Hyperoside: A Review of Its Structure, Synthesis, Pharmacology, Pharmacokinetics and Toxicity
Abstract
:1. Introduction
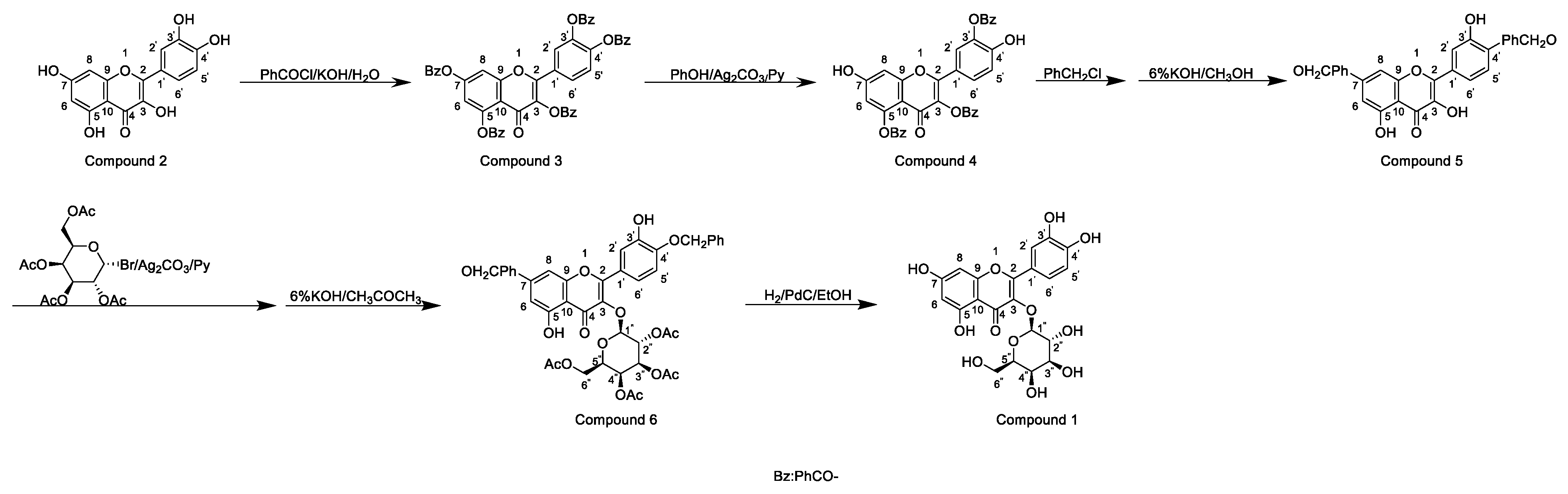
2. Structure and Synthesis
2.1. Chemical Synthesis
2.2. Biosynthesis
3. Pharmacology
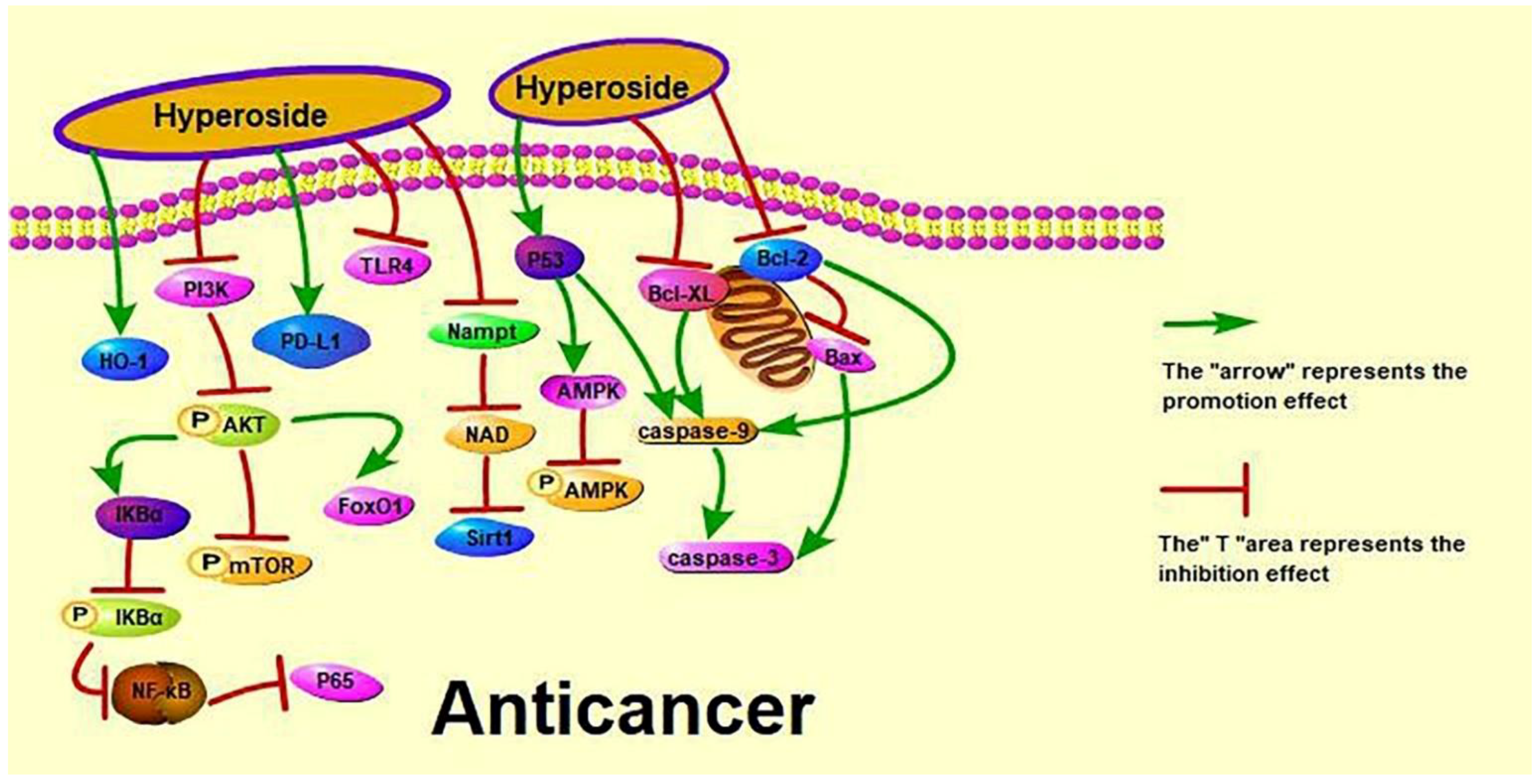
3.1. Anti-Cancer Activity
3.2. Effect on Lung Cancer
3.3. Effect on Cervical Cancer
3.4. Effect on Liver Cancer
3.5. Effect on Breast Cancer
3.6. Effect on Stomach Cancer
3.7. Effect on Other Cancers
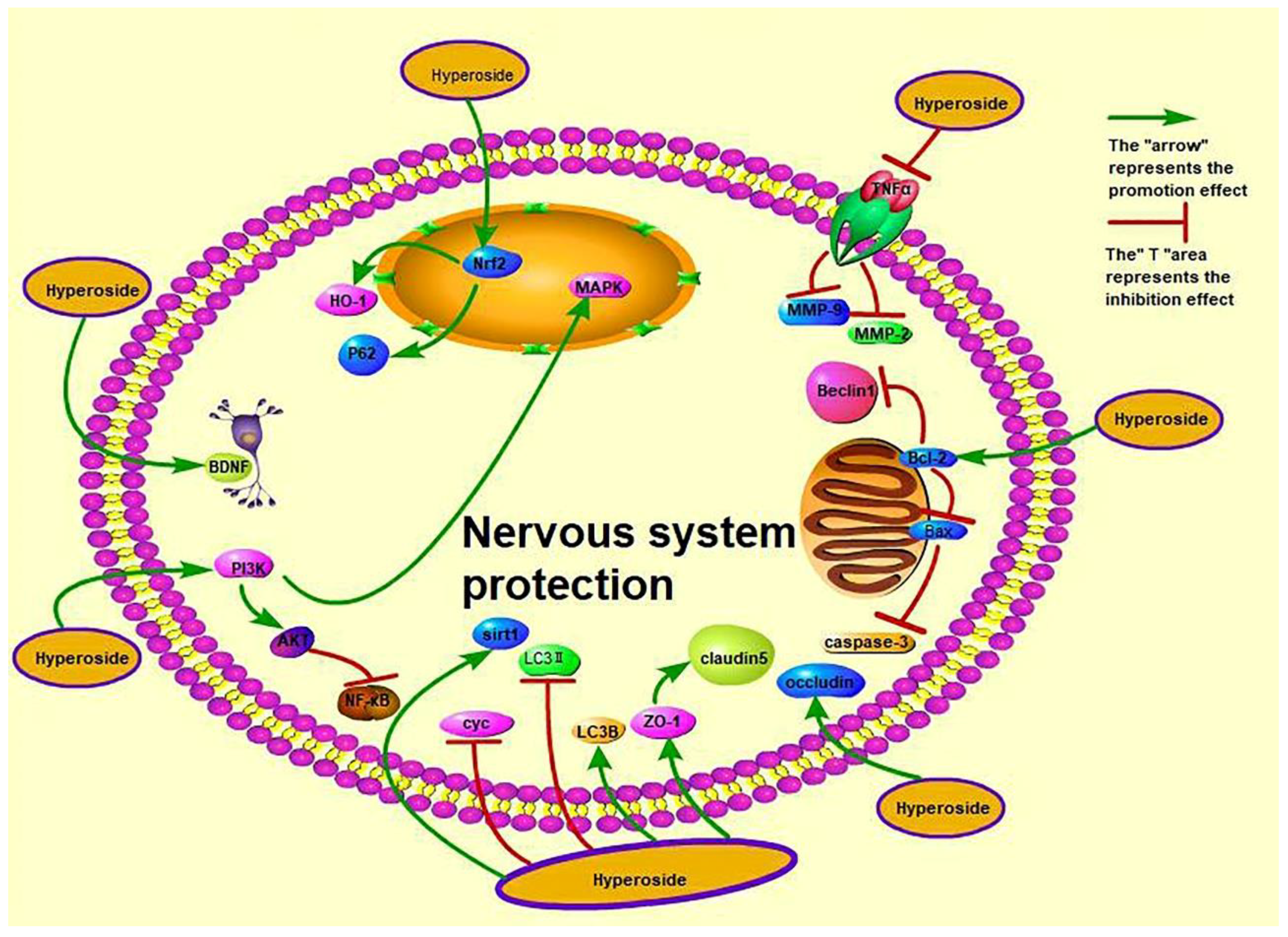
3.8. Brain Protection Activity
3.9. Neuroprotective Effect
3.10. Cardioprotective Activity
3.11. Hepatoprotective Activity
3.12. Renal Protective Activity
3.13. Protective Activity of Bone and Joint Diseases
3.14. Others
4. Pharmacokinetics
5. Toxicity
6. Conclusions and Perspective
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wang, J.; Peng, S.L.; Wang, M.K.; Chen, N.Y.; Ding, L.S. Chemical constituents of Hyperricum monogynum. China J. Chin. Mater. Med. 2002, 27, 43–45. [Google Scholar]
- Wu, Z.; Zhang, X. Simultaneous determination of isoquercetin and hyperoside in Hawthorn by HPLC. Jilin J. Tradit. Chin. Med. 2019, 39, 949–952. [Google Scholar]
- Xu, X.Y.; Zheng, Y.M.; Fu, S.Q.; Wang, A.L. Quantitative determination of hyperoside and quercitrin and luteolin in Polygonum aviculare by HPLC. Lishizhen Med. Mater. Med. Res. 2006, 4, 563–564. [Google Scholar]
- Feng, R.Y.; Zhu, X.Y.; Zhang, M.M. Research progress on pharmacological function of Hawthorn in the past five years. Chin. J. Basic Med. Tradit. Chin. Med. 2019, 25, 715–718. [Google Scholar]
- Yang, J.L.; Huang, L.D.; Zhang, Y.Z.; Wu, J. Research advances of Polygonum aviculare L. Anhui Med. Pharm. J. 2016, 20, 1025–1029. [Google Scholar]
- Wei, S.; Sun, Y.; Wang, L.; Zhang, T.F.; Hu, W.D.; Bao, W.X.; Mao, L.; Chen, J.X.; Li, H.J.; Wen, Y.K.; et al. Hyperoside suppresses BMP-7-dependent PI3K/AKT pathway in human hepatocellular carcinoma cells. Ann. Transl. Med. 2021, 9, 1233. [Google Scholar] [CrossRef]
- Jerzmanowska, Z. Hyperin, a glucoside of Hypericum perforatum L. Chem. Abstr. 1937, 64, 527. [Google Scholar]
- Gu, N.; Qiu, C.; Zhao, L.G.; Zhang, L.H.; Pei, J.J. Efficient production hyperoside from quercetin in escherichia coli through increasing UDP-galactose supply and recycling of resting cell. Catal. Lett. 2021, 151, 1202–1211. [Google Scholar] [CrossRef]
- Zeng, H.Y.; Zhou, P.H.; Pei, G. Studies on chemical constituents of Hypericum sampsonii. Nat. Prod. Res. Dev. 2002, 5, 50–53. [Google Scholar]
- Gao, T. Crataegus pinnatia Bge Chemical Composition and Quality Specification Research. Master’s Thesis, Shenyang Pharmaceutical University, Shenyang, China, 2009. [Google Scholar]
- Chen, J.W.; Zhu, Z.Q.; Hang, K.; Yang, X.N. Relationship between structure and activity of eight natural flavonoids against oxidation. J. East China Norm. Univ. Nat. Sci. 2002, 01, 90–95. [Google Scholar]
- Zhou, J. Synthesis of New Type Analgesic Drug-Hyperin and Its Derivatives. Master’s Thesis, Zhejiang University, Hangzhou, China, 2002. [Google Scholar]
- Wu, D.; Tang, L.; Duan, R.; Hu, X.; Geng, F.; Zhang, Y.; Peng, L.X.; Li, H. Interaction mechanisms and structure-affinity relationships between hyperoside and soybean beta-conglycinin and glycinin. Food Chem. 2021, 347, 129052. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.J.; Li, M.; Liu, F.G.; Peng, F.; Li, F.; Lou, X.M.; Jin, Y.; Wang, J.; Xu, H.D. Fabrication and characterization of zein-tea polyphenols-pectin ternary complex nanoparticles as an effective hyperoside delivery system: Formation mechanism, physicochemical stability, and in vitro release property. Food Chem. 2021, 364, 130335. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.J.; Peng, F.; Liu, F.G.; Xiao, Y.Q.; Li, F.; Lei, H.J.; Wang, J.; Li, M.; Xu, H.D. Zein-pectin composite nanoparticles as an efficient hyperoside delivery system: Fabrication, characterization, and in vitro release property. Lwt 2020, 133, 109869. [Google Scholar] [CrossRef]
- Li, Y.; Jiang, Y.; Liu, Q.; Zhai, G. Progress in research on biological activity of quercetin derivatives. Chin. Pharm. J. 2021, 56, 175–180. [Google Scholar]
- Qi, J.; Li, J.; Chen, L.; Liu, X.; Tan, H.; Zhu, W. Synthesis and SAR studies on hyperin and its analogues as 3CL protease inhibitors of human coronavirus 229E. Chin. J. Med. Chem. 2007, 17, 288–294. [Google Scholar]
- Hrhammer, L.; Wagner, H.; Arndt, H.G.; Dirscherl, R.; Farkas, L. Ber die synthese von quercetin-3-glykosiden; I. synthese und strukturbeweis von isoquercitrin, hyperosid und quercitrin. Eur. J. Inorg. Chem. 1968, 101, 450–453. [Google Scholar]
- Jiang, Z.L.; Zhu, Z.Y.; Wu, Y.H.; Wang, Z.; Zhou, W.S. Study on synthesis of hyperoside. Acta Pharm. Sin. 1994, 29, 874–876. [Google Scholar]
- Wang, Z.L.; Tang, W.Z.; Lin, F.R. Effects of hyperoside on apoptosis and antioxidative capacity of cervical cancer Hela cells. Chin. J. Coal Ind. Med. 2019, 22, 402–407. [Google Scholar]
- Bian, B.F.; Zhang, L.J.; Chen, S.H.; Wang, L.H.; Yang, L. Effect of hyperoside on the proliferation and migration of Hela cells via Nampt/NAD/Sirt1 pathway. Chin Pharm. 2021, 24, 624–629. [Google Scholar]
- Guo, W.K.; Yu, H.; Zhang, L.; Chen, X.W.; Liu, Y.D.; Wang, Y.X.; Zhang, Y.Y. Effect of hyperoside on cervical cancer cells and transcriptome analysis of differentially expressed genes. Cancer Cell Int. 2019, 19, 235. [Google Scholar] [CrossRef] [Green Version]
- Jiang, S.; Xiong, H.S.; Wu, H.K.; Wen, J.; Liang, L. Regulatory effect of hyperoside on proliferation and apoptosis of hepatic carcinoma cell HepG2 via mitochondrial P53/Caspase signaling pathway. Chin. J. Immunol. 2018, 34, 1832–1836. [Google Scholar]
- Dong, J.W.; Kuang, Z.A.; Yin, M.X.; Liu, X.J.; Liu, Y.; Deng, H.B. Hyperoside exerts its anti-tumor activity by reducing the PD-L1 level in non-small cell lung cancer. Acta Pharm. Sin. 2021, 56, 1–16. [Google Scholar]
- Chen, D.; Wu, Y.X.; Qiu, Y.B.; Wan, B.B.; Liu, G.; Chen, J.L.; Lu, M.D.; Pang, Q.F. Hyperoside suppresses hypoxia-induced A549 survival and proliferation through ferrous accumulation via AMPK/HO-1 axis. Phytomedicine 2020, 67, 153138. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.Y.; Zhao, P.J.; Xu, H.F. Hyperoside exhibits anticancer activity in non-small cell lung cancer cells with T790M mutations by upregulating FoxO1 via CCAT1. Oncol. Rep. 2020, 43, 617–624. [Google Scholar] [CrossRef] [PubMed]
- Li, J.P.; Liao, X.H.; Xiang, Y.; Yao, A.; Song, R.H.; Zhang, Z.J.; Huang, F.; Dai, Z.T.; Zhang, T.C. Hyperoside and let-7a-5p synergistically inhibits lung cancer cell proliferation via inducing G1/S. phase arrest. Gene 2018, 679, 232–240. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.X.; Zhang, T.; Zhu, X.Y.; Yang, C.; Wang, Y.X.; Zhou, N.; Ju, B.X.; Zhou, T.H.; Deng, G.Z.; Qiu, C.W. Hyperoside induces breast cancer cells apoptosis via ROS-mediated NF-kappa B signaling pathway. Int. J. Mol. Sci. 2020, 21, 131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, T.; Liu, Y.Y.; Li, M.D.; Yu, H.H.; Piao, H.Z. Administration with hyperoside sensitizes breast cancer cells to paclitaxel by blocking the TLR4 signaling. Mol. Cell. Probes 2020, 53, 101602. [Google Scholar] [CrossRef]
- Liu, H.W. Hyperoside Induces Apoptosis of Gastric Cancer Cell Line MKN-45 and Its Mechanism. Master’s Thesis, Jinzhou Medical University, Jinzhou, China, 2018. [Google Scholar]
- Wang, F.; Zhang, Y.M.; Qu, X.J.; Wang, Z.D.; Yang, T. Effect of hyperoside on the proliferation and apoptosis of human gastric cancer cells by inhibiting the nf-kb pathway. Acta Med. Mediterr. 2020, 36, 471–475. [Google Scholar]
