Advanced Development of Molecularly Imprinted Membranes for Selective Separation
Abstract
:1. Introduction
2. Development of Molecular Imprinting Membranes
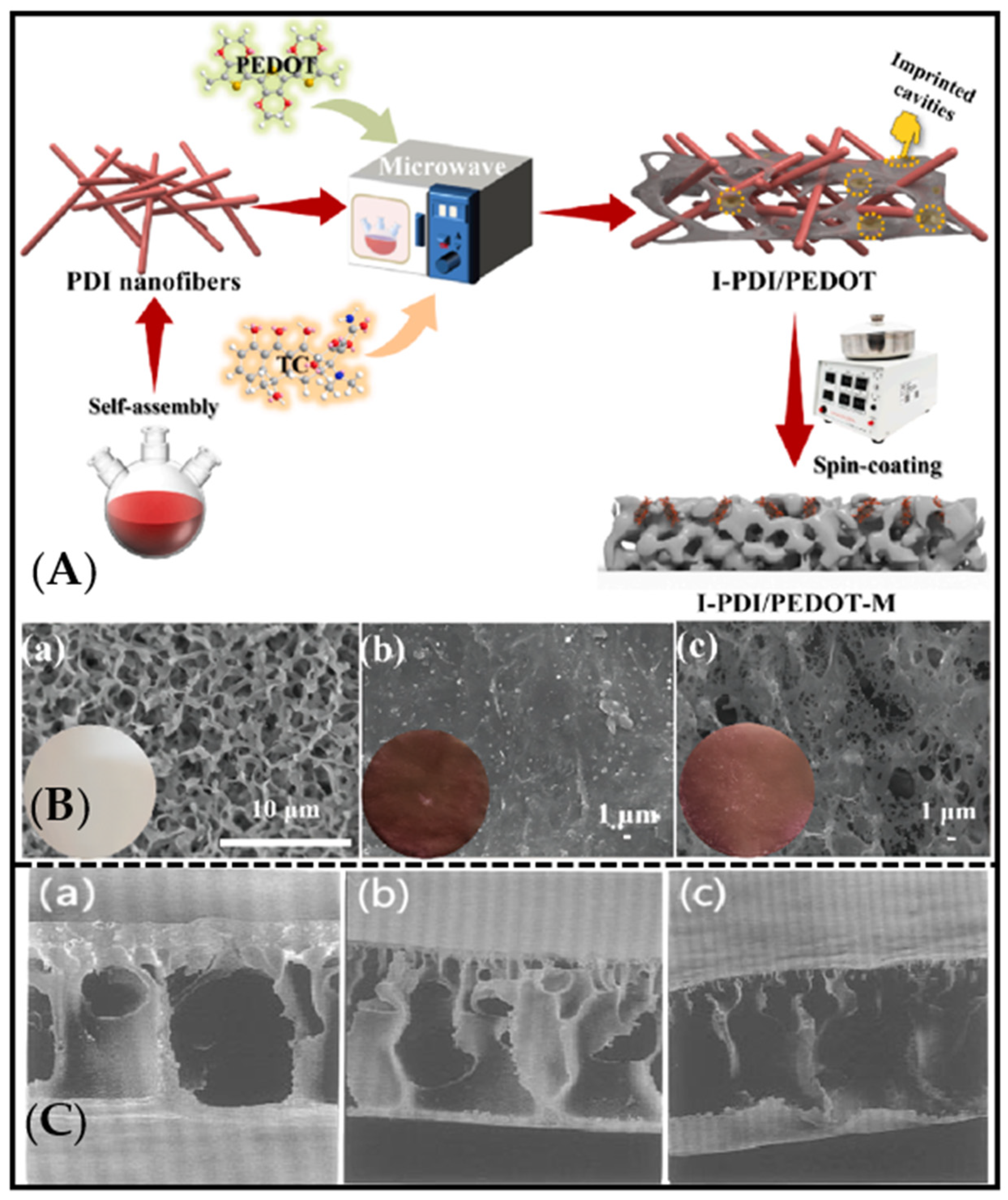
3. Preparation Method for Molecularly Imprinted Membrane
3.1. In Situ Polymerization Method
3.2. Phase-Inversion Method
| Method | Characteristics | Preparation Process | Ref. |
|---|---|---|---|
| In situ polymerization method | Simple preparation process. The obtained membrane has high rigidity and low porosity, but also high level of thickness, poor membrane permeability, and difficulty in diffusion of template molecules out of the imprinted membrane | The support membrane is immersed in the casting solution made of template molecule, functional monomer, and crosslinking agent, and the template molecules are eluted after the polymerization reaction | [59] |
| Phase-inversion method | Improves membrane flux and allows the acquisition of MIM-recognition sites directly in polymeric materials without the need for additional imprinted molecules | A certain amount of template molecules and functional polymers are dissolved on the carrier in a suitable solvent and placed in a coagulation bath or an inert gas atmosphere | [65,66,67] |
| Coating method | It is simple to operate and has good application prospects, but the prepolymer solution must have a suitable concentration | The prepolymer solution is dispersed in the solvent and uniformly dispersed in the substrate film. The composite layer is fixed on the surface of the substrate by chemical-crosslinking method | [70,71] |
| Electrochemical process | Fast preparation speed, controllable thickness of imprinted film can be prepared directly on the electrode surface, low level of film thickness, and solves the problem of contact between the film and sensor interface | Selection and cleaning of the appropriate electrode, preparation of molding fluid, and elution of imprinted molecules after electrochemical polymerization | [72,73] |
| Sol–gel process | Excellent selectivity, adsorption rate, and kinetic properties | Hydrolysis of precursors, condensation, gelation, and thermal treatment of the sol–gel material after drying | [74,75,76] |
3.3. Coating Method
3.4. Electrochemical Process
3.5. Sol–gel Process
4. Important Parameters in the Separation Application of the MIM
4.1. Selective Separation
4.1.1. Permselectivity/Flux
4.1.2. Mechanisms for Selective Transport
- (1)
- “retarded permeation”
- (2)
- “facilitated permeation”
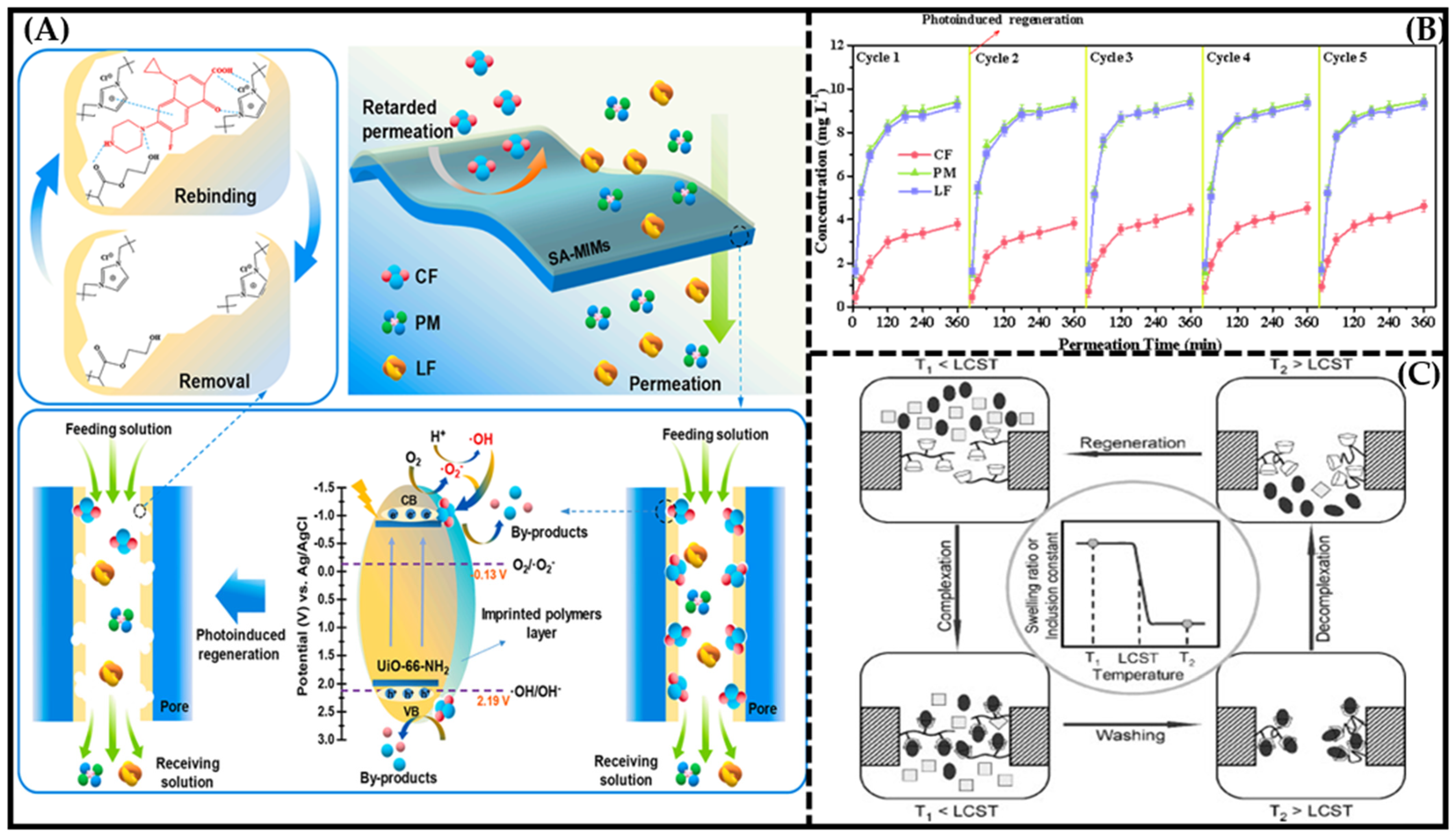
4.2. Regeneration Performance
4.3. Antifouling Performance
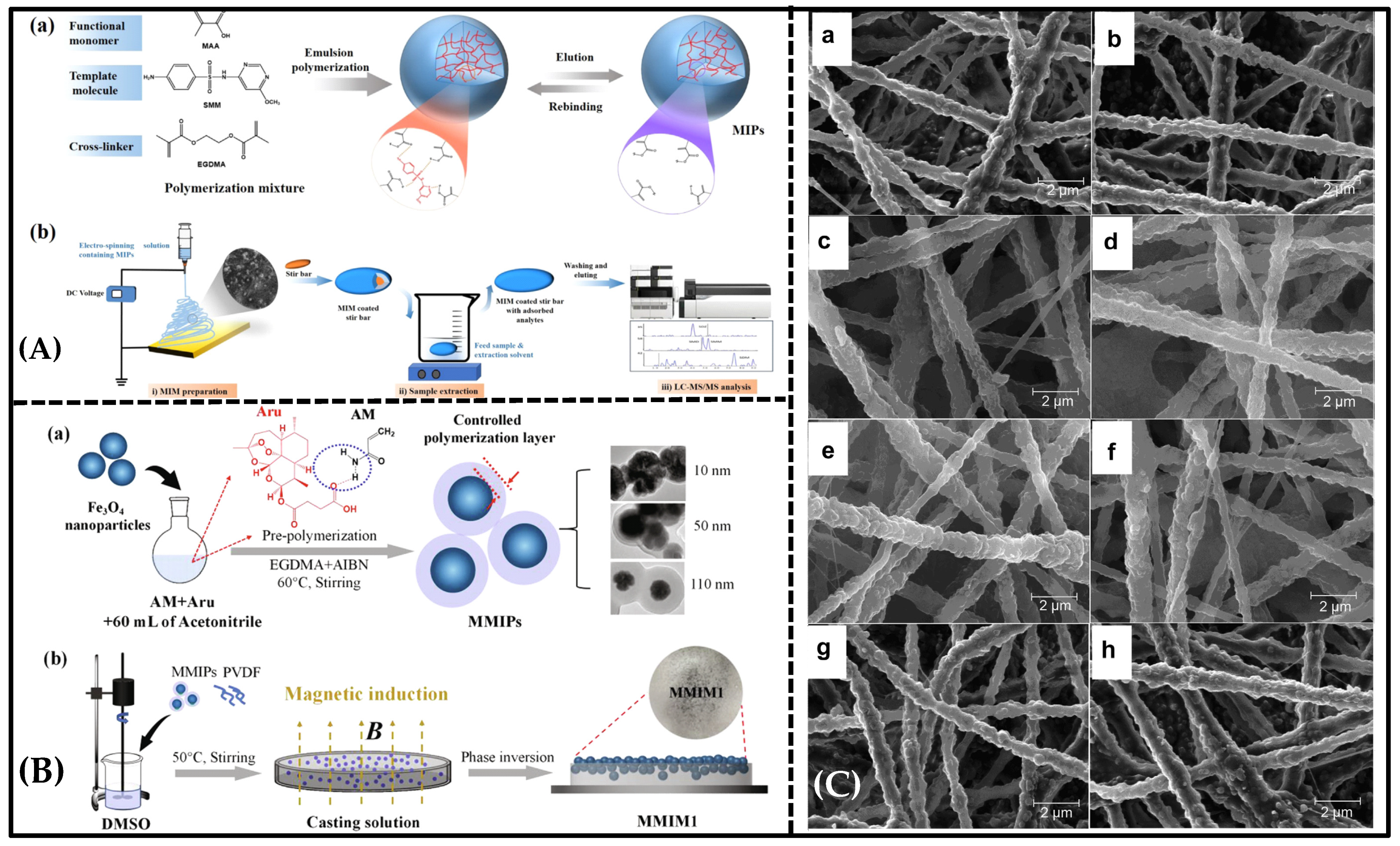
5. Emerging Molecularly Imprinted Membranes in Separation
5.1. Molecularly Imprinted Nanofiber Membranes
5.2. New Phase-Inversion Molecularly Imprinted Membrane
5.3. Metal–Organic-Framework-Material-Based Molecularly Imprinted Membrane
6. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tzschucke, C.C.; Markert, C. Modern Separation Techniques for the Efficient Workup in Organic Synthesis. Angew. Chem. Int. Ed. 2002, 41, 3964–4000. [Google Scholar] [CrossRef]
- Marchetti, P.; Jimenez Solomon, M.F. Molecular separation with organic solvent nanofiltration: A critical review. Chem. Rev. 2014, 114, 10735–10806. [Google Scholar] [CrossRef] [PubMed]
- El-Bourawi, M.S.; Ding, Z. A framework for better understanding membrane distillation separation process. J. Membr. Sci. 2006, 285, 4–29. [Google Scholar] [CrossRef]
- Hartl, J.; Marr, R. Extraction processes for bioproduct separation. Sep. Sci. Technol. 1993, 28, 805–819. [Google Scholar] [CrossRef]
