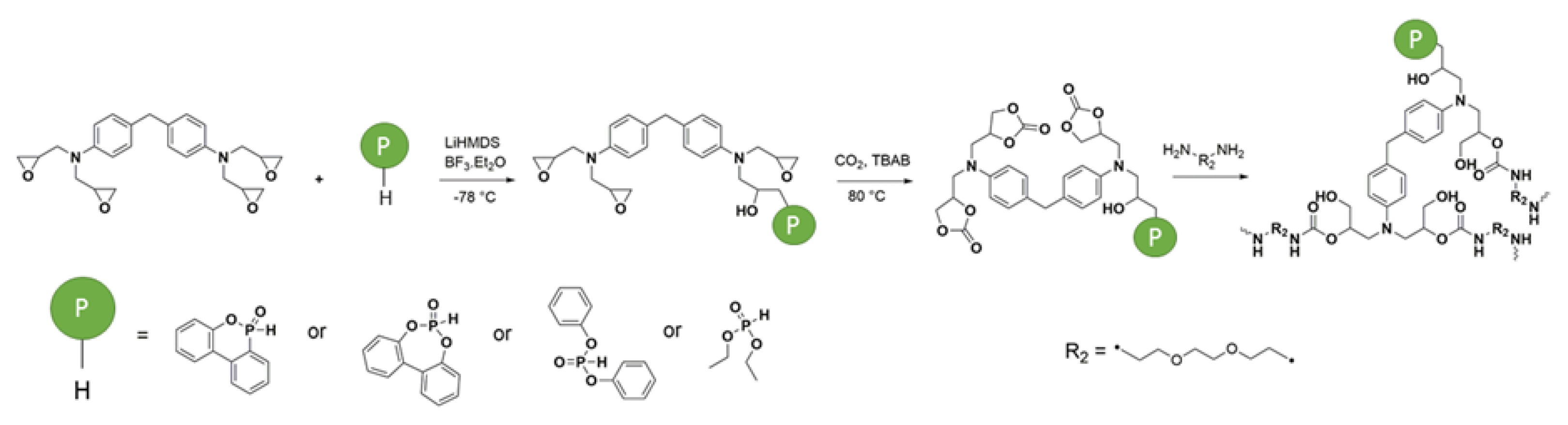
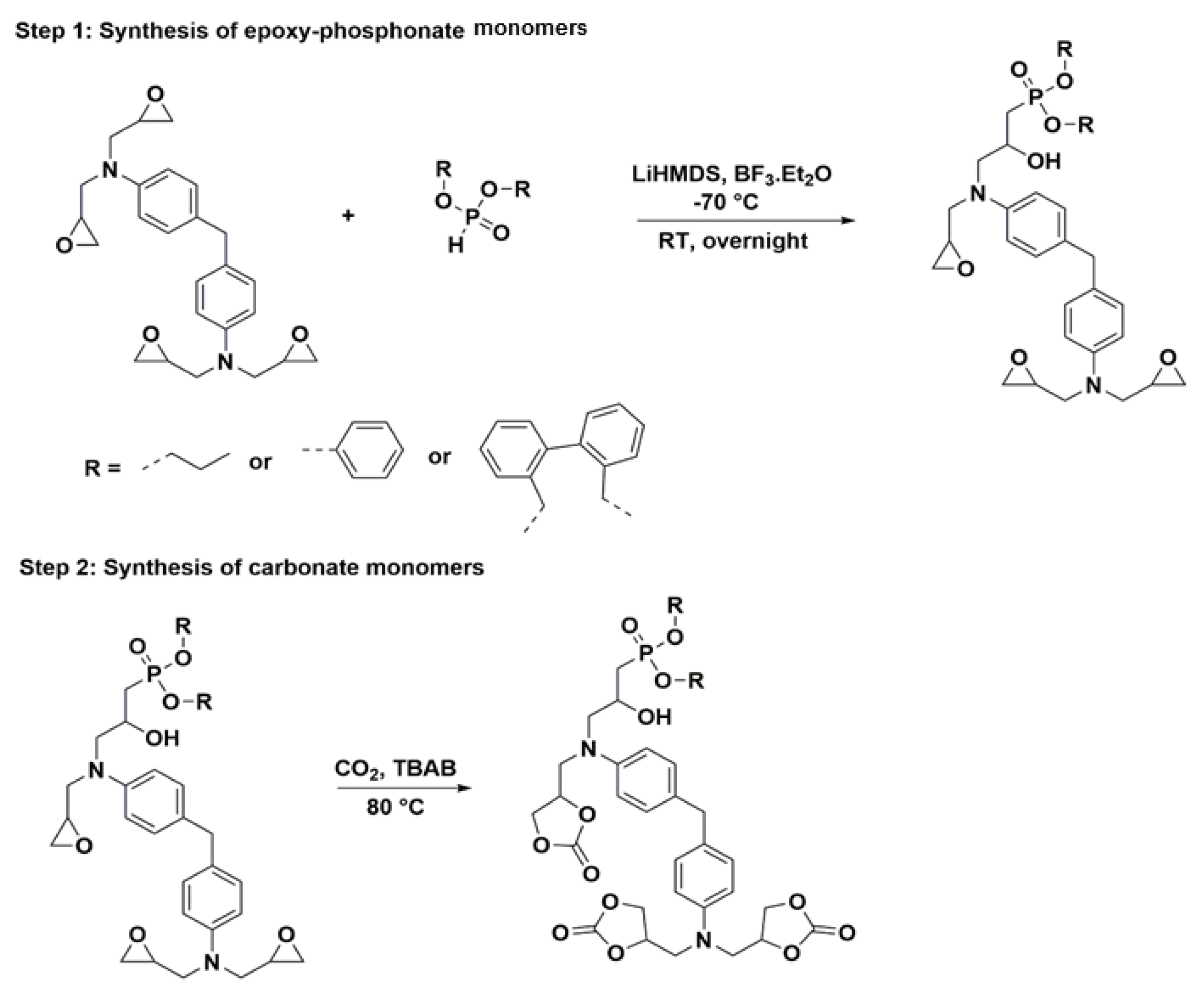
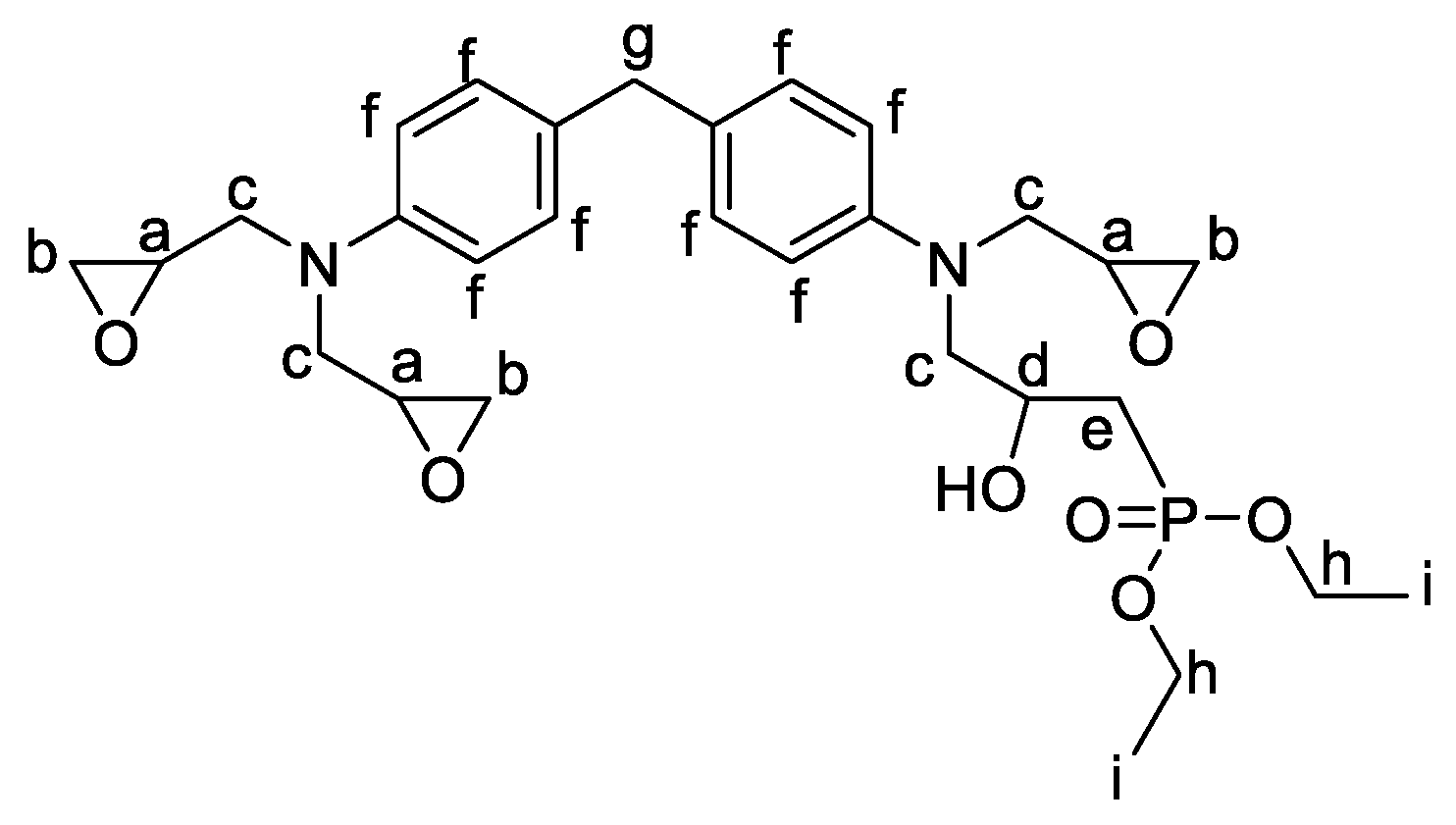
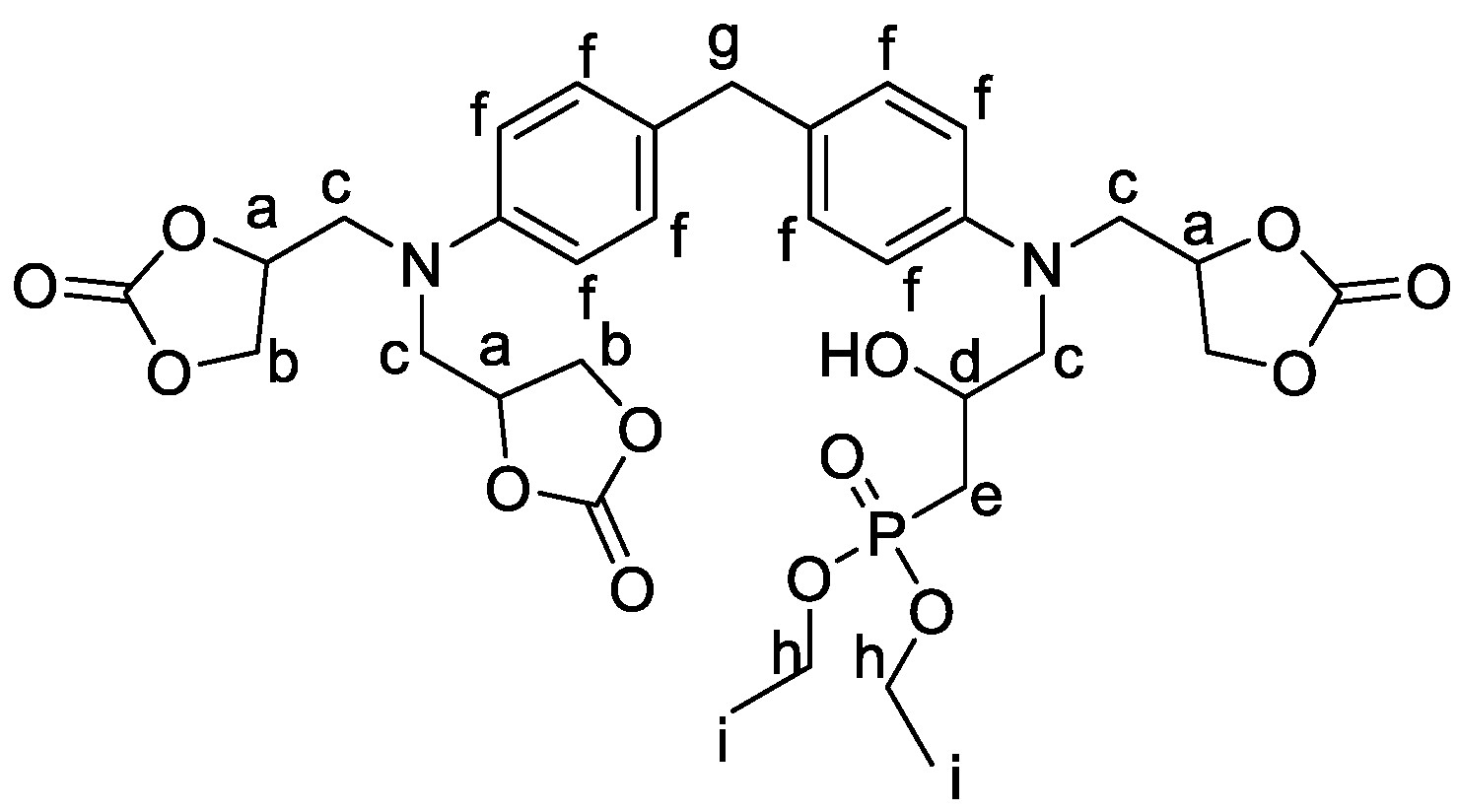
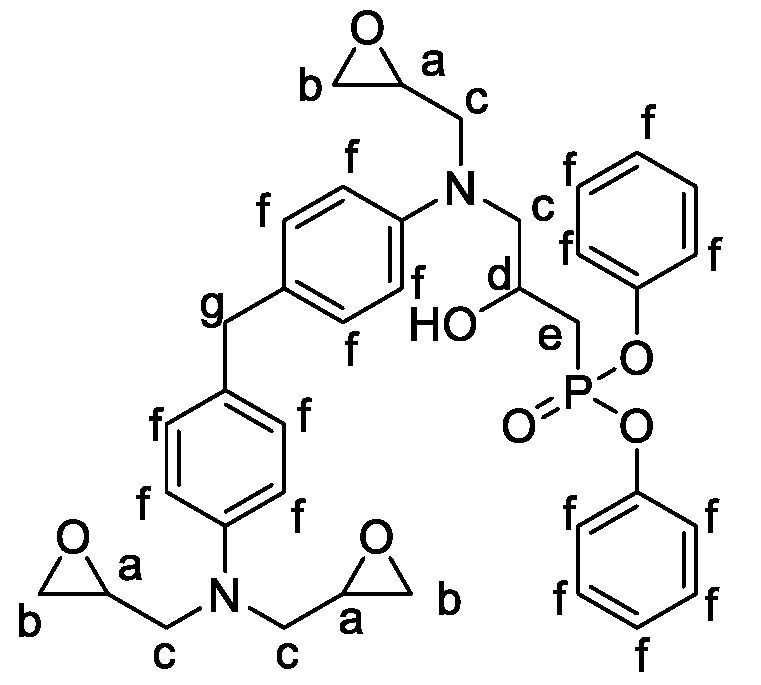
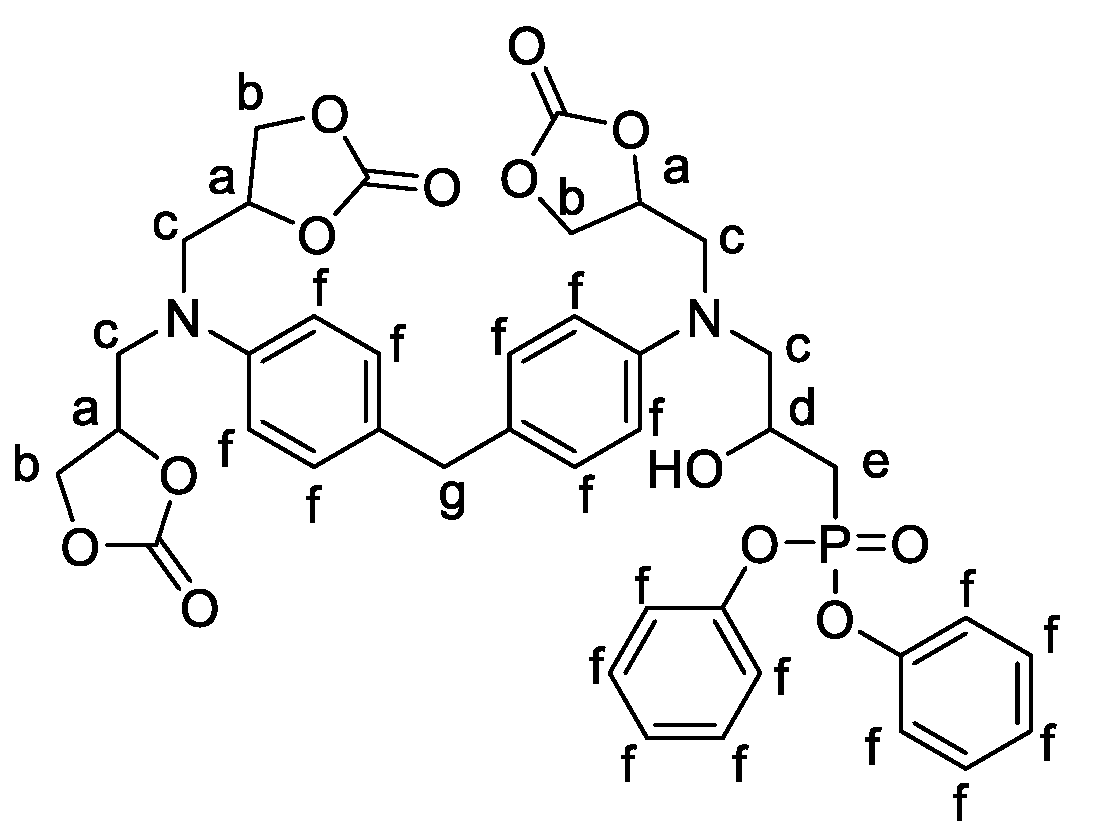
