Potentiating Biosynthesis of Alkaloids and Polyphenolic Substances in Catharanthus roseus Plant Using ĸ-Carrageenan
Abstract
:1. Introduction
2. Results
2.1. Effect of Carrageenan Spraying on Growth Parameters of Catharanthus Roseus
2.2. Effect of Carragennan Spraying on Chemical Parameters of Catharanthus roseus
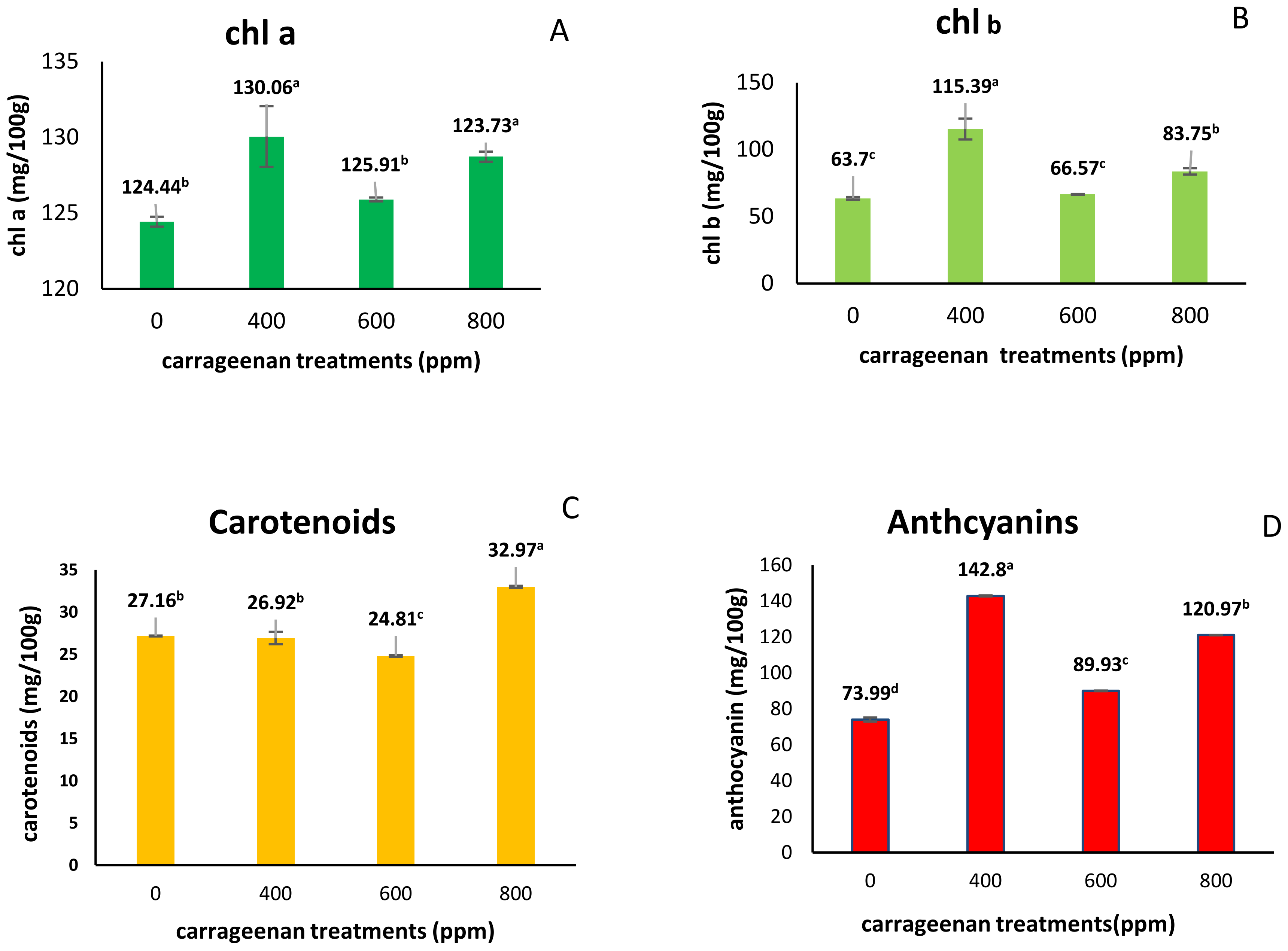
2.3. Effect of Carrageenan Treatments on Pigments
2.4. Effect of Carrageenan Treatments on Elements Content
2.4.1. Macro Elements (N, P, K and Ca)
2.4.2. Micro and Essential Elements (Mn, Cu, Zn and Se)
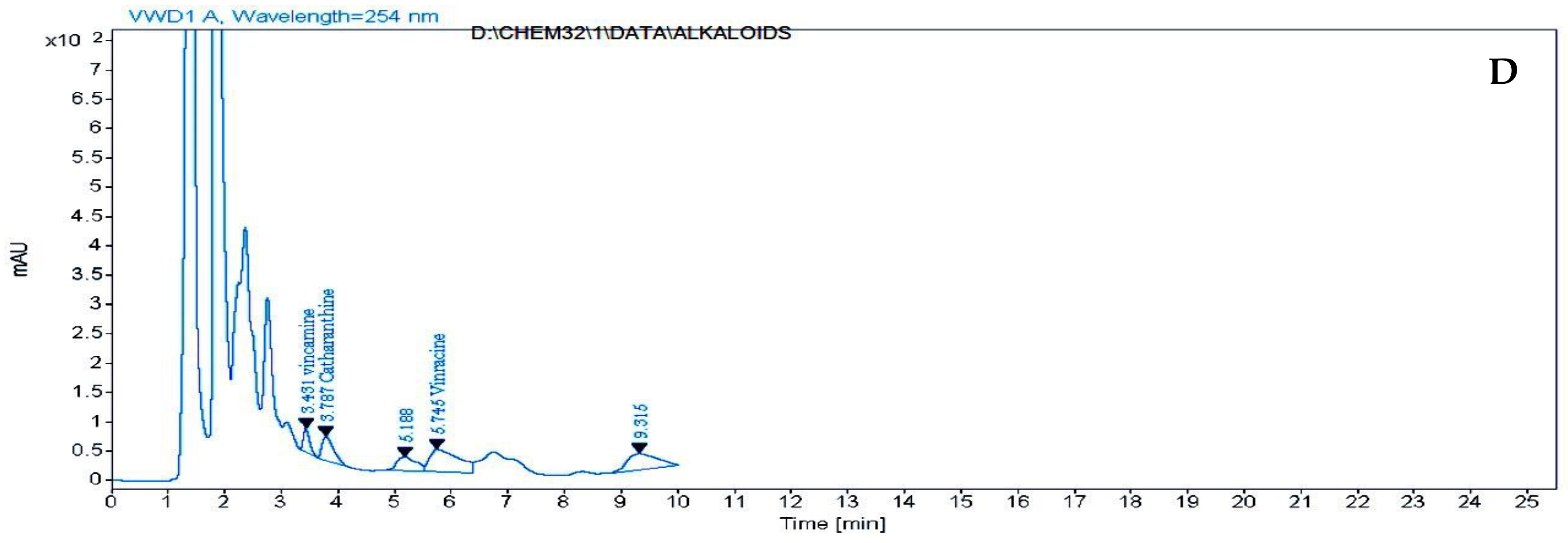
2.5. HPLC Analysis of Free Amino Acids, Phenolic Compounds and Alkaloids of Catharanthus roseus Leaves Treated with k-Carrageenan
3. Discussion
3.1. Effect of Carrageenan Spraying on Growth Parameters of Catharanthus roseus
3.2. Effect of Carragennan Spraying on Chemical Parameters and Phytochemical Screening of Catharanthus roseus
3.3. Effect of Carrageenan Treatments on Pigment
3.4. Effect of Carrageenan Treatments on Elements Content
3.5. HPLC Analysis of Free Amino Acids, Phenolic Compounds and Alkaloids of C. roseus Leaves Treated with ĸ-Carrageenan
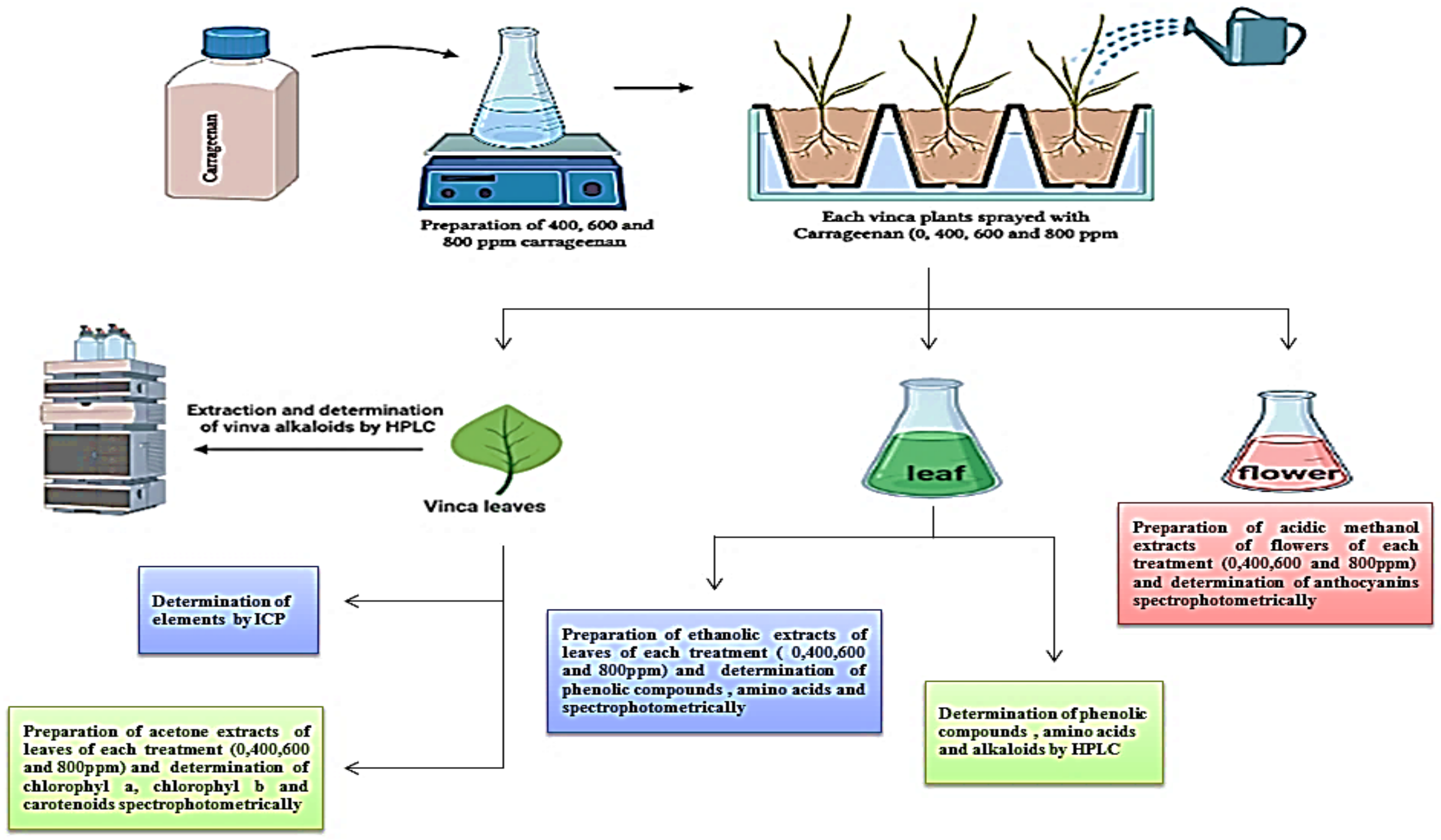
4. Material and Methods
4.1. Plant Material, Field Experiment and Treatments
4.2. Preparation of 80% Ethanolic Extract
4.3. Phytochemical Screening of the Catharanthus roseus Leaf Extracts
4.3.1. Quantitative Analysis of Some Phytochemicals in the Catharanthus roseus Leaf
Total Phenols Content (TP)
Flavonoids Content (F)
Free Amino Acids Content (FAA)
4.3.2. Total Alkaloids Content
4.3.3. Chl a, Chl b and Carotenoids Contents
4.3.4. Anthocyanin Content
4.3.5. Elements Contents
4.4. Free Amino Acids Separation by HPLC
4.5. Phenolic Compounds Separation by HPLC
4.6. Alkaloids (Vincamine, Catharanthine, Vincracine (Vincristine) and Vinblastine) Analysis by HPLC
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Falcão, M.A.; Scopel, R.; Almeida, R.N.; Santo, A.T.D.E.; Franceschini, G.; Garcez, J.J.; Vargas, R.M.; Cassel, E. Supercritical fluid extraction of vinblastine from Catharanthus roseus. J. Supercrit. Fluids 2017, 129, 9–15. [Google Scholar] [CrossRef]
- Leonti, M.; Stafford, G.I.; Cero, M.D.; Cabras, S.; Castellanos, M.E.; Casu, L.; Weckerle, C.S. Reverse ethnopharmacology and drug discovery. J. Ethnopharmacol. 2017, 198, 417–431. [Google Scholar] [CrossRef]
- Speed, M.P.; Fenton, A.; Jones, M.G.; Ruxton, G.; Brockhurst, M. Coevolution can explain defensive secondary metabolite diversity in plants. New Phytol. 2015, 208, 1251–1263. [Google Scholar] [CrossRef]
- Ćujić, N.; Šavikin, K.; Janković, T.; Pljevljakušić, D.; Zdunić, G.; Ibrić, S. Optimization of polyphenols extraction from dried chokeberry using maceration as traditional technique. Food Chem. 2016, 194, 135–142. [Google Scholar] [CrossRef]
