Synthesis of Benzocycloalkanone-Based Michael Acceptors and Biological Activities as Antimalarial and Antitrypanosomal Agents
Abstract
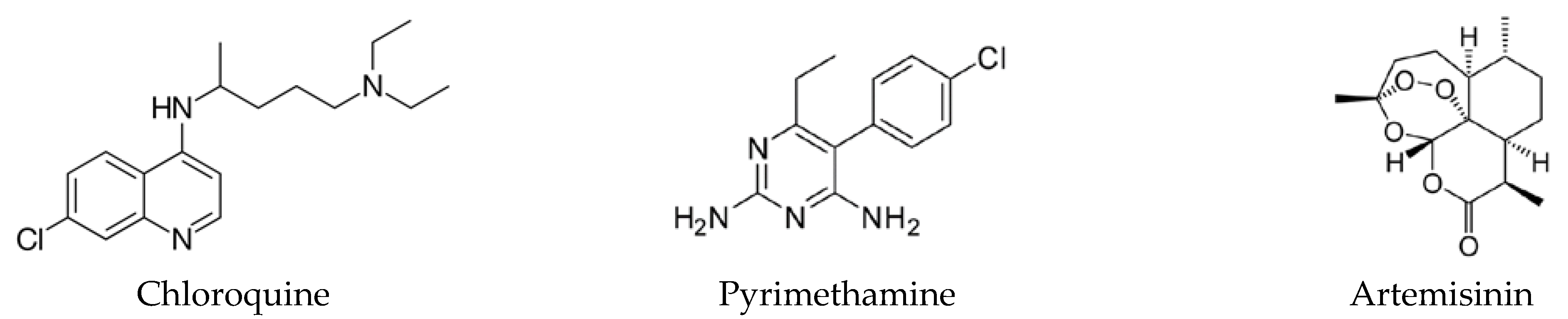
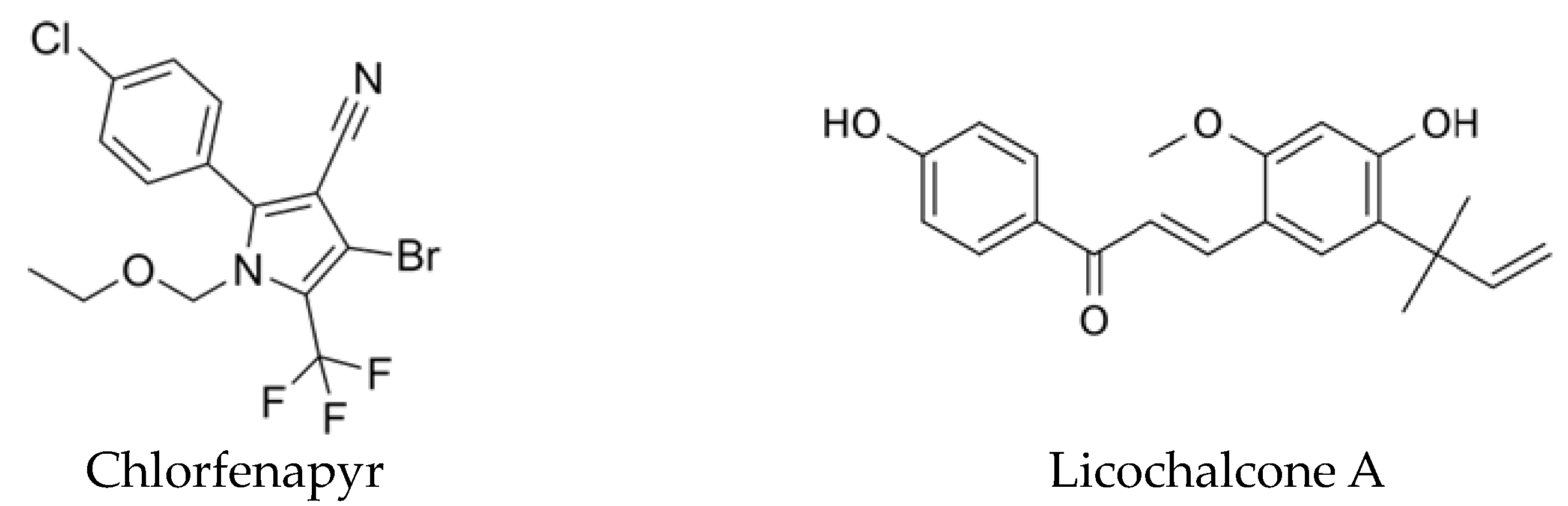
:1. Introduction
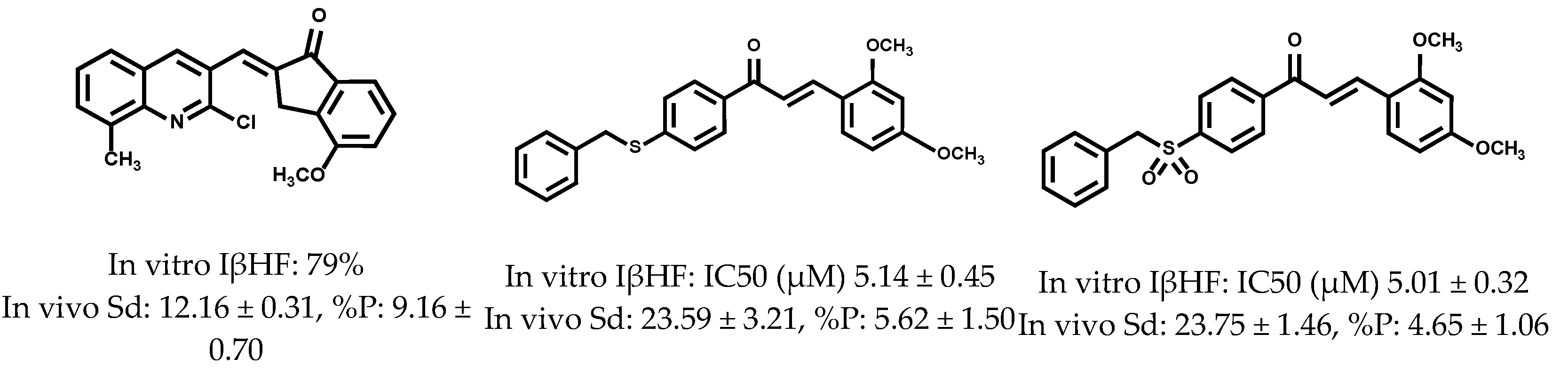
2. Results and Discussion
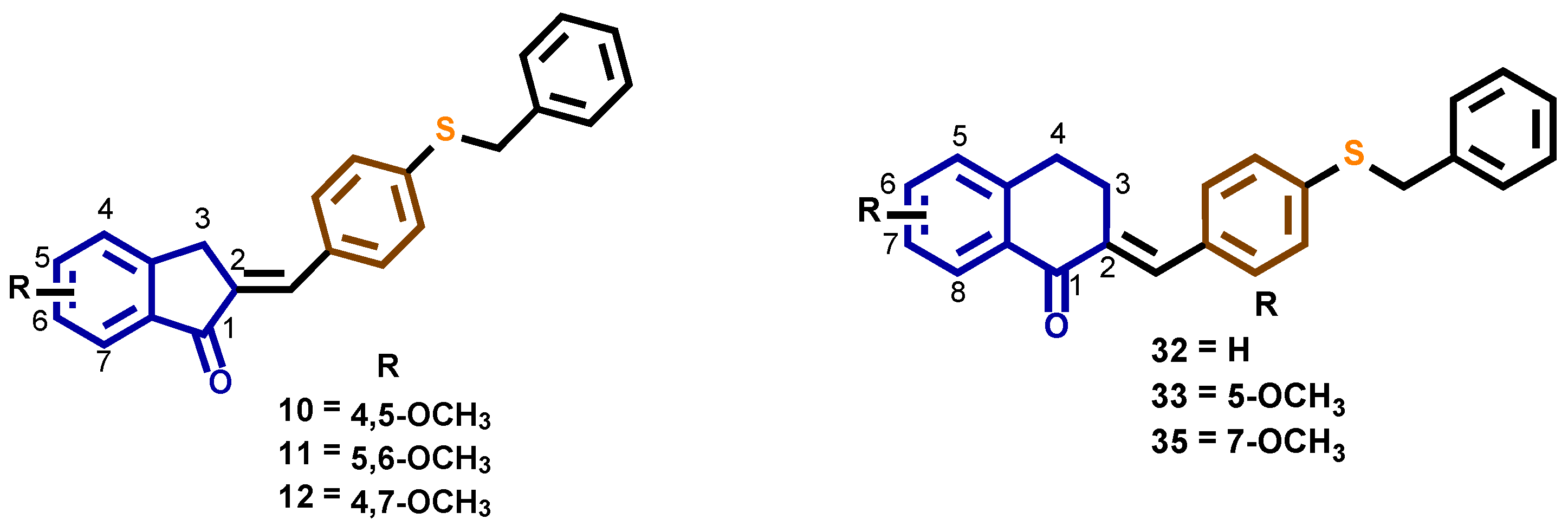
2.1. Synthesis of 4-Benzylsulfanyl and 4-Benzyloxybenzocycloalcanone Derivatives 7–39
2.2. Antiprotozoal Activity
3. Materials and Methods
3.1. Synthesis of 4-(Benzylthio)benzaldehyde 3
3.2. General Procedure for the Synthesis of (E)-2-[4-(Benzylthio)benzylidene or (E)-2-[4-(Benzyloxy)benzylidene]-Benzocycloalkanone Derivatives 7–39
3.2.1. (E)-2-[4-(Benzylthio)benzylidene]-2,3-dihydroinden-1-one 7
3.2.2. (E)-2-[4-(Benzylthio)benzylidene]-2,3-dihydro-5-methoxyinden-1-one 8
3.2.3. (E)-2-[4-(Benzylthio)benzylidene]-2,3-dihydro-6-methoxyinden-1-one 9
3.2.4. (E)-2-[4-(Benzylthio)benzylidene]-2,3-dihydro-4,5-dimethoxyinden-1-one 10
3.2.5. (E)-2-[4-(Benzylthio)benzylidene]-2,3-dihydro-5,6-dimethoxyinden-1-one 11
3.2.6. (E)-2-[4-(Benzylthio)benzylidene]-2,3-dihydro-4,7-dimethoxyinden-1-one 12
3.2.7. (E)-2-[4-(Benzylthio)benzylidene]-2,3-dihydro-5,7-dimethoxyinden-1-one 13
3.2.8. (E)-2-[4-(Benzylthio)benzylidene]-2,3-dihydro-5,6-methylendioxyinden-1-one 14
3.2.9. (E)-2-[4-(Benzylthio)benzylidene]-2,3-dihydro-4-methylinden-1-one 15
3.2.10. (E)-2-[4-(Benzylthio)benzylidene]-5-chloro-2,3-dihydroinden-1-one 16
