1. Introduction
Recently, the inhibition of proprotein convertase subtilisin kexin 9 (PCSK9) has emerged as a promising therapeutic target for reducing low density lipoprotein (LDL) levels. PCSK9 binds to the extracellular domain of the low-density lipoprotein receptor (LDLR), resulting in the receptor not recycling back to the cell membrane and degrading instead in the lysosome [
1]. Currently marketed PCSK9 monoclonal antibodies (mAbs), alirocumab and evolocumab have been proven to considerably lower LDL-C levels and subsequent cardiovascular events. However, FDA-approved humanized PCSK9 mAbs are cost-ineffective and are only administered as subcutaneous injections once or twice a month. Thus, several small-molecule orally active PCSK9 inhibitors are being discovered and developed as feasible alternatives to treat atherosclerosis.
There are concerns about the possible negative consequences of very low serum cholesterol on brain health [
2,
3,
4]. PCSK9 inhibitors have been scrutinized since their public launch over a decade ago for their ability to reduce LDL-C levels far below any statin. However, it has been demonstrated that average LDL-C levels of approximately 120–140 mg/dL could be safely reduced to 30–60 mg/dL [
2,
3]. One of the main issues raised is the connection between low lipoprotein levels and neurocognitive decline [
4]. Dysregulation of brain cholesterol metabolism has been tied to many neurodegenerative malignancies. This led to concerns that PCSK9-targeting entities that lower lipids might negatively affect neuronal function or development. However, cholesterol homeostasis in the brain is thought to be independent of the rest of the body as lipoproteins do not cross the blood brain barrier [
4]. Circulating PCSK9 mAbs are unable to cross the blood-brain barrier, and thus, potential therapeutic mAbs that reduce PCSK9 are not expected to directly reduce PCSK9 expression levels in the brain [
5]. So far, there is no enough evidence to link PCSK9 mAb inhibitors to serious adverse events [
6].
It is estimated that marine species comprise 25% of the total species on the planet [
7]. In response to their extremely difficult and harsh environments, marine organisms produce potent secondary metabolites with unique chemical structures and bioactivity. These secondary metabolites derived from the abundance of marine species represent a fundamentally untapped resource for drug discovery [
8]. Even though discoveries of potential marine-derived therapeutics have been steadily increasing since the advent of SCUBA diving gear in the 1950′s, the number of marine natural products that are under development or marketed as approved drugs are rather few. This can mainly be attributed to difficulties in collecting source organisms, challenges for large-scale production of marine natural products, high potential toxicity of numerous marine compounds, and the lack of scalable extraction procedures resulting in limited quantities insufficient for high-throughput bioassays and subsequent developments [
9].
Pseurotin A (PsA) is a spiroheterocyclic γ-lactam alkaloid (
Scheme 1,
Supplementary Materials: Figures S1 and S2) first isolated as a fermentation product from
Pseudeurotium ovalis (strain S2269/F) and later reported in numerous marine and terrestrial
Aspergillus and
Penicillium species [
10]. PsA competitively inhibited fungal chitin synthase, induced pheochromocytoma PC12 cells differentiation, and was patented as apomorphine antagonist and bacteriostatic food preservative [
11]. PsA also showed anti-inflammatory and weak cytotoxic activities. PsA inhibited IgE production and showed anti-seizure activity in animal models. PsA also inhibited osteoclasts and prevented osteoporosis [
11]. This novel natural product has been recently reported as a dual inhibitor of PCSK9 secretion and PCSK9 LDLR protein–protein interaction (PPI) [
11]. PsA possesses a rare oxygenated 1-oxa-7-azaspiro[4.4]non-2-ene-4,6-dione structural skeleton that is highly substituted with 5 chiral centers [
12]. The unique PsA molecular structure mimics a drug-like high selectivity profile at its possible molecular target(s), which was consistent with results of virtual ADMET studies [
11,
12].
One of the key risk factors for the development of atherosclerosis is elevated LDL levels. The persistent elevation of LDL cholesterol (LDL-C) is a major cause for atherosclerotic cardiovascular disease, and several studies have linked increased serum cholesterol levels with a higher risk of developing cancers [
13,
14]. However, there are also observations that suggest no association exists between cancer progression and cholesterol [
15,
16]. This discrepancy reveals that the relationship between cholesterol and cancer might not be a simple two-factor association, and that hypercholesterolemia may only be a contributing factor in some types of cancers but not all. Prostate cancer and breast cancer are two types of cancer with a plethora of clinical analyses and animal experiments that support the relationship between serum cholesterol levels and higher risk of cancer progression and recurrence [
17,
18,
19,
20,
21]. A meta-analysis highlighted that a non-linear relationship exists between higher dietary cholesterol intake and increased risk of developing breast cancer [
22]. Nude mice fed on a high-fat diet (11% fat) developed several-fold higher breast and prostate cancer volumes [
11,
23]. Studies have revealed PsA’s ability to suppress the progression of hormone dependent BT-474 breast cancer cells in a nude mouse xenograft model [
11]. PsA has also been proven in vivo to suppress the progression and recurrence of prostate cancer using a xenograft nude mice model [
23].
