; Yu, X.H.; Tu, G.H.; He, R.F.; Li, J.F.; Xie, D.; Gui, Q.J.; Fu, Y.C.; Jiang, Z.S.; et al. Apolipoprotein A-I inhibits LPS-induced atherosclerosis in ApoE(-/-) mice possibly via activated STAT3-mediated upregulation of tristetraprolin. Acta Pharmacol. Sin. 2013, 34, 837–846. [Google Scholar] [CrossRef] [Green Version]
- Haddad, D.; Al Madhoun, A.; Nizam, R.; Al-Mulla, F. Role of Caveolin-1 in Diabetes and Its Complications. Oxidative Med. Cell. Longev. 2020, 2020, 9761539. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Zhang, X.; Kong, W.; Wang, S. Reconstitution of Caveolin-1 into Artificial Lipid Membrane: Characterization by Transmission Electron Microscopy and Solid-State Nuclear Magnetic Resonance. Molecules 2021, 26, 6201. [Google Scholar] [CrossRef]
- Lin, H.; Zeng, Y.; Wang, Z. Maternal chronic intermittent hypoxia in rats causes early atherosclerosis with increased expression of Caveolin-1 in offspring. Sleep Breath. 2019, 23, 1071–1077. [Google Scholar] [CrossRef]
- Ramírez, C.M.; Zhang, X.; Bandyopadhyay, C.; Rotllan, N.; Sugiyama, M.G.; Aryal, B.; Liu, X.; He, S.; Kraehling, J.R.; Ulrich, V.; et al. Caveolin-1 Regulates Atherogenesis by Attenuating Low-Density Lipoprotein Transcytosis and Vascular Inflammation Independently of Endothelial Nitric Oxide Synthase Activation. Circulation 2019, 140, 225–239. [Google Scholar] [CrossRef]
- Pavlides, S.; Gutierrez-Pajares, J.L.; Iturrieta, J.; Lisanti, M.P.; Frank, P.G. Endothelial caveolin-1 plays a major role in the development of atherosclerosis. Cell Tissue Res. 2014, 356, 147–157. [Google Scholar] [CrossRef] [Green Version]
- Frank, P.G. Endothelial caveolae and caveolin-1 as key regulators of atherosclerosis. Am. J. Pathol 2010, 177, 544–546. [Google Scholar] [CrossRef] [PubMed]
- Frank, P.G.; Lee, H.; Park, D.S.; Tandon, N.N.; Scherer, P.E.; Lisanti, M.P. Genetic ablation of caveolin-1 confers protection against atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 98–105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Engel, D.; Beckers, L.; Wijnands, E.; Seijkens, T.; Lievens, D.; Drechsler, M.; Gerdes, N.; Soehnlein, O.; Daemen, M.J.; Stan, R.V.; et al. Caveolin-1 deficiency decreases atherosclerosis by hampering leukocyte influx into the arterial wall and generating a regulatory T-cell response. FASEB J. 2011, 25, 3838–3848. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, D.X.; Pan, Y.Q.; Liu, B.; Dai, L. Cav-1 promotes atherosclerosis by activating JNK-associated signaling. Biochem. Biophys. Res. Commun. 2018, 503, 513–520. [Google Scholar] [CrossRef]
- Fernandez-Hernando, C.; Yu, J.; Suarez, Y.; Rahner, C.; Davalos, A.; Lasuncion, M.A.; Sessa, W.C. Genetic evidence supporting a critical role of endothelial caveolin-1 during the progression of atherosclerosis. Cell Metab. 2009, 10, 48–54. [Google Scholar] [CrossRef] [Green Version]
- Fernandez-Hernando, C.; Yu, J.; Davalos, A.; Prendergast, J.; Sessa, W.C. Endothelial-specific overexpression of caveolin-1 accelerates atherosclerosis in apolipoprotein E-deficient mice. Am. J. Pathol. 2010, 177, 998–1003. [Google Scholar] [CrossRef]
- Wu, C.C.; Wang, S.H.; Kuan, I.I.; Tseng, W.K.; Chen, M.F.; Wu, J.C.; Chen, Y.L. OxLDL upregulates caveolin-1 expression in macrophages: Role for caveolin-1 in the adhesion of oxLDL-treated macrophages to endothelium. J. Cell. Biochem. 2009, 107, 460–472. [Google Scholar] [CrossRef]