3.2.11. (E)-2-[4-(Benzylthio)benzylidene]-6-chloro-2,3-dihydroinden-1-one 17
3.2.12. (E)-2-[4-(Benzylthio)benzylidene]-4,6-dichloro-2,3-dihydroinden-1-one 18
3.2.13. (E)-2-[4-(Benzylthio)benzylidene]-5,7-difluoro-2,3-dihydroinden-1-one 19
3.2.14. (E)-2-[4-(Benzyloxy)benzylidene]-2,3-dihydroinden-1-one 20
3.2.15. (E)-2-[4-(Benzyloxy)benzylidene]-2,3-dihydro-5-methoxyinden-1-one 21
3.2.16. (E)-2-[4-(Benzyloxy)benzylidene]-2,3-dihydro-6-methoxyinden-1-one 22
3.2.17. (E)-2-[4-(Benzyloxy)benzylidene]-2,3-dihydro-4,5-dimethoxyinden-1-one 23
3.2.18. (E)-2-[4-(Benzyloxy)benzylidene]-2,3-dihydro-5,6-dimethoxyinden-1-one 24
3.2.19. (E)-2-[4-(Benzyloxy)benzylidene]-2,3-dihydro-4,7-dimethoxyinden-1-one 25
3.2.20. (E)-2-[4-(Benzyloxy)benzylidene]-2,3-dihydro-5,6-methylendioxyinden-1-one 26
3.2.21. (E)-2-[4-(Benzyloxy)benzylidene]-2,3-dihydro-4-methylinden-1-one 27
3.2.22. (E)-2-[4-(Benzyloxy)benzylidene]-5-chloro-2,3-dihydroinden-1-one 28
3.2.23. (E)-2-[4-(Benzyloxy)benzylidene]-6-chloro-2,3-dihydroinden-1-one 29
3.2.24. (E)-2-[4-(Benzyloxy)benzylidene]-4,6-dichloro-2,3-dihydroinden-1-one 30
3.2.25. (E)-2-[4-(Benzyloxy)benzylidene]-5,7-difluoro-2,3-dihydroinden-1-one 31
3.2.26. (E)-2-[4-(Benzylthio)benzylidene]-3,4-dihydronaphthalen-1(2H)-one 32
3.2.27. (E)-2-[4-(Benzylthio)benzylidene]-3,4-dihydro-5-methoxynaphthalen-1(2H)-one 33
