Efficient 2,3-Butanediol Production from Ethanol by a Modified Four-Enzyme Synthetic Biosystem
Abstract
1. Introduction
2. Results and Discussion
2.1. Selection of Mutational Residues
2.2. Single-Point and Combinatorial Mutation of FLS
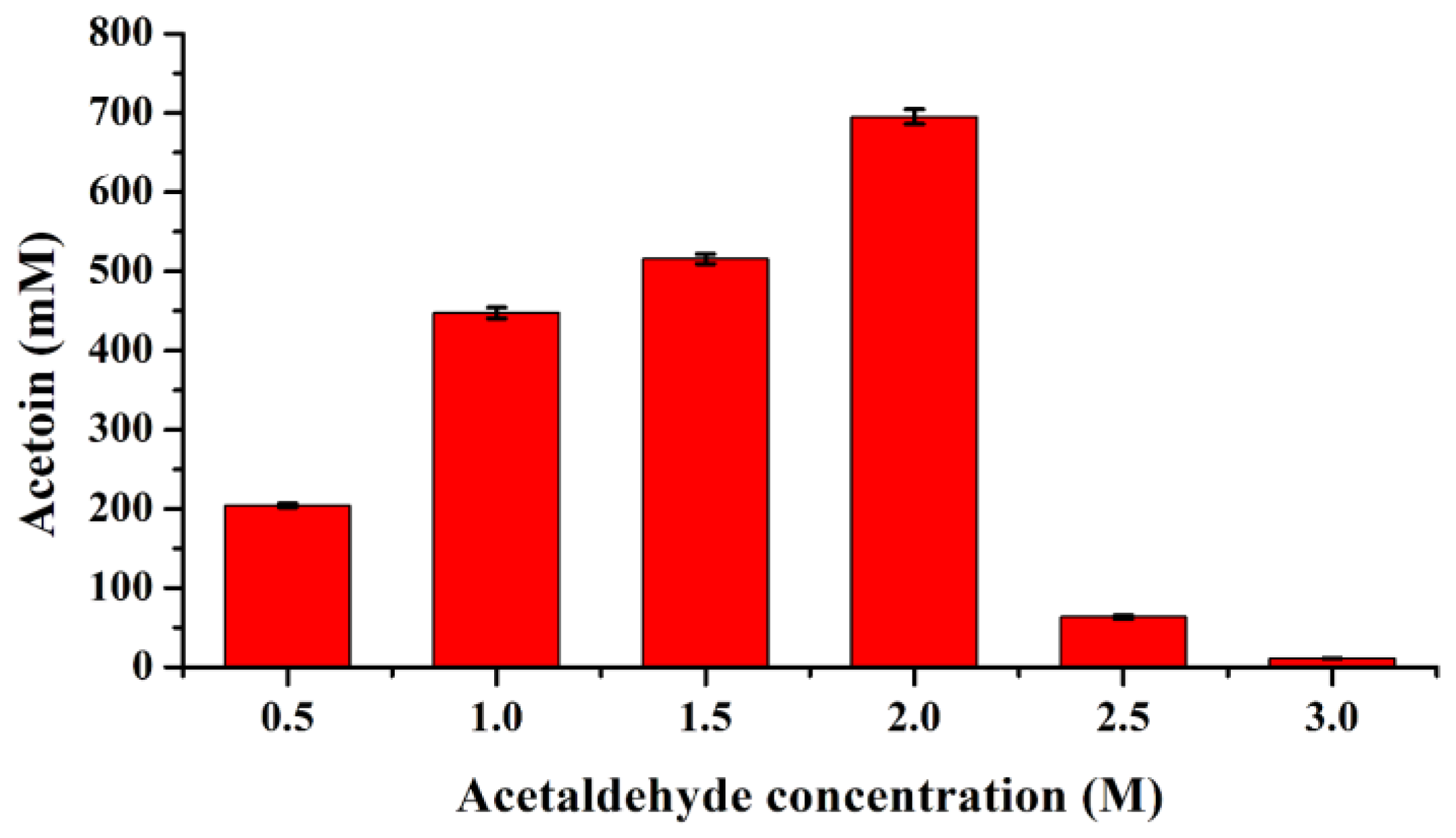
2.3. Acetaldehyde Tolerance Evaluation of FLS and Its Variants
2.4. Kinetic Parameters
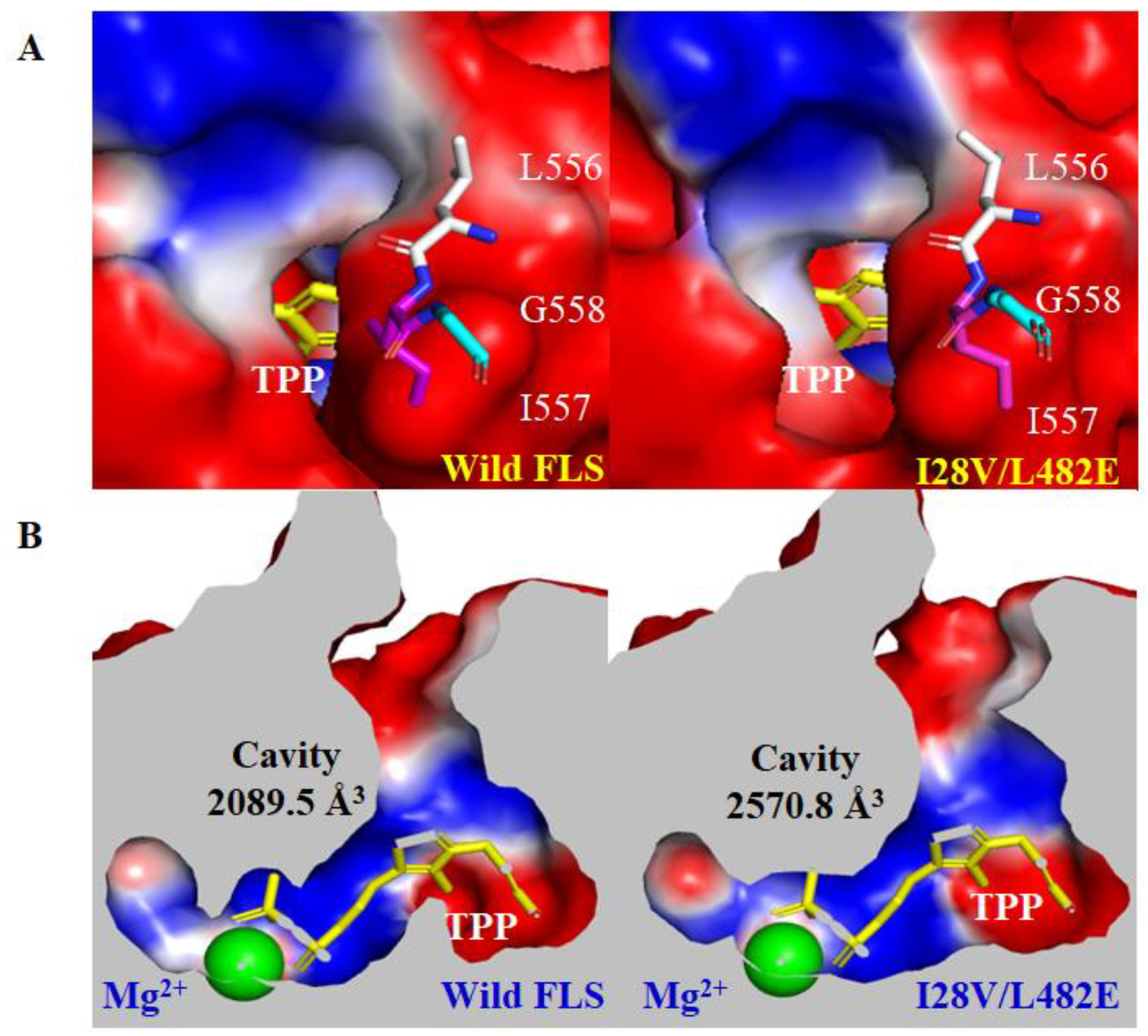
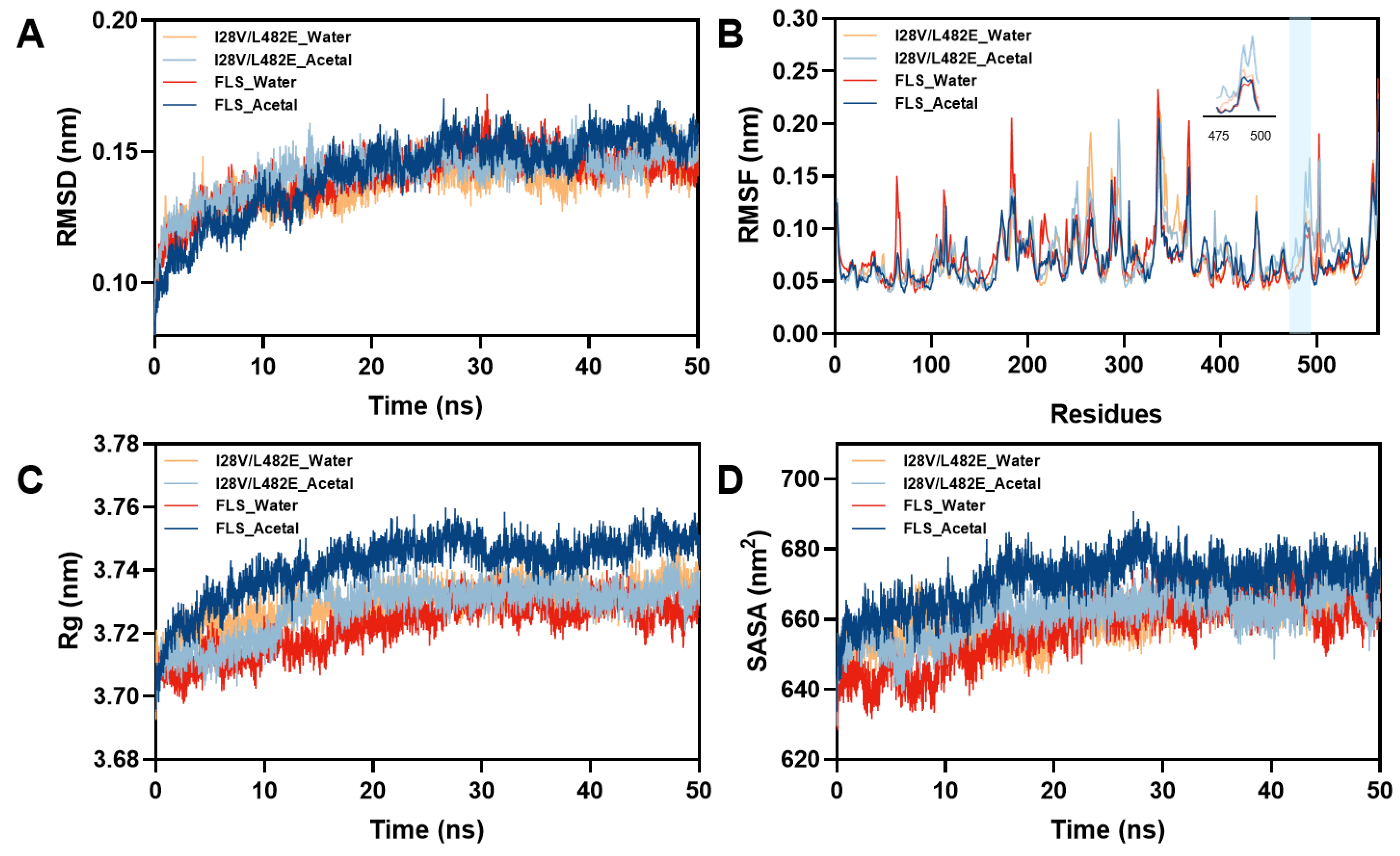
2.5. In Silico Structure Analysis of Wild FLS and Its Variant I28V/L482E
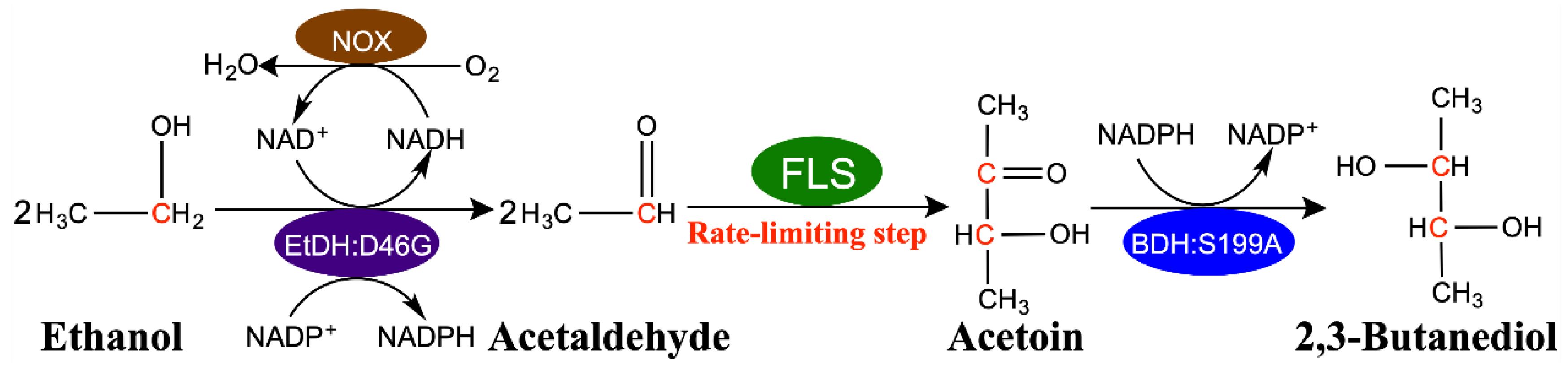
2.6. Fed-Batch Synthesis of 2,3-Butanediol from Ethanol by a Modified Four-Enzyme Synthetic Biosystem
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Bacterial Growth, Recombinant Protein Expression and Purification
3.3. Mutant Library Construction for FLS
3.4. Preliminary Screening of Mutant Library
3.5. Substrate Tolerance Analysis of Beneficial Variants
3.6. Enzyme Activity and Kinetic Parameter Assays
3.7. Fed-Batch Synthesis of 2,3-BD from Ethanol and GC Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ji, X.J.; Huang, H.; Ouyang, P.K. Microbial 2,3-butanediol production: A state-of-the-art review. Biotechnol. Adv. 2011, 29, 351–364. [Google Scholar] [CrossRef]