- Xu, L.; Zhao, C.; Zhang, Y.W. Effects of hypericin on proliferation, apoptosis, migration and invasion of ovarian cancer cells. Chin. Tradit. Pat. Med. 2018, 40, 702–706. [Google Scholar]
- Xue, C.J.; Zhou, Y.; Xu, T.; Lu, X.T.; Zheng, L.; Zhou, Z.H. Enhancement effects of hyperoside on killing activity of human NK cells against pancreatic cancer PANC1 cells. Chin. J. Pancreatol. 2018, 18, 324–327. [Google Scholar]
- Kong, Y.H.; Sun, W.G.; Wu, P.F. Hyperoside exerts potent anticancer activity in skin cancer. Front. Biosci. (Landmark Ed) 2020, 25, 463–479. [Google Scholar]
- Liu, J.J.; Zhang, D.X.; Cao, J.W.; Yan, B.C. Neuroprotective effect and mechanism of hyperoside pretreatment on mice with cerebral ischemia-reperfusion injury. Shandong Med. J. 2021, 61, 15–19. [Google Scholar]
- Yu, Y.; Cai, J. Hypericin alleviates early brain injury following subarachnoid hemorrhage through regulating Akt/GSK-3β signaling pathway in rats. Zhejiang Med. J. 2020, 42, 2030–2036. [Google Scholar]
- Zhang, T.; Song, X.F.; Du, X.N.; Liu, Z.M. Protective effects of Chinese hawthorn leaf hyperoside against high glucose-induced injury in SH-SY5Y cells. Nat. Prod. Res. Dev. 2019, 31, 142. [Google Scholar]
- Zheng, M.Z.; Fan, Y.J.; Pan, Y.; Shi, D.F.; Liu, C.M. Studies on the antidepressant-like effect of hyperoside on the possible mechanism of 5-HT system. J. Changchun Norm. Uni. 2018, 37, 83–87. [Google Scholar]
- Cao, J.W.; Tang, C.; Gao, M.M.; Rui, Y.G.; Zhang, J.; Wang, L.; Wang, Y.; Xu, B.; Yan, B.C. Hyperoside alleviates epilepsy-induced neuronal damage by enhancing antioxidant levels and reducing autophagy. J. Ethnopharmacol. 2020, 257, 112884. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Famurewa, A.C.; Tang, J.; Olatunde, O.O.; Olatunji, O.J. Hyperoside attenuates neuroinflammation, cognitive impairment and oxidative stress via suppressing TNF-alpha/NF-kappa B/caspase-3 signaling in type 2 diabetes rats. Nutr. Neurosci. 2021, 24, 1–11. [Google Scholar]
- Fan, H.J.; Li, Y.R.; Sun, M.Y.; Xiao, W.S.; Song, L.J.; Wang, Q.; Zhang, B.; Yu, J.Z.; Jin, X.M.; Ma, C.G.; et al. Hyperoside reduces rotenone-induced neuronal injury by suppressing autophagy. Neurochem. Res. 2021, 46, 3149–3158. [Google Scholar] [CrossRef]
- Feng, Y.; Wang, D.X.; Wang, Q.; Li, Z.F.; Yang, S.L.; Feng, Y.L.; Luo, T.; Li, Y. Protective effects and mechanism of hyperoside in PC12 cells against oxidative stress injury induced by hydrogen peroxide. Nat. Prod. Commun. 2021, 16, 1–8. [Google Scholar] [CrossRef]
- Huang, J.; Zhou, L.; Chen, J.L.; Chen, T.B.; Lei, B.; Zheng, N.D.; Wan, X.Q.; Xu, J.G.; Wang, T.H. Hyperoside attenuate inflammation in HT22 cells via upregulating SIRT1 to activities Wnt/beta-catenin and sonic hedgehog pathways. Neural Plast. 2021, 2021, 8706400. [Google Scholar]
- Kwon, S.H.; Lee, S.R.; Park, Y.J.; Ra, M.; Lee, Y.; Pang, C.; Kim, K.H. Suppression of 6-hydroxydopamine-induced oxidative stress by hyperoside via activation of Nrf2/HO-1 signaling in dopaminergic neurons. Int. J. Mol. Sci. 2019, 20, 5832. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, C.Y.; Bai, K.; Liu, X.H.; Zhang, L.M.; Yu, G.R. Hyperoside protects the blood-brain barrier from neurotoxicity of amyloid beta 1–42. Neural Regen. Res. 2018, 13, 1974–1980. [Google Scholar] [PubMed]
- Orzelska-Gorka, J.; Szewczyk, K.; Gawronska-Grzywacz, M.; Kedzierska, E.; Glowacka, E.; Herbet, M.; Dudka, J.; Biala, G. Monoaminergic system is implicated in the antidepressant-like effect of hyperoside and protocatechuic acid isolated from impatiens glandulifera royle in mice. Neurochem. Int. 2019, 128, 206–214. [Google Scholar] [CrossRef] [PubMed]
- Li, G.D.; Xue, T.; Huang, X.F. Cardioprotective effects of hyperoside on myocardial ischemia-reperfusion injury in rats. Chin. J. Clin. Pharmacol. Ther. 2020, 36, 3721–3724. [Google Scholar]
- Liu, Z.X.; Yuan, Y.; Zhao, W.T.; Hu, L.P. Study on the protective effect of hypericin on diabetic myocardial injury. Asia-Pac. Tradit. Med. 2020, 16, 25–28. [Google Scholar]
- Wang, C.; Li, X.; Liu, Z.; Han, M.L.; Hou, Y.L.; Guo, C.L. The effect and mechanism of hyperoside on high glucose-induced Oxidative Stress Injury of Myocardial Cells. J. Sichuan Univ. (Med. Sci.) 2018, 49, 518–523. [Google Scholar]
- Wang, S.F. The Effect and Mechanism of Hyperoside Onattenuating Myocardial Ischemia-Reperfusion Injuryvia Activating PKC/mitoKATP Signaling Pathway Inrats. Master’s Thesis, WanNan Medical College, Wuhu, China, 2020. [Google Scholar]
- Wang, C.Y.; Li, J. Protective effects of hyperoside against myocardial injury in severely burned rats and its underlying mechanism. J. Shanxi Med. Univ. 2019, 50, 1718–1724. [Google Scholar]
- Xia, J.; Yao, X.L.; Liu, T.; Liu, B.Y.; Fang, Z.C. Mechanism of hyperoside improving myocardial injury in model rats with sepsis. China Pharm. 2021, 30, 21–25. [Google Scholar]
- Yang, Y.; Li, J.; Rao, T.; Zhang, J. Hyperoside ameliorates the injury of heart and thoracic aorta in mice with myocardial infarction by regulating autophagy pathway. Chin. J. Clin. Pharmacol. Ther. 2021, 26, 601–608. [Google Scholar]
- Zhang, C.Y.; Yang, Q. Effects of hyperoside on the myocardical activities of ATPases and expressions of Cx43 and Kir2.1 in arrhythmia rats induced by ischemia-reperfusion. Chin. Tradit. Pat. Med. 2018, 40, 254–260. [Google Scholar]
- Zhou, H.J.; Wang, F.F.; Bai, B.B.; Liu, P.Y.; Ji, Z.L. The protective effects of hyperoside on cardiac hypertrophy induced by transverse aortic constriction. Prog. Mod. Biomed. 2021, 21, 27–33. [Google Scholar]
- Ma, W.S.; Guo, W.G.; Shang, F.J.; Teng, J.W.; Ma, C. Effect of hyperin on ischemia reperfusion injury in H9C2 cells. Chin. J. Geriatr. Heart Brain Vessel Dis. 2019, 21, 294–298. [Google Scholar]
- Guo, X.; Zhang, Y.T.; Lu, C.H.; Qu, F.X.; Jiang, X.Y. Protective effect of hyperoside on heart failure rats via attenuating myocardial apoptosis and inducing autophagy. Biosci. Biotechnol. Biochem. 2020, 84, 714–724. [Google Scholar] [CrossRef] [PubMed]
- He, S.Y.; Yin, X.Q.; Wu, F.; Zeng, S.J.; Gao, F.; Xin, M.; Wang, J.; Chen, J.; Zhang, L.; Zhang, J.B. Hyperoside protects cardiomyocytes against hypoxia-induced injury via upregulation of microRNA-138. Mol. Med. Rep. 2021, 23, 286. [Google Scholar] [CrossRef]
- Wang, X.F.; Liu, Y.; Xiao, L.L.; Li, L.; Zhao, X.Y.; Yang, L.L.; Chen, N.; Gao, L.; Zhang, J.Y. Hyperoside protects against pressure overload-induced cardiac remodeling via the AKT signaling pathway. Cell Physiol. Biochem. 2018, 51, 827–841. [Google Scholar] [CrossRef]
- Yang, Y.K.; Li, J.; Rao, T.C.; Fang, Z.R.; Zhang, J.Y. The role and mechanism of hyperoside against myocardial infarction in mice by regulating autophagy via NLRP1 inflammation pathway. J. Ethnopharmacol. 2021, 276, 114187. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, Y.J.; Liu, L.J. Hyperoside prevents sepsis-associated cardiac dysfunction through regulating cardiomyocyte viability and inflammation via inhibiting miR-21. Biomed. Pharmacother. 2021, 138, 111524. [Google Scholar] [CrossRef]
- Cai, Y.Q.; Li, B.; Peng, D.; Wang, X.F.; Li, P.; Huang, M.C.; Xing, H.Y.; Chen, J.H. Crm1-dependent nuclear export of Bach1 is involved in the protective effect of hyperoside on oxidative damage in hepatocytes and CCl4-induced acute liver injury. J. Inflamm. Res. 2021, 14, 551–565. [Google Scholar] [CrossRef]
- Guo, X.; Zhu, C.Z.; Liu, X.J.; Ge, Y.P.; Jiang, X.Y.; Zhao, W. Hyperoside protects against heart failure-induced liver fibrosis in rats. Acta Histochem. 2019, 121, 804–811. [Google Scholar] [CrossRef]
- Jiang, Z.T.; Wang, J.C.; Liu, C.; Wang, X.; Pan, J.H. Hyperoside alleviated N-acetyl-para-amino-phenol-induced acute hepatic injury via Nrf2 activation. Int. J. Clin. Exp. Pathol. 2019, 12, 64–76. [Google Scholar]
