2.1. Sorption Isotherms and Their Mathematical Approximations
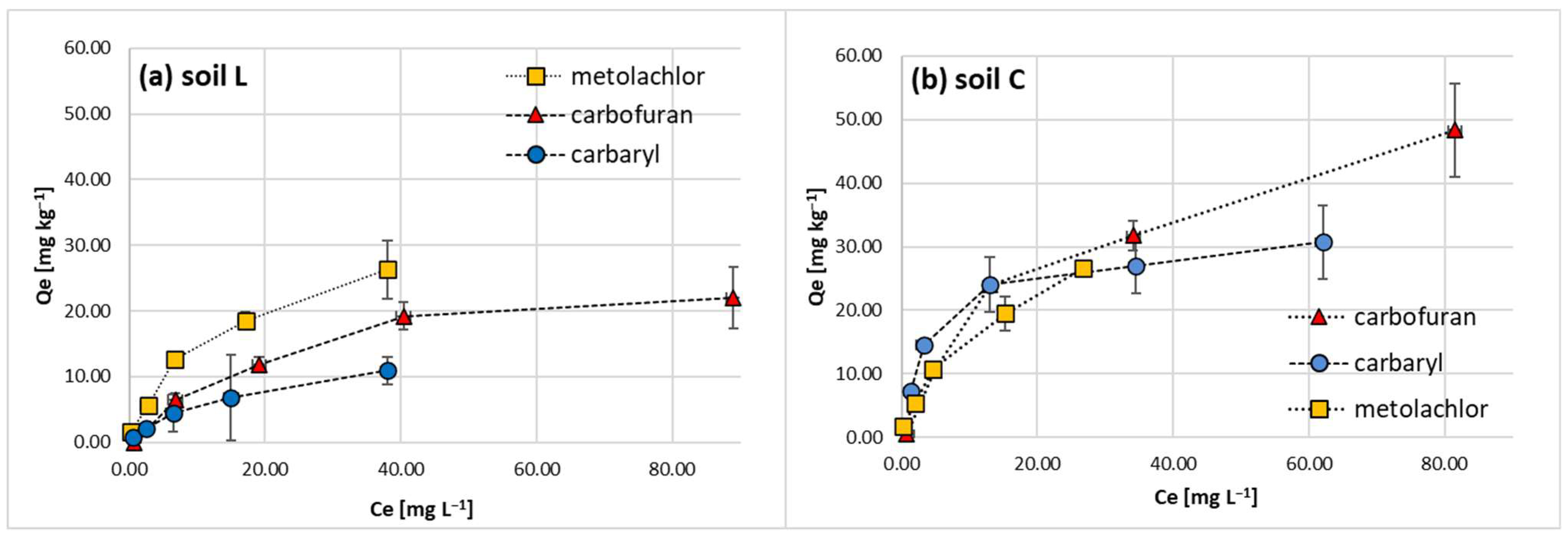
Tested pesticide classes were subjected to sorption studies on sandy loam (L) and loam (C) topsoil materials and resulting isotherms (
Figure 1) were approached with Langmuir, Freundlich Temkin and Dubinin–Radushkievich (D–R) models (
Figure 2). Linear sorption parameters calculated for the obtained Langmuir-type [
43] isotherms are summarized in
Table 1. Applied models revealed high correlation coefficients (R
2 > 0.950), except for the D–R model. The resulting L-shape of the isotherms is due to adsorption site limitations with the increasing pesticide concentration and presumably the energetic heterogeneity of the studied soils, as in the case of most solids [
44]. In fact, the experimental isotherms approached a saturation upper limit but did not reach a complete plateau. This implies that at high adsorbate concentrations, weak intermolecular forces among the studied non-ionic pesticides might occur and participate in multilayer formation [
45,
46].
The maximum monolayer adsorption capacity Qmax (mg kg−1) of the Langmuir model, evaluated for the hydrophobic pesticides on soil L, correlated well with their Freundlich Kf constants and followed the increasing order of carbaryl (15.25) < carbofuran (27.93) < metolachlor (32.47). C soil Qmax parameters for carbaryl and carbofuran were doubled compared to L soil, whereas no significant change in case of metolachlor was noticed.