- Haider, J.; Harvianto, G.R.; Qyyum, M.A.; Lee, M. Cost- and energy-efficient butanol-based extraction-assisted distillation designs for purification of 2,3-butanediol for use as a drop-in fuel. ACS Sustain. Chem. Eng. 2018, 6, 14901–14910. [Google Scholar] [CrossRef]
- Sathesh-Prabu, C.; Kim, D.; Lee, S.K. Metabolic engineering of Escherichia coli for 2,3-butanediol production from cellulosic biomass by using glucose-inducible gene expression system. Bioresour. Technol. 2020, 309, 123361. [Google Scholar] [CrossRef] [PubMed]
- Hazeena, S.H.; Sindhu, R.; Pandey, A.; Binod, P. Lignocellulosic bio-refinery approach for microbial 2,3-Butanediol production. Bioresour. Technol. 2020, 302, 122873. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Liu, S.; Osire, T.; Zhang, X.; Xu, M.; Yang, S.T.; Yang, T.; Rao, Z. Engineering the 2,3-BD pathway in Bacillus subtilis by shifting the carbon flux in favor of 2,3-BD synthesis. Biochem. Eng. J. 2021, 169, 107969. [Google Scholar] [CrossRef]
- Xie, S.Q.; Li, Z.X.; Zhu, G.D.; Song, W.L.; Yi, C.H. Clearner production and downstream processing of bio-based 2,3-butanediol: A review. J. Clean. Prod. 2022, 343, 131033. [Google Scholar] [CrossRef]
- Ge, Y.; Li, K.; Li, L.; Gao, C.; Zhang, L.; Ma, C.; Xu, P. Contracted but effective: Production of enantiopure 2,3-butanediol by thermophilic and GRAS Bacillus licheniformis. Green Chem. 2016, 18, 4693–4703. [Google Scholar] [CrossRef]