- Wang, Z.-Q.; Wang, X. Pillararene-based supramolecular polymers for adsorption and separation. Adv. Mater. 2023, 2301721. [Google Scholar] [CrossRef]
- Cisternas, L.A.; Vásquez, C.M. On the design of crystallization-based separation processes: Review and extension. AlChE J. 2006, 52, 1754–1769. [Google Scholar] [CrossRef]
- Coskun, O. Separation techniques: Chromatography. North Clin. Istanb. 2016, 3, 156–160. [Google Scholar] [CrossRef]
- Adhikari, S.; Fernando, S. Hydrogen membrane separation techniques. Ind. Eng. Chem. Res. 2006, 45, 875–881. [Google Scholar] [CrossRef]
- Sirkar, K.K. Membrane Separation Technologies: Current Developments. Chem. Eng. Commun. 1997, 157, 145–184. [Google Scholar] [CrossRef]
- Cassano, A.; Conidi, C. Fractionation of olive mill wastewaters by membrane separation techniques. J. Hazard. Mater. 2013, 248–249, 185–193. [Google Scholar] [CrossRef]
- Hong, J.-M.; Anderson, P.E. Selectively-Permeable Ultrathin Film Composite Membranes Based on Molecularly-Imprinted Polymers. Chem. Mater. 1998, 10, 1029–1033. [Google Scholar] [CrossRef]
- Ripperger, S.; Altmann, J. Crossflow microfiltration—State of the art. Sep. Purif. Technol. 2002, 26, 19–31. [Google Scholar] [CrossRef]
- Tran, D.H.; Ulbricht, M. Cellulose-cellulose composite membranes for ultrafiltration. J. Membr. Sci. 2023, 672, 121426. [Google Scholar] [CrossRef]
- Mohammad, A.W.; Teow, Y.H. Nanofiltration membranes review: Recent advances and future prospects. Desalination 2015, 356, 226–254. [Google Scholar] [CrossRef]
- Joo, S.H.; Tansel, B. Novel technologies for reverse osmosis concentrate treatment: A review. J. Environ. Manag. 2015, 150, 322–335. [Google Scholar] [CrossRef]
- Bowry, S.K. Dialysis Membranes Today. Int. J. Artif. Organs 2002, 25, 447–460. [Google Scholar] [CrossRef]
- Bazinet, L.; Lamarche, F. Bipolar-membrane electrodialysis: Applications of electrodialysis in the food industry. Trends Food Sci. Technol. 1998, 9, 107–113. [Google Scholar] [CrossRef]
- McKeown, N.B.; Budd, P.M. Polymers of intrinsic microporosity (PIMs): Organic materials for membrane separations, heterogeneous catalysis and hydrogen storage. Chem. Soc. Rev. 2006, 35, 675–683. [Google Scholar] [CrossRef]
- Huang, L.; Zhang, M. Graphene-Based Membranes for Molecular Separation. J. Phys. Chem. Lett. 2015, 6, 2806–2815. [Google Scholar] [CrossRef]
- Yoshikawa, M.; Tanioka, A. Molecularly imprinted nanofiber membranes. Curr. Opin. Chem. Eng. 2011, 1, 18–26. [Google Scholar] [CrossRef]
- Song, J.; Klymov, A. Electrospun nanofibrous silk fibroin membranes containing gelatin nanospheres for controlled delivery of biomolecules. Adv. Healthc. Mater. 2017, 6, 1700014. [Google Scholar] [CrossRef] [PubMed]
- BelBruno, J.J. Molecularly Imprinted Polymers. Chem. Rev. 2019, 119, 94–119. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Xu, S. Recent advances in molecular imprinting technology: Current status, challenges and highlighted applications. Chem. Soc. Rev. 2011, 40, 2922–2942. [Google Scholar] [CrossRef]
- Wulff, G.; Sarhan, A. The use of polymers with enzyme-analogous structures for the resolution of racemates. Angew. Chem. Int. Ed. 1972, 11, 341–346. [Google Scholar]
- Mosbach, K.; Ramström, O. The Emerging Technique of Molecular Imprinting and Its Future Impact on Biotechnology. Bio/Technology 1996, 14, 163–170. [Google Scholar] [CrossRef]
- Zhang, H.; Ye, L. Non-covalent molecular imprinting with emphasis on its application in separation and drug development. J. Mol. Recognit. 2006, 19, 248–259. [Google Scholar] [CrossRef]
- Andersson, L.I.; Mosbach, K. Enantiomeric resolution on molecularly imprinted polymers prepared with only non-covalent and non-ionic interactions. J. Chromatogr. A 1990, 516, 313–322. [Google Scholar] [CrossRef]