3.2.28. (E)-2-[4-(Benzylthio)benzylidene]-3,4-dihydro-6-methoxynaphthalen-1(2H)-one 34
3.2.29. (E)-2-[4-(Benzylthio)benzylidene]-3,4-dihydro-7-methoxynaphthalen-1(2H)-one 35
3.2.30. (E)-2-[4-(Benzyloxy)benzylidene]-3,4-dihydronaphthalen-1(2H)-one 36
3.2.31. (E)-2-[4-(Benzyloxy)benzylidene]-3,4-dihydro-5-methoxynaphthalen-1(2H)-one 37
3.2.32. (E)-2-[4-(Benzyloxy)benzylidene]-3,4-dihydro-6-methoxynaphthalen-1(2H)-one 38
3.2.33. (E)-2-[4-(Benzyloxy)benzylidene]-3,4-dihydro-7-methoxynaphthalen-1(2H)-one 39
3.3. Biology
3.3.1. Inhibition of β-Haematin Formation In Vitro
3.3.2. Plasmodium Berghei and Experimental Host Maintenance
3.3.3. Four-Day Suppressive Test
3.3.4. Trypanocidal Activity
3.3.5. Cytotoxicity Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Centers for Disease Control and Prevention. March 2022. Available online: https://www.cdc.gov/parasites/about.html (accessed on 5 July 2023).
- Barrow, P.; Dujardin, J.C.; Fasel, N.; Greenwood, A.D.; Osterrieder, K.; Lomonossof, G.; Fiori, P.L.; Atterbury, R.; Rossi, M.; Lalle, M. Viruses of protozoan parasites and viral therapy: Is the time now right? Virol. J. 2020, 17, 142. [Google Scholar] [CrossRef] [PubMed]
- Ending the Neglect to Attain the Sustainable Development Goals: A Road Map for Neglected Tropical Diseases 2021–2030. Geneva: World Health Organization; 2020. License: CC BY-NC-SA 3.0 IGO. Available online: https://www.who.int/publications/i/item/9789240010352 (accessed on 5 June 2023).