- Guo, Z.W.; Ou, X.Y.; Xu, P.; Gao, H.F.; Zhang, L.Y.; Zong, M.H.; Lou, W.Y. Energy- and cost-effective non-sterilized fermentation of 2,3-butanediol by an engineered Klebsiella pneumoniae OU7 with an anti-microbial contamination system. Green Chem. 2020, 22, 8584–8593. [Google Scholar] [CrossRef]
- Kim, D.G.; Yoo, S.W.; Kim, M.; Ko, J.K.; Um, Y.; Oh, M.K. Improved 2,3-butanediol yield and productivity from lignocellulose biomass hydrolysate in metabolically engineered Enterobacter aerogenes. Bioresour. Technol. 2020, 309, 123386. [Google Scholar] [CrossRef]
- Kay, J.E.; Jewett, M.C. Lysate of engineered Escherichia coli supports high-level conversion of glucose to 2,3-butanediol. Metab. Eng. 2015, 32, 133–142. [Google Scholar] [CrossRef]
- He, Y.; Chen, F.; Sun, M.; Gao, H.; Guo, Z.; Lin, H.; Chen, J.; Jin, W.; Yang, Y.; Zhang, L.; et al. Efficient (3S)-acetoin and (2S,3S)-2,3-butanediol production from meso-2,3-butanediol using whole-cell biocatalysis. Molecules 2018, 23, 691. [Google Scholar] [CrossRef] [PubMed]
- Schilling, C.; Ciccone, R.; Sieber, V.; Schmid, J. Engineering of the 2,3-butanediol pathway of Paenibacillus polymyxa DSM 365. Metab. Eng. 2020, 61, 381–388. [Google Scholar] [CrossRef]
- Li, L.X.; Wang, Y.; Zhang, L.J.; Ma, C.Q.; Wang, A.L.; Tao, F.; Xu, P. Biocatalytic production of (2S,3S)-2,3-butanediol from diacetyl using whole cells of engineered Escherichia coli. Bioresour. Technol. 2012, 115, 111–116. [Google Scholar] [CrossRef]