Analogous trends to those of Q
max were observed on both studied soils for the theoretical monolayer sorption capacity (Q
m) calculated with the D–R approach. It points to a significant increase in the sorption sites active in insecticide retention. In addition, the hydrophobic character and low water solubility of the studied carbamates may favor their diffusion within organic matter structures. According to the literature, diffusion and the following physical entrapment within the soil–organic matter matrix is a common sorption mechanism, especially in the case of pesticides independent of any traditional chemical adsorption mechanisms [
47].
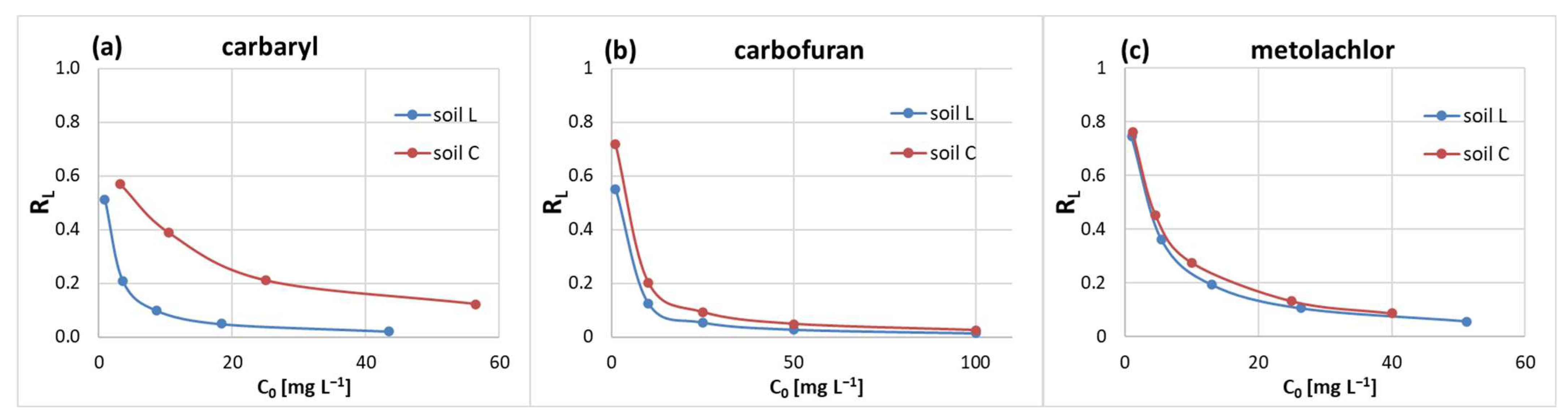
Freundlich and Langmuir isotherm parameters, namely, the adsorption intensity (n
f) and separation factor (R
L), ranged from 0.248 to 0.611 (
Table 1) and up to 0.80 (
Figure 3), respectively. This indicates favorable (0.1 < n
f < 1.0 [
48]) and reversible (0 < R
L < 1 [
49]) adsorption of the solute and suggests that the pesticide molecules preferred high-energy sites followed by sorption at lower-energy sites [
50,
51].
The Temkin isotherm constant related to the heat of adsorption (b
T), and the binding constant of the pesticide to a soil (A) exhibited decreasing and increasing trends, respectively, when comparing soils L and C (
Table 1). According to the model assumptions, b
T would more often decrease than increase with increasing coverage [
52]. Hence, it can be concluded that the lower values of the constant obtained for loamy soil indicate a higher degree of surface coverage with the pesticides compared to sandy loam soil material. Furthermore, from the b
T value, the nature of the process may be elucidated [
53]. In the case of physical sorption, it should be lower than 4.184 kJ mol
−1 (1 kcal mol
−1), while for chemical sorption it is above 41,840 kJ mol
−1 (10 kcal mol
−1). Thus, the values of the b
T parameter (
Table 1) suggest that the sorption of carbamates and metolachlor in L and C soils is of a physical nature. This was also confirmed by the E
a values obtained within D–R isotherms (
Table 1), which were lower than 8 kJ mol
−1 and may be partly due to the van der Waals interaction between the pesticides and the hydrophobic soil components.
Freundlich K
f parameters, which revealed the highest variation when comparing both studied soils, were used for the calculation of the standard Gibb’s free energy change (ΔG°) of the tested pesticides (
Table 2). Prior to ΔG° calculations, K
f parameters were converted to the dimensionless standard equilibrium constants using the appropriate equations, as suggested in the literature [
54,
55,
56].