- World Health Organization. Available online: http://www.who.int/chagas/en/ (accessed on 15 February 2023).
- Castillo-Riquelme, M. Chagas disease in non-endemic countries. Lancet Glob. Health 2017, 5, e379–e380. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Malaria Report. An In-Depth Update on Global and Regional Malaria Data and Trends. Available online: https://www.who.int/teams/global-malaria-programme/reports/world-malaria-report-2022 (accessed on 15 February 2023).
- Short, E.E.; Caminade, C.; Bolaji, N.; Thomas, B.N. Climate change contribution to the emergence or re-emergence of parasitic diseases. Infect. Dis. Res. Treat. 2017, 10, 1178633617732296. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ridley, R.G. Medical need, scientific opportunity and the drive for antimalarial drugs. Nature 2002, 415, 686–693. [Google Scholar] [CrossRef] [PubMed]
- Ashley, E.; Dhorda, M.; Fairhurst, R.; Amaratunga, C.; Lim, P.; Suon, S.; Sreng, S.; Anderson, J.; Mao, S.; Sam, B.; et al. Spread of artemisinin resistance in Plasmodium falciparum malaria. N. Engl. J. Med. 2014, 371, 411–423. [Google Scholar] [CrossRef] [Green Version]
- World Health Organization. WHO Recommends Groundbreaking Malaria Vaccines for Children at Risk. 2021. Available online: https://www.who.int/news/item/06-10-2021-who-recommends-groundbreaking-malaria-vaccine-for-children-at-risk (accessed on 2 May 2023).