To the best of our knowledge, the present study is the first to investigate the potential acute toxicity of orally-administered pseurotin A in vivo before it can be validated for efficacy and safety in human clinical trials. Marketed PCSK9 mAb inhibitors evolocumab and alirocumab are currently being used for the treatment of homozygous familial hypercholesterolemia proving the therapeutic potential of targeting PCSK9 [
24,
25,
26]. This study shed light on the safety thresholds and animal exposure limits of PsA, the recently uncovered PCSK9-LDLR axis modulator, which will aid its subsequent validity as a novel orally active small molecule prostate and breast cancer recurrence modulatory entity. This study investigated the in vitro cytotoxicity and acute influence of purified orally-administered PsA treatments on multiple biochemical, hematological, and histopathological parameters in Swiss albino mice.
3. Discussion
Lipid-lowering therapies, such as statins, continue to be at the forefront of treatment and prevention of cardiovascular events; however, patient’s resistance to continued therapy as well as statin-associated muscle symptoms (SAMS) [
36] have spurred the discovery of novel molecular targets and molecules. PCSK9 inhibitors traditionally used to control hypercholesterolemia by reducing LDL-C. PCSK9 proved playing oncogenic driving role in gastric, lung, colon, breast, liver, ovarian, and skin cancers, validating it as a novel molecular target in cancer [
11,
23]. The marine and terrestrial fermentation product PsA was recently reported as the first small molecule dual inhibitor of PCSK9 expression and PPI [
11,
23]. Though expectations are high, there are valid concerns over possible adverse side effects of new PCSK9 inhibitors, especially on liver, the main PCSK9 biosynthesizing machinery in the human body. This study, to the best of our knowledge, is the first to analyze and report the acute safety profile in vivo of PsA, the novel PCSK9-LDLR axis modulator. The objectives of this study were to gauge potential toxic effects of PsA through in vitro testing as well as single oral administration in Swiss albino mice; therefore, it preliminarily assessed its prospective safety profile for subsequent developmental stages.
Biosafety appraisal of pharmacologically-validated hits and leads is a critical endpoint at the early stages of the drug development pipeline. In vitro cytotoxicity assays are routinely deployed in cell cultures by assessing cellular death response to tested molecules. Current cellular assays mostly rely on assessing the cell viability as a function of either membrane integrity, mitochondrial functionality, oxidative stress, or cell death [
