Metabolic Profiling for Unveiling Mechanisms of Kushenol F against Imiquimod-Induced Psoriasis with UHPLC/MS Analysis
Abstract
:1. Introduction
2. Results and Discussion
2.1. KSCF Mitigated the Psoriatic Dermatitis Induced by IMQ
2.2. Effect of KSCF on Inflammatory Reactions in Psoriasis-like Mice
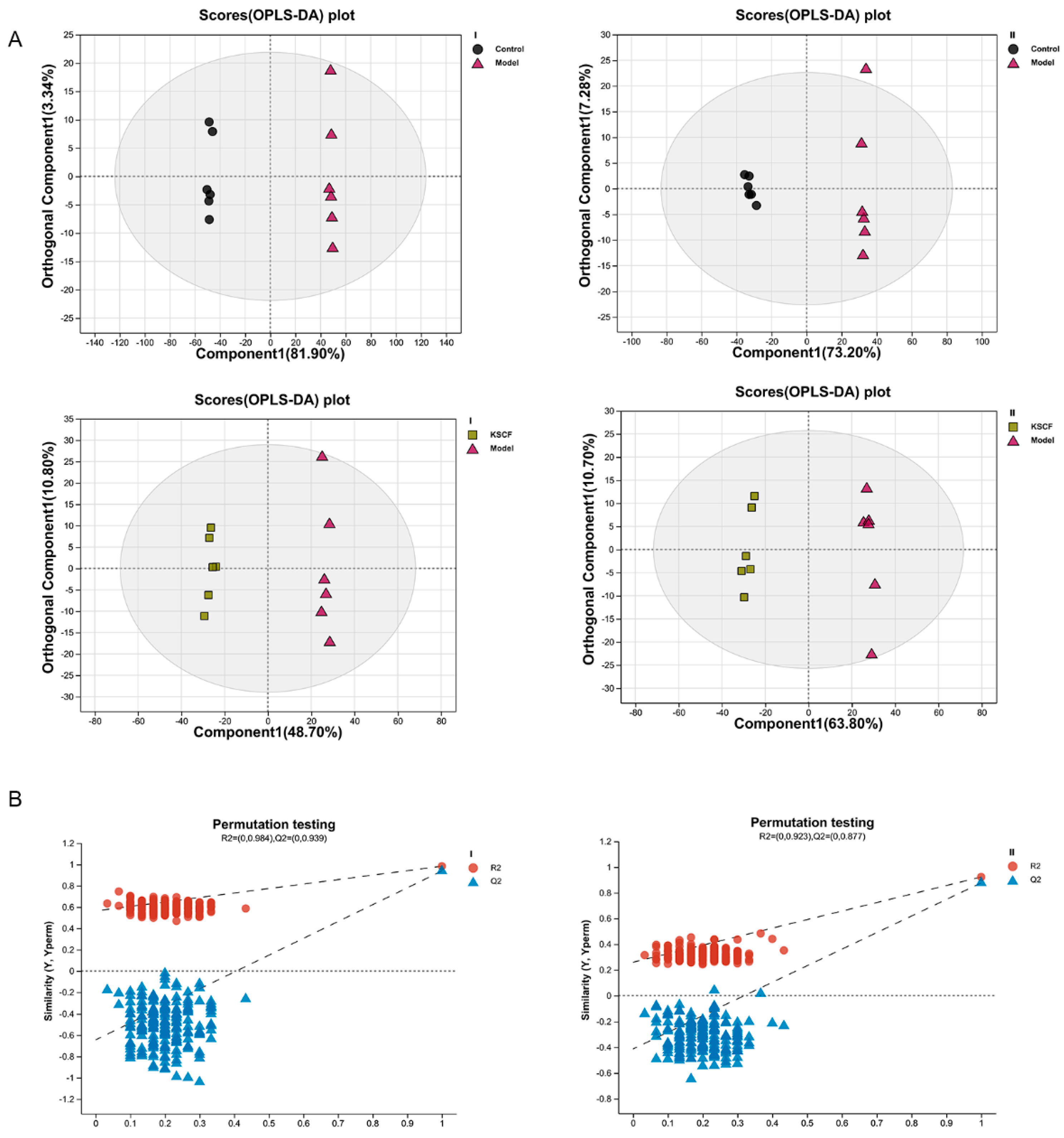
2.3. Effect of KSCF on Metabolites in the Injured Skin of Psoriasis-like Mice
2.4. Identification of Differential Metabolites
2.5. Metabolic Pathway Analysis
3. Materials and Methods
3.1. Chemicals, Reagents, and Materials
3.2. Animals and Experimental Protocol
3.3. Psoriasis Area and Severity Index (PASI) Assessment
3.4. Image Acquisition and Skin Sample Collection
3.5. Histopathology Analyses
3.6. Measurement of Skin Inflammatory Factors
3.7. Metabolomic Analysis
3.7.1. Sample Pre-Treatment
3.7.2. UHPLC/MS Analysis
3.8. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wu, J.J.; Kavanaugh, A.; Lebwohl, M.G.; Gniadecki, R.; Merola, J.F. Psoriasis and metabolic syndrome: Implications for the management and treatment of psoriasis. J. Eur. Acad. Dermatol. Venereol. 2022, 36, 797–806. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, A.W.; Read, C. Pathophysiology, Clinical Presentation, and Treatment of Psoriasis: A Review. JAMA 2020, 19, 1945–1960. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Kim, M. Challenges and future trends in the treatment of psoriasis. Int. J. Mol. Sci. 2023, 24, 13313. [Google Scholar] [CrossRef] [PubMed]
- Tokuyama, M.; Mabuchi, T. New treatment addressing the pathogenesis of psoriasis. Int. J. Mol. Sci. 2020, 21, 7488. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Jin, R.; Hao, J.; Zeng, J.; Yin, D.; Yi, Y.; Zhu, M.; Mandal, A.; Hua, Y.; Ng, C.K.; et al. Consumption of the fish oil high-fat diet uncouples obesity and mammary tumor growth through induction of reactive oxygen species in protumor macrophages. Cancer Res. 2020, 80, 2564–2574. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.; Jin, R.; Zeng, J.; Hua, Y.; Yorek, M.S.; Liu, L.; Mandal, A.; Li, J.; Zheng, H.; Sun, Y.; et al. Consumption of fish oil high-fat diet induces murine hair loss via epidermal fatty acid binding protein in skin macrophages. Cell Rep. 2022, 41, 111804. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.H.; Na, K.M.; Oh, I.; Song, I.H.; Lee, Y.S.; Shin, J.; Kim, T.Y. Kurarinone regulates immune responses through regulation of the JAK/STAT and TCR-mediated signaling pathways. Biochem. Pharmacol. 2013, 85, 1134–1144. [Google Scholar] [CrossRef]
- Griffiths, C.E.M.; Armstrong, A.W.; Gudjonsson, J.E.; Barker, J.N.W.N. Psoriasis. Lancet 2021, 397, 1301–1315. [Google Scholar] [CrossRef] [PubMed]