- Mosha, J.F.; Kulkarni, M.A.; Lukole, E.; Matowo, N.S.; Pitt, C.; Messenger, L.A.; Mallya, E.; Jumanne, M.; Aziz, T.; Kaaya, R.; et al. Effectiveness and cost-effectiveness against malaria of three types of dual-active-ingredient long-lasting insecticidal nets (LLINs) compared with pyrethroid-only LLINs in Tanzania: A four-arm, cluster-randomized trial. Lancet 2022, 399, 1227–1241. [Google Scholar] [CrossRef]
- Njoroge, M.; Njuguna, N.M.; Mutai, P.; Ongarora, D.S.B.; Smith, P.W.; Chibale, K. Recent approaches to chemical discovery and development against malaria and the Neglected Tropical Diseases human African trypanosomiasis and schistosomiasis. Chem. Rev. 2014, 114, 11138–11163. [Google Scholar] [CrossRef]
- Njogu, P.M.; Guantai, E.M.; Pavadai, E.; Chibale, K. Computer-aided drug discovery approaches against the tropical infectious diseases malaria, tuberculosis, trypanosomiasis, and leishmaniasis. ACS Infect. Dis. 2016, 2, 8–31. [Google Scholar] [CrossRef] [PubMed]
- Goupil, L.; McKerrow, J. Introduction: Drug discovery and development for neglected diseases. Chem. Rev. 2014, 114, 11131–11137. [Google Scholar] [CrossRef] [PubMed]
- Devine, W.; Thomas, S.M.; Erath, J.; Bachovchin, K.A.; Lee, P.J.; Leed, S.E.; Rodriguez, A.; Sciotti, R.J.; Mensa-Wilmot, K.; Pollastri, M.P. Antiparasitic lead discovery: Toward optimization of a chemotype with activity against multiple protozoan parasites. ACS Med. Chem. Lett. 2017, 8, 350–354. [Google Scholar] [CrossRef] [Green Version]
- Noronha, M.; Pawar, V.; Prajapati, A.; Subramanian, R. A literature review on traditional herbal medicines for malaria. S. Afr. J. Bot. 2020, 128, 292–303. [Google Scholar] [CrossRef]
- Kapishnikov, S.; Hempelmann, E.; Elbaum, M.; Als-Nielsen, J.; Leiserowitz, L. Malaria pigment crystals: The achilles’ heel of the malaria parasite. ChemMedChem. 2021, 16, 1515–1532. [Google Scholar] [CrossRef] [PubMed]
- Mishra, N.; Arora, P.; Kumar, B.; Mishra, L.; Bhattacharya, A.; Awasthi, S.; Bhasin, V. Synthesis of novel substituted 1,3-diaryl propenone derivatives and their antimalarial activity in vitro. Eur. J. Med. Chem. 2008, 43, 1530–1535. [Google Scholar] [CrossRef] [PubMed]
- Shih, H.; Deng, L.; Carrera, C.; Adachi, S.; Cottam, H.; Carson, D. Rational design, synthesis, and structure-activity relationships of antitumor (E)-2-benzylidene-1-tetralones and (E)-2-benzylidene-1-indanones. Bioorg. Med. Chem. Lett. 2000, 10, 487–490. [Google Scholar] [CrossRef] [PubMed]
- Beteck, R.M.; Legoabe, L.J.; Isaacs, M.; Khanye, S.D.; Laming, D.; Hoppe, H.C. Anti-Trypanosomal and antimalarial properties of tetralone derivatives and structurally related benzocycloalkanones. Medicina 2019, 55, 206. [Google Scholar] [CrossRef] [Green Version]