37]. Herein, we adopted three of these approaches to assess the cytotoxic effects of PsA on two non-tumorigenic human epithelial cell lines: the RWPE-1 prostatic and the CCD 841 CoN colon cells. We have previously reported the recurrence suppressing effects of PsA on prostate cancer in animal models [
23]. Therefore, the immortalized RWPE-1 human prostatic cells were chosen to mimic the target human prostatic gland. Moreover, the CCD 841 CoN human colon cells also chosen to mimic the GIT cells, since PsA was the first oral small molecule modulator of the PCSK9-LDLR axis [
11,
23]. Previous in vitro results on prostate cancer cells showed relatively low cytotoxicity, 100–150 µM, but was impressive in suppressing breast and prostate cancers motility and recurrences [
11,
23]. Thus, we chose doses at effective and relatively higher concentrations to determine the selective effects of PsA (cancer versus immortalized human cells) and to monitor its cytotoxicity at higher concentrations for subsequent preclinical studies. PsA showed no significant effects on the viability of RWPE-1 cells from the concentration range of 0.005 to 0.75 mM (
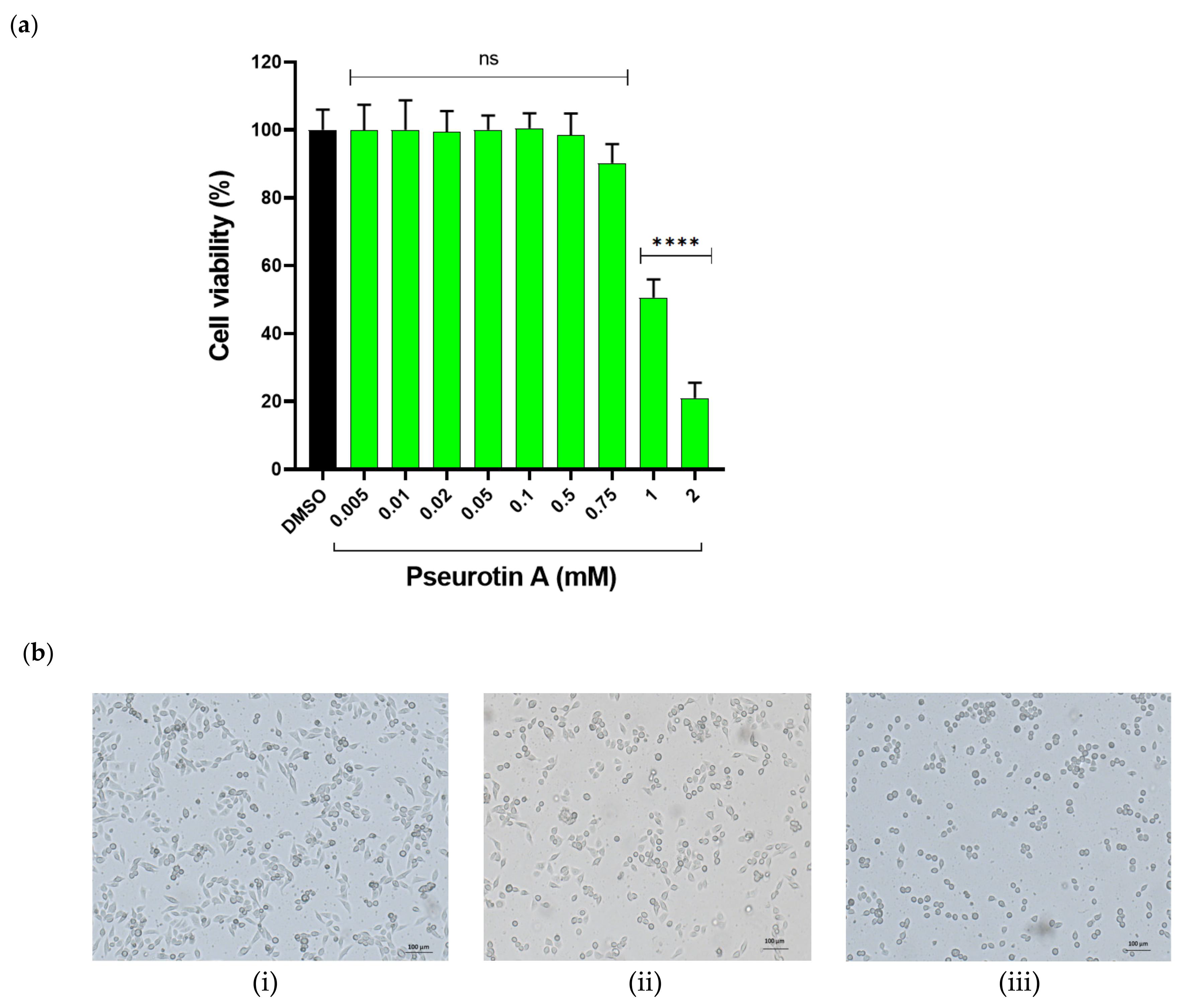
Figure 1), which indicated that PsA lacks cytotoxicity to normal prostate cells at reported anticancer concentrations. Meanwhile, high concentrations at 1 and 2 mM induced significant cell death, indicated by the reduction of cell viability to 49% and 19%, respectively. Moreover, RWPE-1 cells morphological changes were detected at 1 and 2 mM PsA, including cell rounding and cytoplasmic shrinkage (
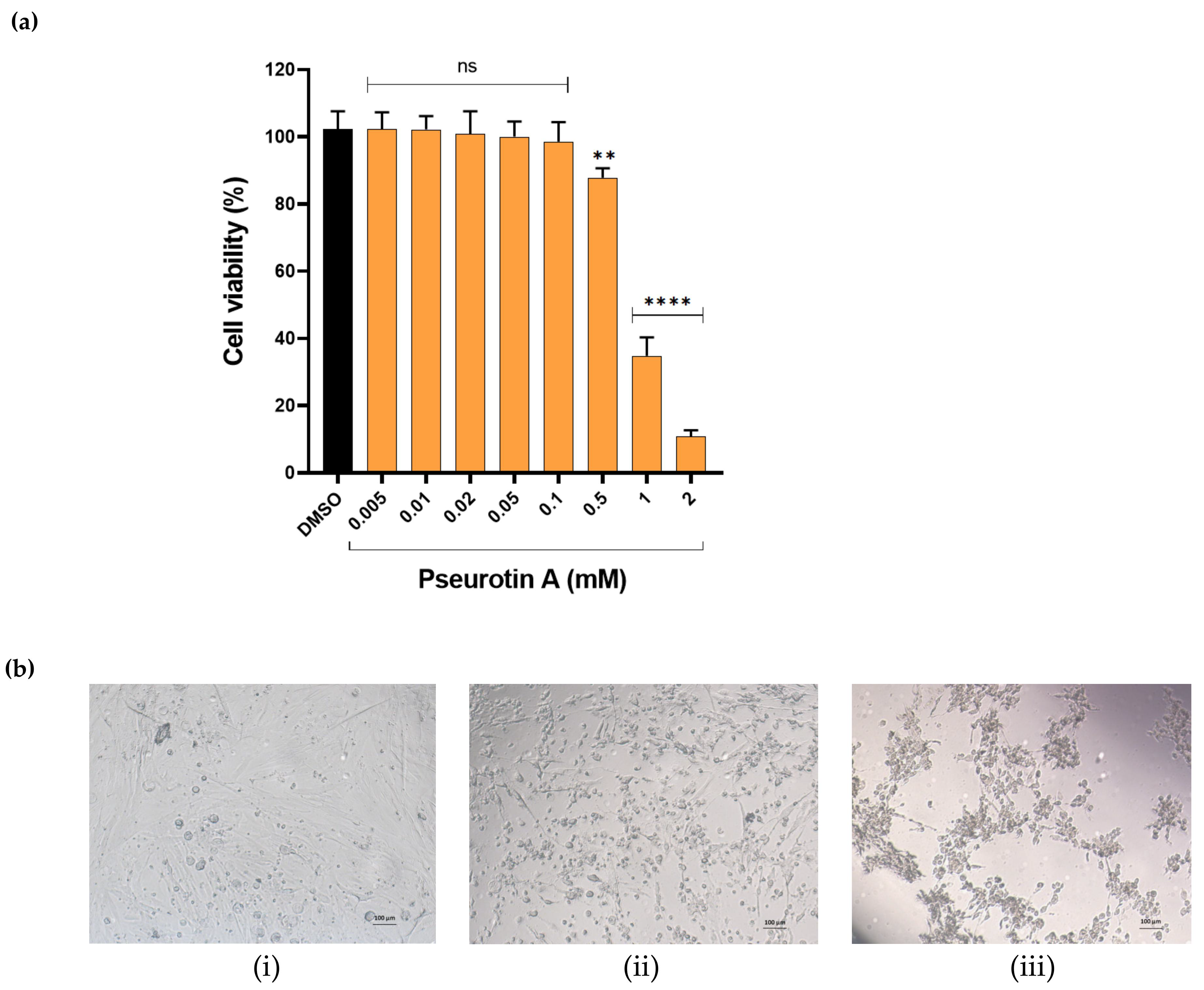
Figure 2), which suggests apoptosis as the main mechanism of cellular death. Furthermore, the viability of CCD 841 CoN colon cells was not much affected when treated with PsA up to 100 µM. PsA 500 µM treatment showed a slight decrease in cell viability (12.3%). Additionally, 1 and 2 mM of PsA induced a significant toxic effect indicated by 38 and 9% cell viability, respectively, at these treatment concentrations. Taken together, this data highlights the broad safety profile of PsA at the cellular level in different human non-tumorigenic cell lines.
To access for changes in mitochondrial functionality induced by PsA, a bioluminescence assay that measures intracellular ATP was utilized. Adenosine triphosphate is the main source of energy for all life on the planet. In cells, the hydrolysis of ATP to ADP releases energy that gets transferred to other molecules. Cellular response to stress can drastically change intracellular ATP, and ATP biosynthesis will be completely halted following cell death. This makes the measuring of intracellular ATP a primary indicator of cell viability. There are multiple validated methods to measure ATP concentration; however, bioluminescence ATP assays are seen as the most rapid, sensitive, and selective [