2.1.2. Formulations of Phosphorylated-PHU Materials
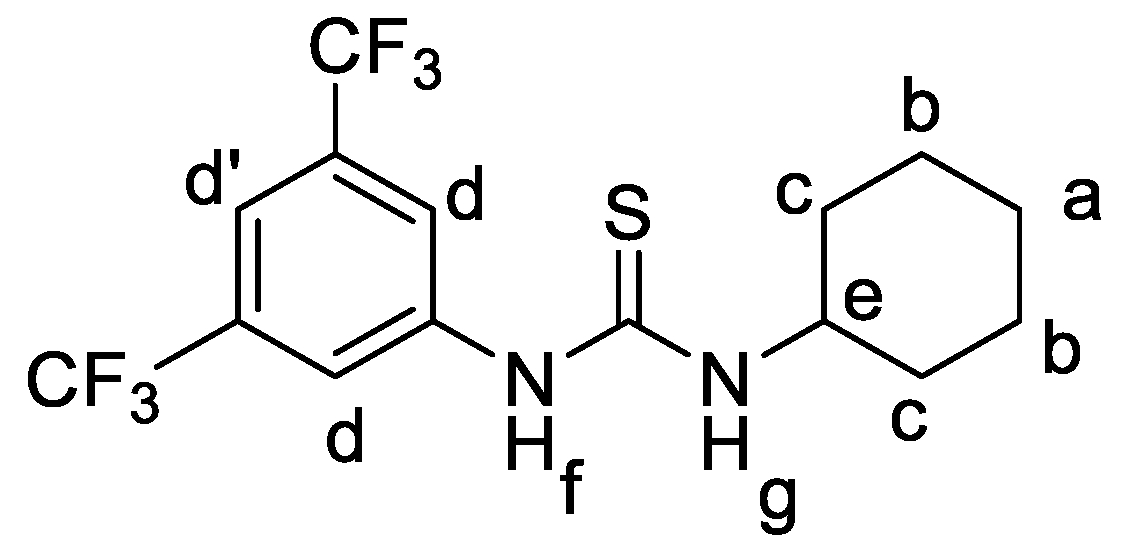
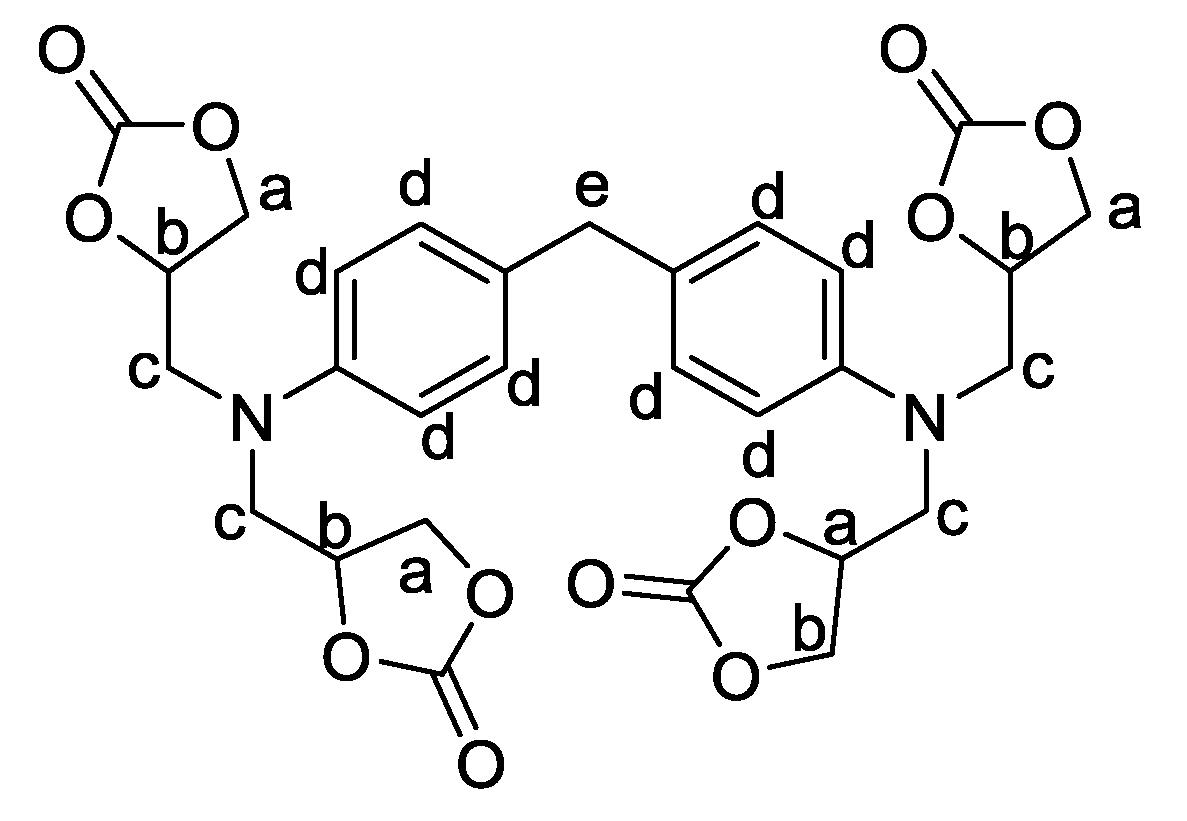
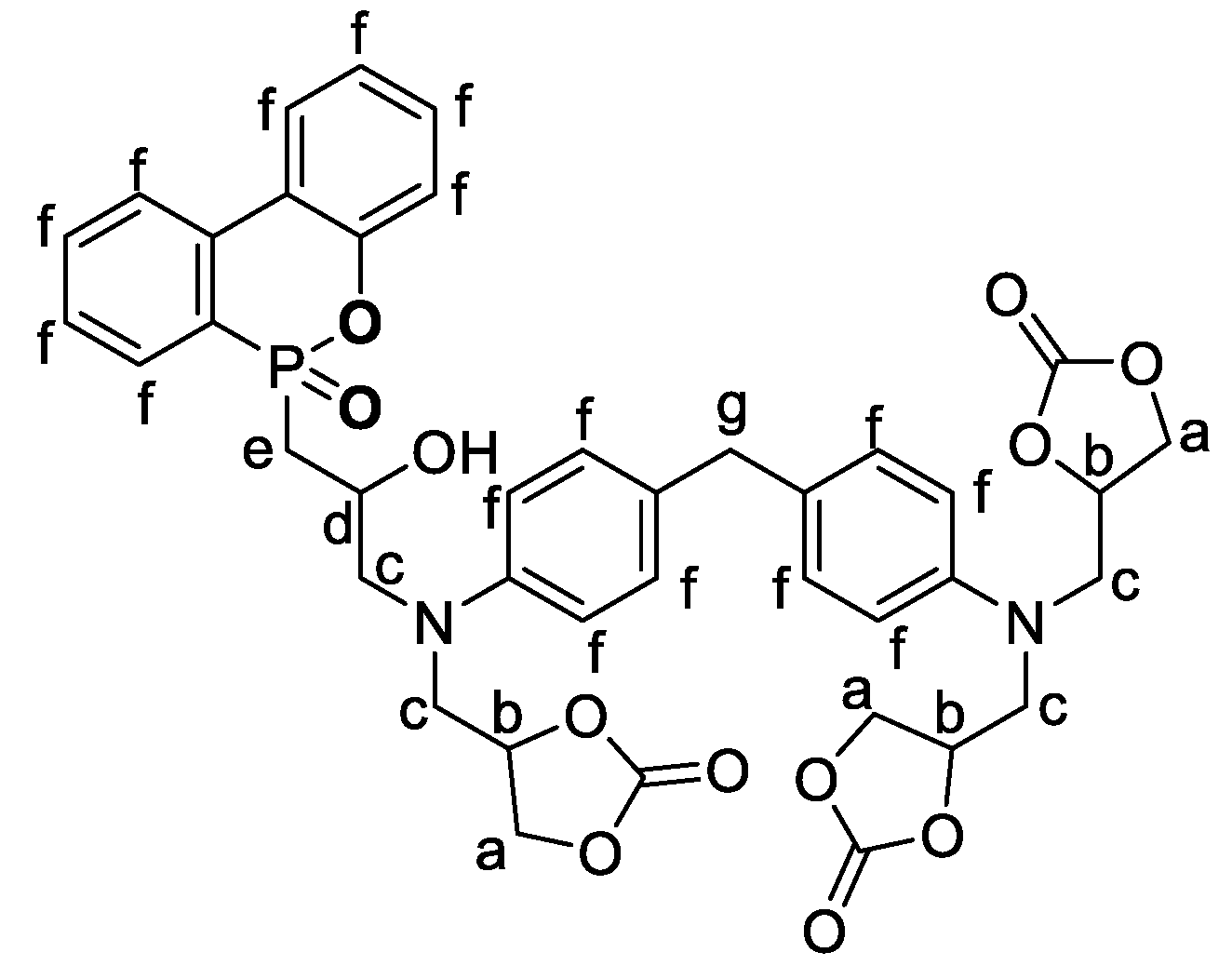
In this study, nine different PHU formulations have been studied. They were prepared at 80 °C by the reaction between the different phosphorylated carbonates and EDR 148. The catalyst has been chosen to further previous work in the literature [
46]. A 1:1 ratio amine:cyclic carbonate/phosphorylated cyclic carbonate is used in the formulations. Before any formulation, all the solids have been turned into fine powder. First, the carbonate and the phosphorylated carbonate have been mixed 1 min with PPO DC and the thiourea catalyst. Then, the amine was introduced and the formulation was mixed 30 additional seconds.