The negative ΔG° values obtained for all studied pesticides on soils L and C point to the spontaneous character of the sorption. The significant increase and the highest negative ΔG° value obtained for carbaryl sorption in C soil indicates that in the case of this insecticide, the adsorption capacity would be higher for clay than for sandy soil, as for carbofuran (
Table 2). Meanwhile, ΔG° of much similar magnitude was obtained for metolachlor sorption in both studied soils. The values of this thermodynamic parameter calculated for the studied pesticides are comparable to those found in the literature, although a limited number of papers performing ΔG° calculations using dimensionless K
c can still be found [
55,
57,
58].
In addition, K
f parameters were used to calculate organic carbon partition coefficient (K
oc) and groundwater ubiquity score (GUS, [
59]) indexes, providing information about pesticide mobility and leachability, respectively (
Table 3). All calculated K
oc values (except for carbaryl sorption in C soil) were below 500, which points to minimum or no adsorption of the chemicals into the soil and indicates high probability of their runoff or leaching [
1,
60]. A similar mobility ranking by FAO [
61] based on log K
oc values classifies all of the pesticides as of moderate mobility, achieving log K
oc values in the range of two to three. Interestingly, despite the fact that the metolachlor sorption magnitude is higher in the loam than in the sandy loam soil under study, the K
oc value for herbicides in C soil is lower than in L soil. This might suggest that organic matter is not the main soil constituent responsible for metolachlor immobilization in soil.
Calculated ground ubiquity score (GUS) indexes point to the following decreasing order of leaching threat in L soil: carbofuran > carbaryl > metolachlor and C soil: carbofuran > metolachlor > carbaryl. The lowest risk of soil C percolation (GUS value ≤ 1.8) was obtained for carbaryl and metolachlor and the chloroacetanilide pesticide in L soil. Carbofuran potential for movement toward groundwater was moderate in soil C (2.47), approaching high (> 2.80) in loamy sand soil.
The retention of carbaryl, carbofuran and metolachlor on the investigated loamy sand and loam soil materials was evaluated by comparing their sorption-desorption magnitudes. Studied pesticide trends in L and C soils are presented in
Figure 4. They vary in the magnitude of sorption on loam and sandy loam soil studied for a given hydrophobic agrochemical. Nevertheless, their common feature is a decline in sorption magnitude with the increase in initial concentration, which confirms the resulting L-type shape of isotherms (
Figure 1).
In general, carbaryl was adsorbed up to 44.51% and 71.07% in L and C soils, respectively (
Figure 4a). Carbofuran was characterized by a similar pattern; however, its maximum sorption magnitude was 33.50% of pesticide initial dose introduced to soil L and 55.0% in the case of soil C (
Figure 4b). Both carbamates revealed a great discrepancy in sorption magnitude among sandy loam and loam soils. On the contrary, metolachlor sorption in both soils was very similar, with the highest magnitude among the studied pesticides (84.06% and 71.96% for soils C and L, respectively,
Figure 4c).
Meanwhile, desorption of carbamates and metolachlor achieved higher values in L rather than the C soil studied (
Figure 5, expressed as a mass initially sorbed and desorbed). Carbaryl and carbofuran release from loamy sand soil was increasing along with the initial pesticide dose introduced to soil, achieving a maximum value of 62.6% and 87.0% of the pesticide dose initially sorbed, respectively.
Desorption in C soil was lower in the case of carbaryl (20.50–69.06% of the initial dose) and much higher for carbofuran in loam soil (from 49.76% of the lowest dose applied, up to approx. 74.70% in case of the highest initial concentration), making its sorption reversible [
63] and enhancing a great threat of carbofuran percolation in soil. These results are consistent with the calculated GUS indexes (
Table 3), confirming the low and slightly moderate potential of carbaryl to leach in soils C and L, respectively, and moderate potential for carbofuran in both studied soils. Metolachlor desorption in L soil was in a range of 31.20% to 45.89% and from 10.49% to 13.87% in C soil. This acetanilide pesticide release was the lowest among the group of the uncharged pesticides studied, as were the calculated GUS values (1.46 and 1.60 in soils L and C, respectively), suggesting its scarce leaching potential in both studied soils (
Table 3). The strong adsorption and low desorption characteristics of metolachlor suggest that for the soils studied, the migration risk to groundwater should be lower than for the other uncharged pesticides examined.