38]. Intracellular ATP levels were significantly downregulated in PsA-treated RWPE-1 cells compared to control cells (
Figure 3). This was especially evident in the 4 mM and 8 mM concentrations with a percent intracellular ATP concentration of only 1.75% and 3.03%, respectively, compared to the control. Cells treated with 1 mM PsA showed more pronounced levels of intracellular ATP (44.71%) versus the control, while the 0.5 mM PsA treatment measured ATP 91.84% of the control cells (
Figure 3). This data highlights the relative safety of PsA to mitochondrial integrity and function at concentrations below 1 mM.
To determine if apoptotic death is induced by PsA, molecular signaling pathways regulating apoptosis were investigated by a Western blot analysis. It is well documented that the Bcl-2 family of prosurvival proteins regulates apoptosis by controlling the release of cytochrome-c from the inner mitochondrial membrane, which will activate several adaptor proteins such as APAF-1 to form apoptosomes. Bcl-2 was downregulated in PsA-treated cells in comparison to its expression level in control cells indicating an active process of apoptosis at indicated concentrations (
Figure 4a). In the same sense, the Bax is recognized for its pro-apoptotic function via opening the voltage-dependent anion channel (VDAC), and thus increasing the permeability of the mitochondrial inner membrane to release cytochrome-c. Immunoblot results of lysates pooled from treated cells revealed the significant upregulation of Bax protein at 1 and 2 mM of PsA when compared to the control cells, proving a dynamic apoptotic response at indicated concentrations of PsA (
Figure 4). Furthermore, the expression level of caspases 3 and 7 were monitored in the cell lysate of PsA-treated and control cells. Caspases 3 and 7 are highly specific proteases involved in execution phase of cell apoptosis through proteolytic cleavage of target proteins involved in DNA fragmentation, chromatin condensation, and cytoskeletal damage. Our results revealed the significant upregulation of both caspases in a concentration-dependent manner (
Figure 4), which emphasizes the apoptotic toxicity of PsA at indicated high doses. However, it is important to point out that the apoptotic effect of PsA on RWPE-1 prostatic cells is seen at approximately 10 and 20-fold its effective anticancer concentrations, signaling the broad safety profile of PsA at a cellular level [
23].