- Kamata, M.; Tada, Y. Crosstalk: Keratinocytes and immune cells in psoriasis. Front. Immunol. 2023, 14, 1286344. [Google Scholar] [CrossRef]
- Sun, P.; Zhao, W.; Wang, Q.; Chen, L.; Sun, K.; Zhan, Z.; Wang, J. Chemical diversity, biological activities and traditional uses of and important Chinese herb Sophora. Phytomedicine 2022, 100, 154054. [Google Scholar] [CrossRef]
- Zhang, Z.; Pan, J.; Zhu, T.; Malewicz, N.; Ye, K.; Rong, J.; Luo, Y.; Situ, Y.; Verkhratsky, A.; Wang, Y.; et al. Oxymatrine screened from Sophora flavescens by cell membrane immobilized chromatography relieves histamine-independent itch. J. Pharm. Pharmacol. 2021, 73, 1617–1629. [Google Scholar] [CrossRef] [PubMed]
- Sun, P.; Zhao, X.; Zhao, W.; Chen, L.; Liu, X.; Zhan, Z.; Wang, J. Sophora flavescens-Angelica sinensis in the treatment of eczema by inhibiting TLR4/MyD88/NF-κB pathway. J. Ethnopharmacol. 2024, 322, 117626. [Google Scholar] [CrossRef] [PubMed]
- Cha, J.D.; Moon, S.E.; Kim, J.Y.; Jung, E.K.; Lee, Y.S. Antibacterial activity of sophoraflavanone G isolated from the roots of Sophora flavescens against methicillin-resistant staphylococcus aureus. Phytother. Res. 2009, 23, 1326–1331. [Google Scholar] [CrossRef]
- Li, Z.Y.; Huang, W.C.; Tu, R.S.; Gu, P.Y.; Lin, C.F.; Liou, C.J. Sophoraflavanone G induces apoptosis in human leukemia cells and blocks MAPK activation. Am. J. Chin. Med. 2016, 44, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wei, S.; Marabada, D.; Wang, Z.; Huang, Q. Research progress of natural matrine compounds and synthetic matrine derivatives. Molecules 2023, 31, 5780. [Google Scholar] [CrossRef] [PubMed]
- Tashiro, T.; Sawada, Y. Psoriasis and systemic inflammatory disorders. Int. J. Mol. Sci. 2022, 23, 4457. [Google Scholar] [CrossRef]
- Guo, L.; Jin, H. Research progress of metabolomics in psoriasis. Chin. Med. J. 2023, 136, 1805–1816. [Google Scholar] [CrossRef] [PubMed]
- Kanda, N. Psoriasis: Pathogenesis, comorbidities, and therapy updated. Int. J. Mol. Sci. 2021, 22, 2979. [Google Scholar] [CrossRef]
- Gao, J.; Chen, F.; Fang, H.; Mi, J.; Qi, Q.; Yang, M. Daphnetin inhibits proliferation and inflammatory response in human HaCaT keratinocytes and ameliorates imiquimod-induced psoriasis-like skin lesion in mice. Biol. Res. 2020, 53, 48. [Google Scholar] [CrossRef]
- Fletcher, J.M.; Moran, B.; Petrasca, A.; Smith, C.M. IL-17 in inflammatory skin diseases psoriasis and hidradenitis suppurativa. Clin. Exp. Immunol. 2020, 201, 121–134. [Google Scholar] [CrossRef]