The crosslinking degree was determined by the measurement of the swelling rate and insoluble content. Finally, thermal properties were studied by DSC and TGA. To determine the influence of the structures of phosphorus-containing compounds on the material combustion behavior, cone calorimeter and PCFC were used.
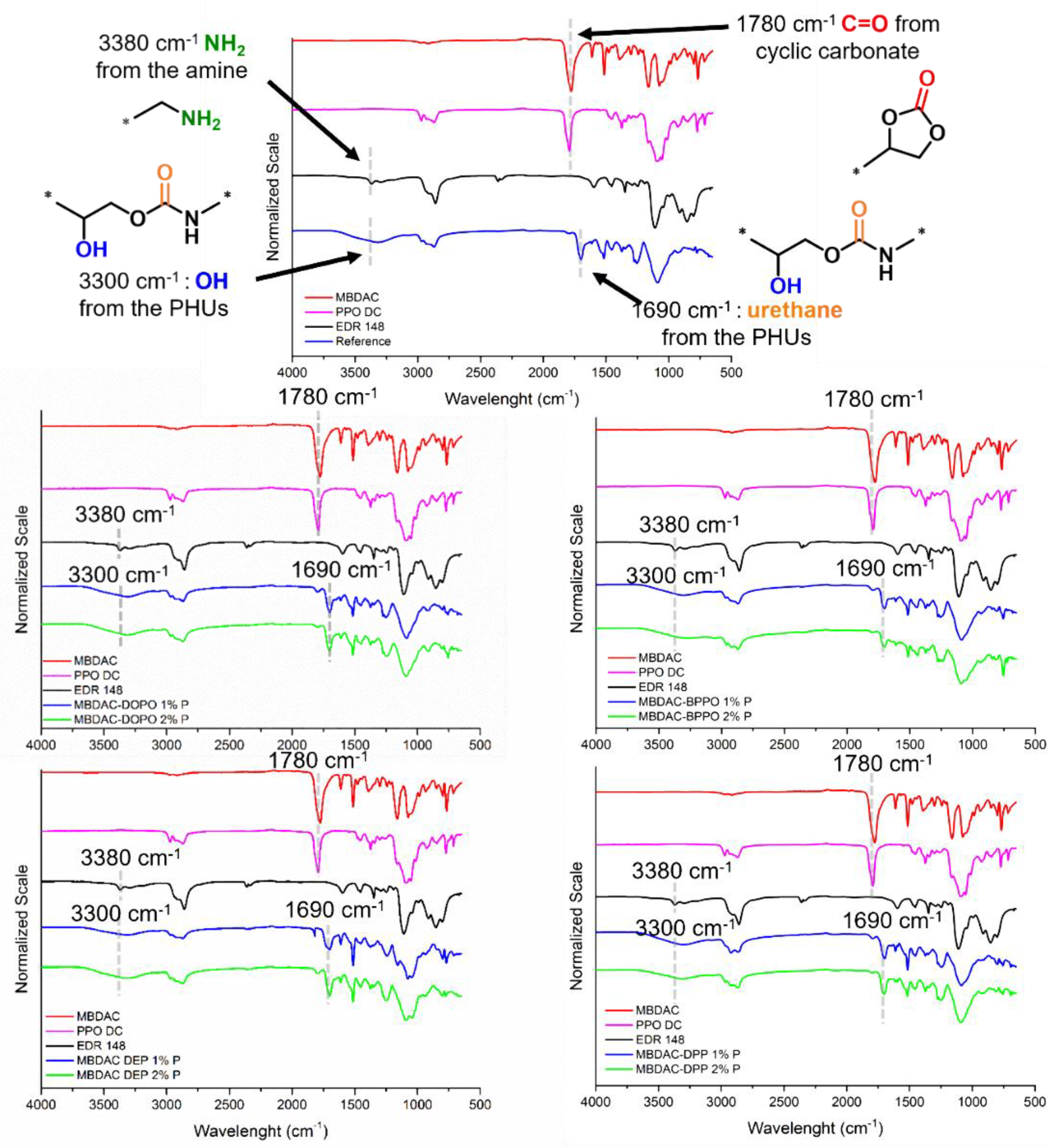
FTIR analyses were performed to first characterize the monomers. The cyclic carbonate stretching was identified at 1780 cm
−1. The amine stretching was observed around 3380 cm
−1. After completion of the reaction of the amine with the MBDAC and PPO DC, the signal assigned to the cyclic carbonate disappeared and that of the C=O from the urethane appeared. The hydroxyl group from the PHU is also observed around 3300 cm
−1.
Figure 1 shows the different FTIR spectra for all the materials and evidences the vanishing of the C=O in the cyclic carbonates on all the thermosets.
To confirm the formation of the polymer matrix, gel content analyses were carried out. The results showed high gel contents, up to 84 wt%, for most of the thermosets, except for MBDAC-BPPO 2 wt% P (
Table 1). Indeed, the lowest gel contents were obtained for MBDAC-BPPO 1 wt% and 2 wt% P. The steric hindrance induced by the large BPPO group may reduce the reactivity and thus may lower the gel content.