The Up-and-Down procedure determined the LD
50 for PsA to be greater than 550 mg/kg. This procedure was also utilized as a range finder to help determine which concentrations would be used in the 14-day single dose acute study. Complications with solubility prevented testing lethality of PsA at 2000 mg/kg. However, the therapeutic dose for prevention of tumor recurrence was determined to be 10 mg/kg body weight in a previous study [
23], so the concentration chosen as the highest dose for this acute study was 50× higher, indicating a good safety. Both sexes of mice were implemented into this study as well as the 14-day acute toxicity study. This is in conjunction with the National Institutes of Health (NIH) guidelines (NOT-OD-16-006) that require the use of both sexes in screening of acute organ toxicity unless it is well justified. Gender-based differences in the toxicity of pharmaceuticals is a question that must be addressed to establish an accurate safety profile for all novel therapeutics.
The first noticeable indicators of possible toxicity in any mammalian safety assessment are changes in behavioral, autonomic, or neurological responses. Though slight hunching, reduced mobility, and anti-social behavior was noted within the first 1–2 h observation in a few of the male 500 mg/kg mice, only 3 h after dosing, the mice’s behavior returned to normal and remained healthy over the remainder of the 14-day experimental period. The assessment of animal welfare in safety studies often lacks experience and objectivity. Thus, there is the need for well-defined and implemented systems for recording animal indicators of distress [
39,
40,
41]. To account for this, the mouse grimace scale was implemented to quantify pain and discomfort more reliably [
28]. A senior toxicologist with over 20 years of animal testing supervised the initial 1–2 h observation after PsA dosing to monitor for changes in response.
A significant finding of the study was the extreme body weight loss of the female Swiss albino mice in the 500 mg/kg group over the 14-day safety study. Loss of body weight is widely accepted as a potential early marker for organ toxicity, and research shows a high empirical association between the loss of body weight and pathological findings [
41]. Although many scientists define a body-weight reduction greater than 20% as a potential parameter for humane sacrifice, other recent studies strongly suggest a more flexible implementation of weight loss as a humane endpoint criteria that stresses using additional criteria for evaluating pain and puts more emphasis on the unique differences in experimental design [
42]. In accordance with directive 2010/63/EU [
43], which promotes the 3R’s (replacement, reduction, and refinement), the female mice 500 mg/kg group were closely monitored for the remainder of the study for behavior responses, neuromuscular responses, and autonomic responses. This group of female mice displayed no signs of pain and no changes in behavior from the time of initial dosing to the day of sacrifice. This guided the decision not to humanely sacrifice the mice after the weighing on day four showed >20% decrease in weight observed across all the animals in the group. Weight loss is an expected therapeutic outcome for PCSK9-targeting drugs. The phase IV clinical trial of the FDA-approved humanized mAb Repatha observed notable weight loss for people who have been taking the drug for 6–12 months, with the weight loss being especially prominent in females [
44]. Another observation of note was that upon dissection, the PsA 250 mg/kg and 500 mg/kg treatment groups had no observable fat surrounding their G.I. tract, while the 10 mg/kg and control groups showed noticeable white fat surrounding their G.I. tract. This clearly indicates effective PsA PCSK9 axis-targeting outcomes at single high doses.
The AST and ALT are among the most reliable markers of liver cell injury/necrosis. The liver transaminases AST and ALT were both significantly increased in the female 250 mg/kg group compared to the control. The same increase was seen in the males but not to a significant degree. ALT is seen as the more specific marker for hepatotoxicity due to its high expression in hepatocytes and low expression in other cells of the body. In contrast, AST has mitochondrial and cytosolic forms present in the tissues of the kidneys, pancreas, lungs, brain, heart, skeletal muscle, and liver. AST and ALT levels can be elevated to levels exceeding 2000 U/L in cases of major hepatocyte injury or necrosis; however, increases not exceeding 5-fold the normal ranges are much more commonly seen in primary care medicine [
45]. One study stated that serum ALT reference intervals for healthy Swiss albino mice are 46.15 ± 5.62 U/L for males and 59.03 ± 9.58 U/L for females [
46]. Another study that measured the clinical markers for over 5000 healthy mice of different breeds found the average ALT for males to be 30 U/L and females to be 26 U/L, which are very close to the values observed in the control groups [
47]. The average level of serum ALT for the 250 mg/kg female group was 49.5 ± 4.1. This was less than a two-fold increase over the serum ALT of the female control group (28.5 ± 3.6) and lower than the healthy reference level of 59.03 ± 9.58 U/L. Meanwhile, a cross-sectional study found an association between low LDL-C levels and elevated serum transaminases [
48]. The histopathology of the liver tissue sections showed no signs of steatosis, necrosis, cirrhosis, inflammation, or other signs of toxicity to the hepatocytes. This leads to the belief that the increase in AST and ALT are not due to overt hepatocyte injury/necrosis.