- Shi, Y.P.; Qiu, X.X.; Dai, M.J.; Zhang, X.B.; Jin, G.X. Hyperoside attenuates hepatic ischemia-reperfusion injury by suppressing oxidative stress and inhibiting apoptosis in rats. Transplant. Proc. 2019, 51, 2051–2059. [Google Scholar] [CrossRef]
- Sun, B.; Zhang, R.T.; Liang, Z.C.; Fan, A.Q.; Kang, D.M. Hyperoside attenuates non-alcoholic fatty liver disease through targeting Nr4A1 in macrophages. Int. Immunopharmacol. 2021, 94, 107438. [Google Scholar] [CrossRef] [PubMed]
- Xing, H.Y.; Fu, R.Q.; Cheng, C.Y.; Cai, Y.Q.; Wang, X.F.; Deng, D.M.; Gong, X.Y.; Chen, J.H. Hyperoside protected against oxidative stress-induced liver injuryviathe PHLPP2-AKT-GSK-3 beta signaling pathway in vivo and in vitro. Front. Pharmacol. 2020, 11, 1065. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.L.; Wang, M.M.; Dong, H.H.; Yu, X.M.; Zhang, J.F. Anti-hypoglycemic and hepatocyte-protective effects of hyperoside from Zanthoxylum bungeanum leaves in mice with high-carbohydrate/high-fat diet and alloxan-induced diabetes. Int. J. Mol. Med. 2018, 41, 77–86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, F.F.; Chen, Z.Z.; Xu, Y.W.; Wan, H.F.; Lin, H.Y. Study on mechanism of hyperoside protecting cerebral ischemia reperfusion injury in rats. Chin. J. Mod. Appl. Pharm. 2021, 38, 1448–1453. [Google Scholar]
- Xu, H.H. Mechanism of Hyperoside Regulating Cerebrovascular trpv4 through ip3/pkc Signaling Pathway to Ameliorate Ischemic Cerebral Apoplexy. Master’s Thesis, WanNan Medical College, Wuhu, China, 2019. [Google Scholar]
- Zhou, L.; Xiong, Y.; Peng, A.L. Brain protective effect of Lilium lancifolium hypericin on rats with cerebral ischemia reperfusion injury. Mod. Food Sci. Technol. 2019, 35, 1–6. [Google Scholar]
- He, J.T.; Li, H.Q.; Li, G.F.; Yang, L. Hyperoside protects against cerebral ischemia-reperfusion injury by alleviating oxidative stress, inflammation and apoptosis in rats. Biotechnol. Biotechnol. Equip. 2019, 33, 798–806. [Google Scholar] [CrossRef] [Green Version]
- Yildiz Deniz, G.; Altun, S. Evaluation of nickel-induced brain injuries in rats via oxidative stress and apoptosis: Attenuating effects of hyperoside. Turk. J. Zool. 2020, 44, 104–113. [Google Scholar] [CrossRef]
- Gong, C.Z. Protective Effect of Hyperoside on LPS-Induced Acute Kidney Injury and the Underlying Mechanisms. Ph.D. Thesis, Shandong University, Jinan, China, 2019. [Google Scholar]
- Liu, B.H. Hyperoside Attenuates Renal Ageing Induced by D-galactose via Regulating AMPK-ULK1 Signalling-Mediated Autophagy. Master’s Thesis, Nanjing University of Chinese Medicine, Nanjing, China, 2019. [Google Scholar]
- Chen, Y.L.; Ye, L.H.; Li, W.J.; Li, D.Z.; Li, F. Hyperoside protects human kidney-2 cells against oxidative damage induced by oxalic acid. Mol Med Rep. 2018, 18, 486–494. [Google Scholar] [CrossRef] [Green Version]
- Liu, B.H.; Tu, Y.; He, W.M.; Liu, Y.L.; Wu, W.; Fang, Q.J.; Tang, H.T.; Tang, R.M.; Wan, Z.Y.; Sun, W.; et al. Hyperoside attenuates renal aging and injury induced by D-galactose via inhibiting AMPK-ULK1 signaling-mediated autophagy. Aging-Us 2018, 10, 4197–4212. [Google Scholar] [CrossRef]
- Wu, L.; Li, Q.; Liu, S.M.; An, X.F.; Huang, Z.M.; Zhang, B.; Yuan, Y.G.; Xing, C.Y. Protective effect of hyperoside against renal ischemia-reperfusion injury via modulating mitochondrial fission, oxidative stress, and apoptosis. Free Radic. Res. 2019, 53, 727–736. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Dai, Q.; Hu, L.L.; Yu, H.; Qiu, J.; Zhou, J.Y.; Long, M.; Zhou, S.W.; Zhang, K.B. Hyperoside alleviates high glucose-induced proliferation of mesangial cells through the inhibition of the ERK/CREB/miRNA-34a signaling pathway. Int. J. Endocrinol. 2020, 2020, 1361924. [Google Scholar] [CrossRef]
- Zhou, J.B.; Zhang, S.; Sun, X.Y.; Lou, Y.; Bao, J.J.; Yu, J.Y. Hyperoside ameliorates diabetic nephropathy induced by STZ via targeting the miR-499-5p/APC axis. J. Pharmacol. Sci. 2021, 146, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.X.; Li, L.M.; Shi, J.Q. Effects and mechanism of hyperin against bleomycin-induced pulmonary fibrosis in mice. Chin. J. Public Health 2018, 34, 1237–1241. [Google Scholar]
- Gao, Y.; Fan, X.Y.; Gu, W.J.; Ci, X.X.; Peng, L.P. Hyperoside relieves particulate matter-induced lung injury by inhibiting AMPK/mTOR-mediated autophagy deregulation. Pharmacol. Res. 2021, 167, 105561. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.Z.; Tong, X.; Zhang, L.; Zhang, Y.; Wang, L.; Wang, D.G.; Zhang, S.J.; Fan, H. Hyperoside attenuates bleomycin-induced pulmonary fibrosis development in mice. Front. Pharmacol. 2020, 11, 550955. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Zhao, Y.H.; Lu, J.M.; Chen, S.H.; Zhang, X.G.; Mao, W.W. Hyperoside inhibits proinflammatory cytokines in human lung epithelial cells infected with Mycoplasma pneumoniae. Mol. Cell. Biochem. 2019, 453, 179–186. [Google Scholar] [CrossRef]
- Liu, C.; Min, D.Y.; Zhu, J.H.; Yuan, Y.; Guan, X.J.; Hu, L.P. Experimental study of hyperoside on process of atherosclerosis by regulating vascular endothelium via NOS/NO system. Drug Eval. Res. 2021, 44, 971–977. [Google Scholar]
- Wang, C.; Guo, C.L.; Li, X.; Liu, Z.; Han, M.L.; Hou, Y.L. The antihypertensive effect of hyperoside as a inhibitor of ACE. Pharmacol. Clin. Chin. Mater. Med. 2018, 34, 33–39. [Google Scholar]
- Jang, S.A.; Park, D.W.; Sohn, E.H.; Lee, S.R.; Kang, S.C. Hyperoside suppresses tumor necrosis factor alpha-mediated vascular inflammatory responses by downregulating mitogen-activated protein kinases and nuclear factor-kappa B signaling. Chem. Biol. Interact. 2018, 294, 48–55. [Google Scholar] [CrossRef]
- Wang, X.B.; Zuo, R.T. Effects of hyperin on sacroiliacjoint of mice with IL-1β-induced chondrocyte injury. Chin. Tradit. Pat. Med. 2021, 43, 369–373. [Google Scholar]
- Chen, Y.Q.; Dai, F.; He, Y.; Chen, Q.; Xia, Q.; Cheng, G.; Lu, Y.X.; Zhang, Q. Beneficial effects of hyperoside on bone metabolism in ovariectomized mice. Biomed. Pharmacother. 2018, 107, 1175–1182. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.C.; Li, B.; Wu, W.L.; Liu, H.C.; Jiang, Y.P. Protective effect of hyperoside against hydrogen peroxide-induced dysfunction and oxidative stress in osteoblastic MC3T3-E1 cells. Artif. Cells Nanomed. Biotechnol. 2020, 48, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zhang, X.F. Hyperoside decreases the apoptosis and autophagy rates of osteoblast MC3T3-E1 cells by regulating TNF-like weak inducer of apoptosis and the p38mitogen activated protein kinase pathway. Mol. Med. Rep. 2019, 19, 41–50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fu, Q.; Jin, X.N.; Gao, Y.Z.; Wang, J.; Yan, P. Effect of hyperoside on synoviocytes of rheumatoid arthritis and its mechanism. J. Jinzhou Med. Uni. 2020, 41, 7–12. [Google Scholar]
- Jin, X.N.; Gao, W.; Feng, X.; Sui, H.J.; Fu, Q. Therapeutic effect of hyperoside on mice with collagen-induced arthritis. J. Pract. Med. 2021, 37, 2199–2203. [Google Scholar]
- Lin, Y.H.; Kong, F.Y.; Liang, R.M. Effect of Hyperoside on LPS-induced osteoarthritis in vitro. J. Snake 2020, 32, 420–423. [Google Scholar]
- Sun, K.; Luo, J.H.; Jing, X.Z.; Xiang, W.; Guo, J.C.; Yao, X.D.; Liang, S.; Guo, F.J.; Xu, T. Hyperoside ameliorates the progression of osteoarthritis: An in vitro and in vivo study. Phytomedicine 2021, 80, 153387. [Google Scholar] [CrossRef]
- Yu, X.P.; Cai, Y.Q.; Zhou, S.Y.; Cheng, H.; Zhou, W.Y.; Li, X.L. Therapeutic effect of hyperoside in rats with ulcerative colitis and its mechanism. Immunol. J. 2021, 37, 417–424. [Google Scholar]
- Ma, W.R.; Tan, Y. The effect and mechanism of hyperin on ovarian reserve of tripterygium glycosides-induced POI mice. J. Sichuan Univ. Med. Sci. 2021, 52, 458–466. [Google Scholar]