- Elpa, D.P.; Chiu, H.Y.; Wu, S.P.; Urban, P.L. Skin metabolomics. Trends Endocrinol. Metab. 2021, 32, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Qi, C.; Feng, F.; Wang, Y.; Di, T.; Meng, Y.; Wang, Y.; Zhao, N.; Zhang, X.; Li, P.; et al. Combining network pharmacology, RNA-seq, and metabolomics strategies to reveal the mechanism of cimicifugae rhizome—Smilax glabra roxb herb pair for the treatment of psoriasis. Phytomedicine 2022, 105, 154384. [Google Scholar] [CrossRef]
- Bocheńska, K.; Gabig-Cimińska, M. Unbalanced sphingolipid metabolism and its implications for the pathogenesis of psoriasis. Molecules 2020, 25, 1130. [Google Scholar] [CrossRef] [PubMed]
- Matwiejuk, M.; Mysliwiec, H.; Chabowski, A.; Flisiak, I. The role of sphingolipids in the pathogenesis of psoriasis. Metabolites 2022, 12, 1171. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Yang, Q.; Zheng, J.; Gu, H.; Chen, K.; Jin, H.; He, C.; Xu, A.E.; Xu, J.; Zhang, J.L.; et al. Efficacy and safety of a topical moisturizer containing linoleic acid and ceramide for mild-to-moderate psoriasis vulgaris: A multicenter randomized controlled trial. Dermatol. Ther. 2023, 33, e14263. [Google Scholar] [CrossRef]
- Slominski, A.; Zbytek, B.; Nikolakis, G.; Manna, P.R.; Skobowiat, C.; Zmijewski, M.; Li, W.; Janjetovic, Z.; Postlethwaite, A.; Zouboulis, C.C.; et al. Steroidogenesis in the skin: Implications for local immune functions. J. Steroid Biochem. Mol. Biol. 2013, 137, 107–123. [Google Scholar] [CrossRef] [PubMed]
- Jin, R.; Luo, L.; Zheng, J. The trinity of skin: Skin homeostasis as a neuro-endocrine-immune organ. Life 2022, 12, 725. [Google Scholar] [CrossRef]
- Li, N.; Zhao, J.; Di, T.; Meng, Y.; Wang, M.; Li, X.; Liu, Z.; Zhai, C.; Zhang, L.; Ma, C.; et al. Matrine alleviates imiquimod-induced psoriasiform dermatitis in BALB/c mice via dendritic cell regulation. Int. J. Clin. Exp. Pathol. 2018, 11, 5232–5240. [Google Scholar]
- Xiang, X.; Tu, C.; Li, Q.; Wang, W.; Huang, X.; Zhao, Z.; Xiong, H.; Mei, Z. Oxymatrine ameliorates imiquimod-induced psoriasis pruritus and inflammation through inhibiting heat shock protein 90 and heat shock protein 60 expression in keratinocytes. Toxicol. Appl. Pharmacol. 2020, 405, 115209. [Google Scholar] [CrossRef]
- Jiang, W.W.; Wang, Y.M.; Wang, X.Y.; Zhang, Q.; Zhu, S.M.; Zhang, C.L. Role and mechanism of matrine alone and combined with acitretin for HaCaT cells and psoriasis-like murine models. Chin. Med. J. 2019, 132, 2079–2088. [Google Scholar] [CrossRef]






| Retention Time (min) | Molecular Weight (Da) | VIP | Potential Biomarker | Formula | Change Trend |
|---|---|---|---|---|---|
| ESI− | |||||
| 4.843 | 308.124 | 2.604 | Glutaminyltyrosine | C14H19N3O5 | DOWN |
| 6.468 | 453.202 | 2.054 | 2-Hydroxy-imipramine glucuronide | C25H32N2O7 | UP |