- Farhat, N.; Thorin-Trescases, N.; Voghel, G.; Villeneuve, L.; Mamarbachi, M.; Perrault, L.P.; Carrier, M.; Thorin, E. Stress-induced senescence predominates in endothelial cells isolated from atherosclerotic chronic smokers. Can. J. Physiol. Pharmacol. 2008, 86, 761–769. [Google Scholar] [CrossRef]
- Majkova, Z.; Toborek, M.; Hennig, B. The role of caveolae in endothelial cell dysfunction with a focus on nutrition and environmental toxicants. J. Cell Mol. Med. 2010, 14, 2359–2370. [Google Scholar] [CrossRef]
- Li, J.; Feng, X.; Ge, R.; Li, J.; Li, Q. Protective Effect of 2,4′,5′-Trihydroxyl-5,2′-dibromo diphenylmethanone, a New Halophenol, against Hydrogen Peroxide-Induced EA.hy926 Cells Injury. Molecules 2015, 20, 14254–14264. [Google Scholar] [CrossRef] [Green Version]
- Yuan, H.X.; Feng, X.E.; Liu, E.L.; Ge, R.; Zhang, Y.L.; Xiao, B.G.; Li, Q.S. 5,2′-dibromo-2,4′,5′-trihydroxydiphenylmethanone attenuates LPS-induced inflammation and ROS production in EA.hy926 cells via HMBOX1 induction. J. Cell Mol. Med. 2019, 23, 453–463. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Cai, H.H.; Feng, X.E.; Li, Q.S. A novel marine halophenol derivative attenuates lipopolysaccharide-induced inflammation in RAW264.7 cells via activating phosphoinositide 3-kinase/Akt pathway. Pharmacol. Rep. PR 2020, 72, 1021–1031. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.S.; Feng, X.E.; Ban, S.R. The Application of Polyhydroxyl Bromo-Diphenylketone Compounds and their Derivatives in the Treatment and Prevention of Atherosclerosis; State Intellectual Property Office of the P.R.C.: Beijing, China, 2014. [Google Scholar]
- Lyu, S.; Wang, W. Spectroscopic methodologies and computational simulation studies on the characterization of the interaction between human serum albumin and astragalin. J. Biomol. Struct. Dyn. 2021, 39, 2959–2970. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Niu, L.; Liu, X.; Zhou, H.; Dong, H.; Kong, D.; Li, Y.; Li, Q. Spectroscopic studies and molecular docking on the interaction of organotin antitumor compound bis[2,4-difluoro-N-(hydroxy-kappaO)benzamidato-kappaO]diphenyltin(IV) with human cytochrome P450 3A4 protease. Spectrochim Acta A Mol. Biomol. Spectrosc. 2016, 163, 154–161. [Google Scholar] [CrossRef]
- Mai, J.; Li, Y.; Qiao, X.; Ji, X.; Li, Q. Exploration on the Interaction Ability of Antitumor Compound Bis-[2,6-difluoro-N-(hydroxyl-<kappa>O)benzamidato-<kappa>O]dibutylitin(IV) with Human Peroxisome Proliferator-Activated Receptor hPPARgamma. Bioinorg. Chem. Appl. 2018, 2018, 3063271. [Google Scholar] [CrossRef] [Green Version]
- Ross, P.D.; Subramanian, S. Thermodynamics of Protein Association Reactions: Forces Contributing to Stability. Biochemistry 1981, 20, 3096–3102. [Google Scholar] [CrossRef]
- Luo, J.; Wang, X.; Jiang, X.; Liu, C.; Li, Y.; Han, X.; Zuo, X.; Li, Y.; Li, N.; Xu, Y.; et al. Rutaecarpine derivative R3 attenuates atherosclerosis via inhibiting NLRP3 inflammasome-related inflammation and modulating cholesterol transport. FASEB J. 2020, 34, 1398–1411. [Google Scholar] [CrossRef] [Green Version]
- Jiang, T.; Jiang, D.; You, D.; Zhang, L.; Liu, L.; Zhao, Q. Agonism of GPR120 prevents ox-LDL-induced attachment of monocytes to endothelial cells. Chem.-Biol. Interact. 2020, 316, 108916. [Google Scholar] [CrossRef]
- Soysal, P.; Arik, F.; Smith, L.; Jackson, S.E.; Isik, A.T. Inflammation, Frailty and Cardiovascular Disease. Adv. Exp. Med. Biol. 2020, 1216, 55–64. [Google Scholar] [CrossRef] [Green Version]