In general, studied pesticides sorbed less to loamy sand than the loam soil (
Figure 4), which contained more organic carbon and clay fractions.The highest negative standard Gibb’s free energy value obtained for carbaryl in soil C (
Table 2) together with its lowest GUS index (1.12,
Table 3) may indicate that the forces holding the insecticide in soil are relatively strong. Similarly, the relatively high and negative ΔG°, the lowest desorption magnitude (
Figure 5e,f) and the highest log
p value among the studied pesticides predestinate metolachlor to be efficiently retained in both soils.
Taking into account the parameters approximated from sorption isotherms as well as the results of desorption studies, it can be concluded that the molecular interactions between the studied pesticides and the active soil constituents are of a physical nature. In general, this type of sorption for uncharged pesticides in soils is primarily governed by van der Waals forces, hydrophobic interactions and hydrogen bonding among the suitable, complementary functional groups of both interacting moieties [
15,
18,
41].
2.2. Influence of Fractional SOM Composition on Carbaryl, Carbofuran and Metolachlor Retention in Studied Soils
According to the previous study on adsorption of the investigated uncharged pesticides in L and C soils amended with various soil colloids, agrochemical retention was attributed to their distribution in soil organic matter and inorganic colloids [
10]. Both soils revealed similar mineralogical compositions according to which montmorillonite and kaolinite were major clay minerals. However, substantial variations were revealed in the fractional compositions of organic matter of soils L and C (
Table 4). According to them, loamy sand material is rich in humic acid fractions (CHAs), which constitutes 40.07% of total organic carbon (TOC), whereas in loam soil residual carbon fractions (CRs, also called stable organic matter or humin (HN) fraction) represent 59.71% with a dominant share in TOC. This discrepancy in the mutual share of particular SOM fractions and their chemodiversity will have a direct impact on the magnitude and stability of interactions with pesticides [
16]. Humic substances (HS) may react with uncharged pesticides via different modes, of which hydrophobic retention is regarded a common adsorption mechanism [
18,
64]. This may occur on hydrophobic active sites of HS where the competition for water is minimized [
64]. These regions include aliphatic side-chains (common in humin fraction) or lipid constituents, as well as lignin-derived moieties with a high carbon content and a small number of polar groups. Van der Waals interactions also play an important role in the adsorption of non-ionic pesticides on complementary sites of HA. Since these forces are additive, their contribution increases with the size of the interacting molecule and with its capacity to adapt to the HA surface. This SOM fraction presumably consists of micelles of a polymeric nature, where the basic structure consists of an aromatic ring (di- or trihydroxy-phenyl type) bridged by -O-, -NH-, -N=, -S- and other functional groups that contain both free -OH and -COOH groups and the double linkages of quinones [
18]. These functionalities may be involved in adsorption of non-ionic polar pesticides directly via hydrogen bonding or indirectly by affecting the hydrophilic/hydrophobic balance of the SOM surface [
64]. Therefore, it is well documented that natural SOM fractions (humic and fulvic acids, humin), as well as different biopolymers (e.g., cellulose, lignin, cutin), due to the structural differences, may exhibit various affinities to organic contaminants, which can be also evaluated by their various K
oc coefficients [
23].
When we compare the calculated K
oc values for carbamates in the investigated L and C soils (
Table 3), an increase in the soil C affinity to the pesticides can be elucidated. This is particularly evident in the case of carbaryl, where an over 5.7 times higher K
oc value for loamy soil is obtained, whereas for carbofuran the increase is by a factor of 1.25. In addition, the maximum monolayer adsorption capacities Q
max of carbamates approximated for C soil were twice as much as for L soil, which correlates well to the doubled (compared to L soil) content of the residual carbon fraction of C soil (
Table 4). Since the diverse share of OC fractions of the studied soils will evoke structural changes in SOM, this should be also reflected in their degree of sorption site heterogeneity [
65]. According to a dual-mode sorption model by Xing and Pignatello [
66], the more “glassy” or condensed nature of the organic matter, such as in the case of its humin (HN) or residual carbon (CR) fraction, the higher the nonlinearity of the sorption isotherm. Furthermore, a lower n
f value for carbaryl sorption in C soil, compared to sandy soil, points to a higher degree of sorption site heterogeneity of loam soil components active in insecticide uptake, which in turn may be due to its high CR fraction content. Since the pH of both tested soil materials was similar, the enhancement of C soil’s adsorption observed for the two carbamates can be related to its residual carbon and partly to humic acid carbon contents. This may be due to the pronounced hydrophobic retention of the carbamate by the CR fraction, supported by hydrogen bonding via the carbaryl’s amide carbonyl group and carboxylic groups of the HA fraction [
67].