| 6.183 | 581.300 | 2.676 | Serylarginine | C9H19N5O4 | UP |
| 2.626 | 263.076 | 1.478 | 3,4,5-trihydroxy-6-[(2-methylpropanoyl)oxy]oxane-2-carboxylic acid | C10H16O8 | DOWN |
| 2.821 | 374.095 | 2.080 | 7-Hydroxygliclazide | C15H21N3O4S | DOWN |
| 2.829 | 299.070 | 1.075 | Methionyl-Glutamate | C10H18N2O5S | DOWN |
| 4.270 | 315.163 | 2.005 | 3-[8-hydroxy-2-methyl-2-(4-methylpent-3-en-1-yl)-2H-chromen-5-yl]propanoic acid | C19H24O4 | DOWN |
| 4.791 | 165.059 | 1.301 | 3-Mercapto-2-methyl-1-butanol | C5H12OS | DOWN |
| 6.240 | 480.249 | 2.770 | Dynorphin A (6–8) | C18H37N9O4 | UP |
| 6.278 | 457.197 | 2.666 | Deoxypyridinoline | C18H28N4O7 | UP |
| 6.308 | 469.161 | 2.372 | Melleolide B | C24H32O7 | UP |
| 6.308 | 667.171 | 1.090 | Hydroxychlorpromazine | C17H19ClN2OS | UP |
| 6.347 | 455.181 | 2.248 | Armillane | C23H32O7 | UP |
| 6.378 | 437.170 | 2.123 | Nevskin | C24H32O5 | UP |
| 6.431 | 177.023 | 1.656 | 3,3′-Thiobispropanoic acid | C6H10O4S | UP |
| 6.442 | 295.031 | 2.518 | Cis,trans-5′-Hydroxythalidomide | C13H10N2O5 | UP |
| 6.456 | 421.175 | 2.033 | Euglobal IIb | C23H30O5 | UP |
| 6.462 | 465.202 | 1.909 | Homofukinolide | C25H34O6 | UP |
| 6.493 | 486.187 | 1.533 | 4-Hydroxyvalsartan | C24H29N5O4 | UP |
| 5.583 | 451.185 | 1.071 | 7-[(2,6-dihydroxy-2,5,5,8a-tetramethyl-decahydronaphthalen-1-yl)methoxy]-6-hydroxy-2H-chromen-2-one | C24H32O6 | UP |
| 6.601 | 467.217 | 2.471 | Colupdox a | C25H36O6 | UP |
| 6.630 | 553.297 | 1.078 | Phytolaccinic acid | C31H48O6 | DOWN |
| 6.969 | 492.117 | 2.575 | 2-amino-4-({1-[(carboxymethyl)-C-hydroxycarbonimidoyl]-2-[(2,3-dihydroxy-1-phenylpropyl)sulfanyl]ethyl}-C-hydroxycarbonimidoyl)butanoic acid | C19H27N3O8S | UP |
| 7.023 | 609.361 | 1.208 | Lactapiperanol C | C16H26O4 | DOWN |
| 7.434 | 810.583 | 1.023 | PE(18:0/20:0) | C43H86NO8P | DOWN |
| 7.480 | 606.500 | 2.048 | 1,1′-(1,4-Dihydro-4-nonyl-3,5-pyridinediyl)bis[1-dodecanone] | C38H69NO2 | DOWN |
| 8.936 | 453.202 | 1.579 | Colupox a | C25H36O5 | UP |
| 8.915 | 701.421 | 1.880 | PG(a-13:0/a-15:0) | C34H67O10P | UP |
| 8.846 | 465.314 | 1.130 | (2β,3α,9α,24R)-Ergosta-7,22-diene-2,3,9-triol | C28H46O3 | DOWN |
| 7.712 | 865.522 | 1.182 | PG(18:1(11Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) | C46H77O10P | DOWN |
| 7.365 | 820.589 | 1.091 | PE(18:1(11Z)/22:1(13Z)) | C45H86NO8P | DOWN |
| 7.157 | 463.293 | 2.768 | Palmitoyl glucuronide | C22H42O7 | DOWN |
| 7.057 | 361.174 | 0.993 | Perindoprilat | C17H28N2O5 | DOWN |
| 6.541 | 379.218 | 2.167 | 3′-Hydroxystanozolol | C21H32N2O2 | DOWN |
| 6.475 | 439.185 | 2.144 | Lucidone A | C24H34O5 | UP |
| 6.456 | 423.154 | 2.463 | 5,7-dihydroxy-4-(1-hydroxypropyl)-6-(3-methylbut-2-en-1-yl)-8-(2-methylbutanoyl)-2H-chromen-2-one | C22H28O6 | UP |