- Sangha, J.S.; Khan, W.; Ji, X.; Zhang, J.; Mills, A.A.S.; Critchley, A.T.; Prithiviraj, B. Carrageenans, Sulphated Polysaccharides of Red Seaweeds, Differentially Affect Arabidopsis thaliana Resistance to Trichoplusia ni (Cabbage looper). PLoS ONE 2011, 6, e26834. [Google Scholar] [CrossRef]
- González, A.; Castro, J.; Vera, J.; Moenne, A. Seaweed Oligosaccharides Stimulate Plant Growth by Enhancing Carbon and Nitrogen Assimilation, Basal Metabolism, and Cell Division. J. Plant Growth Regul. 2013, 32, 443–448. [Google Scholar] [CrossRef]
- Zia, K.M.; Tabasum, S.; Nasif, M.; Sultan, N.; Aslam, N.; Noreen, A.; Zuber, M. A review on synthesis, properties and applications of natural polymer based carrageenan blends and composites. Int. J. Biol. Macromol. 2017, 96, 282–301. [Google Scholar] [CrossRef]
- El-Beltagi, H.S.; Mohamed, A.A.; Mohamed, H.I.; Ramadan, K.M.A.; Barqawi, A.A.; Mansour, A.T. Phytochemical and Potential Properties of Seaweeds and Their Recent Applications: A Review. Mar. Drugs 2022, 20, 342. [Google Scholar] [CrossRef]
- Prajapati, V.D.; Maheriya, P.M.; Jani, G.K.; Solanki, H.K. RETRACTED: Carrageenan: A natural seaweed polysaccharide and its applications. Carbohydr. Polym. 2014, 105, 97–112. [Google Scholar] [CrossRef]
- Shukla, P.S.; Borza, T.; Critchley, A.T.; Prithiviraj, B. Carrageenans from Red Seaweeds As Promoters of Growth and Elicitors of Defense Response in Plants. Front. Mar. Sci. 2016, 3, 81. [Google Scholar] [CrossRef]
- González, A.; Contreras, R.A.; Moenne, A. Oligo-Carrageenans Enhance Growth and Contents of Cellulose, Essential Oils and Polyphenolic Compounds in Eucalyptus globulus Trees. Molecules 2013, 18, 8740–8751. [Google Scholar] [CrossRef]
- Bi, F.; Iqbal, S.; Arman, M.; Ali, A.; Hassan, M.-U. Carrageenan as an elicitor of induced secondary metabolites and its effects on various growth characters of chickpea and maize plants. J. Saudi Chem. Soc. 2011, 15, 269–273. [Google Scholar] [CrossRef]
- Naeem, M.; Idrees, M.; Aftab, T.; Alam, M.M.; Khan, M.M.A.; Uddin, M.; Varshney, L. Radiation Processed Carrageenan Improves Plant Growth, Physiological Activities, and Alkaloids Production in Catharanthus roseus L. Adv. Bot. 2015, 2015, 150474. [Google Scholar] [CrossRef]
- Mousavi, E.A.; Kalantari, K.M.; Nasibi, F.; Oloumi, H. Effects of carrageenan as elicitor to stimulate defense responses of basil against Cuscuta campestris Yunck. Acta Bot. Croat. 2018, 77, 62–69. [Google Scholar] [CrossRef]
- Jiang, J.-L.; Zhang, W.-Z.; Ni, W.-X.; Shao, J.-W. Insight on structure-property relationships of carrageenan from marine red algal: A review. Carbohydr. Polym. 2021, 257, 117642. [Google Scholar] [CrossRef]
- Stewart, W.M. Sulfur-The 4th Major Nutrient, IPNI Plant Nutrition TODAY, Spring, 2010, 7. Available online: www.sulphurmagazin.com (accessed on 18 April 2023).