- Samuel, N.; Bao, T.; Zhang, X.; Yang, T.W.; Xu, M.J.; Li, X.; Komera, I.; Philibert, T.; Rao, Z.M. Optimized whole cell biocatalyst from acetoin to 2,3-butanediol through coexpression of acetoin reductase with NADH regeneration systems in engineered Bacillus subtilis. J. Chem. Technol. Biotechnol. 2017, 9, 2477–2487. [Google Scholar]
- Tinoco, D.; Freire, D.M.G. Scale-up of 2,3-butanediol production by Paenibacillus peoriae NRRL BD-62 using constant oxygen transfer rate-based strategy. Fuel 2023, 340, 127603. [Google Scholar] [CrossRef]
- Fu, J.; Huo, G.; Feng, L.; Mao, Y.; Wang, Z.; Ma, H.; Chen, T.; Zhao, X. Metabolic engineering of Bacillus subtilis for chiral pure meso-2,3-butanediol production. Biotechnol. Biofuels 2016, 9, 90. [Google Scholar] [CrossRef]
- Zhang, J.K.; Zhao, J.Z.; Fu, Q.B.; Liu, H.Y.; Li, M.; Wang, Z.Y.; Gu, W.; Zhu, X.M.; Lin, R.S.; Dai, L.; et al. Metabolic engineering of Paenibacillus polymyxa for effective production of 2,3-butanediol from polar hydrolysate. Bioresour. Technol. 2024, 392, 130002. [Google Scholar] [CrossRef]
- Cui, Z.; Mao, Y.; Zhao, Y.; Chen, C.; Tang, Y.J.; Chen, T.; Ma, H.; Wang, Z. Concomitant cell-free biosynthesis of optically pure D-(−)-acetoin and xylitol via a novel NAD+ regeneration in two-enzyme cascade. J. Chem. Technol. Biotechnol. 2018, 93, 3444–3451. [Google Scholar] [CrossRef]