- Kandimalla, V.B.; Ju, H. Molecular imprinting: A dynamic technique for diverse applications in analytical chemistry. Anal. Bioanal. Chem. 2004, 380, 587–605. [Google Scholar] [CrossRef]
- Yang, H.; Liu, H.-B. Synthesis, performance, and application of molecularly imprinted membranes: A review. J. Environ. Chem. Eng. 2021, 9, 106352. [Google Scholar] [CrossRef]
- Ratnaningsih, E.; Kadja, G.T.M. Molecularly imprinted affinity membrane: A review. ACS Omega 2022, 7, 23009–23026. [Google Scholar] [CrossRef]
- Donato, L.; Nasser, I.I. Green chemistry and molecularly imprinted membranes. Membranes 2022, 12, 472. [Google Scholar] [CrossRef]
- Piletsky, S.A.; Dubey, I. Substrate-selective polymeric membranes. Sel. Transf. Nucleic Acids Components. Biopolym. Cell 1990, 6, 55–58. [Google Scholar]
- Wang, H.Y.; Kobayashi, T. Molecular Imprint Membranes Prepared by the Phase Inversion Precipitation Technique. Langmuir 1996, 12, 4850–4856. [Google Scholar] [CrossRef]
- Yoshikawa, M. Molecularly imprinted polymeric membranes. Bioseparation 2001, 10, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.Y.; Kobayashi, T. Molecular imprint membranes prepared by the phase inversion precipitation technique. 2. influence of coagulation temperature in the phase inversion process on the encoding in polymeric membranes. Langmuir 1997, 13, 5396–5400. [Google Scholar] [CrossRef]
- Meng, M.; Li, Y. Hydrophilic imprinted MnO2 nanowires “coating” membrane with ultrahigh adsorption capacity for highly selective separation of Artemisinin/Artemether. Chem. Eng. J. 2023, 466, 143020. [Google Scholar] [CrossRef]
- Tse Sum Bui, B.; Haupt, K. Molecularly imprinted polymers: Synthetic receptors in bioanalysis. Anal. Bioanal. Chem. 2010, 398, 2481–2492. [Google Scholar] [CrossRef]
- Piletsky, S.A.; Matuschewski, H. Surface functionalization of porous polypropylene membranes with molecularly imprinted polymers by photograft copolymerization in water. Macromolecules 2000, 33, 3092–3098. [Google Scholar] [CrossRef]
- Wu, Y.; Lu, J. Double-layer-based molecularly imprinted membranes for template-dependent recognition and separation: An imitated core-shell-based synergistic integration design. Chem. Eng. J. 2020, 397, 125371. [Google Scholar] [CrossRef]
- Piletsky, S.A.; Panasyuk, T.L. Receptor and transport properties of imprinted polymer membranes—A review. J. Membr. Sci. 1999, 157, 263–278. [Google Scholar] [CrossRef]
- Zhu, X.; Su, Q. Molecularly imprinted polymer membranes for substance-selective solid-phase extraction from aqueous solutions. J. Appl. Polym. Sci. 2006, 101, 4468–4473. [Google Scholar] [CrossRef]
- Lehmann, M.; Brunner, H. Selective separations and hydrodynamic studies: A new approach using molecularly imprinted nanosphere composite membranes. Desalination 2002, 149, 315–321. [Google Scholar] [CrossRef]
- Cowell Iii, E.W.; Alimardani, N. Advancing mim electronics: Amorphous metal electrodes. Adv. Mater. 2011, 23, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Ramamoorthy, M.; Ulbricht, M. Molecular imprinting of cellulose acetate-sulfonated polysulfone blend membranes for Rhodamine B by phase inversion technique. J. Membr. Sci. 2003, 217, 207–214. [Google Scholar] [CrossRef]
- Cheong, W.J.; Ali, F. Recent applications of molecular imprinted polymers for enantio-selective recognition. Talanta 2013, 106, 45–59. [Google Scholar] [CrossRef]
- Kochkodan, V.; Weigel, W. Molecularly imprinted composite membranes for selective binding of desmetryn from aqueous solutions. Desalination 2002, 149, 323–328. [Google Scholar] [CrossRef]