Significantly lower levels of serum alkaline phosphatase (ALP) were observed in the female 250 mg/kg group. ALP is a hydrolase enzyme that is highly expressed in the liver, bone, and kidney. Elevated serum levels have commonly been used as a marker for hepatotoxicity. Lower ALP levels are not seen as a marker for hepatotoxicity but, instead, may indicate protein deficiency, malnutrition, or insufficient number of vitamins or minerals. The serum ALP levels of the female 250 mg/kg mice were not considered to be at very low levels (<30 U/L).
Liver is regarded as the organ with the highest levels of PCSK9 expression in the body [
29], making it a primary target for testing PsA cytotoxicity on hepatocytes. The prosurvival Bcl-2 protein was slightly downregulated for the 500 mg/kg dose group in both animal genders, while it remained unchanged for the lower doses. On the other hand, the proapoptotic Bax remained unaffected at 10 and 250 mg/kg dose levels, while being upregulated at 500 mg/kg dose in both sexes (
Figure 8). Taken together, the data strongly suggest that PsA could induce apoptosis in mouse liver tissues at very high doses relative to its previously reported therapeutically recommended dose of 10 mg/kg in nude mice [
11,
23]. In a similar fashion, the executioner caspases 3 and 7 were upregulated at the 500 mg/kg dose, while being unchanged for the 10 and 250 mg/kg doses of PsA (
Figure 10). These results propose a broad biosafety margin for the PCSK9 axis-targeting PsA as a prostate cancer recurrence suppressor, with hepatocyte cell death observed only at 50-fold the recommended anticancer therapeutic dose [
11,
23].
Another sensitive indicator of potential drug toxicity is its effect on relative organ weight [
49]. No significant changes (
p > 0.05) on the relative liver, kidneys, spleen, lungs, heart, and brain weights were observed for the treatment and control groups of both mouse sexes. Similarly, no organ variations in the size, color, or overall appearance noticed in each treated group versus vehicle control.
Hematological parameters continue to be important markers in the detection of injury or disease in animals and humans. It has been reported that deleterious hematological results in animals have one of the highest concordances with negative results in humans [
50]. The average RBC-C, HGB, HCT, MCV, and MCHC was unchanged amongst the treatment and control groups, indicating no signs of aplastic or hemolytic anemia. The platelet count increased in a step-wise manner in the 10 mg/kg and 250 mg/kg treatment groups, for both genders, versus vehicle control before falling slightly below the control levels in the 500 mg/kg treatment group. The PLT levels in the 500 mg/kg dosing group were too high to be considered thrombocytopenia [
51]. The only significant finding among the hematological data was the increase in platelet count for the male 250 mg/kg group (
p > 0.05). PsA treatment is causing thrombocytosis, an increase in platelet count, up to a certain dose; however, a high platelet count is not very indicative of serious injury and most times does not cause any symptoms. Possible causes of thrombocytosis are inflammation, anemia, or cancer [
52].
The results of this 14-day acute toxicity study, when viewed together, strongly suggest that acute doses up to 500 mg/kg of PsA present no major toxicity to the organs analyzed. The decrease in body weight for the female 500 mg/kg group was the most significant finding and signaled potential organ toxicity. However, the relative organ weight saw no change compared to the control and the results of the histopathological examination revealed no signs of damage to the liver, kidney, heart, brain, and lungs. The weight loss effect of PsA is expected in PCSK9-targeting entities. The hematological and biochemical parameters tested also showed miniscule change compared to the control, with the exception of the liver transaminases in the female 250 mg/kg dosing group. Future repeated dose studies will test the sub-acute and sub-chronic effects associated with PsA dosing. Moreover, HPLC detection of PsA in Swiss albino mouse sera inevitably confirmed systemic effects (
Figure S3). The novel marine natural product PsA is a prospective entity appropriate for subsequent preclinical development of prostate and breast cancer recurrence controls.
4. Materials and Methods
4.1. Cell Lines and Culture Conditions
Non-tumorigenic human prostatic cells RWPE-1 and colon CCD 841 CoN epithelial cells were purchased from the American Type Culture Collection (ATCC, Manassas, VA, USA). RWPE-1 cells were maintained in Gibco keratinocyte serum-free basal medium (Thermo Scientific, Rockford, IL, USA) supplemented with 5 µg/mL bovine pituitary extract (BPE), and 5 ng/mL recombinant human EGF. CCD 841 CoN cells were cultured in EMEM (ATCC, Manassas, VA, USA) supplemented with 10% fetal bovine serum (R&D Systems, Inc, Minneapolis, MN, USA). Cells were regularly sub-cultured upon confluency using trypsin EDTA (Corning, Glendale, AZ, USA) and fresh growth media. Cells were maintained in a humidified incubator kept at 37 °C with 5% CO2.