- Charris, J.; Domínguez, J.; Gamboa, N.; Rodrigues, J.; Angel, J. Synthesis and antimalarial activity of E-2-quinolinylbenzocycloalcanones. Eur. J. Med. Chem. 2005, 40, 875–881. [Google Scholar] [CrossRef]
- Charris, J.; Lobo, G.; Camacho, J.; Ferrer, R.; Barazarte, A.; Domínguez, J.; Gamboa, N.; Rodrigues, J.; Angel, J. Synthesis and antimalarial activity of (E) 2-(2′-Chloro-3′-quinolinylmethylidene)-5,7-dimethoxyindanones. Lett. Drug Des. Dis. 2007, 4, 49–54. [Google Scholar] [CrossRef]
- Ferrer, R.; Lobo, G.; Gamboa, N.; Rodrigues, J.; Abramjuk, C.; Jung, K.; Lein, M.; Charris, J. Synthesis of [(7-chloroquinolin-4-yl)amino]chalcones: Potential antimalarial and anticancer agents. Sci. Pharm. 2009, 77, 725–742. [Google Scholar] [CrossRef] [Green Version]
- Domínguez, J.; León, C.; Rodrigues, J.; Gamboa, N.; Gut, J.; Rosenthal, P. Synthesis of chlorovinyl sulfones as structural analogs of chalcones and their antiplasmodial activities. Eur. J. Med. Chem. 2009, 44, 1457–1462. [Google Scholar] [CrossRef]
- Bahekar, S.; Hande, S.; Agrawal, N.; Chandak, H.; Bhoj, P.; Goswami, K.; Reddy, M. Sulfonamide chalcones: Synthesis and in vitro exploration for therapeutic potential against Brugia malayi. Eur. J. Med. Chem. 2016, 124, 262–269. [Google Scholar] [CrossRef]
- Li, R.; Kenyon, G.; Cohen, F.; Chen, X.; Gong, B.; Domínguez, J.; Davidson, E.; Kurzban, G.; Miller, R.; Nuzum, E.; et al. In vitro antimalarial activity of chalcones and their derivatives. J. Med. Chem. 1995, 38, 5031–5037. [Google Scholar] [CrossRef]
- Zhuang, C.; Zhang, W.; Sheng, C.; Zhang, W.; Xing, C.; Miao, Z. Chalcone: A privileged structure in medicinal chemistry. Chem. Rev. 2017, 117, 7762–7810. [Google Scholar] [CrossRef] [PubMed]
- Sahu, N.; Balbhadra, S.; Choudhary, J.; Kohli, D. Exploring the pharmacological significance of chalcone scaffold: A review. Curr. Med. Chem. 2012, 19, 209–225. [Google Scholar] [CrossRef]
- Mahapatra, D.; Bharti, S.; Asati, V. Chalcone scaffolds as anti-infective agents: Structural and molecular target perspectives. Eur. J. Med. Chem. 2015, 101, 496–524. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, C.; Gomes, J.; Gomes, P. Falcipains, Plasmodium falciparum cysteine proteases as key drug targets against malaria. Curr. Med. Chem. 2011, 18, 1555–1572. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Go, M.; Liu, M.; Wilairat, P.; Rosenthal, P.; Saliba, K.; Kirk, K. Antiplasmodial chalcones inhibit sorbitol-induced hemolysis of Plasmodium falciparum-infected erythrocytes. Antimicrob. Agents Chemother. 2004, 48, 3241–3245. [Google Scholar] [CrossRef] [Green Version]