- Narayan, O.P.; Kumar, P.; Yadav, B.; Dua, M.; Johri, A.K. Sulfur nutrition and its role in plant growth and development. Plant Signal. Behav. 2022, e2030082. [Google Scholar] [CrossRef]
- Leustek, T.; Martin, M.N.; Bick, J.A.; Davies, J.P. Pathways and regulation of sulfur metabolism revealed through molecular and genetic studies. Annu. Rev. Plant Physiol. Plant Mol. Biol. 2000, 51, 141–165. [Google Scholar] [CrossRef]
- Lewandowska, M.; Sirko, A. Recent advances in understanding plant response to sulfur-deficiency stress. Acta Biochim. Pol. 2008, 55, 457–471. [Google Scholar] [CrossRef]
- Li, Q.; Gao, Y.; Yang, A. Sulfur Homeostasis in Plants. Int. J. Mol. Sci. 2020, 21, 8926. [Google Scholar] [CrossRef]
- Scherer, H.W.; Pacyna, S.; Spoth, K.R.; Schulz, M. Low levels of ferredoxin, ATP and leghemoglobin contribute to limited N2 fixation of peas (Pisum sativum L.) and alfalfa (Medicago sativa L.) under S deficiency conditions. Biol. Fertil. Soils 2008, 44, 909–916. [Google Scholar] [CrossRef]
- Bloom, A.J. The increasing importance of distinguishing among plant nitrogen sources. Curr. Opin. Plant Biol. 2015, 25, 10–16. [Google Scholar] [CrossRef]
- Hemerly, A. Genetic controls of biomass increase in sugarcane by association with beneficial nitrogen-fixing bacteria. In Proceedings of the Plant and Animal Genome XXIV Conference, Singapore, 6–8 June 2016. [Google Scholar]
- Ahmad, S.S.; Tahir, I. Regulatory role of phenols in flower development and senescence in the genus Iris. Indian J. Plant Physiol. 2017, 22, 135–140. [Google Scholar] [CrossRef]
- Chepel, V.; Lisun, V.; Skrypnik, L. Changes in the Content of Some Groups of Phenolic Compounds and Biological Activity of Extracts of Various Parts of Heather (Calluna vulgaris (L.) Hull) at Different Growth Stages. Plants 2020, 9, 926. [Google Scholar] [CrossRef]
- Dreccer, M.F.; Van Oijen, M.; Schapendonk, A.H.; Pot, C.S.; Rabbinge, R. Dynamics of Vertical Leaf Nitrogen Distribution in a Vegetative Wheat Canopy. Impact on Canopy Photosynthesis. Ann. Bot. 2000, 86, 821–831. [Google Scholar] [CrossRef]
- Sharma-Natu, P.; Ghildiyal, M. Potential targets for improving photosynthesis and crop yield. Curr. Sci. 2005, 88, 1918–1928. [Google Scholar]
- Ahmed, F.F.; Abdelaal, S.A.H.M.; El-Masry, E.M.A.; Farag, W.B.M.M. Response of superior grapevines to foliar application of some micronutrients, calcium, amino acids and salicylic acids. World Rural. Observ. 2014, 6, 57–64. [Google Scholar]
- Madian, A.M.; Refaai, M.M. The synergistic effect of using B vitamins with two amino acids tryptophane and methionine in Thompson seedless grapevines. Minia J. Agric. Res. Develop. 2011, 31, 100–121. [Google Scholar]
- Ahmad, S.; Tahir, I. Storage protocol for improving the postharvest performance in cut scapes of Iris versicolor. Acta Horti. 2015, 1060, 71–79. [Google Scholar] [CrossRef]
- White, P.J. Selenium metabolism in plants. Biochim. Biophys. Acta Gen. Subj. 2018, 1862, 2333–2342. [Google Scholar] [CrossRef]
- Schiavon, M.; Lima, L.W.; Jiang, Y.; Hawkesford, M.J. Effects of Selenium on Plant Metabolism and Implications for Crops and Consumers. In Selenium in Plants: Molecular, Physiological, Ecological and Evolutionary Aspects; Pilon-Smits, E.A.H., Winkel, L.H.E., Lin, Z.-Q., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 257–275. ISBN 978-3-319-56249-0. [Google Scholar]
- Gantait, S.S.; Pal, P. Anthocyanin content of spray Chrysanthemum cultivars under polyhouse and open field conditions. IJNPR 2010, 1, 236–242. [Google Scholar]
- Tanaka, Y.; Sasaki, N.; Ohmiya, A. Biosynthesis of plant pigments: Anthocyanins, betalains and carotenoids. Plant J. 2008, 54, 733–749. [Google Scholar] [CrossRef]
- Dong, J.Z.; Wang, Y.; Wang, S.H.; Yin, L.P.; Xu, G.J.; Zheng, C.; Lei, C.; Zhang, M.Z. Selenium increases chlorogenic acid, chlorophyll and carotenoids of Lycium chinense leaves. J. Sci. Food Agric. 2013, 93, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Thiruvengadam, M.; Chung, I.-M. Selenium, putrescine, and cadmium influence health-promoting phytochemicals and molecular-level effects on turnip (Brassica rapa ssp. rapa). Food Chem. 2015, 173, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Scherer, H. Sulphur in crop production—Invited paper. Eur. J. Agron. 2001, 14, 81–111. [Google Scholar] [CrossRef]
- Singh, V.; Rathore, S.S. Effect of applied potassium and sulphur on yield, oil content and their uptake by linseed. J. Pot. Res. 1994, 10, 407–410. [Google Scholar]
- Razmjoo, K.; Henderlong, P.R. Effect of potassium, sulfur, boron, and molybdenum fertilization on alfalfa production and herbage macronutrient contents. J. Plant Nutr. 1997, 20, 1681–1696. [Google Scholar] [CrossRef]
- Umar, S.; Debnath, G.; Bansal, S.K. Groundnut pod yield and leaf spot disease as affected by potassium and sulphur nutrition. Ind. J. Plant Physiol. 1997, 2, 59–64. [Google Scholar]
- Babhulkar, P.S.; Kar, D.; Badole, W.P.; Balpande, S.S. Effect of sulphur and zinc on yield, quality and nutrient uptake by safflower in Vertisol. J. Lnd. Soc. Soil Sci. 2000, 48, 541–543. [Google Scholar]