- Onat, D.; Brillon, D.; Colombo, P.C.; Schmidt, A.M. Human vascular endothelial cells: A model system for studying vascular inflammation in diabetes and atherosclerosis. Curr. Diab. Rep. 2011, 11, 193–202. [Google Scholar] [CrossRef] [Green Version]
- Ahn, K.; Pan, S.; Beningo, K.; Hupe, D. A permanent human cell line (EA.hy926) preserves the characteristics of endothelin converting enzyme from primary human umbilical vein endothelial cells. Life Sci. 1995, 56, 2331–2341. [Google Scholar] [CrossRef]
- Bouïs, D.; Hospers, G.A.P.; Meijer, C.; Molema, G.; Mulder, N.H. Endothelium in vitro: A review of human vascular endothelial cell lines for blood vessel-related research. Angiogenesis 2001, 4, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Edgell, C.J.; McDonald, C.C.; Graham, J.B. Permanent cell line expressing human factor VIII-related antigen established by hybridization. Proc. Natl. Acad. Sci. USA 1983, 80, 3734–3737. [Google Scholar] [CrossRef] [Green Version]
- Suggs, J.E.; Madden, M.C.; Friedman, M.; Edgell, C.J. Prostacyclin expression by a continuous human cell line derived from vascular endothelium. Blood 1986, 68, 825–829. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.C.; Li, C.C.; Yang, Y.C.; Chiu, T.H.; Liu, K.L.; Lii, C.K.; Chen, H.W. Andrographis paniculata diterpenoids and ethanolic extract inhibit TNFalpha-induced ICAM-1 expression in EA.hy926 cells. Phytomedicine 2019, 52, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.; Zhang, X.; Miao, J.; Yuan, H.; Liu, E.; Liang, T.; Li, Q. Farrerol Directly Targets GSK-3beta to Activate Nrf2-ARE Pathway and Protect EA.hy926 Cells against Oxidative Stress-Induced Injuries. Oxidative Med. Cell. Longev. 2020, 2020, 5967434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaissarian, N.; Kang, J.; Shu, L.; Ferraz, M.J.; Aerts, J.M.; Shayman, J.A. Dissociation of globotriaosylceramide and impaired endothelial function in alpha-galactosidase-A deficient EA.hy926 cells. Mol. Genet. Metab. 2018, 125, 338–344. [Google Scholar] [CrossRef]
- Ladurner, A.; Atanasov, A.G.; Heiss, E.H.; Baumgartner, L.; Schwaiger, S.; Rollinger, J.M.; Stuppner, H.; Dirsch, V.M. 2-(2,4-dihydroxyphenyl)-5-(E)-propenylbenzofuran promotes endothelial nitric oxide synthase activity in human endothelial cells. Biochem. Pharmacol. 2012, 84, 804–812. [Google Scholar] [CrossRef] [Green Version]
- Schurr, J.R.; Young, E.; Byrne, P.; Steele, C.; Shellito, J.E.; Kolls, J.K. Central role of toll-like receptor 4 signaling and host defense in experimental pneumonia caused by Gram-negative bacteria. Infect. Immun. 2005, 73, 532–545. [Google Scholar] [CrossRef] [Green Version]
- Villacorta, L.; Chang, L.; Salvatore, S.R.; Ichikawa, T.; Zhang, J.; Petrovic-Djergovic, D.; Jia, L.; Carlsen, H.; Schopfer, F.J.; Freeman, B.A.; et al. Electrophilic nitro-fatty acids inhibit vascular inflammation by disrupting LPS-dependent TLR4 signalling in lipid rafts. Cardiovasc. Res. 2013, 98, 116–124. [Google Scholar] [CrossRef]
- Steven, S.; Hausding, M.; Kroller-Schon, S.; Mader, M.; Mikhed, Y.; Stamm, P.; Zinssius, E.; Pfeffer, A.; Welschof, P.; Agdauletova, S.; et al. Gliptin and GLP-1 analog treatment improves survival and vascular inflammation/dysfunction in animals with lipopolysaccharide-induced endotoxemia. Basic Res. Cardiol. 2015, 110, 6. [Google Scholar] [CrossRef] [PubMed]