- Wang, X.X.; Fan, G.M.; Wei, F.M.; Bu, Y.; Huang, W.H. Hyperoside protects rat ovarian granulosa cells against hydrogen peroxide-induced injury by sonic hedgehog signaling pathway. Chem. Biol. Interact. 2019, 310, 108759. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.B.; Zhang, S.; Sun, X.Y.; Lou, Y.; Yu, J.Y. Hyperoside protects HK-2 cells against high glucose-induced apoptosis and inflammation via the miR-499a-5p/NRIP1 pathway. Pathol. Oncol. Res. 2021, 27, 629829. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Jin, H.L.; Jang, D.S.; Jeong, K.W.; Choung, S.Y. Hyperoside (quercetin-3-O-beta-D-galactopyranoside) protects A2E-laden retinal pigmented epithelium cells against UVA and blue light-induced apoptosis in vitro and in vivo. J. Funct. Foods 2018, 40, 426–437. [Google Scholar] [CrossRef]
- Wu, W.; Xie, Z.L.; Zhang, Q.; Ma, Y.Q.; Bi, X.T.; Yang, X.; Li, B.; Chen, J.H. Hyperoside ameliorates diabetic retinopathy via anti-oxidation, inhibiting cell damage and apoptosis induced by high glucose. Front. Pharmacol. 2020, 11, 797. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Wu, X.; Zhou, Z.; Ye, Y.; Yan, C.T.; Zhuge, N.S.; Yu, J.H. Hyperoside ameliorates periodontitis in rats by promoting osteogenic differentiation of BMSCs via activation of the NF-kappa B pathway. Febs. Open Bio. 2020, 10, 1843–1855. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Yao, X.Y.; Li, H.B. Study on the inhibitory effects of hyperoside on lipopolysaccharides-induced inflammatory response in mouse macrophage RAW264.7. Chin. J. Basic Med. Tradit. Chin. Med. 2021, 27, 944–949. [Google Scholar]
- Zhou, Y.Q.; Zhao, Y.T.; Zhao, X.Y.; Liang, C.; Xu, Y.W.; Li, L.; Liu, Y.; Yang, H.B. Hyperoside suppresses lipopolysaccharide-induced inflammation and apoptosis in human umbilical vein endothelial cells. Curr. Med. Sci. 2018, 38, 222–228. [Google Scholar] [CrossRef]
- Zhang, Y.L.; Yu, X.M.; Wang, M.M.; Ding, Y.; Guo, H.; Liu, J.K.; Cheng, Y. Hyperoside from Z. bungeanum leaves restores insulin secretion and mitochondrial function by regulating pancreatic cellular redox status in diabetic mice. Free Radic. Biol. Med. 2021, 162, 412–422. [Google Scholar] [CrossRef]
- Berkoz, M. Effect of hyperoside on the inhibition of adipogenesis in 3t3-L1 adipocytes. Acta Endocrinol. (Buchar. Rom.) 2019, 15, 165–172. [Google Scholar] [CrossRef]
- Zheng, C.C.; Yang, Y.; Wei, Y.; Zhou, X.M.; Gao, J.Y. Study on the effects and mechanism of hyperoside on anti-fatigue in mice. Sci. Technol. Food Ind. 2021, 42, 351–355. [Google Scholar]
- Zhou, Y.; Gao, X.N.; Lv, X.T.; Sun, L.Q.; Chen, L.; Zheng, L.; Zhou, Z.H. Enhancement of NK cells proliferation and function by hyperoside. Chin. J. Immunol. 2019, 35, 1569–1572. [Google Scholar]
- Wei, A.W.; Song, Y.L.; Ni, T.T.; Xiao, H.D.Z.; Wan, Y.R.; Ren, X.X.; Li, H.J.; Xu, G.L. Hyperoside attenuates pregnancy loss through activating autophagy and suppressing inflammation in a rat model. Life Sci. 2020, 254, 117735. [Google Scholar] [CrossRef]
- Wei, A.W.; Xiao, H.D.Z.; Xu, G.L.; Yu, X.L.; Guo, J.J.; Jing, Z.Q.; Shi, S.Q.; Song, Y.L. Hyperoside protects human umbilical vein endothelial cells against anticardiolipin antibody-induced injury by activating autophagy. Front. Pharmacol. 2020, 11, 762. [Google Scholar] [CrossRef]
- Chen, W.; Zheng, R.; Baade, P.D.; Zhang, S.; Zeng, H.; Bray, F.; Jemal, A.; Yu, X.Q.; He, J. Cancer statistics in China, 2015. CA Cancer J. Clin. 2016, 66, 115–132. [Google Scholar] [CrossRef] [Green Version]
- Schiller, J.H.; David, H.; Belani, C.P.; Corey, L.; Alan, S.; James, K.; Junming, Z.; Johnson, D.H.; Group, E. Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. N. Engl. J. Med. 2018, 346, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Carlos, S.J.; De, A.; Dias, G.; Chada, B.E. Cervical cancer: What’s new? Rev. Assoc. Med. Bras. 2015, 61, 536–542. [Google Scholar]
- Arakawa, A.; Ichikawa, H.; Kubo, T.; Motoi, N.; Kumamoto, T.; Nakajima, M.; Yonemori, K.; Noguchi, E.; Sunami, K.; Shiraishi, K.; et al. Vaginal transmission of cancer from mothers with cervical cancer to infants. N. Engl. J. Med. 2021, 384, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.L. Diet and health care of patients with liver cancer. Diet and Health 2020, 18, 20–22. [Google Scholar]
- Han, J.X. Hyperoside Inhibits Tumor Progress by Targeting YY1-p65 Complex. Master’s Thesis, Nankai University, Tianjin, China, 2018. [Google Scholar]
- Hu, C.; Chen, Y.; Cao, Y.Y.; Jia, Y.Q.; Zhang, J.Q. Metabolomics analysis reveals the protective effect of quercetin-3-O-galactoside (Hyperoside) on liver injury in mice induced by acetaminophen. J. Food Biochem. 2020, 44, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.T.; Shen, S.J.; Sun, Q. Current situation and progress of breast cancer screening in China. Chin. J. Front. Med. Sci. 2020, 12, 6–11. [Google Scholar]
- Esteva, A.; Kuprel, B.; Novoa, R.A.; Ko, J.; Swetter, S.M.; Blau, H.M.; Thrun, S. Dermatologist-level classification of skin cancer with deep neural networks. Nature 2017, 542, 115–118. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Zhang, J.; Ma, C. Protective effect of hyperin on cerebral infarction in rats. Chin. J. Chin. Miner. Med. 1998, 23, 51–53. [Google Scholar]
- Chen, Z.; Ma, C.; Zhao, W. Protective effect of hyperin against cerebral ischemia-reperfusion injury. Acta Pharm. Sin. 1998, 33, 15–18. [Google Scholar]
- Haas, J.S.; Stolz, E.D.; Betti, A.H.; Stein, A.C.; Schripsema, J.; Poser, G.L.; Rates, S.M. The anti-immobility effect of hyperoside on the forced swimming test in rats is mediated by the D2-like receptors activation. Planta Med. 2011, 77, 334–339. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Yang, Y.; Chen, X.; Yang, M.; Huang, D.; Yang, R.; Zhou, L.; Li, C.; Xiong, Q.; Xiong, Z. Hyperoside protects against chronic mild stress-induced learning and memory deficits. Biomed. Pharmacother. 2017, 91, 831–840. [Google Scholar] [CrossRef] [PubMed]
- Butterweck, V.; Hegger, M.; Winterhoff, H. Flavonoids of St. John’s Wort reduce HPA axis function in the rat. Planta Med. 2004, 70, 1008–1011. [Google Scholar] [CrossRef]
- Han, J.; Xuan, J.L.; Hu, H.R.; Chen, Z.W. Protective effect against myocardial ischemia reperfusion injuries induced by hyperoside preconditioning and its relationship with PI3K/Akt signaling pathway in rats. Zhongguo Zhong Yao Za Zhi 2015, 40, 126–131. [Google Scholar]
- Xing, H.; Fu, R.; Cheng, C.; Cai, Y.; Wang, X.; Deng, D.; Gong, X.; Chen, J. Hyperoside protected against oxidative stress-induced liver injury via the PHLPP2-AKT-GSK-3β signaling pathway in vivo and in vitro. Front Pharmacol. 2020, 11, 1065. [Google Scholar] [CrossRef]
- Zhu, W.; Xu, Y.F.; Feng, Y.; Peng, B.; Che, J.P.; Liu, M.; Zheng, J.H. Prophylactic effects of quercetin and hyperoside in a calcium oxalate stone forming rat model. Urolithiasis 2014, 42, 519–526. [Google Scholar] [CrossRef]
- Yan, Y.; Feng, Y.; Li, W.; Che, J.P.; Wang, G.C.; Liu, M.; Zheng, J.H. Protective effects of quercetin and hyperoside on renal fibrosis in rats with unilateral ureteral obstruction. Exp. Ther. Med. 2014, 8, 727–730. [Google Scholar] [CrossRef] [Green Version]
- Liu, C.; Yuan, Y.; Zhao, W.T.; Hu, L.P. Prediction of target and mechanism of hyperoside in atherosclerosis treatment by using network pharmacology methods. Mod. Chin. Med. 2018, 20, 684–690. [Google Scholar]
- Wang, T.T.; Su, M.T.; Gao, H.; Liang, J.L.; Du, B.L.; Cao, Z.Q.; Zheng, Y.Z. Effects of hypericin on blood lipid and arterial plaque formation in ApoE -/- mice. Chin. Pract. Med. 2018, 13, 196–197. [Google Scholar]