| 6.451 | 455.217 | 2.656 | Cavipetin D | C25H38O5 | UP |
| 6.431 | 475.163 | 2.153 | α-CEHC glucuronide | C22H30O10 | UP |
| 6.378 | 500.167 | 2.330 | 2-amino-4-({1-[(carboxymethyl)-C-hydroxycarbonimidoyl]-2-{[4-(2,6-dihydroxyphenyl)-3-hydroxy-2-methylbutan-2-yl]sulfanyl}ethyl}-C-hydroxycarbonimidoyl)butanoic acid | C21H31N3O9S | UP |
| 6.371 | 453.165 | 2.610 | Diosbulbin H | C23H30O7 | UP |
| 6.354 | 439.186 | 1.945 | 1α-O-Methylquassin | C23H32O6 | UP |
| 6.347 | 507.174 | 2.465 | L-Nicotianine | C10H12N2O4 | UP |
| 6.284 | 520.193 | 2.082 | Hydroxynefazodone | C25H32ClN5O3 | UP |
| 6.240 | 452.218 | 1.926 | Chondroitin sulfate E (GalNAc4,6diS-GlcA), precursor 5a | C7H9N3O | UP |
| 6.222 | 369.211 | 1.248 | 3-keto-Digoxigenin | C23H32O5 | DOWN |
| 6.218 | 476.279 | 1.123 | LysoPE(0:0/18:2(9Z,12Z)) | C23H44NO7P | DOWN |
| 6.134 | 457.160 | 1.105 | Valechlorin | C22H31ClO8 | UP |
| 5.718 | 371.226 | 2.685 | Digoxigenin | C23H34O5 | DOWN |
| 5.114 | 348.156 | 1.934 | (+)-O-Methylarmepavine | C20H25NO3 | DOWN |
| 5.065 | 329.179 | 2.313 | 12,20-Dioxo-leukotriene B4 | C20H28O5 | DOWN |
| 4.974 | 357.211 | 1.896 | 17-Hydroxy-E4-neuroprostane | C22H32O5 | DOWN |
| 4.589 | 357.222 | 1.037 | Kinetensin 1-3 | C15H30N6O4 | UP |
| 2.829 | 365.143 | 1.110 | Valproic acid glucuronide | C14H24O8 | DOWN |
| 2.814 | 296.106 | 1.004 | 7-Aminonitrazepam | C15H13N3O | DOWN |
| 1.685 | 295.040 | 1.398 | N-Acetyldjenkolic acid | C9H16N2O5S2 | UP |
| 1.527 | 183.044 | 1.218 | Mevalonic acid | C6H12O4 | UP |
| 6.371 | 383.161 | 1.501 | 21-Hydroxy-5b-pregnane-3,11,20-trione | C21H30O4 | DOWN |
| 6.278 | 581.263 | 1.973 | S-(3-Methylbutanoyl)-dihydrolipoamide-E | C13H25NO2S2 | UP |
| 8.922 | 689.351 | 1.131 | Zeranol | C18H26O5 | UP |
| ESI+ | |||||
| 6.444 | 453.200 | 3.257 | Pro Ser Ser Tyr | C20H28N4O8 | UP |
| 4.727 | 828.534 | 0.976 | 3-O-Sulfogalactosylceramide (d18:1/18:1(9Z)) | C42H79NO11S | DOWN |
| 3.285 | 404.222 | 1.210 | Ser Ala Ala Arg | C15H29N7O6 | UP |
| 4.253 | 317.177 | 1.919 | Capillartemisin A | C19H24O4 | DOWN |
| 5.069 | 331.193 | 2.827 | 16-Hydroxy-4-oxoretinoic acid | C20H26O4 | DOWN |
| 6.224 | 371.225 | 1.960 | 17-phenyl trinor-13,14-dihydro Prostaglandin A2 | C23H30O4 | DOWN |
| 6.254 | 395.252 | 1.328 | (3R, 6′Z)-3,4-Dihydro-8-hydroxy-3-(6-pentadecenyl)-1H-2-benzopyran-1-one | C24H36O3 | UP |
| 6.304 | 565.350 | 1.002 | PG(22:2(13Z,16Z)/0:0) | C28H53O9P | UP |
| 6.324 | 313.077 | 2.457 | Inosine-5′-carboxylate | C11H12N4O7 | UP |
| 7.031 | 626.533 | 1.139 | Muricatenol | C37H68O6 | UP |
| 6.338 | 301.077 | 2.967 | Temazepam | C16H13ClN2O2 | UP |