4.2. In Vitro Cytotoxicity of Pseurotin A
4.2.1. MTT-Based Cell Cytotoxicity Assay
RWPE-1 cells were plated in a 96-well microplate at a density of 3 × 104 cells per well. Cells were allowed to recover and attach for 24 h post seeding, while CCD 841 CoN were plated at a cell density of 2 × 104 per well and allowed 12 h to adhere. Upon attaching, media were removed, and serum-free media containing different treatment concentration of PsA (from a stock solution of 100 mM in sterile DMSO) were added to each well. Cells were then incubated with treatment concentrations for 24 h, and then media were gently removed, and 100 µL of MTT solution (0.5 mg/mL final concentration) were added for each well. Plates were then incubated for 3 h and monitored for the formation of formazan crystals. After the incubation period, media were gently removed, and crystals were solubilized in 100 µL DMSO before measuring the optical absorbance at 570 nm using a Synergy 2 microplate reader (Bio Tek Instruments Inc., Winooski, VT, USA).
4.2.2. ATP-Based Cell Cytotoxicity Assay
RWPE-1 cells were plated in a 96-well microplate at a density of 3 × 104 cells per well. Cells were allowed to recover and attach for 24 h post seeding and 3 replicates/groups were utilized. Upon attaching, media were removed, and serum-free media containing different treatment concentration of PsA (from a stock solution of 500 mM in sterile DMSO) added to each well. Cells were then incubated with treatment concentrations for 24 h after which ATP release was determined using a Promega CellTiter-Glo® 2.0 Assay kit (Promega Corporation, Madison, WI, USA). An amount of 95 μL of CellTiter-Glo® 2.0 Reagent (equal to the volume of cell culture media resent in each well) was added to each well containing cells. After 2 min on an orbital shaker followed by another 10 min incubation at rt, the luminescence of each well was measured using a Synergy H1 hybrid reader (BioTek Instruments Inc., Winooski, VT, USA).
4.2.3. Western Blot Analysis
Cells treated with different concentrations of PsA and liver tissues isolated from animals of different treatment and control groups were lysed using RIPA buffer (Thermo Scientific, Rockford, IL, USA) supplemented with a mammalian protease inhibitor cocktail (G-Biosciences, St. Louis, MO, USA). Protein concentrations in cell and tissue lysates were determined by Pierce BCA assay (Thermo Scientific, Rockford, IL, USA) as per manufacturer protocol. Lysates were mixed with equal volumes of 2X Laemmli buffer (Bio-Rad, Hercules, CA, USA) containing 5% β-mercaptoethanol before heat blocking in a water bath at 90 °C for 5 min. About 30 ug of cell and tissue lysates were resolved on 10 and 12% SDS-PAGE using Tris/Glycine/SDS as resolving buffer. Resolved protein bands were then transferred to PVDF membranes using Tris/Glycine buffer in 20% methanol/water at 4 °C overnight. Membranes were then blocked by rocking in 5% BSA/TBST for 2 h. Blocked membranes were then incubated overnight with different primary antibodies at 4 °C. Tubulin, Bax, Bcl-2 antibodies were purchased from Cell Signaling (Danvers, MA, USA) and caspases 3 and 7 were obtained from ProteinTech (Rosemont, IL, USA). Primary antibodies were used per vendor recommended dilutions in 5% BSA/TBST. Membranes were then washed 3–4 times with TBST before incubating with Cell Signaling HRP-linked antirabbit secondary antibody for an additional 2 h. Finally, membranes were washed 3–4 times with TBST before adding Super Signal West Femto prior to the imaging with ChemiDoc Touch Imaging System (Bio-Rad, Hercules, CA, USA). Densitometric analyses accomplished using the Image J software (NIH, Bethesda, MD, USA). Photographed protein blots were then normalized to β-tubulin for a quantitative comparison relative to untreated control cells and tissues.
4.3. Experimental Animals
Twenty male and twenty female Swiss albino mice, 4–5 weeks old, were purchased from Envigo (Indianapolis, IN, USA). The animals were given a 2-week acclimatization period and maintained under clean room conditions with a relative humidity of 55–65%, a temperature of 22 ± 2 °C, a 12:12 h light/dark cycle, Alpha-Dri bedding, and free access to drinking water and pelleted rodent chow (no. 7012, Envigo/Teklad, Madison, WI, USA). Animals were housed in group cages (male; n = 5 and female; n = 5) and kept in the same environmental conditions. All animal experiments were approved by the Institutional Animal Care and Use Committee (IACUC), University of Louisiana at Monroe, and were conducted in strict accordance with good animal practice as defined by NIH guidelines (Protocol # 21DEC-KES-03).