- Zhao, Q.; Ansorge-Schumacher, M.B.; Haag, R.; Wu, C. Living whole-cell catalysis in compartmentalized emulsion. Bioresour. Technol. 2020, 295, 122221. [Google Scholar] [CrossRef] [PubMed]
- Korman, T.P.; Opgenorth, P.H.; Bowie, J.U. A synthetic biochemistry platform for cell free production of monoterpenes from glucose. Nat. Commun. 2017, 8, 15526. [Google Scholar] [CrossRef]
- Meng, D.; Wei, X.; Bai, X.; Zhou, W.; You, C. Artificial in Vitro Synthetic Enzymatic Biosystem for the One-Pot Sustainable Biomanufacturing of Glucosamine from Starch and Inorganic Ammonia. ACS Catal. 2020, 10, 13809–13819. [Google Scholar] [CrossRef]
- Cui, Z.; Mao, Y.; Zhao, Y.; Zheng, M.; Wang, Z.; Ma, H.; Chen, T. One-pot efficient biosynthesis of (3R)-acetoin from pyruvate by a two-enzyme cascade. Catal. Sci. Technol. 2020, 10, 7734–7744. [Google Scholar] [CrossRef]
- Gao, C.; Zhang, L.; Xie, Y.; Hu, C.; Zhang, Y.; Li, L.; Wang, Y.; Ma, C.; Xu, P. Production of (3S)-acetoin from diacetyl by using stereoselective NADPH-dependent carbonyl reductase and glucose dehydrogenase. Bioresour. Technol. 2013, 137, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Bornemann, S.; Crout, D.H.G.; Dalton, H.; Hutchinson, D.W.; Dean, G.; Thomson, N.; Turner, M.M. Stereochemistry of the formation of lactaldehyde