- Steven, S.; Dib, M.; Roohani, S.; Kashani, F.; Munzel, T.; Daiber, A. Time Response of Oxidative/Nitrosative Stress and Inflammation in LPS-Induced Endotoxaemia-A Comparative Study of Mice and Rats. Int. J. Mol. Sci. 2017, 18, 2176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kanoh, H.; Iwashita, S.; Kuraishi, T.; Goto, A.; Fuse, N.; Ueno, H.; Nimura, M.; Oyama, T.; Tang, C.; Watanabe, R.; et al. cGMP signaling pathway that modulates NF-kappaB activation in innate immune responses. iScience 2021, 24, 103473. [Google Scholar] [CrossRef] [PubMed]
- Fan, B.; Liu, X.; Chen, X.; Xu, W.; Zhao, H.; Yang, C.; Zhang, S. Periostin Mediates Condylar Resorption via the NF-kappaB-ADAMTS5 Pathway. Inflammation 2020, 43, 455–465. [Google Scholar] [CrossRef]
- Chidlow, J.H., Jr.; Sessa, W.C. Caveolae, caveolins, and cavins: Complex control of cellular signalling and inflammation. Cardiovasc. Res. 2010, 86, 219–225. [Google Scholar] [CrossRef] [Green Version]
- Qin, L.; Zhu, N.; Ao, B.X.; Liu, C.; Shi, Y.N.; Du, K.; Chen, J.X.; Zheng, X.L.; Liao, D.F. Caveolae and Caveolin-1 Integrate Reverse Cholesterol Transport and Inflammation in Atherosclerosis. Int. J. Mol. Sci. 2016, 17, 429. [Google Scholar] [CrossRef] [Green Version]
- Zhao, W.; Feng, X.; Ban, S.; Lin, W.; Li, Q. Synthesis and biological activity of halophenols as potent antioxidant and cytoprotective agents. Bioorganic Med. Chem. Lett. 2010, 20, 4132–4134. [Google Scholar] [CrossRef]


| T(K) | Ksv (L·mol−1) × 106 | Kq (L·mol−1·s−1) × 1014 | R2 |
|---|---|---|---|
| 298 | 3.48 | 3.48 | 0.95 |
| 310 | 1.94 | 1.94 | 0.93 |
| T(K) | Kb(L·mol−1) | n | R2 |
|---|---|---|---|
| 298 | 1.09 × 109 | 1.3907 | 0.96 |
| 310 | 6.11 × 104 | 0.7436 | 0.96 |
| Gene | Forward Primer Sequence (5′-3′) | Reverse Primer Sequence (5′-3′) |
|---|---|---|
| GAPDH (human) | TGGTGAAGACGCCAGTGGA | GCACCGTCAAGGCTGAGAAC |
| TLR4 (human) | TAAGGTTGCCGCTTTCACTT | TGACCGAGCAGTTTCTGAGG |
| MCP-1 (human) | TCAGCCAGATGCAATCAATG | AGATCTCCTTGGCCACAATG |
| IL-6 (human) | GTGTGAAAGCAGCAAAGAG | CTCCAAAAGACCAGTGATG |
| TNF-α (human) | CCTGTGAGGAGGACGAACAT | TTTGAGCCAGAAGAGGTTGAG |
| IL-1β (human) | TGGCAGAAAGGGAACAGAAA | CTGGCTGATGGACAGGAGAT |
| GAPDH (mouse) | AGGTCGGTGTGAACGGATTTG | TGTAGACCATGTAGTTGAGGTCA |
| TLR4 (mouse) | GAGCCGGAAGGTTATTGTGGTAGTG | TCAAGGACAATGAAGATGATGCCAGAG |
| MCP-1 (mouse) | CCACTCACCTGCTGCTACTCATTC | CTGCTGCTGGTGATCCTCTTGTAG |
| IL-6 (mouse) | GATGGATGCTACCAAATGGA | CCAGGTAGCTATGGTACTCCAGAA |
| TNF-α (mouse) | CCTCTAGCCCACGTCGTAGC | AGCAATGACTCCAAAGTAGACC |
| IL-1β (mouse) | AGCAGCACATCAACAAGAGC | AGGTGCTCATGTCCTCATCC |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yuan, H.; Hou, Q.; Feng, X.; Zhang, Y.; Yang, F.; Ge, R.; Li, Q. 5,2′-Dibromo-2,4′,5′-trihydroxydiphenylmethanone Inhibits LPS-Induced Vascular Inflammation by Targeting the Cav1 Protein. Molecules 2022, 27, 2884. https://doi.org/10.3390/molecules27092884
Yuan H, Hou Q, Feng X, Zhang Y, Yang F, Ge R, Li Q. 5,2′-Dibromo-2,4′,5′-trihydroxydiphenylmethanone Inhibits LPS-Induced Vascular Inflammation by Targeting the Cav1 Protein. Molecules. 2022; 27(9):2884. https://doi.org/10.3390/molecules27092884
Chicago/Turabian StyleYuan, Hongxia, Qianyi Hou, Xiue Feng, Yuanlin Zhang, Fan Yang, Rui Ge, and Qingshan Li. 2022. "5,2′-Dibromo-2,4′,5′-trihydroxydiphenylmethanone Inhibits LPS-Induced Vascular Inflammation by Targeting the Cav1 Protein" Molecules 27, no. 9: 2884. https://doi.org/10.3390/molecules27092884