- Ichikawa, Y.; Hayami, H.; Sugiyama, T.; Amann, M.; Schöpp, W. Forecast of Sulfur Deposition in Japan for Various Energy Supply and Emission Control Scenarios. Water Air Soil Pollut. 2001, 130, 301–306. [Google Scholar] [CrossRef]
- Messick, D.L.; Fan, M.X.; De Brey, C. Global sulfur requirement and sulfur fertilizers. FAL Agric. Res. 2005, 283, 97–104. [Google Scholar]
- Baqir, H.A.; Zeboon, N.H.; Al-behadili, A.A.J. The role and importance of amino acids within plants: A review. Plant Arch. 2019, 19 (Suppl. 2), 1402–1410. [Google Scholar]
- Park, C.H.; Yeo, H.J.; Kim, Y.J.; Nguyen, B.V.; Park, Y.E.; Sathasivam, R.; Kim, J.K.; Park, S.U. Profiles of secondary metabolites (phenolic acids, carotenoids anthocyanins, and galantamine) and primary metabolites (carbohydrates, amino acids, and organic acids) during flower development in Lycoris radiata. Biomolecules 2021, 11, 248. [Google Scholar] [CrossRef] [PubMed]
- Borghi, M.; Fernie, A.R. Floral Metabolism of Sugars and Amino Acids: Implications for Pollinators’ Preferences and Seed and Fruit Set. Plant Physiol. 2017, 175, 1510–1524. [Google Scholar] [CrossRef]
- Gaufichon, L.; Marmagne, A.; Belcram, K.; Yoneyama, T.; Sakakibara, Y.; Hase, T.; Grandjean, O.; Clément, G.; Citerne, S.; Boutet-Mercey, S.; et al. ASN1-encoded asparagine synthetase in floral organs contributes to nitrogen filling in Arabidopsis seeds. Plant J. 2017, 91, 371–393. [Google Scholar] [CrossRef] [PubMed]
- Le, B.H.; Cheng, C.; Bui, A.Q.; Wagmaister, J.A.; Henry, K.F.; Pelletier, J.; Kwong, L.; Belmonte, M.; Kirkbride, R.; Horvath, S.; et al. Global analysis of gene activity during Arabidopsis seed development and identification of seed-specific transcription factors. Proc. Natl. Acad. Sci. USA 2010, 107, 8063–8070. [Google Scholar] [CrossRef]
- Guan, M.; Møller, I.S.; Schjoerring, J.K. Two cytosolic glutamine synthetase isoforms play specific roles for seed germination and seed yield structure in Arabidopsis. J. Exp. Bot. 2014, 66, 203–212. [Google Scholar] [CrossRef]
- Bernsdorff, F.; Doering, A.-C.; Gruner, K.; Schuck, S.; Braeutigam, A.; Zeier, J. Pipecolic acid orchestrates plant systemic acquired resistance and defense priming via salicylic acid-dependent and -independent pathways. Plant Cell 2016, 28, 102–129. [Google Scholar] [CrossRef]
- Arruda, P.; Barreto, P. Lysine Catabolism Through the Saccharopine Pathway: Enzymes and Intermediates Involved in Plant Responses to Abiotic and Biotic Stress. Front. Plant Sci. 2020, 11, 587. [Google Scholar] [CrossRef]
- Wang, W.; Xu, M.; Wang, G.; Galili, G. New insights into the metabolism of aspartate-family amino acids in plant seeds. Plant Reprod. 2018, 31, 203–211. [Google Scholar] [CrossRef]
- Batista-Silva, W.; Heinemann, B.; Rugen, N.; Nunes-Nesi, A.; Araújo, W.L.; Braun, H.-P.; Hildebrandt, T.M. The role of amino acid metabolism during abiotic stress release. Plant Cell Environ. 2019, 42, 1630–1644. [Google Scholar] [CrossRef]
- Rouhier, K.A.; Perrett, M.E.; Goldfarb, M.H.; Reinhart, P.E.; Wilczewski-Shirai, K.C.; Raymond-Schmidt, L. Isoleucine serves as a precursor for the synthesis of β-alanine in plants. FASEB J. 2019, 33, 486. [Google Scholar] [CrossRef]
- Townsend, D.M.; Tew, K.D.; Tapiero, H. Sulfur containing amino acids and human disease. Biomed. Pharmacother. 2004, 58, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Kandi, A.A.; Sharief, A.E.M.; Seadh, S.E.; Altai, D.S.K. Role of humic acid and amino acids in limiting loss of nitrogen fertilizer and increasing productivity of some wheat cultivars grown under newly reclaimed sandy soil. Int. J. Adv. Res. Biol. Sci. 2016, 3, 123–136. [Google Scholar]
- Biancucci, M.; Mattioli, R.; Forlani, G.; Funck, D.; Costantino, P.; Trovato, M. Role of proline and GABA in sexual reproduction of angiosperms. Front. Plant Sci. 2015, 6, 680. [Google Scholar] [CrossRef] [PubMed]
- Chiang, H.-H.; Dandekar, A. Regulation of proline accumulation in Arabidopsis thaliana (L.) Heynh during development and in response to desiccation. Plant Cell Environ. 1995, 18, 1280–1290. [Google Scholar] [CrossRef]
- Carter, C.; Shafir, S.; Yehonatan, L.; Palmer, R.G.; Thornburg, R. A novel role for proline in plant floral nectars. Sci. Nat. 2006, 93, 72–79. [Google Scholar] [CrossRef]
- Koslowsky, S.; Riegler, H.; Bergmüller, E.; Zrenner, R. Higher biomass accumulation by increasing phosphoribosylpyrophosphate synthetase activity in Arabidopsis thaliana and Nicotiana tabacum. Plant Biotechnol. J. 2008, 6, 281–294. [Google Scholar] [CrossRef]
- Doyle, S.M.; Rigal, A.; Grones, P.; Karady, M.; Barange, D.K.; Majda, M.; Pařízková, B.; Karampelias, M.; Zwiewka, M.; Pěnčík, A.; et al. A role for the auxin precursor anthranilic acid in root gravitropism via regulation of PIN-FORMED protein polarity and relocalisation in Arabidopsis. New Phytol. 2019, 223, 1420–1432. [Google Scholar] [CrossRef]
- Hozayn, M.; Abd El-Monem, A.A. Alleviation of the potential impact of climate change on wheat productivity using arginine under irrigated Egyptian agriculture. Option Mediterr. 2010, 95, 95–100. Available online: http://om.ciheam.org/article.php?IDPDF=801332 (accessed on 18 April 2023).