- Lu, Z.; Li, B. NMP-induced surface self-corrosion-assisted rapid spin-coating method for synthesizing imprinted heterojunction photocatalyst anchored membrane towards high-efficiency selective degradation tetracycline. Sep. Purif. Technol. 2023, 314, 123609. [Google Scholar] [CrossRef]
- Wang, H.Y.; Kobayashi, T. Surface molecular imprinting on photosensitive dithiocarbamoyl polyacrylonitrile membranes using photograft polymerization. J. Chem. Technol. Biotechnol. 1997, 70, 355–362. [Google Scholar] [CrossRef]
- Dong, C.; Shi, H. Molecularly imprinted polymers by the surface imprinting technique. Eur. Polym. J. 2021, 145, 110231. [Google Scholar] [CrossRef]
- Hensel, R.; Finn, A. Biologically inspired omniphobic surfaces by reverse imprint lithography. Adv. Mater. 2014, 26, 2029–2033. [Google Scholar] [CrossRef]
- Barahona, F.; Díaz-Álvarez, M. Molecularly imprinted polymer-coated hollow fiber membrane for the microextraction of triazines directly from environmental waters. J. Chromatogr. A 2016, 1442, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Meng, M.; Feng, Y. Optimization of surface imprinted layer attached poly(vinylidene fluoride) membrane for selective separation of salicylic acid from acetylsalicylic acid using central composite design. Chem. Eng. J. 2013, 231, 132–145. [Google Scholar] [CrossRef]
- Bures, P.; Huang, Y. Surface modifications and molecular imprinting of polymers in medical and pharmaceutical applications. J. Control. Release 2001, 72, 25–33. [Google Scholar] [CrossRef]
- Hilal, N.; Kochkodan, V. Surface modified microfiltration membranes with molecularly recognising properties. J. Membr. Sci. 2003, 213, 97–113. [Google Scholar] [CrossRef]
- Lee, B.; Park, Y.-H. Ultralow-k nanoporous organosilicate dielectric films imprinted with dendritic spheres. Nat. Mater. 2005, 4, 147–150. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Fornasiero, F. Fabrication of flexible, aligned carbon nanotube/polymer composite membranes by in-situ polymerization. J. Membr. Sci. 2014, 460, 91–98. [Google Scholar] [CrossRef]
- Shen, H.; Wang, S. Preparation of polyamide thin film nanocomposite membranes containing silica nanoparticles via an in-situ polymerization of SiCl4 in organic solution. J. Membr. Sci. 2018, 565, 145–156. [Google Scholar] [CrossRef]
- Nasreen, S.A.A.N.; Sundarrajan, S. In situ polymerization of PVDF-HEMA polymers: Electrospun membranes with improved flux and antifouling properties for water filtration. Polym. J. 2014, 46, 167–174. [Google Scholar] [CrossRef]
- Sergeyeva, T.A.; Piletsky, S.A. In Situ Formation of Porous Molecularly Imprinted Polymer Membranes. Macromolecules 2003, 36, 7352–7357. [Google Scholar] [CrossRef]
- Xu, T.; Woo, J.-J. In situ polymerization: A novel route for thermally stable proton-conductive membranes. J. Membr. Sci. 2008, 325, 209–216. [Google Scholar] [CrossRef]
- Tao, M.; Liu, F. Hydrophilic poly(vinylidene fluoride) (PVDF) membrane by in situ polymerisation of 2-hydroxyethyl methacrylate (HEMA) and micro-phase separation. J. Mater. Chem. 2012, 22, 9131–9137. [Google Scholar] [CrossRef]
- Huang, Z.; Zhang, P. Preparing molecularly imprinted membranes by phase inversion to separate kaempferol. Polym. Adv. Technol. 2017, 28, 373–378. [Google Scholar] [CrossRef]
- Yu, X.; Mi, X. Fouling resistant CA/PVA/TiO2 imprinted membranes for selective recognition and separation salicylic acid from waste water. Front Chem. 2017, 5, 2. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, S.; Nematollahzadeh, A. Molecularly imprinted polymer membrane for the removal of naphthalene from petrochemical wastewater streams. Adv. Polym. Technol. 2018, 37, 2288–2293. [Google Scholar] [CrossRef]