The postulated particularly high affinity of carbaryl to the soil residual carbon fraction is in agreement with the studies of Murthy and Raghu (1991, [
68]), who found that nearly 78.9% of carbaryl was accumulated in the humin fraction of sandy loam soil, followed by the fulvic acid fraction (19.2%), whereas the least amount of pesticide was measured in the humic acid fraction (1.9%). Worth mentioning is the pH (6.8) of the investigated soil, which was very similar to that of the L soil studied herein. To further corroborate this finding, according to the literature, among the OC fractions the residual stable lignin fraction (of aromatic backbone, containing 64–65% of carbon) followed by CFA and CHA accounted for greater adsorption of some pesticides, including carbaryl [
27]. In another study [
41] it was also proven that carbaryl sorption on humic acid as a single sorbent was scarce in comparison to a more aromatic biochar, where a hydrophobic effect together with van der Waals forces were postulated to be responsible for insecticide effective retention, similar to the interactions claimed herein for soil C. Hence, the high CR content of C soil may significantly contribute to the generally higher sorption magnitude of carbaryl compared to sandy loam material.
According to the literature [
69], carbofuran predominantly incorporates into the recalcitrant humin fraction of SOM, constituting approximately 50% of the initial insecticide dose introduced into the soil. In a previous study [
41], it was proven that retention of non-ionic carbamates on HA was much weaker (35.4% of carbofuran and 10.2% of carbaryl initial dose) than on more hydrophobic wheat-straw biochar. Therefore, hydrophobic attraction may also govern carbofuran uptake by the particular SOM fractions. In addition, studies performed on the same L and C soils investigated herein [
10] have also shown that carbofuran retention was mainly controlled by goethite, expandable CMs, followed by HA (a slight but significant sorption increase) on sandy loam soil. This is in accordance with the study of Singh et al. [
70], who found that carbofuran adsorption was strongly correlated with clay content followed by the organic matter content of soils. However, insecticide uptake on the loam soil material was attributed solely to montmorillonite, presumably due to the enhanced physical diffusion of the pesticide into Mt interlayers, governed by van der Waals forces [
71]. It is commonly observed that the particulate organic (or organo-mineral) soil fractions form stable microaggregates where the retention of pesticides could depend more on steric factors and diffusion mechanisms than on the type and amounts of oxygen-containing functional groups [
72]. Therefore, both organic (mainly CR) and inorganic (Mt) soil colloids contribute to carbofuran retention in the studied soils via hydrophobic bonding and van der Waals forces. However, care should be taken when assessing their mutual superiority and share in the insecticide uptake by soils. Nevertheless, the results obtained herein clearly demonstrate that carbofuran molecules were bound weakly to loam and sandy loam soils, confirming the physical nature of insecticide sorption and a great leaching potential.