- Xing, A.; Last, R.L. A Regulatory Hierarchy of the Arabidopsis Branched-Chain Amino Acid Metabolic Network. Plant Cell 2017, 29, 1480–1499. [Google Scholar] [CrossRef]
- Parthasarathy, A.; Savka, M.A.; Hudson, A.O. The Synthesis and Role of β-Alanine in Plants. Front. Plant Sci. 2019, 10, 921. [Google Scholar] [CrossRef]
- Tzin, V.; Galili, G. New Insights into the Shikimate and Aromatic Amino Acids Biosynthesis Pathways in Plants. Mol. Plant 2010, 3, 956–972. [Google Scholar] [CrossRef]
- Schenck, C.A.; Maeda, H.A. Tyrosine biosynthesis, metabolism, and catabolism in plants. Phytochemistry 2018, 149, 82–102. [Google Scholar] [CrossRef] [PubMed]
- Hildebrandt, T.M.; Nesi, A.N.; Araújo, W.L.; Braun, H.-P. Amino Acid Catabolism in Plants. Mol. Plant 2015, 8, 1563–1579. [Google Scholar] [CrossRef] [PubMed]
- Datta, A.; Bhattacharyya, D.; Singh, S.; Ghosh, A.; Schmidtchen, A.; Malmsten, M.; Bhunia, A. Role of Aromatic Amino Acids in Lipopolysaccharide and Membrane Interactions of Antimicrobial Peptides for Use in Plant Disease Control. J. Biol. Chem. 2016, 291, 13301–13317. [Google Scholar] [CrossRef] [PubMed]
- Dar, S.A.; Rather, B.A.; Wani, A.R.; Ganie, M.A. Chemical Science Review and Letters Resistance against Insect Pests by Plant Phenolics and their Derivative Compounds. Chem. Sci. Rev. Lett. 2017, 6, 1941–1949. [Google Scholar]
- De la Rosa, L.A.; Moreno-Escamilla, J.O.; Rodrigo-García, J.; Alvarez-Parrilla, E. Phenolic compounds. In Postharvest Physiology and Biochemistry of Fruits and Vegetables; Yahia, E., Carrillo-Lopez, A., Eds.; Woodhead Publishing: Sawston, UK; Elsevier: Amsterdam, The Netherlands, 2018; pp. 253–271. ISBN 9780128132784. [Google Scholar]
- Clé, C.; Hill, L.M.; Niggeweg, R.; Martin, C.R.; Guisez, Y.; Prinsen, E.; Jansen, M.A. Modulation of chlorogenic acid biosynthesis in Solanum lycopersicum; consequences for phenolic accumulation and UV-tolerance. Phytochemistry 2008, 69, 2149–2156. [Google Scholar] [CrossRef]
- Srinivasan, M.; Sudheer, A.R.; Menon, V.P. Ferulic acid: Therapeutic potential through its antioxidant property. J. Clin. Biochem. Nutr. 2007, 40, 92–100. [Google Scholar] [CrossRef]
- Shahidi, F.; Yeo, J. Bioactivities of Phenolics by Focusing on Suppression of Chronic Diseases: A Review. Int. J. Mol. Sci. 2018, 19, 1573. [Google Scholar] [CrossRef]
- Hawkesford, M.J.; DE Kok, L.J. Managing sulphur metabolism in plants. Plant Cell Environ. 2006, 29, 382–395. [Google Scholar] [CrossRef]
- Fontecave, M.; Atta, M.; Mulliez, E. S-adenosylmethionine: Nothing goes to waste. Trends Biochem. Sci. 2004, 29, 243–249. [Google Scholar] [CrossRef]
- Roje, S. S-Adenosyl-l-methionine: Beyond the universal methyl group donor. Phytochemistry 2006, 67, 1686–1698. [Google Scholar] [CrossRef]
- Badiaa, O.; Yssaad, H.A.R.; Topcuoglu, B. Effect of heavy metals (copper and zinc) on proline, polyphenols and flavonoids content of tomato (Lycopersicon esculentum MILL.). Plant Arch. 2020, 20, 2125–2137. [Google Scholar]
- Bazl, S.; Dashti, F.; Delshad, M. Effect of different levels of sulpur and selenium on some morphological and antioxidant properties on onion (Allium cepa L.) cv. Germez Azarshahr. Iran. J. Hortic. Sci. 2017, 48, 623–633. [Google Scholar] [CrossRef]
- Skrypnik, L.; Novikova, A.; Tokupova, E. Improvement of Phenolic Compounds, Essential Oil Content and Antioxidant Properties of Sweet Basil (Ocimum basilicum L.) Depending on Type and Concentration of Selenium Application. Plants 2019, 8, 458. [Google Scholar] [CrossRef]
- Chomchan, R.; Siripongvutikorn, S.; Puttarak, P.; Rattanapon, R. Influence of selenium bio-fortification on nutritional compositions, bioactive compounds content and anti-oxidative properties of young ricegrass (Oryza sativa L.). Funct. Foods Health Dis. 2017, 7, 195. [Google Scholar] [CrossRef]
- Li, L.; Yu, J.; Yuan, H.; Zha, S.; Deng, K.; Xiao, X.; Luo, Y.; Cheng, S.; Cheng, H. High-Density Kinetic Analysis of the Metabolomic and Transcriptomic Response of Ginkgo biloba Flavonoids Biosynthesis to Selenium Treatments. Not. Bot. Horti Agrobot. Cluj Napoca 2019, 47, 792–803. [Google Scholar] [CrossRef]
- Wildermuth, M.C. Variations on a theme: Synthesis and modification of plant benzoic acids. Curr. Opin. Plant Biol. 2006, 9, 288–296. [Google Scholar] [CrossRef] [PubMed]
- Widhalm, J.R.; Dudareva, N. A Familiar Ring to It: Biosynthesis of Plant Benzoic Acids. Mol. Plant 2015, 8, 83–97. [Google Scholar] [CrossRef] [PubMed]
- Barch, D.H.; Rundhaugen, L.M.; Stoner, G.D.; Pillay, N.; Rosche, W.A. Structure-function relationships of the dietary anticarcinogen ellagic acid. Carcinogenesis 1996, 17, 265–269. [Google Scholar] [CrossRef]
- Pari, L.; Sivasankari, R. Effect of ellagic acid on cyclosporine A-induced oxidative damage in the liver of rats. Fundam. Clin. Pharmacol. 2008, 22, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Cozzi, R.; Ricordy, R.; Bartolini, F.; Ramadori, L.; Perticone, P.; De Salvia, R. Taurine and ellagic acid: Two differently-acting natural antioxidants. Environ. Mol. Mutagen. 1995, 26, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Zeng, L.; Chen, Y.; Wang, X.; Liao, Y.; Xiao, Y.; Fu, X.; Yang, Z. Metabolism of Gallic Acid and Its Distributions in Tea (Camellia sinensis) Plants at the Tissue and Subcellular Levels. Int. J. Mol. Sci. 2020, 21, 5684. [Google Scholar] [CrossRef] [PubMed]
- Karamaæ, M.; Kosinska, A.; Pegg, R.B. Comparison of radical–scavenging activities of selected phenolic acids. Pol. J. Food Nutr. Sci. 2005, 14, 165–170. [Google Scholar]
- Abe, T.; Masai, E.; Miyauchi, K.; Katayama, Y.; Fukuda, M. Tetrahydrofolate- Dependent O-Demethylase, LigM, Is Crucial for Catabolism of Vanillate and Syringate in Sphingomonas paucimobilis SYK-6. J. Bacteriol. 2005, 187, 2030–2037. [Google Scholar] [CrossRef]
- El-Beltagi, H.S.; Mohamed, H.I.; Abdelazeem, A.S.; Youssef, R.; Safwat, G. GC-MS Analysis, Antioxidant, Antimicrobial and Anticancer Activities of Extracts from Ficus sycomorus Fruits and Leaves. Not. Bot. Horti Agrobot. Cluj Napoca 2019, 47, 493–505. [Google Scholar] [CrossRef]
- Kundu, A.; Vadassery, J. Chlorogenic acid-mediated chemical defence of plants against insect herbivores. Plant Biol. 2019, 21, 185–189. [Google Scholar] [CrossRef]
- Zawoznik, M.; Garrido, L.M.; Del Pero Martinez, M.A.; Tomaro, M.L. Occurrence and role of Vanillin in root exudates of Peanut (Arachis hypogaea). Symbiosis 2004, 36, 257–268. Available online: http://hdl.handle.net/10222/78056 (accessed on 18 April 2023).