DSC analyses were carried out to highlight the glass transition temperatures (T
g) of the thermosets (
Table 1). The reference containing only MBDAC exhibited a glass transition temperature at around 2 °C. This value is the lowest T
g compared to the other formulations. Thus, phosphorus moiety seems to favor higher T
g. For MBDAC-DOPO, MBDAC-BPPO and MBDAC-DPP (
Figures S29, S30 and S32) it is explained by the aromatic ring contained in the FR molecule. The OH group of the opened epoxy could also explain the slight increase [
47,
48]. Indeed, OH groups promote H-bonding between polymer chains which can explain the T
g increase. The increase in phosphorus amount showed various effects on T
g. For example, with the MBDAC-DOPO, MBDAC-BPPO and MBDAC-DPP the T
g is lowered with 2 w% P. This can be due to a lower crosslinking density. However, using DEP, the T
g was improved. Interestingly the DPP exhibits two T
g meaning that the thermoset is heterogeneous. The second T
g inflexion is higher with 2 w% P meaning that it corresponds to the DPP FR.
The swelling rates were calculated and the values are presented in
Table 1. The reference had a swelling rate of 190%, which was the lowest value obtained. Indeed, the MBDAC sample had the highest concentration in crosslinking nodes since all the cyclic carbonates were available to react. Thus, the solvent could not penetrate as much as with a lower concentration of crosslinking nodes. This trend is observed with the other thermosets. In fact, when DOPO, BPPO, DPP or DEP were grafted onto the MBDA, one epoxy is converted. This enables us to get three cyclic carbonates available to react instead of 4. Thus, the resulting thermosets had a lower density of crosslinking nodes. That is why when 1 w% P is used, the swelling rate is higher than that of the reference. Hence, 2 w% P led to a higher swelling rate than 1% P for all the samples. The swelling rate for the DOPO thermosets is significantly higher than for the other FR-thermosets.
- 2.
Thermal and flame-retardancy analyses
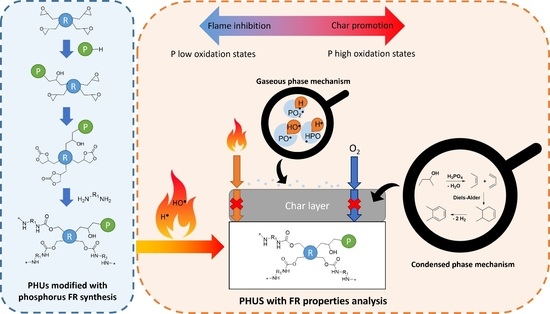
Thermal and flame-retardancy properties were investigated in this part to highlight the influence of phosphorus structures on the material formulations and their modes of action. Indeed, phosphinate-containing molecule (DOPO) was compared to phosphonate-containing molecules (BPPO, DEP and DPP). Moreover, different phosphonate compounds were used in order to determine the influence of the structure on the thermal and flame-retardant properties.
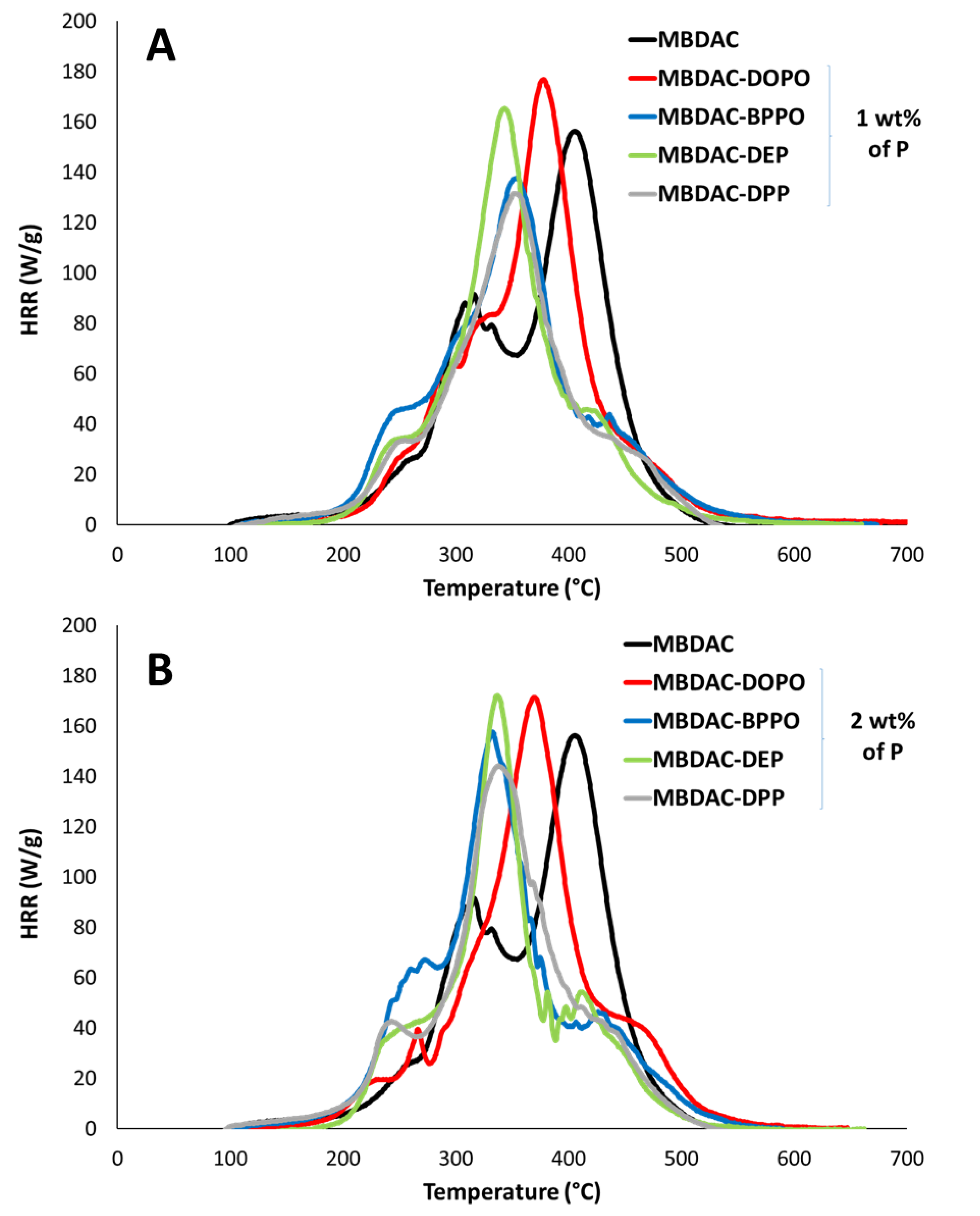
The thermogravimetric analyses (TGA) under nitrogen flow, allowed us to determine the decomposition behavior of the different materials.