In the present study, metolachlor uptake by soil C was slightly but significantly higher than that of soil L (
Figure 4c). Its affinity for organic carbon (K
oc) of soil C (
Table 3) decreased nearly 1.5 times in comparison to sandy soil. Taking that into account, it can be concluded that the higher herbicide K
f calculated for C soil (
Table 1) indicates that apart from particular SOM fractions, other soil constituents must also contribute to its retention in loamy soil. This is in agreement with the studies of Weber et al. [
73], who related metolachlor uptake by soil to its organic matter, humic matter (HM) and clay contents. In general, they postulated that the herbicide sorption by soil was of a physical nature and was based on the following mechanisms: hydrophobic bonding to lipophilic SOM sites, charge–transfer interactions, van der Waals forces and H-bonds on polar surfaces of clay minerals. It is in line with other studies where it was proven that metolachlor exhibits a great affinity for the HA polar functional groups, which renders the formation of H-bonding via the carbonyl site and for hydrophobic bonding sites such as aromatic rings [
41], which may even preclude H-bonding [
18,
74]. The recent results of the herbicide sorption on Chernozem soil as well as its HA and HN fractions [
75] corroborate the higher affinity of the herbicide for humic acids. Kozak et al. [
76] studied the adsorption of metolachlor by various SOM constituents, including humic substances (HA and FA), humin and nonoxidizable soil organic matter. Based on the estimated partition coefficients normalized to organic carbon, they proved that fulvic and humic acids exhibited the highest affinity for the herbicide among the soil OC constituents. On the contrary, studies of Ding et al. [
65] revealed that the humin fraction was superior to humic acids in metolachlor uptake. However, the HN fractions they isolated from soils were, uncommonly, much more aromatic than the HA, and the qualitative differences in their structural characteristics were correlated with the long-term tillage management of the soil. This finding is in contradiction with the latest studies on the nature of humin, which demonstrate that its major components are predominantly aliphatic hydrocarbon functionalities [
77]. Nevertheless, it must be kept in mind that the type of extraction or isolation method will strongly influence the chemodiversity of this stable organic matter fraction [
16,
75,
78], as well as the results of the studies conducted on the single-sorbent systems. Among the inorganic soil constituents, particularly clay minerals, bentonites and montmorillonites were found to be effective sorbents of metolachlor [
79]. According to the literature, the herbicide sorption takes place dominantly as molecular species on hydrophobic microsites of Mt surfaces [
8,
80]. Presumably, metolachlor is adsorbed at the uncharged and hydrophobic siloxane groups of the clays silicate surface and held there by accepting hydrogen bonds from water molecules around the interlayer cations. Hence, in the present study, it can be postulated that in the case of C soil, the overall sorption magnitude of herbicide can be attributed in part to the humic acid fraction content (decreased by a factor of two in relation to L soil) and increased clay minerals content (mainly Mt [
10]).
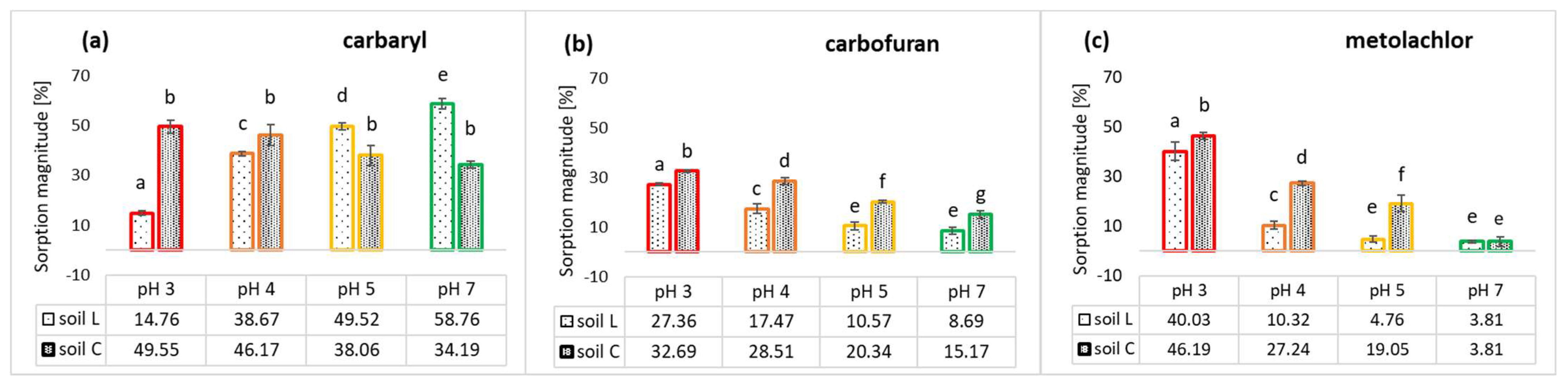
2.3. pH-Dependent Sorption of Carbamates and Metolachlor on L and C Soils
Sorption of carbaryl by soil L was noted to increase along with the soil pH, appreciably up to pH seven (
Figure 6a). According to some studies, carbaryl sorption is due, in part, to site-specific interactions between carbamate functional group and exchangeable cations [
27,
81], e.g., via Ca
2+ bridging. Thus, the trend observed for sandy loam material may be explained by the increasing competition between hydrogen and Ca
2+ ions for active sites on the adsorbent surface with pH decline. In such case, available adsorption domains for carbaryl on clay minerals (kaolinite and montmorillonite) and SOM fractions (
Table 4) on soil L will decrease. Furthermore, it was found that an increase in soil pH decreases the amount of extractable carbaryl residues [
68], increasing bound residues at the same time, which can be retained by the CR fraction of soil OC. The sorption magnitude found for carbaryl on soil C is in a narrow range of 34.19% to 49.55% and seemingly declines with a pH increase, but the differences are not statistically significant (
Figure 6a). The higher sorption magnitude observed for carbaryl on C soil of a pH of three to four compared to L soil is presumably related to its higher organic carbon content, as well as SOM composition (
Table 4). Over the range of experimental pH values, some part of surface functional groups (e.g., carbonyl, hydroxyl, carboxyl) should become more protonated with a pH decrease. This results in less negatively charged SOM fractions abundant in hydrophilic moieties at a lower pH [
82], which together with the high share of CR (59.71% of TOC) may favor adsorption of the 1-naphthyl-N-methyl moiety of carbaryl to the active sorption sites via hydrophobic bonding [
28].