- Agati, G.; Azzarello, E.; Pollastri, S.; Tattini, M. Flavonoids as antioxidants in plants: Location and functional significance. Plant Sci. 2012, 196, 67–76. [Google Scholar] [CrossRef]
- Davies, K.M.; Albert, N.W.; Schwinn, K.E. From landing lights to mimicry: The molecular regulation of flower colouration and mechanisms for pigmentation patterning. Funct. Plant Biol. 2012, 39, 619–638. [Google Scholar] [CrossRef]
- Amić, D.; Davidović-Amić, D.; Bešlo, D.; Trinajstić, N. Structure-radical scavenging activity relationships of flavonoids. Croat. Chem. Acta 2003, 76, 55–61. [Google Scholar]
- Terao, J.; Piskula, M.K.; Yao, Q. Protective Effect of Epicatechin, Epicatechin Gallate, and Quercetin on Lipid Peroxidation in Phospholipid Bilayers. Arch. Biochem. Biophys. 1994, 308, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Ryan, K.G.; Swinny, E.E.; Winefield, C.; Markham, K.R. Flavonoids and UV Photoprotection in Arabidopsis Mutants. Z. Naturforschung. Sect. C 2001, 56, 745–754. [Google Scholar] [CrossRef] [PubMed]
- Davies, K.M.; Jibran, R.; Zhou, Y.; Albert, N.W.; Brummell, D.A.; Jordan, B.R.; Bowman, J.L.; Schwinn, K.E. The Evolution of Flavonoid Biosynthesis: A Bryophyte Perspective. Front. Plant Sci. 2020, 11, 7. [Google Scholar] [CrossRef]
- Kianersi, F.; Abdollahi, M.R.; Mirzaie-Asl, A.; Dastan, D.; Rasheed, F. Identification and tissue-specific expression of rutin biosynthetic pathway genes in Capparis spinosa elicited with salicylic acid and methyl jasmonate. Sci. Rep. 2020, 10, 8884. [Google Scholar] [CrossRef]
- Fini, A.; Brunetti, C.; Di Ferdinando, M.; Ferrini, F.; Tattini, M. Stress-induced flavonoid biosynthesis and the antioxidant machinery of plants. Plant Signal. Behav. 2011, 6, 709–711. [Google Scholar] [CrossRef]
- Hong, G.; Wang, J.; Hochstetter, D.; Gao, Y.; Xu, P.; Wang, Y. Epigallocatechin-3-gallate functions as a physiological regulator by modulating the jasmonic acid pathway. Physiol. Plant. 2015, 153, 432–439. [Google Scholar] [CrossRef]
- Afify, A.M.R.; El-Beltagi, H.S.; Aly, A.A.; El-Ansary, A.E. Antioxidant enzyme activities and lipid peroxidation as biomarker for potato tuber stored by two essential oils from Caraway and Clove and its main component carvone and eugenol. Asian Pac. J. Trop. Biomed. 2012, 2, S772–S780. [Google Scholar] [CrossRef]
- Ramadan, K.M.A.; El-Beltagi, H.S.; Bendary, E.S.A.; Ali, H.M. Experimental evaluation of the antioxidant and antitumor activities of thyme and basil essential oils and their phenolic constituents: Theoretical antioxidant evaluation. Chem. Biol. Technol. Agric. 2022, 9, 102. [Google Scholar] [CrossRef]
- El-Beltagi, H.S.; Dhawi, F.; Ashoush, I.S.; Ramadan, K. Antioxidant, anti-cancer and ameliorative activities of Spirulina platensis and pomegranate juice against hepatic damage induced by CCl4. Not. Bot. Horti Agrobot. Cluj Napoca 2020, 48, 1941–1956. [Google Scholar] [CrossRef]
- Ramadan, K.M.A.; El-Beltagi, H.S.; Mohamed, H.I.; Shalaby, T.A.; Galal, A.; Mansour, A.T.; Fotouh, M.M.A.; Bendary, E.S.A. Antioxidant, Anti-Cancer Activity and Phytochemicals Profiling of Kigelia pinnata Fruits. Separations 2022, 9, 379. [Google Scholar] [CrossRef]
- Moctezuma, C.; Hammerbacher, A.; Heil, M.; Gershenzon, J.; Méndez-Alonzo, R.; Oyama, K. Specific Polyphenols and Tannins are Associated with Defense Against Insect Herbivores in the Tropical Oak Quercus oleoides. J. Chem. Ecol. 2014, 40, 458–467. [Google Scholar] [CrossRef] [PubMed]
- Sohn, S.I.; Pandian, S.; Oh, Y.J.; Kang, H.J.; Cho, W.S.; Cho, Y.S. Metabolic Engineering of Isoflavones: An Updated Overview. Front. Plant Sci. 2021, 12, 670103. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, A.A.; Sameeh, M.Y.; El-Beltagi, H.S. Preparation of Seaweed Nanopowder Particles Using Planetary Ball Milling and Their Effects on Some Secondary Metabolites in Date Palm (Phoenix dactylifera L.) Seedlings. Life 2023, 13, 39. [Google Scholar] [CrossRef]
- Gutiérrez-Grijalva, E.P.; López-Martínez, L.X.; Contreras-Angulo, L.A.; Elizalde-Romero, C.A.; Heredia, J.B. Plant Alkaloids: Structures and Bioactive Properties. In Plant-Derived Bioactives: Chemistry and Mode of Action; Swamy, M.K., Ed.; Springer: Singapore, 2020; pp. 85–117. ISBN 978-981-15-2361-8. [Google Scholar]
- Stander, E.A.; Sepúlveda, L.J.; de Bernonville, T.D.; Carqueijeiro, I.; Koudounas, K.; Cruz, P.L.; Besseau, S.; Lanoue, A.; Papon, N.; Giglioli-Guivarc’h, N.; et al. Identifying Genes Involved in Alkaloid Biosynthesis in Vinca minor through Transcriptomics and Gene Co-Expression Analysis. Biomolecules 2020, 10, 1595. [Google Scholar] [CrossRef]
- Sottomayor, M.; López-Serrano, M.; DiCosmo, F.; Barceló, A.R. Purification and characterization of α-3′,4′-anhydrovinblastine synthase (peroxidase-like) from Catharanthus roseus (L.) G. Don. FEBS Lett. 1998, 428, 299–303. [Google Scholar] [CrossRef]
- Yu, R.; Zhu, J.; Wang, M.; Wen, W. Biosynthesis and regulation of terpenoid indole alkaloids in Catharanthus roseus. Pharmacogn. Rev. 2015, 9, 24–28. [Google Scholar] [CrossRef]
- Mimmo, T.; Tiziani, R.; Valentinuzzi, F.; Lucini, L.; Nicoletto, C.; Sambo, P.; Scampicchio, M.; Pii, Y.; Cesco, S. Selenium Biofortification in Fragaria × ananassa: Implications on Strawberry Fruits Quality, Content of Bioactive Health Beneficial Compounds and Metabolomic Profile. Front. Plant Sci. 2017, 8, 1887. [Google Scholar] [CrossRef]
- Kalaei, M.H.R.; Abdossi, V.; Danaee, E. Evaluation of foliar application of selenium and flowering stages on selected properties of Iranian Borage as a medicinal plant. Sci. Rep. 2022, 12, 12568. [Google Scholar] [CrossRef]