- Lee, S.; Jung, S.H.; Lee, Y.S.; Yamada, M.; Kim, B.K.; Ohuchi, K.; Shin, K.H. Antiinflammatory activity of hyperin from Acanthopanax chiisanensis roots. Arch. Pharm. Res. 2004, 27, 628–632. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Yu, X.; Sun, H.; Zhang, W.; Liu, G.; Zhu, L. Flos lonicerae flavonoids attenuate experimental ulcerative colitis in rats via suppression of NF-κB signaling pathway. Naunyn-Schmiedeb. Arch. Pharmacol. 2020, 393, 2481–2494. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Wang, Z.; Guo, Y.Y.; Zhang, X.N.; Xu, Z.H.; Liu, S.B.; Guo, H.J.; Yang, Q.; Zhang, F.X.; Sun, X.L.; et al. A role of periaqueductal grey NR2B-containing NMDA receptor in mediating persistent inflammatory pain. Mol. Pain. 2009, 5, 71. [Google Scholar] [CrossRef] [Green Version]
- Gao, Y.T.; Fang, L.Y.; Wang, X.X.; Lan, R.N.; Wang, M.Y.; Du, G.; Guan, W.Q.; Liu, J.F.; Brennan, M.; Guo, H.X.; et al. Antioxidant activity evaluation of dietary flavonoid hyperoside using saccharomyces cerevisiae as a model. Molecules 2019, 24, 788. [Google Scholar] [CrossRef] [Green Version]
- Zhang, W.Y.; Zhang, W.W.; Zhang, X.; Lu, Q.; Cai, H.B.; Tan, W.S. Hyperoside promotes ex vivo expansion of hematopoietic stem/progenitor cells derived from cord blood by reducing intracellular ROS level. Process Biochem. 2018, 72, 143–151. [Google Scholar] [CrossRef]
- Wang, D.X. Analysis of Different Components in Different Parts of Acanthopanax senticosus and Protective Mechanism of Hyperoside in the Oxidative Damage. Master’s Thesis, Jiangxi University of Traditional Chinese Medicine, Nanchang, China, 2020. [Google Scholar]
- Pan, S.S. Multi-Omics Analysis of the Mechanism in Lipid Metabolism by Hyperinin Mice. Master’s Thesis, Liaoning Normal University, Dalian, China, 2021. [Google Scholar]
- Wang, M.Y. Study on the Preparation of Luteoloside and Hyperoside Reference Materials and Their Antioxidant Activities. Master’s Thesis, Tianjin University of Technology, Tianjin, China, 2020. [Google Scholar]
- Ni, C.R.; Yuan, S.J. Quantitation of hyperoside in rat plasma by LC/MS and its application to pharmacokinetics study. Guiding J. Tradit. Chin. Med. Pharm. 2020, 26, 61–64. [Google Scholar]
- Zhu, X.F. Determination of hyperoside in the extract of Senecionis Scandens herba and Qianbai Biyan capsule by HPLC and its pharmacokinetics in rats. Shandong Chem. Ind. 2019, 48, 71–74. [Google Scholar]
- Chen, S.S.; Wu, D.L.; Zhang, W.; Liu, J.S.; Xu, F.Q.; Wang, T.S. Effects of different compatibilities of Wuzi Yanzong pill on the absorption and distribution of hyperoside in rats. Chin. J. Integr. Tradit. West. Med. 2021, 41, 79–85. [Google Scholar]
- Chen, S.S. Effects of Different Compatibility on the Content and Pharmacokinetics of Hyperoside in Cuscuta chinensis Lam. Master’s Thesis, Anhui University of Traditional Chinese Medicine, Hefei, China, 2019. [Google Scholar]
- Yuan, W.J.; Wang, J.J.; An, X.F.; Dai, M.X.; Jiang, Z.Z.; Zhang, L.Y.; Yu, S.; Huang, X. UPLC-MS/MS method for the determination of hyperoside and application to pharmacokinetics study in rat after different administration routes. Chromatographia 2021, 84, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xu, S.Y.; Jia, Z.; Han, T.; Liu, M.N.; Jia, T.Y.; Qu, W.J.; Xu, X.F.; Li, X.R. UPLC-MS/MS determination of chlorogenic acid, hyperoside and astragalin in plasma and its pharmacokinetic application in liver injury rats. Curr. Pharm. Anal. 2021, 17, 1016–1025. [Google Scholar] [CrossRef]
- Ai, G.; Huang, Z.M.; Wang, D.W.; Zhang, H.C. Acute toxicity and genotoxicity evaluation of hyperoside extracted from Abelmoschus manihot (L.). Medic. J. Chin. Pharm. Sci. 2012, 21, 477–482. [Google Scholar] [CrossRef]
- Ai, G.; Huang, Z.M.; Wang, D.W.; Liu, Z.P. Study on toxicity of hyperoside in rat embryo-fetal development. China J. Chin. Mater. Med. 2012, 37, 2452–2455. [Google Scholar]
- Ai, G.; Huang, Z.M.; Wang, D.W.; Zhang, H.D. Toxicity of hyperoside after long-term oral administration in wistar rats. Chin. J. New Drugs. 2012, 21, 2811–2816. [Google Scholar]





| Detail | Cell Lines/Model | Dose | Ref. |
|---|---|---|---|
| Anti-cancer activity | |||
| Up-regulates caspase-3, caspase-8, Bax, p53 and MDA contents; decreases GSH, SOD and CAT activities; decreases VEGF and Bcl-2 levels; and inhibits cell growth. | HeLa | 100 μmol/L | [20] |
| Down-regulates Nampt, NAD and Sirt1 mRNA and protein expression and inhibits cell proliferation and migration. | HeLa | 400 μg/mL | [21] |
| Inhibits cell proliferation in a dose- and time-dependent manner. | Cervical cancer HeLa and C-33A cells | 0.25, 0.5, 1, 2, 4 and 8 mM | [22] |
| Up-regulates p53, caspase-9 and caspase-3 expression and inhibits cell proliferation and apoptosis. | Human hepatoma HepG2 | 20 and 50 nmol/L | [23] |
| Down-regulates BMP-7 expression, AKT phosphorylation and PI3K expression; induces cell cycle arrest; and inhibits cell proliferation. | Human HepG2 | 5, 10, 20, 40 and 80 μM | [6] |
| Down-regulates PD-L1 and the protein expression of transcription factor c-Myc and prevents tumour formation. | Human non-small cell lung cancer H1975 and HCC827 | 0, 2, 5 and 10 μmol/L | [24] |
| Decreases cell hypoxia-induced survival and proliferation; up-regulates AMPK phosphorylation and HO-1 expression; and inhibits cell survival and proliferation. | Human NSCLC | 10, 50 and 100 μM | [25] |
| Inhibits proliferation, induces apoptosis and up-regulates FoxO1 expression. | Adenocarcinoma lung cancer PC-9 and T790M-positive NSCLC | 150 µM | [26] |
| Induces apoptosis and G1/S phase arrest and inhibits cell proliferation. | Human lung adenocarcinoma A549 | 100 μg/mL | [27] |
| Inhibits cell viability and migration; increases cell apoptosis; down-regulates Bcl-2 and X-linked inhibitor of apoptosis; and up-regulates Bax and cleaved caspase-3. | 4T1 and MCF-7 | 25, 50 or 100µM | [28] |
| Down-regulates Bcl-2; up-regulates Bax and IL-6; may elevate cell sensitivity to paclitaxel. | Human normal breast epithelial cell line MCF-10A and breast cancer cell lines | 5, 10, 50 and 100 μg/mL | [29] |
| Up-regulates caspase-3, Bax and IκBα expression; down-regulates NF-κB, P65 and Bcl-2 expression; induces cell apoptosis; and blocks cell cycle in the G0/G1 phase. | Human gastric cancer MKN-45 | 50 and 100 μg/mL | [30] |
| Reduces cell proliferation; down-regulates P65 and Bcl-2 expression; and up-regulates caspase-3 and IκBα expression. | human gastric cancer MKN-45 | 50, 75 and 100 µg/mL | [31] |
| Inhibits SKOV3 cell proliferation; promotes cell apoptosis; up-regulates cleaved caspase-3 and caspase-9; down-regulates Bcl-2, p65 and p-IκB-α; and decreases cell migration and invasion. | Human ovarian cancer SKOV3 | 5, 15 and 25 μmol/L | [32] |
| Up-regulates perforin and granzyme B expression and increases killing activity of NK cells against PANC 1 cells. | Human NK cells and pancreatic cancer PANC1 | 1.6 and 8 μg/mL | [33] |
| Reduces the changes in phosphorylated levels of PI3K, AKT, mTOR and AMPK while decreasing p38 phosphorylation; inhibits proliferation; and induces apoptosis and autophagy. It may have a therapeutic value in the chemoprophylaxis of skin cancer. | Skin cancer cell lines and DMBA/TPA-induced skin tumours | 25 and 50 μM | [34] |
| Detail | Cell Lines/Model | Dose | Ref. |
|---|---|---|---|
| Increases the number of NeuN-positive cells; decreases the number of GFAP- and PECAM-positive cells; up-regulates ZO-1 and claudin5 protein expression; maintains the integrity of the blood–brain barrier; and may protect neural function in CIR-injured mice. | CIR injury induced by MCAO in mice | 50 mg/kg | [35] |
| Inhibits the activation of microglia and the synthesis of inflammatory factors after SAH; increases the phosphorylation of AKT and GSK-3β; alleviates early brain injury after subarachnoid haemorrhage; and promotes nerve function recovery in rats. | SAH in rats | 50 mg/kg | [36] |