Figure 2 shows the thermograms of the PHU materials with four different phosphorus-containing molecules (DOPO, BPPO, DEP, DPP) at various ratios of phosphorus (1 wt% and 2 wt%) compared to the free-phosphorus reference. Thermal parameters, such as the decomposition temperature at 5% weight loss (T
d,
5wt%), temperature at 50% weight loss (T
d,50wt%) and the residue yield at 750 °C, are summarized in
Table 2.
The T
d,5wt% values are reached around the same temperature, i.e., 200 °C for all the materials. Considering the temperature at 50 % weight loss (T
d,50wt%), all the materials exhibited equivalent thermal stability with temperatures ranging between 338 °C and 366 °C. For all the samples, the curves exhibited two main decomposition steps. The first one appeared between 200 and 400 °C and could be attributed to the decomposition of the polymers. For the MBDAC polymer, the second step was observed between 500 and 750 °C; whereas, it was shifted to lower temperatures (400–500 °C) for the polymers containing phosphorus-containing functions. Moreover, as presented in
Figure 2, the second step is more significant for the phosphonate than for the phosphinate compounds and it appeared at lower temperatures for phosphonate compounds. Indeed, the second step occurred between 370 and 400 °C for the PHU containing 2 wt% P phosphonate; whereas, it appeared around 440 °C for the polymer containing 2 wt% P phosphinate. The degradation of PHU containing 2 wt% P DPP seems more complex with a third decomposition step, occurring after 600 °C. Hence, the higher the phosphorus concentration, the slightly lower the temperature of the second step decomposition. The residue yield is improved with the introduction of phosphorus compounds. Moreover, it depends on both the phosphorus oxidation state and the structure of the compounds. Indeed, phosphonate compounds exhibited more residue content than phosphinate compounds. MBDAC-DOPO 2 wt% P exhibited a char yield of 12%, whereas MBDAC-BPPO, MBDAC-DPP and MBDAC-DEP displayed char contents of 22, 23 and 20%, respectively. Moreover, MBDAC-DPP and MBDAC-BPPO materials were composed of two more aromatic rings than MBDAC-DEP, which could explain a slightly higher residue content. These results were consistent with the mode of action of phosphonate compounds that act mainly in a condensed phase [
49,
50,
51]. Indeed, phosphonate compounds may decompose earlier and participate to the formation of a char layer through cyclization, cross-linking and aromatization by dehydration of the polymeric structure [
52,
53]. Then, this carbon layer could insulate the polymer and prevent heat and gas transfer between gas and condensed phases [
54,
55]. Thereby, the carbon layer could protect the remaining polymer from further degradation.
During decomposition, the phosphorus-containing compounds may generate free radicals such as PO
●, HPO
●, PO2
●. These radicals can quench the active radicals generated by the flame (H
● and OH
●) and then, reduce the combustion in the gaseous phase. This mode of action was formerly observed for phosphinate compounds such as DOPO, in the literature [
49,
56]. Nevertheless, such a mode of action cannot be evidenced using TGA.
The flammability of PHUs was investigated using PCFC. HRR curves are shown in
Figure 3 and the main data are listed in
Table 3.
All curves show that HRR starts to increase in the same range of temperatures (after 200 °C) in good agreement with Td,5wt% measured in TGA. The decomposition occurs in several steps for all the PHUs. The first shoulder at a low temperature is more significant for BPPO-based PHUs. This may be ascribed to the lower crosslinking rate of these resins and then a higher amount of unreacted monomers could be released.
The main peak is located at a higher temperature, for reference (403 °C), than for phosphorus-based PHUs. Moreover, the temperature at pHRR is also slightly higher for DOPO-based PHUs than for phosphonate-based PHUs (>360 °C versus 330–350 °C) which is also expected. The intensity is in the range 130–170 W.g−1 for all resins, including the reference one. It may be slightly lower for the DPP compound.
Residue content is slightly enhanced for phosphorus-based PHUs, and slightly higher for phosphonate compounds than for phosphinate ones. Nevertheless, these residues are still lower than those noted from TGA analyses. The reason is unclear and may be due to the dependence of decomposition on the heating rate. Nevertheless, an intumescent char is sometimes observed, especially for DEP-based PHUs.
The heat of complete combustion is also in the same range, i.e., around 20 (±2) kJ.g−1. It is noteworthy that flame inhibition (which is the main mode of action of DOPO, for example) cannot be observed in standard conditions in PCFC, because the combustion is forced to be complete.
While the heat of combustion is rather constant and residue does not increase strongly, THR is also in the same range (16–19 kJ.g−1) for all PHUs. DEP seems to provide the lowest values due to the combination of a higher residue content and lower heat of combustion.
Fire behavior at bench-scale was studied using q cone calorimeter (external heat flux 35 kW·m
−2).