- Vázquez-Flota, F.; Hernandez-Dominguez, E.; de Lourdes Miranda-Ham, M.; Monforte-González, M. A differential response to chemical elicitors in Catharanthus roseus in vitro cultures. Biotechnol. Lett. 2009, 31, 591–595. [Google Scholar] [CrossRef]
- Rijhwani, S.K.; Shanks, J.V. Effect of Elicitor Dosage and Exposure Time on Biosynthesis of Indole Alkaloids by Catharanthus roseus Hairy Root Cultures. Biotechnol. Prog. 1998, 14, 442–449. [Google Scholar] [CrossRef] [PubMed]
- Martino, E.; Casamassima, G.; Castiglione, S.; Cellupica, E.; Pantalone, S.; Papagni, F.; Rui, M.; Siciliano, A.M.; Collina, S. Vinca alkaloids and analogues as anti-cancer agents: Looking back, peering ahead. Bioorg. Med. Chem. Lett. 2018, 28, 2816–2826. [Google Scholar] [CrossRef] [PubMed]
- Carlson, K.-S.; Ocean, A.J. Peripheral Neuropathy with Microtubule-Targeting Agents: Occurrence and Management Approach. Clin. Breast Cancer 2011, 11, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Triarico, S.; Romano, A.; Attinà, G.; Capozza, M.A.; Maurizi, P.; Mastrangelo, S.; Ruggiero, A. Vincristine-Induced Peripheral Neuropathy (VIPN) in Pediatric Tumors: Mechanisms, Risk Factors, Strategies of Prevention and Treatment. Int. J. Mol. Sci. 2021, 22, 4112. [Google Scholar] [CrossRef]
- Duca, A.; Sturza, A.; Moacă, E.-A.; Negrea, M.; Lalescu, V.-D.; Lungeanu, D.; Dehelean, C.-A.; Muntean, D.-M.; Alexa, E. Identification of Resveratrol as Bioactive Compound of Propolis from Western Romania and Characterization of Phenolic Profile and Antioxidant Activity of Ethanolic Extracts. Molecules 2019, 24, 3368. [Google Scholar] [CrossRef]
- Marinova, D.; Ribarova, F.; Atanassova, M. Total phenolic and total flavonoids in bulgarian fruits and vegetables. J. Univ. Chem. Tech. Metall. 2005, 40, 255–260. [Google Scholar]
- Jayaraman, J. Laboratory Manual in Biochemistry; Wiley Eastern Ltd.: New Delhi, India, 1985; p. 107. [Google Scholar]
- Shamsa, F.; Monsef, H.; Ghamooshi, R.; Verdian-rizi, M. Spectrophotometric determination of total alkaloids in some Iranian medicinal plants. Thai J. Pharm. Sci. 2008, 32, 17–20. [Google Scholar]
- Sumanta, N.; Haque, C.I.; Nishika, J.; Suprakash, R. Spectrophotometric analysis of chlorophylls and carotenoids from commonly grown fern species by using various extracting solvents. Res. J. Chem. Sci. 2014, 4, 63–69. [Google Scholar]
- Giusti, M.M.; Wrolstad, R.E. Characterization and Measurement of Anthocyanins by UV-Visible Spectroscopy. Curr. Protoc. Food Anal. Chem. 2001, 1042, F1.2.1–F1.2.13. [Google Scholar] [CrossRef]
- Cottenie, A.; Verloo, M.; Kiekens, L.; Velgh, G.; Camerlynech, R. Chemical Analysis of Plants and Soils; Article No. 42, 80-284; Laboratory of Analytical Agrochemistry State University: Gent, Belgium, 1982. [Google Scholar]
- A.O.A.C. Official Method of Analysis of Association of Official Analytical Chemists, 12th ed.; Association of Official Analytical Chemists: Washington, DC, USA, 1975. [Google Scholar]
- Benton, J.J. Laboratory Guide for Conducting Soil Test and Plant Analysis; CRC Press: Boca Raton, FL, USA, 2001. [Google Scholar]
- Wang, L.; Xu, R.; Hu, B.; Li, W.; Sun, Y.; Tu, Y.; Zeng, X. Analysis of free amino acids in Chinese teas and flower of tea plant by high performance liquid chromatography combined with solid-phase extraction. Food Chem. 2010, 123, 1259–1266. [Google Scholar] [CrossRef]
- Henderson, J.; Brooks, A. Improved Amino Acid Methods Using Agilent ZORBAX Eclipse Plus C18 Columns for a Variety of Agilent LC Instrumentation and Separation Goals; Agilent Technologies Inc: Wilmington, DE, USA, 2010; Available online: https://www.agilent.com/cs/library/applications/5990-4547EN (accessed on 18 April 2023).
- Gökbulut, A. Validated RP-HPLC Method for Quantification of Phenolic Compounds in Methanol Extracts of Aerial Parts and Roots of Thymus sipyleus and Evaluation of Antioxidant Potential. Trop. J. Pharm. Res. 2015, 14, 1871–1877. [Google Scholar] [CrossRef]
- Liu, Z.; Wu, H.-L.; Li, Y.; Gu, H.-W.; Yin, X.-L.; Xie, L.-X.; Yu, R.-Q. Rapid and simultaneous determination of five vinca alkaloids in Catharanthus roseus and human serum using trilinear component modeling of liquid chromatography–diode array detection data. J Chromatogr B Anal. Technol Biomed Life Sci. 2016, 1026, 114–123. [Google Scholar] [CrossRef] [PubMed]
- SAS. SAS/STAT User’s Guide, Release 6.03 ed.; SAS Institute Inc.: Cary, NC, USA, 1988. [Google Scholar]


| Carrageenan Treatments | TPC | TFC | TAC | FAA |
|---|---|---|---|---|
| μg Gallic/g FW | μg Quercetin/g FW | μg Atropine/g DW | mg Lysine/g FW | |
| 0 | 2476.5 d ± 5.711 | 271.7 d ± 1.815 | 1359 d ± 2.645 | 132.05 c ± 4.173 |
| 400 ppm | 3066.4 c ± 1.742 | 466.1 c ± 2.775 | 2535 c ± 0.577 | 195.57 a ± 3.115 |
| 600 ppm | 3425.6 b ± 3.009 | 663.6 b ± 3.671 | 3064 b ± 4.509 | 138.07 bc ± 3.684 |
| 800 ppm | 3948.6 a ± 3.492 | 754.6 a ± 3.329 | 3518 a ± 0.046 | 145.49 bc ± 1.061 |
| Elements Concentration | Carrageenan Treatments | |||
|---|---|---|---|---|
| 0 ppm | 400 ppm | 600 ppm | 800 ppm | |
| N (mg/Kg DW) | 15.58 b ± 0.196 | 13.36 d ± 0.213 | 14.66 c ± 0.125 | 16.42 a ± 0.1457 |
| P (g/100 g DW) | 0.063 a ± 0.000 | 0.066 a ± 0.001 | 0.060 a ± 0.000 | 0.067 a ± 0.0002 |
| K (g/100 g DW) | 0.29 d ± 0.004 | 0.33 b ± 0.009 | 1.38 a ± 0.010 | 1.23 c ± 0.0059 |
| Ca (g/100 g DW) | 0.25 c ± 0.003 | 1.54 b ± 0.002 | 1.65 a ± 0.042 | 1.63 a ± 0.0295 |
| Mn (mg/Kg DW) | 76.98 c ± 0.027 | 46.14 d ± 0.380 | 154.56 a ± 0.169 | 128.79 b ± 0.7794 |
| Cu (mg/Kg DW) | 7.48 c ± 0.397 | 78.79 a ± 0.417 | 13.92 b± 0.4769 | 12.50 b ± 0.4392 |
| Zn (mg/Kg DW) | 15.00 c ± 0.987 | 54.22 a ± 0.129 | 43.80 b ± 0.2810 | 43.96 b ± 0.8215 |
| Se (mg/Kg DW) | 32.43 d ± 0.282 | 117.17 a ± 0.075 | 92.46 b ± 1.0016 | 85.40 c ± 0.0139 |