| Increases cell survival rate; decreases LDH release; reduces cleared ROS level, MDA content and caspase-3 activity; increases SOD and CAT activities and GSH content; increases SIRT1 gene expression; down-regulates NF-κB mRNA and protein expression; and protects against high glucose-induced oxidative damage of human neuroblastoma cells. | SH-SY5Y | 50 and 100 μm L/L | [37] |
| Significantly shortens the cumulative immobility time of forced swimming and tail suspension mice; can act as an anti-depressant. | Depressive ICR mice | 20 mg/kg | [38] |
| Targets the PI3K/AKT and MAPK pathways to increase antioxidant levels and decrease the levels of autophagy-related proteins; protects the hippocampal CA3 region from neuronal damage caused by epilepsy. | Neuronal damage in a mouse model | 50 mg/kg | [39] |
| Decreases TC, TG and LDL-C levels and prevents cognitive dysfunction, neuroinflammation and oxidative stress in diabetic rats. | Type 2 diabetes rats | 50, 200 and 400 mg/kg/day | [40] |
| Reverses the regulation of Beclin1, LC3, Bax, cleaved caspase-3, Cyc and Bcl-2 expression in rat SNpc tissues and SH-SY5Y cells; promotes the regulation of P62 and α-synuclcin; and exerts a neuroprotective effect. | Rotenone was used to induce Parkinson’s disease rat model and SH-SY5Y cell injury model | 100 and 200 mg/kg 0.5, 1 and 2 μmol/L | [41] |
| Activates the PI3K, AKT and LC3B pathways; deactivates the NF-κB, Bax, caspase 3 and P62 pathways; and inhibits oxidative stress damage. | Hyperoside on rat pheochromocytoma (PC12) cells | 12.5, 25 and 50 μmol/L | [42] |
| Down-regulates IL-1β, IL-6, IL-8, TNF-α, ROS, MDA, Bax and caspase-3 levels; increases CAT, SOD and GSH activities; up-regulates Bcl-2, BDNF, TrkB, SIRT1 and NGF expression; reduces LPS-induced inflammation, oxidative stress and apoptosis; and protects against neuroinflammation. | HT22 | 20 μM | [43] |
| Significantly improves neuronal cell viability loss, lactate dehydrogenase release, excessive ROS accumulation and mitochondrial membrane potential dysfunction and inhibits neuronal death. | Human dopaminergic neuroblastoma SH-SY5Y | 0.5, 1 and 2 µM | [44] |
| Decreases Bax/Bcl-2 ratio, down-regulates cytochrome c expression and caspase-3 activity, up-regulates zonula occludens-1 and claudin-5 expression; and minimises damage to the blood–brain barrier. | Fibrillar Aβ1–42-induced disruption in an in vitro blood-brain barrier model | 200 or 500 µM | [45] |
| Decreases the immobility of prefrontal lobe and caudate putamen and exerts anti-depressant effects. | Forced swimming test and tail suspension test in mice | 1.875, 3.75 and 7.5 mg/kg | [46] |
| Detail | Cell Lines/Model | Dose | Ref. |
|---|---|---|---|
| Decreases the levels of AST, CK, CK-MB and c-TnT in rats; the rate of cardiomyocyte apoptosis; and the relative expression of protein and CaMK II protein. Effectively relieves heart failure. | Rat model of myocardial ischaemia–reperfusion injury | 50,100 mg/kg | [47] |
| Decreases fasting blood glucose, cTnI I and MDA contents and increases SOD activity, the value of ejection fraction and short-axis shortened pitch in diabetic mice; improves glycolipid metabolism; and exerts a protective effect on myocardial injury caused by diabetes. | Type 2 diabetic mice induced by high-fat diet combined with low-dose streptozotocin | ND | [48] |
| Increases cell survival rate; decreases cell apoptosis rate and ROS and MDA levels; increases SOD level, PI3K relative expression level and AKT and Nrf2 phosphorylation level; and protects cardiac muscle cells from high sugar-induced oxidative stress damage. | High-glucose treatment simulates oxidative stress injury of cardiomyocytes | 4, 8 and 20 nmol/L | [49] |
| Decreases MDA content and CK-MB activity; increases SOD activity and ATP content; up-regulates Nrf2, PKCε and Kir6.2 expression in cardiomyocytes; and improves cardiac muscle damage. | Myocardial I/R injury in rats | 50 mg/kg | [50] |
| Decreases CK-MB, TNF-α and IL-1β expression in serum; increases SOD and GSH activities in myocardial tissue; up-regulates SIRT1 protein expression; down-regulates FoxO1 acetylation level and apoptosis protein cleaved caspase-3 expression; alleviates early myocardial injury caused by antioxidative stress and anti-inflammatory effects in severely burned rats. | Early myocardial injury in severely burned rats | 30 mg/kg | [51] |
| Increases the levels of LVSP, maximum increase rate of left ventricular pressure (+dp/dtmax), maximum decrease rate of left ventricular pressure (−dp/dtmax), HIF-1α and HO-1; decreases the levels of LVEDP, cTnI, BNP, TNF-α and IL-1β; and ameliorates myocardial injury in septic rats. | Myocardial cells of sepsis model in rats | 10 and 20 mg/kg | [52] |
| Decreases serum oxidative stress; improves thoracic aorta remodelling and endothelial dysfunction to a certain extent; exerts a cardiovascular protective effect on mice with myocardial infarction. | Myocardial infarction model | 36 mg/kg | [53] |
| Increases the levels of heart rate, mean arterial pressure and heart rate systolic pressure product; decreases the levels of CK-MB and cTnI; and reduces I/R ventricular arrhythmia in rats. | Myocardial I/R model in rats | 50 mg/kg | [54] |
| Significantly enhances SIRT3 signal expression; alleviates oxidative stress injury of myocardial tissue; inhibits the progression of myocardial fibrosis; and improves pathological myocardial hypertrophy caused by stress load. | Myocardial hypertrophy (mouse) | 50 mg/kg/day | [55] |
| Increases phosphorylated AMPK, phosphorylated mTOR and P62 proteins; decreases apoptosis index, caspase-3 activity and LC3II and Beclin1 expression; and alleviates I/R injury of H9C2 cells. | I/R model | 50 μmol/L | [56] |
| Decreases cardiac myocyte cross-sectional area and cardiac weight/body weight ratio; inhibits autophagy in TAC rats and AngII-induced H9C2 cells and apoptosis; and effectively alleviates heart failure by inhibiting apoptosis and inducing autophagy. | TAC-induced heart failure in rats | 100 and 200 mg/kg | [57] |
| Decreases number of apoptotic cells; down-regulates lytic caspase-3 expression; up-regulates Bcl-2 expression; increases survival of myocardial cells; and alleviates hypoxic injury. | Hypoxia model in H9C2 cells and C57BL/6 mice | 50 µmol/L, 50 mg/kg | [58] |
| Inhibits AngII-induced cardiomyocyte hypertrophy; protects against stress overload-induced cardiac remodelling; | Myocardial hypertrophy model in mice | 20 mg/kg/day | [59] |
| reduces infarct CVF and myocardial hypertrophy; exerts obvious protective effect on heart injury in mice with myocardial infarction. | Myocardial mouse model | 18 and 36 mg/kg | [60] |
| Decreases cell vitality and aggravates inflammation; down-regulates miR-21 expression in cardiomyocytes; increases cell survival rate; decreases inflammatory response; inhibits miR-21; and regulates cardiomyocyte activity and inflammation. | Sepsis mouse model, and myocardial injury cell model | 20 mg/kg, 10 μM | [61] |
| Detail | Cell Lines/Model | Dose | Ref. |
|---|---|---|---|
| Decreases liver index, AST, ALT, MDA and Bach1 complex levels and alleviates the pathological damage of acute liver injury mice. Increases SOD and GSH activities and Nrf2–complex level; up-regulates transporter CRM1 expression; enhances ERK1/2 activity; and reduces cell oxidative stress damage. | Acute liver injury caused by CCl4 Human hepatocytes L02 cells | 100 mg/kg 100 and 200 μM | [62] |