- Geyer, J.; Keenan, S.; Woodard, C.; Thompson, P.; Gerena, L.; Nichols, D.; Gutteridge, C.; Waters, N. Selective inhibition of Pfmrk, a Plasmodium falciparum CDK, by antimalarial 1,3-diaryl-2-propenones. Bioorg. Med. Chem. Lett. 2009, 19, 1982–1985. [Google Scholar] [CrossRef]
- Srivastava, S.; Joshi, S.; Singh, A.; Yadav, S.; Saxena, A.; Ram, V.; Chandra, S.; Saxena, J. Oxygenated chalcones and bischalcones as a new class of inhibitors of DNA topoisomerase II of malarial parasites. Med. Chem. Res. 2008, 17, 234–244. [Google Scholar] [CrossRef]
- Kumar, S.; Guha, M.; Choubey, V.; Maity, P.; Bandyopadhyay, U. Antimalarial drugs inhibiting hemozoin (beta-hematin) formation: A mechanistic update. Life Sci. 2007, 80, 813–828. [Google Scholar] [CrossRef]
- Le Bonniec, S.; Deregnaucourt, C.; Redeker, V.; Banerjee, R.; Grellier, P.; Goldberg, D.; Schrével, J. Plasmepsin II, an acidic hemoglobinase from the Plasmodium falciparum food vacuole, is active at neutral pH on the host erythrocyte membrane skeleton. J. Biol. Chem. 1999, 274, 14218–14223. [Google Scholar] [CrossRef] [Green Version]
- Ribeiro, F.F.; Junior, F.J.; da Silva, M.S.; Scotti, M.T.; Scotti, L. Computational and investigative study of flavonoids active against Trypanosoma cruzi and Leishmania spp. Nat. Prod. Commun. 2015, 10, 917–920. [Google Scholar] [PubMed]
- Magalhaes, E.P.; Barroso Gomes, N.D.; Araújo de Freitas, T.; Pinheiro Silva, B.; Rodrigues Ribeiro, L.; Queiroz Ameida-Neto, F.W.; Machado Marinho, M.; de Lima-Neto, P.; Silva Marinho, E.; Silva dos Santos, H.; et al. Chloride substitution on 2-hydroxy-3,4,6-trimethoxyphenylchalcones improves in vitro selectivity on Trypanosoma cruzi strain Y. Chem. Biol. Interact. 2022, 361, 109920. [Google Scholar] [CrossRef]
- Leitao Cavalcante, C.H.; Queiroz Almeida-Neto, F.W.; Nunes da Rocha, M.; Nogueira Bandeira, P.; Pessoa Bezerra de Menezes, R.R.; Magalhaes, E.P.; Lima Sampaio, T.; Silva Marinho, E.; Machado Marinho, M.; Costa Martins, A.M.; et al. Antichagasic evaluation, molecular docking and ADMET properties of the chalcone (2E)-3-(2-fluorophenyl)-1-(2-hydroxy- 3,4,6-trimethoxyphenyl)prop-2-en1-one against Trypanosoma cruzi. J. Biomol. Struct. Dyn. 2022. online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Ramírez, H.; Domínguez, J.; Fernandez-Moreira, E.; Rodrigues, J.; Rodríguez, m.; Charris, J.E. Synthesis of 4-benzylsulfanyl and 4-benzylsulfonyl chalcones. biological evaluation as antimalarial agents. Farmacia J. 2022, 70, 30–42. [Google Scholar] [CrossRef]
- Baelmans, R.; Deharo, E.; Muñoz, V.; Sauvain, M.; Ginsburg, H. Experimental conditions for testing the inhibitory activity of chloroquine on the formation of beta-hematin. Exp. Parasitol. 2000, 96, 243–248. [Google Scholar] [CrossRef]