| Amino Acids (mg/g FW) | Rt (min) | Carrageenan Treatments | ||||
|---|---|---|---|---|---|---|
| 0 ppm | 400 ppm | 600 ppm | 800 ppm | |||
| 1 | ASP | 2.973 | 1.55 a ± 0.011 | 1.32 b ± 0.004 | 0.91 c ± 0.003 | 0.78 d ± 0.004 |
| 2 | GLU | 4.721 | 4.01 a ± 0.028 | 1.84 d ± 0.010 | 2.18 c ± 0.027 | 2.78 b ± 0.021 |
| 3 | Serine | 8.391 | 5.41 a ± 0.039 | 5.28 b ± 0.034 | 5.06 c ± 0.041 | 5.27 b ± 0.050 |
| 4 | Histidine | 10.248 | 0.23 c ± 0.001 | 0.25 b ± 0.002 | 0.18 d ± 0.001 | 0.26 a ± 0.001 |
| 5 | Glycine | 10.566 | 1.48 c ± 0.009 | 1.60 bc ± 0.008 | 1.70 ab ± 0.007 | 1.85 a ± 0.005 |
| 6 | Threonine | 10.85 | 4.03 d ± 0.054 | 4.58 b ± 0.050 | 4.45 c ± 0.049 | 6.82 a ± 0.060 |
| 7 | Arginine | 12.621 | 9.21 a ± 0.104 | 3.08 c ± 0.047 | 0.614 d ± 0.003 | 4.32 b ± 0.052 |
| 8 | Alanine | 13.122 | 6.85 a ± 0.056 | 4.14 b ± 0.053 | 2.05 c ± 0.017 | 1.23 d ± 0.009 |
| 9 | Tyrosine | 15.52 | 3.14 a ± 0.066 | 2.48 d ± 0.023 | 3.11 b ± 0.031 | 2.73 c ± 0.048 |
| 10 | Cysteine | 17.349 | ---- | ---- | --- | ---- |
| 11 | Valine | 18.783 | 3.52 d ± 0.017 | 3.73 c ± 0.021 | 4.17 b ± 0.041 | 4.93 a ± 0.050 |
| 12 | Methionine | 19.191 | 4.31 c ± 0.029 | 4.34 c ± 0.051 | 4.58 b ± 0.039 | 5.00 a ± 0.051 |
| 13 | Tryptophan | 20.796 | 1.06 d ± 0.008 | 1.11 c ± 0.009 | 1.43 b ± 0.011 | 2.44 a ± 0.014 |
| 14 | Phenylalanine | 21.478 | 3.75 d ± 0.022 | 4.28 a ± 0.044 | 3.84 c ± 0.033 | 4.07 b ± 0.035 |
| 15 | IsoLeucine | 21.817 | 2.51 d ± 0.019 | 3.36 a ± 0.023 | 3.06 c ± 0.043 | 3.16 b ± 0.031 |
| 16 | Leucine | 23.018 | 5.28 d ± 0.031 | 6.32 a ± 0.048 | 5.57 c ± 0.047 | 6.23 b ± 0.051 |
| 17 | Lysine | 24.296 | 1.42 a ± 0.006 | 0.25 d ± 0.001 | 0.82 b ± 0.002 | 0.54 c ± 0.001 |
| 18 | Proline | 29.288 | 56.45 d ± 1.025 | 59.82 c ± 1.030 | 62.98 b ± 1.007 | 65.28 a ± 1.072 |
| Phenolic Compounds (mg/g FW) | Rt min | 0 ppm | 400 ppm | 600 ppm | 800 ppm | |
|---|---|---|---|---|---|---|
| 1 | Gallic acid | 3.526 | 0.93 a ± 0.004 | 0.71 b ± 0.003 | 0.72 b ± 0.003 | 0.72 b ± 0.004 |
| 2 | Chlorogenic acid | 4.311 | 11.18 d ± 0.521 | 15.47 c ± 0.601 | 17.92 b ± 0.624 | 18.97 a ± 0.671 |
| 3 | catechin | 4.622 | --- | --- | --- | --- |
| 4 | Methyl gallate | 5.401 | 3.58 b ± 0.021 | 5.16 a ± 0.033 | 2.65 c ± 0.012 | 2.19 d ± 0.012 |
| 5 | Caffeic acid | 5.8820 | 0.64 a ± 0.003 | 0.55 b ± 0.004 | 0.48 c ± 0.002 | 0.40 d ± 0.003 |
| 6 | Syringic acid | 6.611 | 3.13 d ± 0.014 | 4.98 a ± 0.021 | 4.11 c ± 0.024 | 4.26 b ± 0.026 |
| 7 | pyrocatechol | 6.791 | --- | --- | --- | --- |
| 8 | Rutin | 7.839 | 0.33 b ± 0.001 | 1.11 a ± 0.009 | 0.19 c ± 0.001 | 0.20 c ± 0.001 |
| 9 | Ellagic acid | 9.036 | --- | 0.89 a ± 0.002 | 0.30 b ± 0.001 | --- |
| 10 | Coumaric acid | 9.074 | 0.15 c ± 0.001 | 0.15 c ± 0.001 | 0.18 b ± 0.002 | 0.22 a ± 0.002 |
| 11 | Vanillin | 9.774 | 0.10 b ± 0.001 | 0.22 a ± 0.002 | 0.12 b ± 0.001 | 0.23 a ± 0.002 |
| 12 | Ferulic acid | 10.292 | 0.23 d ± 0.001 | 0.30 c ± 0.002 | 0.49 b ± 0.002 | 0.74 a ± 0.004 |
| 13 | Naringenin | 10.587 | 0.11 c ± 0.001 | 0.14 c ± 0.001 | 0.27 b ± 0.001 | 0.30 a ± 0.001 |
| 14 | Daidzein | 12.12 | 0.07 d ± 0.001 | 1.38 c ± 0.025 | 1.59 b ± 0.027 | 1.88 a ± 0.030 |
| 15 | Querectin | 12.545 | 1.10 d ± 0.027 | 1.97 c ± 0.030 | 3.10 b ± 0.040 | 3.73 a ± 0.043 |
| 16 | Cinnamic acid | 14.014 | 0.02 c ± 0.001 | 0.06 c ± 0.001 | 0.16 b ± 0.001 | 0.24 a ± 0.001 |
| 17 | Apigenin | 14.514 | 0.03 a ± 0.001 | --- | --- | --- |
| 18 | kaempferol | 15.024 | --- | --- | --- | --- |
| 19 | hesperetin | 15.606 | --- | --- | --- | --- |
| Alkaloid Compounds μg/g | Treatments | |||
|---|---|---|---|---|
| 0 | 400 ppm | 600 ppm | 800 ppm | |
| vincamine | 24.911 | 12.043 | 16.227 | 18.167 |
| catharanthine | 9.878 | 1.559 | 1.662 | 10.157 |
| Vincracine (Vincristine) | - | 6.574 | 9.129 | 13.522 |
| vinblastine | - | - | - | - |
| Total | 34.789 | 20.176 | 27.018 | 41.846 |
| Time (Min) | A% | B% |
|---|---|---|
| 0 | 98 | 2 |
| 0.84 | 98 | 2 |
| 33.40 | 43 | 57 |
| 33.50 | 0 | 100 |
| 39.30 | 0 | 100 |
| 39.40 | 98 | 2 |
| 40.0 | 98 | 2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Beltagi, H.S.; El-Sayed, S.M.; Abdelhamid, A.N.; Hassan, K.M.; Elshalakany, W.A.; Nossier, M.I.; Alabdallah, N.M.; Al-Harbi, N.A.; Al-Qahtani, S.M.; Darwish, D.B.E.; et al. Potentiating Biosynthesis of Alkaloids and Polyphenolic Substances in Catharanthus roseus Plant Using ĸ-Carrageenan. Molecules 2023, 28, 3642. https://doi.org/10.3390/molecules28083642
El-Beltagi HS, El-Sayed SM, Abdelhamid AN, Hassan KM, Elshalakany WA, Nossier MI, Alabdallah NM, Al-Harbi NA, Al-Qahtani SM, Darwish DBE, et al. Potentiating Biosynthesis of Alkaloids and Polyphenolic Substances in Catharanthus roseus Plant Using ĸ-Carrageenan. Molecules. 2023; 28(8):3642. https://doi.org/10.3390/molecules28083642
Chicago/Turabian StyleEl-Beltagi, Hossam S., Salwa M. El-Sayed, Ahmed. N. Abdelhamid, Karim. M. Hassan, Walaa. A. Elshalakany, Mona Ibrahim Nossier, Nadiyah M. Alabdallah, Nadi Awad Al-Harbi, Salem Mesfir Al-Qahtani, Doaa Bahaa Eldin Darwish, and et al. 2023. "Potentiating Biosynthesis of Alkaloids and Polyphenolic Substances in Catharanthus roseus Plant Using ĸ-Carrageenan" Molecules 28, no. 8: 3642. https://doi.org/10.3390/molecules28083642
APA StyleEl-Beltagi, H. S., El-Sayed, S. M., Abdelhamid, A. N., Hassan, K. M., Elshalakany, W. A., Nossier, M. I., Alabdallah, N. M., Al-Harbi, N. A., Al-Qahtani, S. M., Darwish, D. B. E., Abbas, Z. K., & Ibrahim, H. A. (2023). Potentiating Biosynthesis of Alkaloids and Polyphenolic Substances in Catharanthus roseus Plant Using ĸ-Carrageenan. Molecules, 28(8), 3642. https://doi.org/10.3390/molecules28083642