4.4. Up-and-Down Procedure
The procedure for determination of an LD
50 was performed following the Organization of Economic Co-operation and Development (OECD) guideline 425 [
27] for the testing of chemicals. Animals were given a 2-week acclimatization period. The starting dose for the procedure was 175 mg/kg per guideline 425 suggestion for the testing small molecules. The dosing progression was 175–550 mg/kg and was halted there due to limited PsA solubility at higher concentrations. Initially, mice were randomly selected and treated with a single PsA oral dose via oral gavage feeding syringe. After administering the dose, the mice were observed for mortality/morbidity and behavioral signs of pain/toxicity at 1, 2, 4, 6, 12, and 24 h and then once daily for a total of 14 days. If no mortality was observed after 24 h, another animal would receive a 3.2× increased dose, in a step wise manner, until stopping criteria was met (the upper limit of 550 mg/kg was reached). Toxicity was observed for 14 days unless clear signs of grimace and pain were present. In that case, the animal was euthanized in accordance with the 3R’s of toxicity testing. Rats were anesthetized using Isoflurane (2–5%).
4.5. 14-Day Single Oral Dose PsA Acute Toxicity Study Design
For the single oral dose 14-day acute toxicity study, healthy 8-week-old male and female Swiss albino mice were randomly selected and placed in 8 groups (n = 5/sex/group). Graded doses of PsA (10, 250, 500 mg/kg body weight) were administered to the mice formulated in <5% DMSO/0.2% Tween 80 at a volume not exceeding 200 µL by flexible plastic oral gavage (2 mm diameter with stainless steel bite protector, 18-gauge, 3.81 cm long). The mice were observed for mortality, morbidity, and behavioral signs of pain/toxicity at 1, 2, 4, 6, 12, and 24 h and then once daily for a total of 14 days. The body weights of the mice were weighed in grams using a Scout
TM Pro (Ohaus Corp., Pine Brook, NJ, USA) on day 0, 4, 8, 11, 14 after initial treatment. On day 14, all mice were anesthetized using isoflurane (USP-vaporizer method, Matrix VIP-3000, exposure of animals in anesthesia chamber at 3% isoflurane vaporization rate. Exposure time 3 min). Mice were then euthanized by cervical dislocation according to AVMA Guidelines on Euthanasia (2013) and dissected. Mice organs (brain, liver, kidney, heart, lungs, spleen) were excised and weighed for histopathological examination. The organs were stored in 10% formalin for 24 h then transferred to 70% ethanol. This study was performed following the OECD guideline 423 for the testing of chemicals [
53].
4.6. Hematological and Biochemical Evaluation
Sacrificed mice were decapitated and the blood was quickly collected into a green topped Sodium Heparin 3.6 mg blood collection tube (BD Vacutainer REF# 368771) as well as a purple topped K2 EDTA (K2E) 3.6 mg blood collection tube (BD Vacutainer REF# 367841) to ensure that all blood samples were free of clots. The Sodium Heparin samples were immediately centrifuged at 13,000× g for 10 min and the plasma was collected for biochemical analysis. Samples were analyzed and glucose, AST, ALT, ALP, BUN, and creatinine levels were determined with the Beckman AU680 clinical chemistry analyzer system (Beckman Coulter, Atlanta, GA, USA). The EDTA samples were analyzed for hematological parameters and WBC, RBC, Hgb, Hct, MCV, MCHC, and PLT were determined using the Siemens Advia 120 hematology analyzer (Siemens Healthcare Diagnostics Inc., Tarrytown, NY, USA). All blood samples were analyzed at the LSU School of Veterinary Medicine (SVM) Clinical Pathology Laboratory at Baton Rouge, Louisiana.
4.7. Data Analysis
Results for the hematological and biochemical parameters were analyzed separately by a one-way Analysis of Variance (ANOVA) followed by multiple comparison with Dunnett’s test. All statistical analysis was performed using GraphPad Prism version 8 software (San Diego, CA, USA). Data in this study was expressed as a mean ± SD (standard deviation) and mean ± SEM (standard error of mean). A probability value of <0.05 was considered statistically significant (* p < 0.05; ** p < 0.01; *** p < 0.001; and **** p < 0.0001).
4.8. Paraffin Embedding and Staining
The mouse organs were carefully removed and quickly fixed in 10% neutral buffered formalin for 24 h before transfer to 70% ethanol. Organs were then embedded in paraffin before sectioning and staining by hematoxylin and eosin (H&E). All sectioning and staining were done at the AML Laboratories (Jacksonville, FL, USA). The paraffin-embedded tissues were sliced into 5 um-thick sections before being mounted on a positively charged slide. The 5 um-thick sections were then dewaxed using xylene, rinsed using 80–95% ethanol, and rehydrated using H2O. Afterwards they were stained with H&E before being dehydrated again using 95–80% ethanol to xylene.