| 1.543 | 286.157 | 2.191 | Tryptophyl-Valine | C16H21N3O3 | UP |
| 3.194 | 298.150 | 1.266 | Histidinyl-Threonine | C10H16N4O4 | DOWN |
| 3.319 | 281.119 | 1.191 | 6-hydroxy-7E,9E-Octadecadiene-11,13,15,17-tetraynoic acid | C18H16O3 | UP |
| 4.064 | 534.323 | 1.347 | Cys Lys Lys Arg | C21H43N9O5S | DOWN |
| 4.235 | 255.129 | 1.186 | Isoleucyl-Threonine | C10H20N2O4 | DOWN |
| 4.305 | 622.377 | 1.446 | Tyr Arg Lys Arg | C27H47N11O6 | DOWN |
| 4.474 | 277.124 | 1.256 | Isobutyrylcarnitine | C11H22NO4+ | UP |
| 4.604 | 386.246 | 1.473 | Pseudoginsenoside RT3 | C41H70O13 | DOWN |
| 4.654 | 549.172 | 1.514 | Myricatomentoside I | C26H32O10 | DOWN |
| 4.987 | 359.225 | 1.475 | 7,8-epoxy-17S-HDHA | C22H30O4 | DOWN |
| 5.118 | 696.452 | 1.502 | PA(16:1(9Z)/15:0) | C34H65O8P | DOWN |
| 5.170 | 828.533 | 1.338 | PE(MonoMe(11,3)/MonoMe(11,3)) | C43H74NO10P | DOWN |
| 5.994 | 242.134 | 0.972 | Lysyl-Methionine | C11H23N3O3S | UP |
| 6.016 | 548.287 | 1.511 | 11-Hydroxyprogesterone 11-glucuronide | C27H38O9 | UP |
| 6.082 | 465.259 | 1.132 | (−)-Jolkinol B | C29H36O5 | UP |
| 6.136 | 502.163 | 2.001 | 3-(3-hydroxyphenyl)-2-(4-hydroxyphenyl)-5-[(E)-2-(3hydroxyphenyl)ethenyl]-2,3-dihydro-1 benzofuran-6-ol | C28H22O5 | UP |
| 6.149 | 467.275 | 1.316 | 1,25-Dihydroxyvitamin D3-26,23-lactone | C27H40O5 | UP |
| 6.205 | 363.225 | 1.097 | 5α-Pregnan-20α-ol-3-one | C21H34O2 | UP |
| 6.224 | 453.221 | 1.813 | Australigenin | C27H42O4 | UP |
| 6.283 | 534.221 | 1.608 | Cyclocalopin D | C23H32O13 | UP |
| 6.297 | 365.241 | 1.267 | Adipostatin A | C21H36O2 | UP |
| 6.324 | 574.253 | 1.565 | Glaucarubolone 15-O-β-d-glucopyranoside | C26H36O13 | UP |
| 6.360 | 427.276 | 1.508 | Asymmetric dimethylarginine | C8H18N4O2 | UP |
| 6.360 | 455.179 | 3.227 | Gly Glu Ser Tyr | C19H26N4O9 | UP |
| 6.367 | 441.200 | 4.192 | Gly Thr Tyr Thr | C19H28N4O8 | UP |
| 6.416 | 435.294 | 2.760 | Alanyl-Lysine | C9H19N3O3 | UP |
| 6.437 | 527.345 | 2.800 | Pro Val Arg Arg | C22H42N10O5 | UP |
| 6.451 | 355.125 | 2.024 | Meta-O-Dealkylated flecainide | C15H19F3N2O3 | UP |
| 6.472 | 463.182 | 2.851 | PA(8:0/8:0) | C19H37O8P | UP |
| 6.479 | 441.200 | 3.151 | (9S,10S)-10-hydroxy-9-(phosphonooxy)octadecanoate | C18H37O7P | UP |
| 6.500 | 714.509 | 1.058 | PA(18:1(9Z)/18:3(9Z,12Z,15Z)) | C39H69O8P | UP |
| 6.521 | 523.411 | 2.648 | Panaxydol linoleate | C35H54O3 | DOWN |
| 6.604 | 411.225 | 1.903 | Pro Pro Thr Pro | C19H30N4O6 | UP |
| 6.646 | 722.519 | 1.386 | PE(18:4(6Z,9Z,12Z,15Z)/P-18:1(11Z)) | C41H72NO7P | UP |
| 6.660 | 678.492 | 1.312 | PE(18:4(6Z,9Z,12Z,15Z)/P-16:0) | C39H70NO7P | UP |
| 6.674 | 634.465 | 1.471 | Tsugarioside B | C37H60O7 | UP |
| 6.856 | 670.523 | 1.605 | PE(15:0/18:0) | C38H76NO8P | UP |