Meanwhile, desorption of the agrochemical from the two soils studied was the lowest at pH three; however, magnitude of the phenomena varied with the soil (
Table 5). In loam, material carbaryl desorbed up to 25.34% (at pH 7) of the dose initially adsorbed, whereas in loamy sand soil its release ranged from 47.23% to 59.08%. Two conclusions can be drawn from these observations. Firstly, low pH reduces carbaryl desorption from loamy sand and loam soils studied, enhancing its retention. Secondly, carbaryl is more efficiently retained by loamy material, suggesting that the residual carbon fraction may be essential in the stabilization and immobilization of agrochemical residues in the soil environment.
Carbofuran sorption exhibited an inverse trend with a pH increase for both studied soils (
Figure 6b). Its sorption magnitude was significantly higher for loam soil material, achieving values of 27.36% and 32.69% of the carbofuran initial dose introduced for soils L and C, respectively. This discrepancy in the insecticide uptake by soils may be related to their different SOM quantities and compositions (
Table 4), which significantly influence the number of specific potential sorption centers for the adsorbate. However, these results are in contradiction to the work of Rama Krishna and Philip [
35], who found that with a pH increase from two to eight there was an increase in the sorption of carbofuran, methyl parathion and lindane in sandy, red, clayey and compost soils. A similar observation was found by Hsieh and Kao [
12], who reported the enhanced adsorption of carbofuran in lateritic soils with a pH increase. Nevertheless, lateritic soils, rich in aluminum and iron minerals, are formed in wet, hot tropical areas, contrary to the soil materials of temperate climate and low inorganic colloids content investigated herein. Another study by Gupta et al. [
82] revealed the inverse to pH adsorption trend for carbofuran, corroborating the results presented in this paper. Similarly, Salman et al. reported a favorable adsorption of carbofuran in acidic soils. Meanwhile, desorption of the insecticide was higher in sandy loam soil material (up to 98.1% of carbofuran initial dose at pH seven,
Table 5), suggesting that the forces holding it there were weak, enhancing a serious threat of groundwater contamination. Carbofuran release in soil C was significantly lower (maximum of 33.05%), suggesting the high efficiency of soil colloids in its immobilization in soil.
Metolachlor sorption in soil L is also pH dependent, and similarly to carbofuran, it declines inversely with pH. The same behavior of pesticide can be observed in loamy soil (
Figure 6c), which reveals an overall higher sorption capacity under the experimental conditions than the loamy sand soil material. This can be explained by the fact that a lower pH facilitates hydrogen bonds between the carbonyl oxygen of metolachlor and H atoms of carboxyl and hydroxyl groups of the organic moieties and charge–transfer (π-π) bonds between the aromatic nucleus of the herbicide and the aromatic rings on the organic matter surfaces [
83]. Meanwhile, the desorption of agrochemicals (
Table 5) increases with pH in sandy loam soil and is hindered by lower pH (three to five) conditions in case of loam soil material. Nevertheless, its magnitude is much higher in L soil (61.67%), whereas it seems to be limited by some C soil constituents (maximum desorption of 18.84% at pH seven), as they retain agrochemicals more efficiently than loamy sand.