- Zeng, J.; Lv, C. A novel ion-imprinted membrane induced by amphiphilic block copolymer for selective separation of Pt(IV) from aqueous solutions. J. Membr. Sci. 2019, 572, 428–441. [Google Scholar] [CrossRef]
- Algieri, C.; Parisi, O.I. Development of novel hybrid imprinted membranes for selective recovery of theophylline. Sep. Purif. Technol. 2018, 192, 513–519. [Google Scholar] [CrossRef]
- Cui, J.; Zhou, Z. Facile synthesis of degradable CA/CS imprinted membrane by hydrolysis polymerization for effective separation and recovery of Li+. Carbohydr. Polym. 2019, 205, 492–499. [Google Scholar] [CrossRef]
- Chen, R.-R.; Qin, L. Novel surface-modified molecularly imprinted membrane prepared with iniferter for permselective separation of lysozyme. J. Membr. Sci. 2010, 363, 212–220. [Google Scholar] [CrossRef]
- Ma, X.; Chen, R. Preparation of molecularly imprinted CS membrane for recognizing naringin in aqueous media. Polym. Bull. 2011, 66, 853–863. [Google Scholar] [CrossRef]
- Li, H.; Huang, Q. Generation of a molecular imprinted membrane by coating cellulose acetate onto a zro2-modified alumina membrane for the chiral separation of mandelic acid enantiomers. Org. Process Res. Dev. 2018, 22, 278–285. [Google Scholar] [CrossRef]
- Fujikawa, S.; Muto, E. Nanochannel design by molecular imprinting on a free-standing ultrathin titania membrane. Langmuir 2009, 25, 11563–11568. [Google Scholar] [CrossRef] [PubMed]
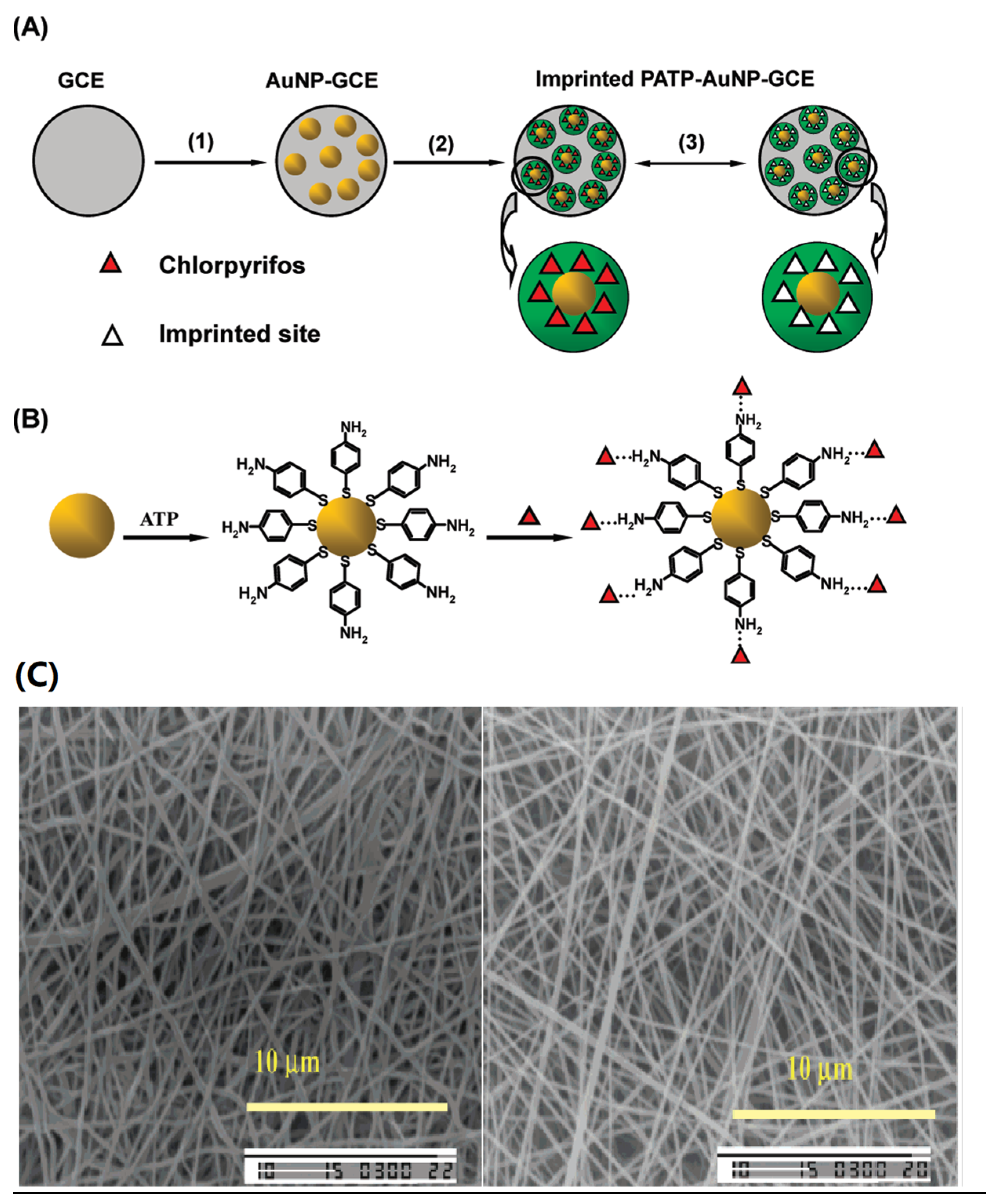
- Xie, C.; Li, H. Surface molecular self-assembly for organophosphate pesticide imprinting in electropolymerized poly(p-aminothiophenol) membranes on a gold nanoparticle modified glassy carbon electrode. Anal. Chem. 2010, 82, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Zhang, J. Electrochemical sensor based on molecularly imprinted membranes at platinum nanoparticles-modified electrode for determination of 17beta-estradiol. Biosens Bioelectron. 2011, 29, 29–33. [Google Scholar] [CrossRef]
- Queiros, R.B.; Silva, S.O. Microcystin-LR detection in water by the Fabry-Perot interferometer using an optical fibre coated with a sol-gel imprinted sensing membrane. Biosens. Bioelectron. 2011, 26, 3932–3937. [Google Scholar] [CrossRef] [PubMed]
- Dunn, B.; Zink, J.I. Optical properties of sol–gel glasses doped with organic molecules. J. Mater. Chem. 1991, 1, 903–913. [Google Scholar] [CrossRef]
- Meng, M.; Liu, Y. Introduction of an ordered porous polymer network into a ceramic alumina membrane via non-hydrolytic sol–gel methodology for targeted dynamic separation. RSC Adv. 2014, 4, 38630–38642. [Google Scholar] [CrossRef]
- Marnadu, R.; Chandrasekaran, J. Influence of metal work function and incorporation of Sr atom on WO3 thin films for MIS and MIM structured SBDs. Superlattices Microstruct. 2018, 119, 134–149. [Google Scholar] [CrossRef]
- Zhang, H.Q.; Jin, Y. The optical and electrical characteristics of PMMA film prepared by spin coating method. IOP Conf. Ser. Mater. Sci. Eng. 2015, 87, 012032. [Google Scholar] [CrossRef]
- Giwa, A.; Dindi, A. Membrane bioreactors and electrochemical processes for treatment of wastewaters containing heavy metal ions, organics, micropollutants and dyes: Recent developments. J. Hazard. Mater. 2019, 370, 172–195. [Google Scholar] [CrossRef]
- Blanco-Lopez, M.C.; Lobo-Castanon, M.J. Voltammetric sensor for vanillylmandelic acid based on molecularly imprinted polymer-modified electrodes. Biosens. Bioelectron. 2003, 18, 353–362. [Google Scholar] [CrossRef]