| 7.010 | 904.712 | 1.184 | PC(o-20:0/22:0) | C50H102NO7P | UP |
| 7.066 | 407.153 | 1.119 | 3b,16a-Dihydroxyandrostenone sulfate | C19H28O6S | UP |
| 8.944 | 425.205 | 4.129 | O-Desmethylcarvedilol | C23H24N2O4 | UP |
| 9.287 | 423.189 | 2.131 | Cys Val Thr Thr | C16H30N4O7S | UP |
| 8.685 | 895.754 | 1.939 | TG(18:1(11Z)/22:4(7Z,10Z,13Z,16Z)/18:3(9Z,12Z,15Z)) | C61H102O6 | DOWN |
| 6.940 | 539.360 | 1.721 | 3-methyl-4-(methylamino)-1,2-diphenylbutan-2-ol | C18H23NO | UP |
| 6.870 | 626.496 | 1.675 | PE(15:0/P-16:0) | C36H72NO7P | UP |
| 6.870 | 614.353 | 1.399 | LysoPC(22:2(13Z,16Z)) | C30H58NO7P | UP |
| 6.849 | 714.550 | 1.546 | PC(14:1(9Z)/18:0) | C40H78NO8P | UP |
| 6.828 | 802.604 | 1.578 | PC(18:0/22:4(7Z,10Z,13Z,16Z)) | C48H88NO8P | UP |
| 6.737 | 696.496 | 1.312 | CL(i-12:0/18:2(9Z,11Z)/i-18:0/i-19:0) | C76H144O17P2 | UP |
| 6.730 | 740.523 | 1.185 | CL(18:0/18:0/16:1(9Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) | C83H148O17P2 | UP |
| 6.646 | 840.578 | 1.939 | PG(18:0/22:4(7Z,10Z,13Z,16Z)) | C46H83O10P | DOWN |
| 6.604 | 163.076 | 1.060 | 4-Hydroxy-2,6,6-trimethyl-3-oxo-1,4-cyclohexadiene-1-carboxaldehyde | C10H12O3 | UP |
| 6.590 | 295.066 | 1.816 | 2-hydroxy-3-[3-(3-methylbut-2-en-1-yl)-4-(sulfooxy)phenyl]propanoic acid | C14H18O7S | UP |
| 6.583 | 459.187 | 2.136 | Ser Glu Ser His | C17H26N6O9 | UP |
| 6.542 | 355.194 | 1.101 | Nigakihemiacetal B | C22H30O6 | DOWN |
| 6.493 | 764.516 | 1.715 | CL(16:0/18:0/18:0/22:5(7Z,10Z,13Z,16Z,19Z)) | C83H152O17P2 | DOWN |
| 6.472 | 700.493 | 1.413 | PA(15:0/18:2(9Z,12Z)) | C36H67O8P | UP |
| 6.472 | 385.136 | 3.206 | 2′,7-Dihydroxy-4′-methoxy-8-prenylflavan | C21H24O4 | UP |
| 6.465 | 351.130 | 1.754 | N-Ribosylhistidine | C11H17N3O6 | UP |
| 6.451 | 521.396 | 1.727 | Ginsenoyne A linoleate | C35H52O3 | DOWN |
| 6.388 | 285.081 | 1.654 | (2S)-2-hydrazinyl-3-(4-hydroxy-3-methoxyphenyl)-2-methylpropanoic acid | C11H16N2O4 | DOWN |
| 6.381 | 439.184 | 3.069 | Gln Glu Tyr | C19H26N4O8 | UP |
| 6.345 | 375.115 | 1.271 | Cyclic N-Acetylserotonin glucuronide | C18H20N2O8 | DOWN |
| 6.338 | 361.135 | 2.023 | 7-Hydroxy-6-methyl-8-ribityl lumazine | C12H16N4O7 | UP |
| 6.324 | 538.255 | 1.523 | Rubraflavone B | C30H34O5 | UP |
| 6.311 | 618.283 | 2.136 | 3′-Deaminofusarochromanone | C15H19NO4 | UP |
| 6.297 | 424.221 | 2.812 | Sphingosine 1-phosphate | C18H38NO5P | UP |
| 6.290 | 508.280 | 2.746 | LysoPE(0:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) | C27H44NO7P | UP |
| 6.283 | 510.296 | 4.312 | LysoPE(0:0/22:5(7Z,10Z,13Z,16Z,19Z)) | C27H46NO7P | UP |
| 6.276 | 583.277 | 3.375 | (4E)-6-hydroxy-1,7-diphenylhept-4-en-3-one | C19H20O2 | UP |