| Decreases ALT, AST and ALP levels; down-regulates α-SMA, type I collagen, fibrotic factor-CTGF, MMP2 and MMP9; inhibits the activation of the transforming growth factor-β-1/Smad pathway and hepatic stellate cells mediated by transforming growth factor-β1; and prevents liver fibrosis. | A model of via aortocaval fistula in rats Hepatic stellate cell line LX-2 | 200 mg/kg 2 mM | [63] |
| Decreases AST, ALT and ALP levels; activates Nrf2 and its downstream genes; decreases SOD, GST and GSH-Px activities; reduces LPO, LDH and ALT production; and prevents acute liver injury. | N-APAP acute hepatic injury LO2 cells | 100 mg/kg 20 μM | [64] |
| Decreases AST/ALT and MDA activities; increases SOD and glutathione peroxidase activities and haem oxygenase 1 and NAD(P)H expression of quinone oxidoreductase 1; down-regulates caspase-3 expression; and prevents hepatic ischaemia–reperfusion injury in rats. | 70% hepatic ischaemia–reperfusion injury model | 50 mg/kg/day | [65] |
| Up-regulates Nr4A1; improves liver steatosis, insulin resistance and inflammation; and may prevent the pathological progression of non-alcoholic fatty liver disease. | NAFLD Primary hepatocytes | 50 mg/kg 1, 5 and 10 μM | [66] |
| Increases SOD activity and MDA level in the body; down-regulates PHLPP2 expression; activates AKT phosphorylation; induces GSK-3 double phosphorylation; and protects against oxidative stress-induced liver damage. | Carbon tetrachloride damages rat liver Human hepatocytes (L02 cells) | 30 and 15 mg/kg 10 and 100 μM | [67] |
| Decreases total cholesterol, triglycerides and low-density lipoprotein cholesterol levels; inhibits the phosphorylation of p65/NF-κB and mitogen-activated protein kinase; activates transcription factor 3 protein expression; and decreases Bax, cytochrome c, caspase-9 and caspase-3 expression. It may be beneficial in the treatment of diabetes. | Diabetic mice induced by high-sugar and high-fat diet and alloxan | 50,100 and 200 mg/kg bw/day | [68] |
| Detail | Cell Lines/Model | Dose | Ref. |
|---|---|---|---|
| Down-regulates TNF-α, IL-1β, IL-6, ICAM-1, VCAM-1, TLR4, COX-2, NF-κB, caspase-3, caspase-9, Bax and Bcl-2 expression and prevents CIR injury. | Middle cerebral artery occlusion/reperfusion rat model | 60 and 120 mg/kg | [69] |
| Decreases LDH activity and MDA, NSE and S100β contents; relaxes the cerebral basilar artery in a dose-dependent manner; up-regulates IP3, PKC, TRPV4, SKca and IKca expression; reduces Ca2+ fluorescence intensity; and ameliorates brain injury in rats with ischaemic stroke. | Global CIR model | 50 mg/kg | [70] |
| Up-regulates BDNF expression in the hippocampus; down-regulates p75NRT; reduces hippocampal neuron and cell damage; improves learning and memory; and protects the hippocampal tissue in rats. | Middle cerebral artery occlusion, MCAO model | 50 and 100 mg/kg | [71] |
| Up-regulates Bcl-2 mRNA, p-PI3K and p-AKT protein expression; down-regulates Bax and caspase-3 mRNA expression; and prevents hepatic IR injury in rats. | Cerebral I/R injury in rats | 50 mg/kg/day | [72] |
| Increases SOD, catalase and glutathione peroxidase activities and improves cell apoptosis after nickel administration. | Nickel-induced brain damage in rats | 50 mg/kg | [73] |
| Detail | Cell Lines/Model | Dose | Ref. |
|---|---|---|---|
| Suppresses NLRP3, caspase-1 and ASC expression and prevents acute kidney injury induced by lipopolysaccharide. | Mouse acute kidney injury model | 25, 50 and 100 mg/kg | [74] |
| Up-regulates Klotho protein expression; down-regulates P53, P21, IL-1, MCP-1, TGF-β, LC3 and Beclinl protein expression; decreases AMPK-ULK1 signalling pathway activity; prevents aging and damage of renal and tubular epithelial cells. | D-gal induces kidney aging and damage model | 10 μg/mL 20 mg/kg/day | [75] |
| Decreases ROS and H2O2 levels and NADPH oxidase and LD activities and shows potential in the treatment of kidney stones and ROS-related diseases. | Human kidney-2 cells | 100 and 200 µM | [76] |
| Up-regulates Klotho expression; down-regulates p53 expression; and prevents age-related kidney damage. | NRK-52E cells | 5 and 10 μg/mL | [77] |
| Inhibits OPA1 hydrolysis, mitochondrial division, oxidative stress and apoptosis and shows new therapeutic potential in the treatment of acute kidney injury. | Renal ischaemia models | 20 mg/kg | [78] |
| Decreases ERK pathway activation and downstream transcription factor CREB phosphorylation; down-regulates miRNA-34a expression; and inhibits high glucose-induced proliferation of mesangial cells. | Mouse glomerular mesangial cell line (SV40-MES13) diabetes model | 50, 100 and 200 μM | [79] |
| Down-regulates APC expression; up-regulates miR-499e5p expression; and improves diabetic nephropathy by targeting the miR-499e5p/APC axis. | Mouse model of diabetic nephropathy | 30 mg/kg | [80] |
| Detail | Cell Lines/Model | Dose | Ref. |
|---|---|---|---|
| Decreases collagen I and III expression, serum TGF-β1 content and alveolar lavage fluid IL-6 levels and effectively improves bleomycin-induced pulmonary fibrosis in mice. | Pulmonary fibrosis model | 100 mg/kg | [81] |
| Decreases cytotoxicity and p-AMPK expression; increases p-mTOR expression; inhibits the AMPK/α signalling pathway; down-regulates TNF-α and IL-6 expression in the alveolar lavage fluid; decreases total number of cells in the alveolar lavage fluid; and inhibits autophagy dysregulation and apoptosis by regulating the AMPK/mTOR pathway to prevent lung injury. | Human bronchial epithelial BEAS-2B | 50 μM | [82] |
| Down-regulates MDA, TNF-α and IL-6 expression; increases SOD activity; inhibits epithelial–mesenchymal transition; and slows the development of pulmonary fibrosis by inhibiting oxidative stress and inflammation in the lung tissues of mice with pulmonary fibrosis. | Pulmonary fibrosis model | 50 mg/kg/day | [83] |
| Increases the number of A549 cells; down-regulates IL-8 and TNF-α expression; and benefits Mycoplasma pneumoniae pneumonia through chemokine ligand 5–chemokine receptor 4 interaction. | Lung cancer A549 | 100, 200 and 400 µg/mL | [84] |
| Detail | Cell Lines/Model | Dose | Ref. |
|---|---|---|---|
| Reduces LDL-C, MDA and IL-6 levels; increases NO and eNOS levels; decreases plaque area in the aortic lumen of mice; improves lipid deposition; down-regulates PARP1, ARG2 and iNOS expression in mouse aortic tissue; and slows down atherosclerosis. | Atherosclerosis model | 200 mg/kg | [85] |
| Decreases blood pressure and vascular tension, vascular remodelling, serum angiotensin-converting enzyme, ALD, U-mAlb, BUN, SCr, ALT and AST; increases fluorescence intensity of ANS-angiotensin-converting enzyme; inhibits vascular remodelling; and lowers blood pressure. | Rat hypertension model | 23.2, 46.4 and 232.2 mg/kg | [86] |
| Decreases the adhesion of monocytes to TNF-α-stimulated VSMCs; down-regulates p38 MAPK, JNK and ERK, NF-κB and TNFR1 expression; inhibits vascular inflammation; and shows potential to prevent atherosclerosis. | Human monocyte U937 cell lines | 1 and 10 µg/mL | [87] |
| Detail | Cell Lines/Model | Dose | Ref. |
|---|---|---|---|
| Increases IL-6 and TNF-α levels; decreases Col-I and Col-III levels; up-regulates MMP-3, MMP-9, p-IκB-α and p-p65 expression; prevents IL-1β-induced chondrocyte injury in the sacroiliac joint of mice; improves cell activity; and inhibits inflammatory factors and extracellular matrix disorder. | BALB/c mouse sacroiliac joint chondrocytes | 50 and 100 μg/mL | [88] |
| Down-regulates RANKL, TRAF6 and IκBα expression; up-regulates NFATC1 and osteoprotegerin expression; and shows potential anti-osteoporotic effect on ovariectomised mice. | Ovariectomised mice | 40 or 80 mg/kg/day | [89] |