- Chronakis, I.S.; Milosevic, B. Generation of Molecular Recognition Sites in Electrospun Polymer Nanofibers via Molecular Imprinting. Macromolecules 2006, 39, 357–361. [Google Scholar] [CrossRef]
- Yoshikawa, M.; Tharpa, K. Molecularly imprinted membranes: Past, present, and future. Chem. Rev. 2016, 116, 11500–11528. [Google Scholar] [CrossRef] [PubMed]
- Xie, R.; Chu, L.Y. Membranes and membrane processes for chiral resolution. Chem. Soc. Rev. 2008, 37, 1243–1263. [Google Scholar] [CrossRef] [PubMed]
- Sueyoshi, Y.; Fukushima, C. Molecularly imprinted nanofiber membranes from cellulose acetate aimed for chiral separation. J. Membr. Sci. 2010, 357, 90–97. [Google Scholar] [CrossRef]
- Zhang, C.; Liu, F. Influence of stereoregularity and linkage groups on chiral recognition of poly(phenylacetylene) derivatives bearing L-leucine ethyl ester pendants as chiral stationary phases for HPLC. J. Polym. Sci. Part A Polym. Chem. 2013, 51, 2271–2278. [Google Scholar] [CrossRef]
- Zhang, F.; He, L. Chiral liquid membrane for enantioselective separation of racemic ibuprofen by l-tartaric acid derivatives. RSC Adv. 2015, 5, 41729–41735. [Google Scholar] [CrossRef]
- Zhao, X.; Jin, J. Structural and optical properties of zirconia thin films deposited by reactive high-power impulse magnetron sputtering. Thin Solid Films 2014, 570, 404–411. [Google Scholar] [CrossRef]
- Zhao, Y.; Song, J. Chiral separation of six tetralone derivative enantiomers using immobilized cellulose chiral stationary phase. Se Pu 2014, 32, 832–836. [Google Scholar] [CrossRef]
- Huang, Q.; Li, H. Chiral separation of (d,l)-lactic acid through molecularly imprinted cellulose acetate composite membrane. Cellulose 2018, 25, 3435–3448. [Google Scholar] [CrossRef]
- Gao, J.; Chen, L. “Nanomagnet-inspired” design on molecularly imprinted nanofiber membrane: Mechanisms for improved transport selectivity of sufficient specific sites. J. Membr. Sci. 2023, 672, 121467. [Google Scholar] [CrossRef]
- Ulbricht, M. Membrane separations using molecularly imprinted polymers. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2004, 804, 113–125. [Google Scholar] [CrossRef]
- Bai, M.; Qiang, L. Upper surface imprinted membrane prepared by magnetic guidance phase inversion method for highly efficient and selective separation of Artemisinin. Chem. Eng. J. 2021, 405, 126899. [Google Scholar] [CrossRef]
- Xing, W.; Yan, Y. MOFs self-assembled molecularly imprinted membranes with photoinduced regeneration ability for long-lasting selective separation. Chem. Eng. J. 2022, 437, 135128. [Google Scholar] [CrossRef]
- Yang, M.; Chu, L.-Y. A Thermoresponsive Membrane for Chiral Resolution. Adv. Funct. Mater. 2008, 18, 652–663. [Google Scholar] [CrossRef]
- Kashani, T.; Jahanshahi, M. Nanopore molecularly imprinted polymer membranes for environmental usage: Selective separation of 2,4-dichlorophenoxyacetic acid as a toxic herbicide from water. Polym. Technol. Eng. 2016, 55, 1700–1712. [Google Scholar] [CrossRef]
- Lu, J.; Qin, Y. Antibacterial, high-flux and 3D porous molecularly imprinted nanocomposite sponge membranes for cross-flow filtration of emodin from analogues. Chem. Eng. J. 2019, 360, 483–493. [Google Scholar] [CrossRef]
- He, Z.; Meng, M. Fabrication of new cellulose acetate blend imprinted membrane assisted with ionic liquid ([BMIM]Cl) for selective adsorption of salicylic acid from industrial wastewater. Sep. Purif. Technol. 2015, 145, 63–74. [Google Scholar] [CrossRef]
- Lu, J.; Qin, Y. Irregular dot array nanocomposite molecularly imprinted membranes with enhanced antibacterial property: Synergistic promotion of selectivity, rebinding capacity and flux. Chem. Eng. J. 2021, 405, 126716. [Google Scholar] [CrossRef]