| 6.241 | 451.279 | 0.944 | PA(19:1(9Z)/0:0) | C22H43O7P | UP |
| 6.231 | 498.259 | 3.159 | PE(18:3/0:0) | C23H42NO7P | UP |
| 6.199 | 437.264 | 1.065 | 1-Oleoyl Lysophosphatidic Acid (sodium salt) | C21H41O7P | UP |
| 6.186 | 175.123 | 2.811 | α-Methyltryptamine (AMT) | C11H14N2 | UP |
| 6.174 | 569.298 | 3.370 | 11-α-O-β-d-Glucopyranosyl-16α-O-methylneoquassin | C29H44O11 | UP |
| 6.075 | 487.359 | 1.085 | Lys Ile Val Lys | C23H46N6O5 | UP |
| 6.005 | 618.340 | 1.903 | Pentasine | C30H49N6O10+ | UP |
| 6.000 | 349.209 | 1.127 | Oryzalexin E | C20H32O2 | UP |
| 5.554 | 183.082 | 1.526 | 2-Methyl-4-propyl-1,3-oxathiane | C8H16OS | UP |
| 5.112 | 740.479 | 1.500 | PS(14:0/20:3(8Z,11Z,14Z)) | C40H72NO10P | DOWN |
| 5.006 | 240.142 | 0.993 | Isopropyl β-d-glucoside | C9H18O6 | UP |
| 4.945 | 336.130 | 0.976 | Gynocardin | C12H17NO8 | DOWN |
| 4.897 | 352.161 | 1.890 | (+/−)-Rollipyrrole | C16H20N2O3 | DOWN |
| 4.615 | 338.145 | 1.145 | N-Acetyl-3-hydroxyprocainamide | C15H23N3O3 | DOWN |
| 4.391 | 233.097 | 1.094 | L-Targinine | C7H16N4O2 | UP |
| 3.611 | 402.242 | 1.022 | N-Didesmethylmifepristone (RU 42848) | C27H31NO2 | DOWN |
| 3.396 | 261.150 | 1.327 | Methyl dihydrophaseate | C16H24O5 | UP |
| 3.222 | 229.123 | 1.660 | Blennin B | C15H20O4 | UP |
| 2.566 | 267.140 | 1.527 | (S)-17-Hydroxy-9,11,13,15-octadecatetraynoic acid | C18H20O3 | UP |
| 1.360 | 217.134 | 1.518 | Girgensonine | C13H16N2O | DOWN |
| 0.013 | 300.208 | 1.040 | 2-hydroxydesipramine | C18H22N2O | UP |
| 6.800 | 524.283 | 1.270 | 11-β-Hydroxyandrosterone-3-glucuronide | C25H38O9 | UP |
| No. | Pathway | p-Value | Details |
|---|---|---|---|
| 1 | Sphingolipid metabolism | 0.001 | KEGG |
| 2 | Linoleic acid metabolism | 0.036 | KEGG |
| 3 | Steroid hormone biosynthesis | 0.021 | KEGG |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, X.; Cheng, J.; Yin, X.; Ao, T.; He, X.; Yang, Y.; Lin, Y.; Chen, Z. Metabolic Profiling for Unveiling Mechanisms of Kushenol F against Imiquimod-Induced Psoriasis with UHPLC/MS Analysis. Molecules 2024, 29, 2410. https://doi.org/10.3390/molecules29112410
Yang X, Cheng J, Yin X, Ao T, He X, Yang Y, Lin Y, Chen Z. Metabolic Profiling for Unveiling Mechanisms of Kushenol F against Imiquimod-Induced Psoriasis with UHPLC/MS Analysis. Molecules. 2024; 29(11):2410. https://doi.org/10.3390/molecules29112410
Chicago/Turabian StyleYang, Xingxin, Jiaoli Cheng, Xunqing Yin, Ting Ao, Xudong He, Yaqin Yang, Yuping Lin, and Zhen Chen. 2024. "Metabolic Profiling for Unveiling Mechanisms of Kushenol F against Imiquimod-Induced Psoriasis with UHPLC/MS Analysis" Molecules 29, no. 11: 2410. https://doi.org/10.3390/molecules29112410