- Peters, W.; Robinson, B. Parasitic Infection Models. In Handbook of Antimalarial Models of Infection; Zak, O., Sande, M., Eds.; Academic Press: London, UK, 1999; p. 757. [Google Scholar]
- Leañez, J.; Nuñez, J.; García-Marchan, Y.; Sojo, F.; Arvelo, F.; Rodriguez, D.; Buscema, I.; Alvarez-Aular, A.; Bello, J.; Kouznetsov, V.; et al. Anti-leishmanial effect of spiro dihydroquinoline-oxindoles on volume regulation decrease and sterol biosynthesis of Leishmania braziliensis. Exp. Parasitol. 2019, 198, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Martín, X.; Payares, G.; Mendoza-León, A. Glibenclamide, a blocker of K+ATP channels, shows antileishmanial activity in experimental murine cutaneous leishmaniasis. Antimicrob. Agents Chemother. 2006, 4, 4214–4216. [Google Scholar] [CrossRef] [PubMed] [Green Version]


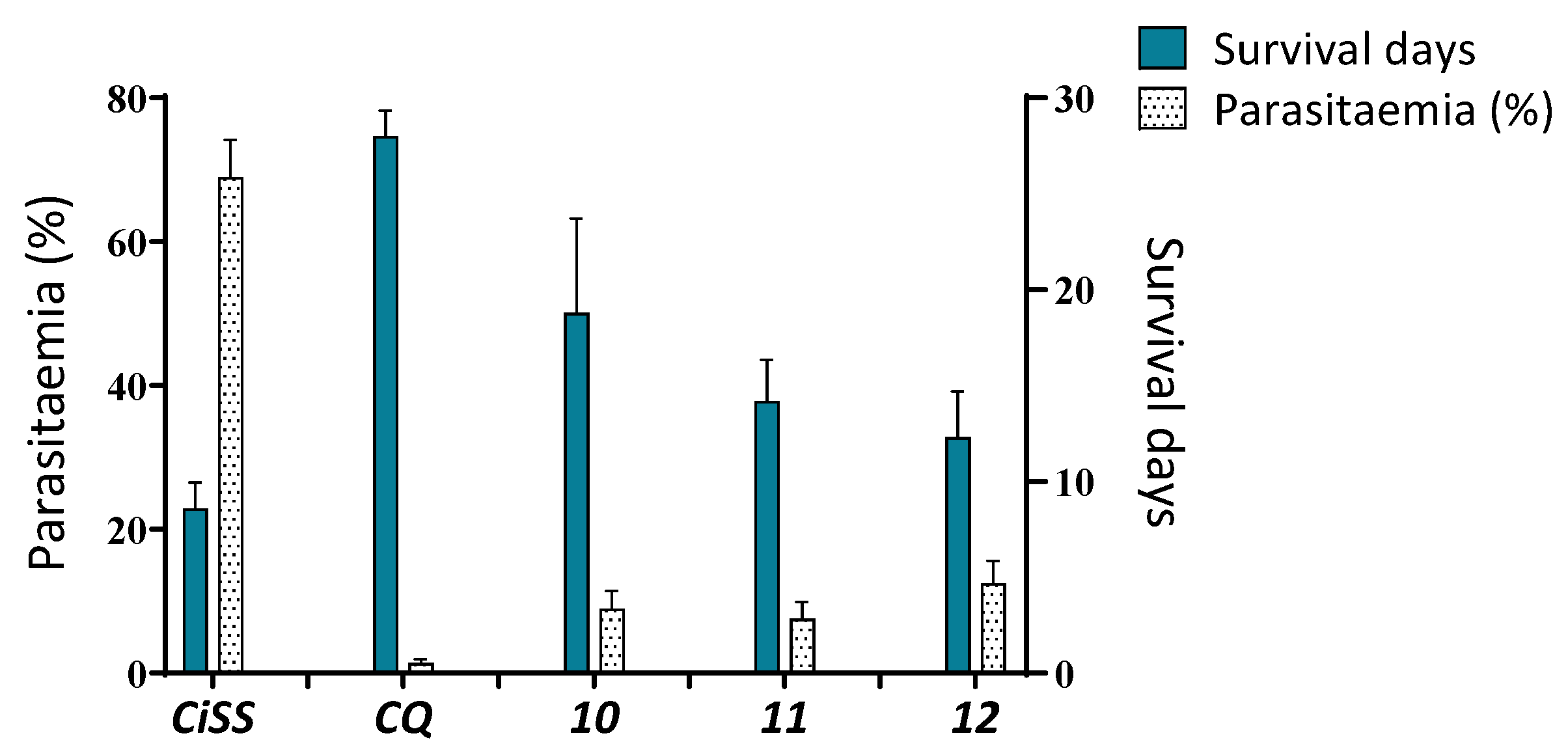
| No | %IβHF (± SEM) a | IC50(µM) (± SEM) b | Sd(± SEM) c | %P(± SEM) d | Survival |
|---|---|---|---|---|---|
| 10 | 75.16 | 33.1 ± 2.5 | 18.8 ± 4.9 * | 9.1 ± 2.44 | 0/6 |
| 11 | 63.02 | 0.82 ± 0.1 | 14.2 ± 2.2 * | 7.6 ± 2.3 | 0/6 |
| 12 | 56.17 | 0.66 ± 0.1 | 12.3 ± 2.4 | 12.5 ± 3.1 | 0/6 |
| CQ | 95.34 | 0.18 ± 0.1 | 28 ± 1.3 | 1.4 ± 0.54 | 5/6 |
| CiSS | - | - | 8.6 ± 1.3 | 69.9 ± 5.17 | 0/6 |
| No | %ITcP (50 µM) a | VERO IC50 µM |
|---|---|---|
| 10 | 32.95 ± 5.6 | >150 |
| 11 | 39.99 ± 8.2 | >150 |
| 12 | 18.50 ± 2.9 | >150 |
| 32 | 43.66 ± 6.7 | >150 |
| 33 | 51.08 ± 3.4 | >150 |
| 35 | 36.25 ± 6.1 | >150 |
| Bnz | 59.99 ± 2.9 | >200 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mijoba, A.; Fernandez-Moreira, E.; Parra-Giménez, N.; Espinosa-Tapia, S.; Blanco, Z.; Ramírez, H.; Charris, J.E. Synthesis of Benzocycloalkanone-Based Michael Acceptors and Biological Activities as Antimalarial and Antitrypanosomal Agents. Molecules 2023, 28, 5569. https://doi.org/10.3390/molecules28145569
Mijoba A, Fernandez-Moreira E, Parra-Giménez N, Espinosa-Tapia S, Blanco Z, Ramírez H, Charris JE. Synthesis of Benzocycloalkanone-Based Michael Acceptors and Biological Activities as Antimalarial and Antitrypanosomal Agents. Molecules. 2023; 28(14):5569. https://doi.org/10.3390/molecules28145569
Chicago/Turabian StyleMijoba, Ali, Esteban Fernandez-Moreira, Nereida Parra-Giménez, Sandra Espinosa-Tapia, Zuleyma Blanco, Hegira Ramírez, and Jaime E. Charris. 2023. "Synthesis of Benzocycloalkanone-Based Michael Acceptors and Biological Activities as Antimalarial and Antitrypanosomal Agents" Molecules 28, no. 14: 5569. https://doi.org/10.3390/molecules28145569