- Kadhem, A.J.; Xiang, S. Photonic molecularly imprinted polymer film for the detection of testosterone in aqueous samples of the article. J. Artic. 2018, 10, 349. [Google Scholar] [CrossRef]
- Dong, Z.; Lu, J. Antifouling molecularly imprinted membranes for pretreatment of milk samples: Selective separation and detection of lincomycin. Food Chem. 2020, 333, 127477. [Google Scholar] [CrossRef]
- Rana, D.; Matsuura, T. Surface Modifications for Antifouling Membranes. Chem. Rev. 2010, 110, 2448–2471. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Maruf, S. Surface patterning of polymeric membranes and its effect on antifouling characteristics. Sep. Sci. Technol. 2016, 52, 240–257. [Google Scholar] [CrossRef]
- Gao, J.; Zhou, S. One pot-economical fabrication of molecularly imprinted membrane employing carbon nanospheres sol coagulation bath with specific separation and advanced antifouling performances. Sep. Purif. Technol. 2019, 218, 59–69. [Google Scholar] [CrossRef]
- Cui, J.; Zhang, Y. Fabrication of lithium ion imprinted hybrid membranes with antifouling performance for selective recovery of lithium. New J. Chem. 2018, 42, 118–128. [Google Scholar] [CrossRef]
- Sun, D.; Meng, M. Synthesis of ion imprinted nanocomposite membranes for selective adsorption of lithium. Sep. Purif. Technol. 2018, 194, 64–72. [Google Scholar] [CrossRef]
- Cui, Y.; Jiang, L. Molecularly imprinted electrospun nanofibre membrane assisted stir bar sorptive extraction for trace analysis of sulfonamides from animal feeds. J. Ind. Eng. Chem. 2021, 96, 382–389. [Google Scholar] [CrossRef]
- Wu, Y.T.; Zhang, Y.H. Selective and simultaneous determination of trace bisphenol A and tebuconazole in vegetable and juice samples by membrane-based molecularly imprinted solid-phase extraction and HPLC. Food Chem. 2014, 164, 527–535. [Google Scholar] [CrossRef]
- Ayyavoo, J.; Nguyen, T.P.N. Protection of polymeric membranes with antifouling surfacing via surface modifications. Colloids Surf. 2016, 506, 190–201. [Google Scholar] [CrossRef]
- Yoshikawa, M.; Izumi, J.-i. Alternative molecular imprinting, a facile way to introduce chiral recognition sites. React. Funct. Polym. 1999, 42, 93–102. [Google Scholar] [CrossRef]
- Sueyoshi, Y.; Utsunomiya, A. Chiral separation with molecularly imprinted polysulfone-aldehyde derivatized nanofiber membranes☆. J. Membr. Sci. 2012, 401–402, 89–96. [Google Scholar] [CrossRef]
- Yan, M.; Wu, Y. Multilevel/hierarchical nanocomposite-imprinted regenerated cellulose membranes for high-efficiency separation: A selective recognition method with Au/PDA-loaded surface. Environ. Sci. Nano 2021, 8, 1978–1991. [Google Scholar] [CrossRef]
- Gao, T.; Guan, G. Electrospun molecularly imprinted sodium alginate/polyethylene oxide nanofibrous membranes for selective adsorption of methylene blue. Int. J. Biol. Macromol. 2022, 207, 62–71. [Google Scholar] [CrossRef] [PubMed]
- Tasselli, F.; Donato, L. Evaluation of molecularly imprinted membranes based on different acrylic copolymers. J. Membr. Sci. 2008, 320, 167–172. [Google Scholar] [CrossRef]
- Bai, M.; Li, Y. “Cobweb locking raindrops” inspired construction of bio-based 3D porous molecularly imprinted membrane with ultrahigh adsorption capacity and selectivity: Effectively avoiding imprinting sites embedment. Surf. Interfaces 2022, 34, 102348. [Google Scholar] [CrossRef]
- Pinar Gumus, Z.; Soylak, M. Metal organic frameworks as nanomaterials for analysis of toxic metals in food and environmental applications. TrAC Trends Anal. Chem. 2021, 143, 116417. [Google Scholar] [CrossRef]