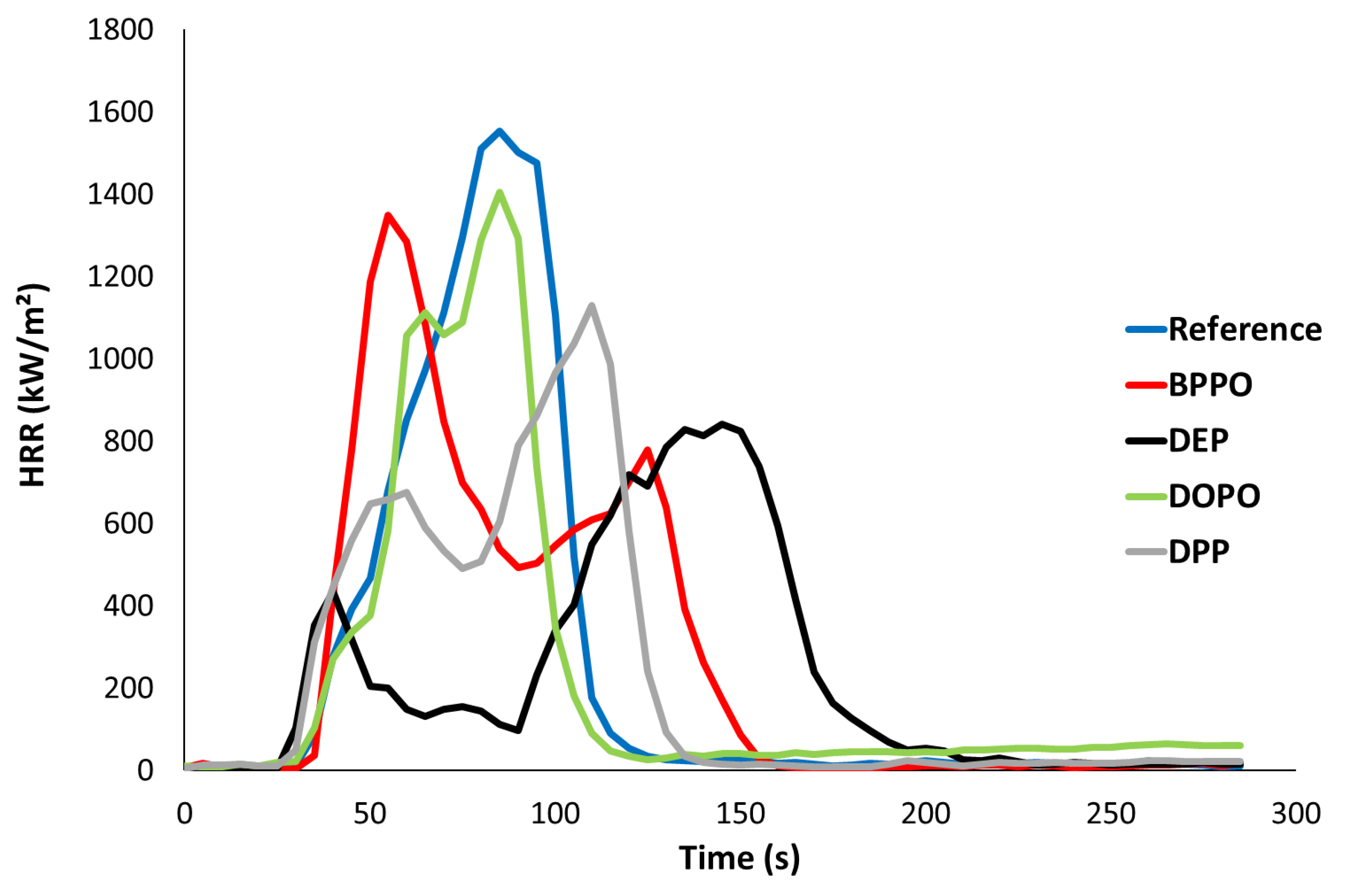
Figure 4 and
Figure 5 exhibit the HRR curves for resins containing 1 and 2 wt% of phosphorus, respectively. The main data are listed in
Table 4 and
Table 5. While residue contents are similar in PCFC and cone calorimeter, combustion efficiency X can be properly calculated as the ratio between effective heat of combustion in cone calorimeter (EHC) and heat of complete combustion in PCFC.
Reference ignites at 36 s and vigorous bubbling is observed at the sample surface. The heat release rate increases continuously up to a very high value (1644 kW·m−2) at 90 s. Then, HRR decreases quickly due to fuel depletion up to flame out. The residue fraction is negligible. This behavior is typical of a thermally thin behavior due to the small dimensions of the sample as well as the lack of a protective char layer formed during burning. Moreover, combustion is close to being complete (X~1).
DOPO-based PHUs exhibit similar behavior (vigorous bubbling and only one pHRR at around 90 s) even if the pHRR is slightly lower (1100–1300 kW·m−2). Flashes are observed before ignition, which occurs at the same time as the reference. Residue content is enhanced (around 10%) but it is not intumescent and charring seems to occur late. It is well known that DOPO is a poor char promoter and is especially efficient in fire tests as LOI or UL94. In these tests, ignition source is removed after few seconds and flame poisoning (the main mode of action of DOPO) may lead to fast flame extinction. On the contrary, under forced burning as in cone calorimeter, DOPO is quite often less efficient. Nevertheless, it should be noted that combustion efficiency is close to 1 even for DOPO-based PHUs. In other words, even flame poisoning does not occur for these DOPO-based PHUs.
Phosphonate-based PHUs exhibit a different behavior with two pHRRs. The first one is observed early, at 40–70 s, while the second one occurs at 120–165 s. Nevertheless, significant differences can be observed between these materials.
TTI is reduced for BPPO-based PHU (with 2 wt% of phosphorus). Vigorous bubbling is still observed for both materials (1 and 2 wt% P) and it explains why the first pHRR is intense. However, a char is formed with limited intumescence and reduces to some extent the HRR. Nevertheless, gases trapped under char are managed to be released leading to enhanced burning, corresponding to the pHRR2. As for DOPO compounds, residue content is close to 10% and combustion is complete.
On the contrary, DEP-based PHUs (and DPP-based PHUs to a lesser extent) show very different behavior. Ignition is reduced for 1 wt% P compounds (in comparison to the reference) but increases for 2 wt% P. No bubbling is observed and char is formed quickly after ignition leading to a low pHRR1. After pHRR1, the intumescent char efficiently traps the gases and the flame is strongly reduced and even vanishes in one case (one test for DEP-based PHU with 1 wt% P—see video in
Supplementary Materials). Nevertheless, after several dozen seconds, gases are released at the bottom of the char and burning resumes forcefully leading to the pHRR2. Intumescence is especially significant for DEP-based PHUs and occurs in two steps corresponding roughly to both pHRRs. Residue fraction remains moderate (slightly higher with DEP compounds) and combustion is close to being complete in all cases (X > 0.9). It is noteworthy that the residue content is similar for all phosphorus-based PHUs. In other words, the cohesion of the char layer (for DEP, char’s interior is finely porous), as well as the time of its formation (early for DEP compounds, later for DOPO or BPPO ones), explains why DEP allows reducing more significantly HRR than other phosphorus compounds.
As for PCFC, DEP-based PHUs exhibit a slightly lower heat of combustion. Together with a higher residue fraction, they lead to a lower THR (around 15.5 kJ·g−1 versus 16.6–20 kJ·g−1 for other resins).
Finally, all phosphorus-based PHUs show enhanced smoke production. Total Smoke Production (TSR) increases from 820 m2·m−2 for the reference to more than 2000 m2·m−2 for DOPO-based PHUs. Smoke production basically depends on the material composition as well as on the burning rate. TSR is the highest for DOPO and BPPO, and lower for DEP and DPP, i.e., for the PHUs showing the lowest HRR. On another hand, TSR increases when phosphorus content increases from 1 to 2 wt% P (except for DEP).
Pictures of some chars from cone calorimeter tests are shown in
Figure 6. The reference does not lead to any cohesive char covering the whole sample surface. Char from DOPO-based PHUs is more consistent but many cracks can be observed preventing its barrier effect. Chars from BPPO and DPP-based PHUs are only slightly expanded but rather cohesive. Nevertheless, chars from DEP-based PHUs are strongly intumescent allowing efficient protection of the underlying material from the heat flux. While the volume is much larger but the residue content is only slightly higher, such chars are much more porous. The inner structure is made of many small pores which must be beneficial for effective insulating protection.