| Increases ALP, Col-I and OCN mRNA expression; decreases the rate of apoptosis, expression of apoptosis-related proteins and levels of MC3T3-E1 phosphorylated JNK and p38; and protects osteoblasts by inhibiting MAPK signalling and oxidative damage of cells. | Osteoblastic MC3T3-E1 cells | 20 and 40 μmol/L | [90] |
| Increases cell viability and proliferation; decreases cell apoptosis and autophagy; and protects osteoblasts from damage induced by Ti particles. | MC3T3-E1 cells | 200 and 400 µg/mL | [91] |
| Detail | Cell Lines/Model | Dose | Ref. |
|---|---|---|---|
| Down-regulates INOS, MMP3 and p38 expression and inhibits the proliferation and migration of IL-1β-induced fibroblast synovial cells. | FLS cells | 50 and 100 μm/mL | [92] |
| Decreases TNF-α and IL-6 contents in the serum; prevents the synovial hyperplasia and inflammatory cell infiltration of the mouse ankle joint; and exerts a certain therapeutic effect on rheumatoid arthritis. | CIA mice | 25 and 50 mg/kg | [93] |
| Down-regulates MMP3 and TNF-a genes; reduces the loss of cell matrix of rat chondrocytes; maintains cell activity; down-regulates the expression of genes related to cell inflammation; reduces inflammation; and exerts a certain therapeutic effect on osteoarthritis in vitro. | SD rat chondrocyte osteoarthritis model | 9 μg/mL | [94] |
| Down-regulates INOS, COX-2, MMPs and ADAMTS5; up-regulates type II collagen, agglutinin and SOX9; and exerts anti-arthritic effects. | Osteoarthritis model | 20 and 40 μM 20 mg/kg | [95] |
| Detail | Cell Lines/Model | Dose | Ref. |
|---|---|---|---|
| Decreases IL-1β, TNF-α, MDA, MPO, NF-κB p65, TRAF6, LC3, Beclin1, p62 levels and cell apoptosis; increases SOD activity; and exerts obvious therapeutic effect on rat ulcerative colitis. | Ulcerative colitis | 50 and 100 mg/kg | [96] |
| Increases serum E2, AMH, SOD and CAT activities; reduces FSH activity; up-regulates Nrf-2, HO-1, p-PI3K, p-AKT and Bcl-2 expression; decreases caspase3, Bax and ROS levels; improves tripterygium glycoside-induced primary ovarian insufficiency and mouse ovarian reserve function decline. | Adult endothelial cells | 75 mg/(kg/day) | [97] |
| Decreases MDA content; increases SOD, GSH-Px and CAT activities; down-regulates Bax expression; up-regulates Bcl-2, SHH, Gli1 and SMO expression; and protects granular cells from H2O2-induced apoptosis and oxidative stress by activating the Sonic hedgehog signalling pathway. | Granular cells | 40 μM | [98] |
| Increases MiR-499a-5p expression and decreases NRIP1 expression in a dose-dependent manner and mitigates apoptosis and inflammatory response induced by high glucose via the miR-499a-5p/NRIP1 axis. | HK-2 cells | 10, 50 and 100 μmol/L | [99] |
| Up-regulates AhR target genes CYP1A1 and CYP1B1 and may prevent age-related macular degeneration. | Human retinal pigmented epithelial ARPE-19 cells and adenocaricinomic human alveolar basal epithelial A549 cells Animals exposed to UVA or blue light | 25, 50 and 100 μM 25, 50 and 100 mg/kg | [100] |
| Down-regulates caspase-3, caspase-9 and Bax expression; up-regulates Bcl-2 expression; and possibly plays a protective role in diabetic retinopathy by reducing oxidative stress induced by high glucose and inhibiting cell damage and apoptosis. | Rat retinal vascular endothelial cells Rats fed with high-fat rat | 10 mg/mL 20, 50 and 100 mg/kg | [101] |
| Increases the proliferation of rat bone mesenchymal stem cells and the number of EdU-positive cells; decreases cell cycle distribution; up-regulates Ki67 and PCNA expression; promotes the proliferation and osteogenic differentiation of bone marrow mesenchymal stem cells; and exerts potential therapeutic effect on periodontitis. | Bone marrow mesenchymal stem cells Rat periodontitis model | 20 mL, 200 mg/mL | [102] |
| Down-regulates NO, TNF-α, IL-1β, IL-6 iNOS, p38 and NF-κB p65 expression; up-regulates Sirt6 expression; and inhibits the release of macrophage proinflammatory factors. | RAW264.7 macrophages | 25, 50 and 100 μmol/L | [103] |
| Down-regulates IL-1β, IL-6, TNF-α and iNOS mRNA cleaved-caspase 8, 9, 3 and Bax expression; up-regulates Bcl-2 expression; and inhibits the inflammatory and apoptotic responses of human umbilical vein endothelial cells. | Human umbilical vein endothelial cells | 20 and 50 μmol/L | [104] |
| Down-regulates TXNIP expression and intracellular calcium concentration; protects pancreatic beta cell function; and prevents type 2 diabetes and promotes its treatment. | Diabetic mice induced by high-fat diet and alloxan | 200 and 400 mg/kg | [105] |
| Inhibits the expression of transcription factors and adipogenic genes and reduces lipid accumulation in adipocytes. Hyperoside at 5 µM inhibits adipogenesis, and hyperoside at 10 µM reduces fat accumulation in mature adipocytes. | 3T3-L1 cells | 10 μM | [106] |
| Decreases LA and BUN contents; increases LG and MG contents; declines ROS and MDA levels; enhances SOD and GSH-Px activities; and regulates the Nrf2 signalling pathway to improve the antioxidant capacity of the body and avoid fatigue. | Exercise-induced fatigue mouse model | 5, 10 and 20 mg/kg | [107] |
| Promotes FasL and IFN-γ expression and significantly promotes NK cell proliferation at 1.6–8.0 μg/mL. | NK cells | 8 μg/mL | [108] |
| Down-regulates mTOR, phosphorylated p70S6 kinase, TLR4, MyD88, NF-κB and p-p65 and shows potential to treat recurrent pregnancy loss. | Rat pregnancy loss model | 40 mg/kg | [109] |
| Down-regulates IL-1β, IL-8 and endothelial adhesion cytokines TF, ICAM1 and VCAM1 in HUVECs. | Human umbilical Vein endothelial cells | 50 μM | [110] |
| Detail | Cmax | Tmax | AUC(0-t) | AUC(0-ꝏ) | T1/2 | MRT | CL | V | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Hyperoside i.p. 100 mg/kg was administered once. | 55,310.9 ng/mL | 0.32 h | ND | 40535.6 h*ng/mL | ND | 0.77 h | 2498.5 mL/h/kg | 7862.0 mL/kg | [143] |
| Hyperoside i.g. 100 mg/kg was administered once. | 149.0 ng/mL | 1.00 h | ND | 305.1 h*ng/mL | ND | 5.11 h | 382,994.1 mL/h/kg | 2,290,414.7 mL/kg | |
| Hyperoside i.v. 2 mg/kg was administered once. | 4630.6 ng/mL | 0.03 h | ND | 748.3 h*ng/mL | ND | 0.64 h | 2786.7 mL/h/kg | 13,835.6 mL/kg | |
| Hyperoside i.g. 2.5 mL/100 g was administered once. | 0.24 ± 0.01 μg/mL | 0.39 ± 0.17 h | 1344.98 ± 62.31 μg/L*h | 1668.05 ± 66.53 μg/L*h | 0.04 ± 0.03 h | 5.72 ± 0.29 h | ND | ND | [142] |
| Hyperoside i.g. 2.5 mL/100 g was administered once a day for seven days. | 0.32 ± 0.02 μg/mL | 2.33 ± 0.52 h | 3169.42 ± 678.46 μg/L*h | 3831.70 ± 705.26 μg/L*h | 0.04 ± 0.01 h | 11.87 ± 2.75 h | ND | ND | |
| Senecio scandens water extract i.g. 10 mg/kg was administered once. | 1.821 ± 0.179 μg/mL | 1 h | 0.006 ± 0.003 μg*h/mL | 0.023 ± 0.002 μg*h/mL | 1.150 ± 0.273 h | ND | ND | ND | [140] |
| Cuscuta chinensis water extract i.g. 2 g/100 g was administered once. | 72.03 ng/mL | 18 min | ND | 5106.86 ng*min/mL | 222.58 min | 248.83 min | ND | ND | [139] |
| Cuscutae Semen water extract i.g. 15 mg/kg was administered once. | 48.44 ± 12.54 μg/L | 1.17 ± 0.26 h | 159.85 ± 28.71 μg/L*h | 177.69 ± 24.88 μg/L*h | 6.63 ± 1.41 h | 6.41 ± 1.73 h | ND | ND | [144] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, S.; Chen, S.; Xia, W.; Sui, H.; Fu, X. Hyperoside: A Review of Its Structure, Synthesis, Pharmacology, Pharmacokinetics and Toxicity. Molecules 2022, 27, 3009. https://doi.org/10.3390/molecules27093009
Xu S, Chen S, Xia W, Sui H, Fu X. Hyperoside: A Review of Its Structure, Synthesis, Pharmacology, Pharmacokinetics and Toxicity. Molecules. 2022; 27(9):3009. https://doi.org/10.3390/molecules27093009
Chicago/Turabian StyleXu, Sijin, Shuaipeng Chen, Wenxin Xia, Hong Sui, and Xueyan Fu. 2022. "Hyperoside: A Review of Its Structure, Synthesis, Pharmacology, Pharmacokinetics and Toxicity" Molecules 27, no. 9: 3009. https://doi.org/10.3390/molecules27093009