- Habila, M.; Alhenaki, B. Metal organic frameworks enhanced dispersive solid phase microextraction of malathion before detection by UHPLC-MS/MS. J. Sep. Sci. 2020, 43, 3103–3109. [Google Scholar] [CrossRef]
- Bakhshizadeh, S.; Sabzehmeidani, M.M. Preparation and characterization of mixed matrix membranes based on PVDF blend and hydrophilic molecularly imprinted MIL-101 (Cr) as filler for efficient selective removal of dye. J. Environ. Chem. Eng. 2022, 10, 108864. [Google Scholar] [CrossRef]
- Ma, F.; Yan, M. Biomimetic dual-layer imprinted UiO-66-Based basswood membranes for ibuprofen recognition and separation. ACS Sustain. Chem. Eng. 2023, 11, 6373–6384. [Google Scholar] [CrossRef]
- Xing, W.; Ma, Z. Novel molecular organic framework composite molecularly imprinted nanofibrous membranes with a bioinspired viscid bead structure for selective recognition and separation of atrazine. ACS Appl. Mater. Interfaces 2021, 13, 28749–28763. [Google Scholar] [CrossRef]

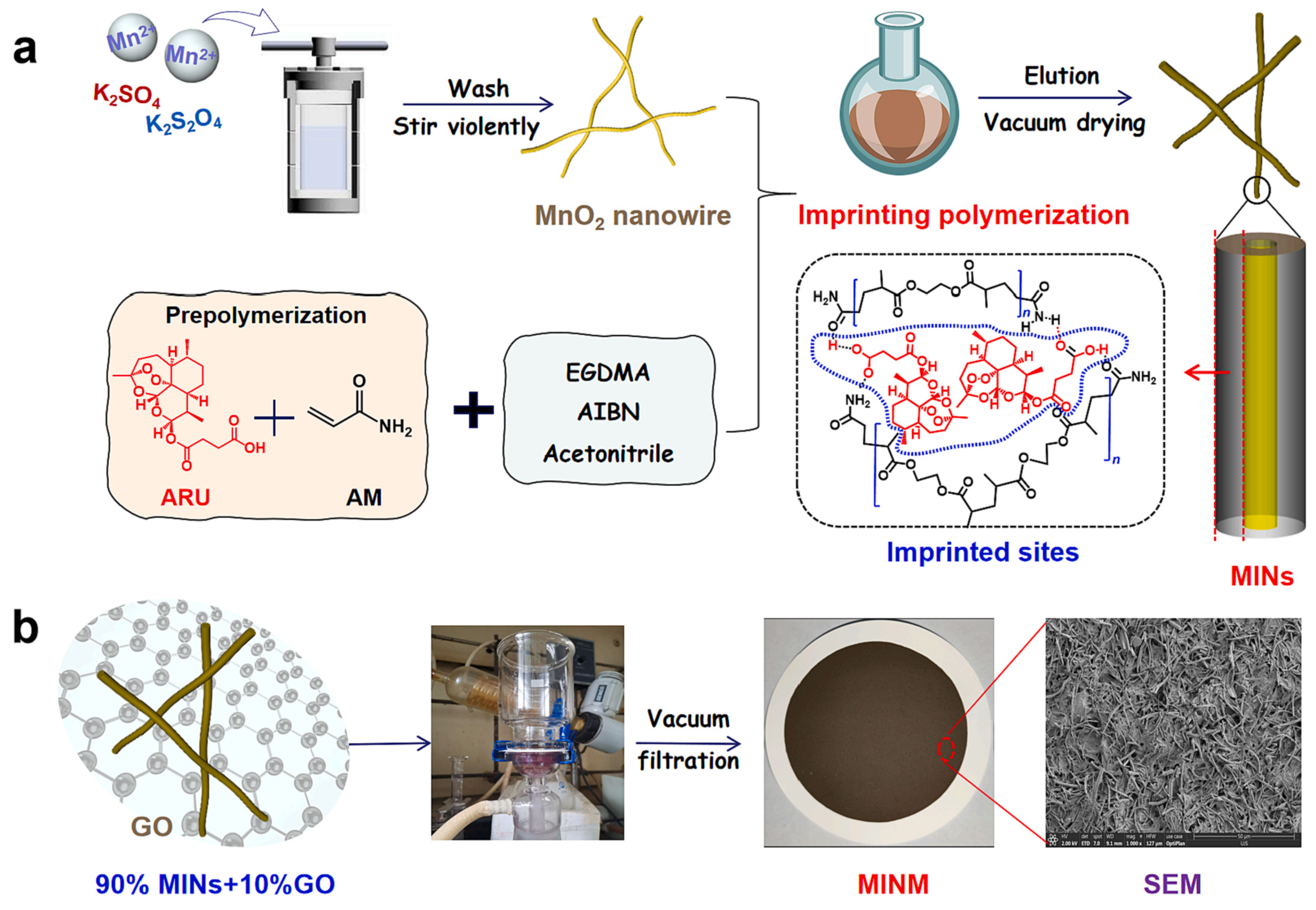
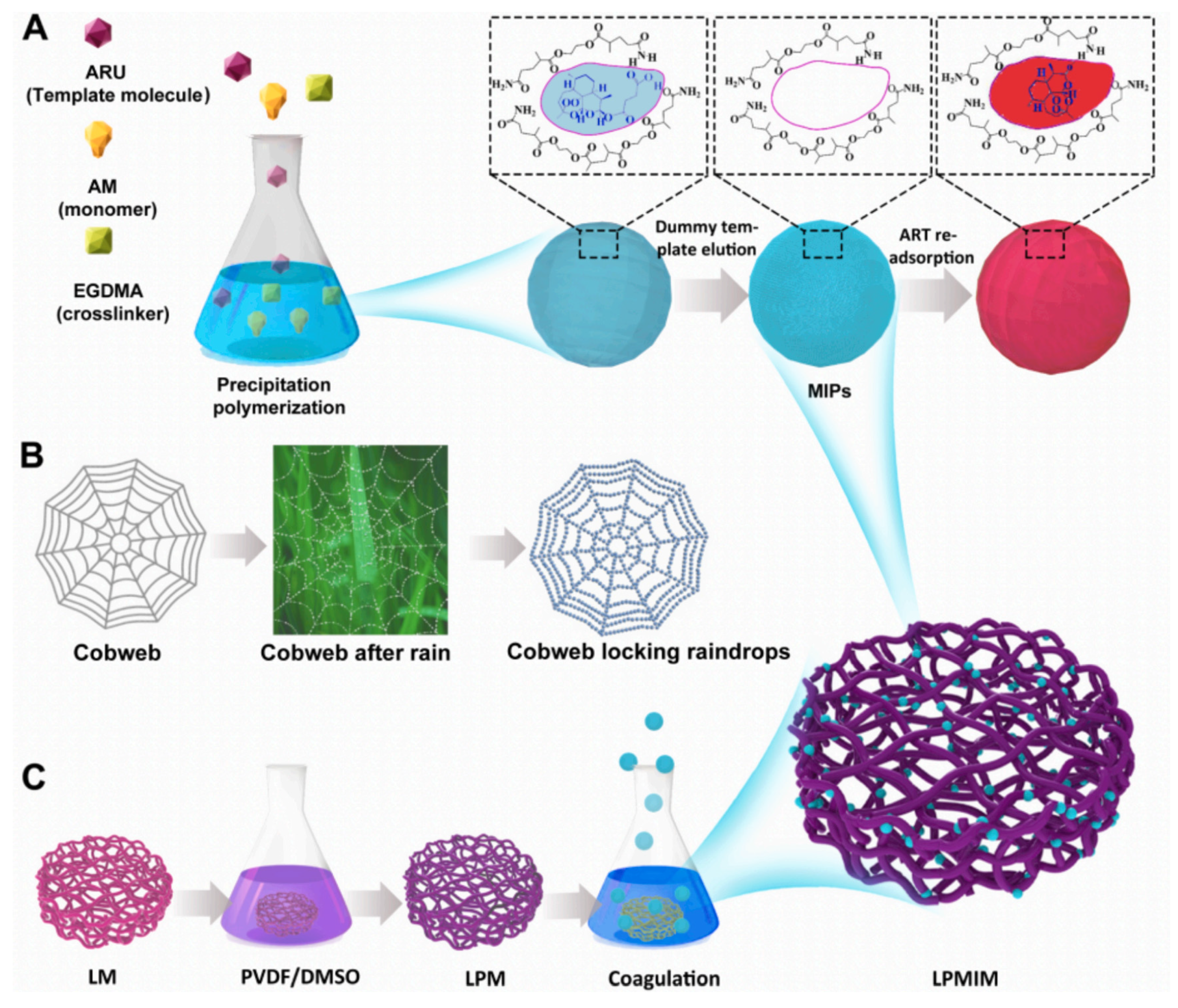
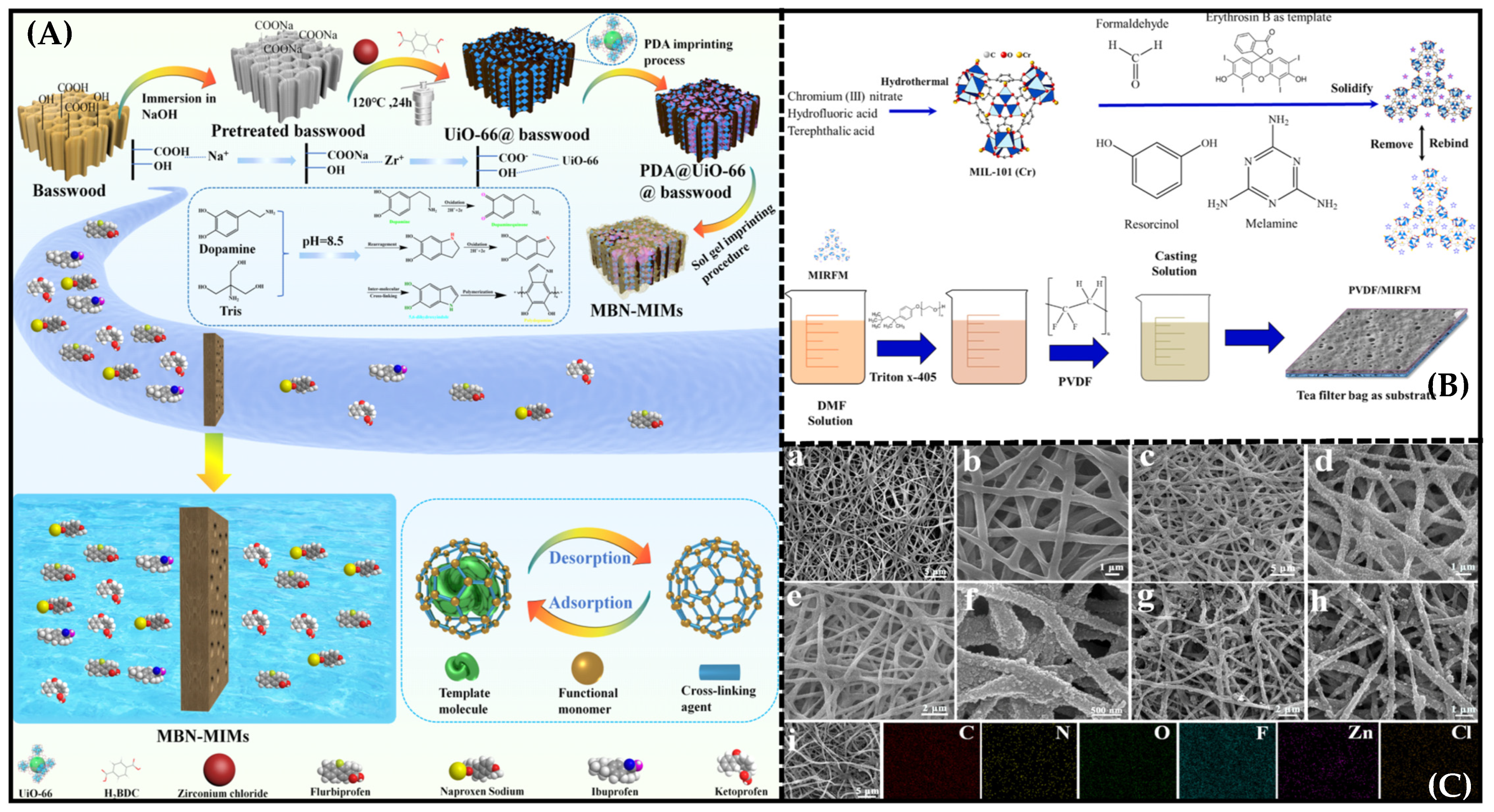
| Coagulation Temp (°C) | [Sb]T (µmol/g of Membrane) | [Sb]C (µmol/g of Membrane) | |
|---|---|---|---|
| 10 | 1.25 | 0.024 | 52 |
| 15 | 0.85 | 0.030 | 28 |
| 30 | 0.48 | 0.026 | 18 |
| 40 | 0.26 | 0.023 | 11 |
| Number | Membrane | Concentration (mmol/L) | Flux (mg/cm2 × min) | α | Ref. |
|---|---|---|---|---|---|
| 1 | Supported liquid membrane | 17.9 | 2.76 × 10−5 | 2 | [85] |
| 2 | Supported liquid membrane | 29.7 | 8.76 × 10−4 | 2.3 | [86] |
| 3 | Supported liquid membrane | 40 | 1.01 × 10−5 | 1.2 | [87] |
| 4 | Porous ceramic disc and hollow-fiber organic membrane | 44.74 | 6 × 10−5 | 15 | [87] |
| 5 | Supported liquid membrane | 44.74 | 6 × 10−7 | 2 | [88] |
| 6 | Molecularly imprinted cellulose membrane | 10 | 0.0198 | 7.3 | [89] |
| 7 | Molecularly imprinted nano-channel membrane | 10 | 0.070 | 8.7 | [89] |
| Membrane | Split Object | Elution Solvent | Regeneration Times | Ref. |
|---|---|---|---|---|
| Antibacterial, high-flux, and 3D porous molecularly imprinted nanocomposite-sponge membranes | Emodining from analogues | A mixture of methanol and acetic acid (95:5, v/v) | 10 | [96] |
| Highly selective cellulose acetate (CA) blend imprinted membranes for salicylic acid (SA) | Template SA | A methanol/acetic acid (9:1, v/v) mixed solvent | 5 | [97] |
| Irregular-dot -array nanocomposite bisphenol A (BPA)-molecularly-imprinted membranes | Bisphenol A | A mixture of methanol and HAc (95:5, v/v) | 10 | [98] |
| Molecularly imprinted polymer (MIP) photonic film | Testosterone | A methanol/acetic acid (9:1, v/v) mixed solvent | 6 | [99] |
| Solvent-driven controllable molecularly imprinted membranes | Bisphenol A | 100% MeOH | 8 | [93] |
| Lincomycin molecularly imprinted membrane | Lincomycin | A mixture ofmethanol and acetic acid (95:5, v/v) | 10 | [100] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, J.; Wei, M.; Meng, M. Advanced Development of Molecularly Imprinted Membranes for Selective Separation. Molecules 2023, 28, 5764. https://doi.org/10.3390/molecules28155764
Chen J, Wei M, Meng M. Advanced Development of Molecularly Imprinted Membranes for Selective Separation. Molecules. 2023; 28(15):5764. https://doi.org/10.3390/molecules28155764
Chicago/Turabian StyleChen, Jiahe, Maobin Wei, and Minjia Meng. 2023. "Advanced Development of Molecularly Imprinted Membranes for Selective Separation" Molecules 28, no. 15: 5764. https://doi.org/10.3390/molecules28155764
APA StyleChen, J., Wei, M., & Meng, M. (2023). Advanced Development of Molecularly Imprinted Membranes for Selective Separation. Molecules, 28(15), 5764. https://doi.org/10.3390/molecules28155764