and acetoin produced by the pyruvate decarboxylases of yeast (Saccharomyces sp.) and Zymomonas mobilis: Different boltzmann distributions between bound forms of the electrophile, acetaldehyde, in the two enzymatic reactions. J. Chem. Soc. Perkin Trans. 1993, 1, 309–311. [Google Scholar]
- Seo, P.W.; Jo, H.J.; Hwang, I.Y.; Jeong, H.Y.; Kim, J.H.; Kim, J.W.; Lee, E.Y.; Park, J.B.; Kim, J.S. Understanding the molecular properties of the E1 subunit (SucA) of α-ketoglutarate dehydrogenase complex from Vibrio vulnificus for the enantioselective ligation of acetaldehydes into (R)-acetoin. Catal. Sci. Technol. 2020, 10, 79–85. [Google Scholar] [CrossRef]
- Siegel, J.B.; Smith, A.L.; Poust, S.; Wargacki, A.J.; Bar-Even, A.; Louw, C.; Shen, B.W.; Eiben, C.B.; Tran, H.M.; Noor, E.; et al. Computational protein design enables a novel one-carbon assimilation pathway. Proc. Natl. Acad. Sci. USA 2015, 112, 3704–3709. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Singh, R.; Sivakumar, D.; Guo, Z.; Li, J.; Chen, F.; He, Y.; Guan, X.; Kang, Y.C.; Lee, J.K. An artificial synthetic pathway for acetoin, 2,3-butanediol, and 2-butanol production from ethanol using cell free multi-enzyme catalysis. Green Chem. 2018, 20, 230–242. [Google Scholar] [CrossRef]
- Moore, C.; Staples, O.; Jenkins, R.; Brooks, T.J.; Semelsberger, T.; Sutton, A. Acetaldehyde as an ethanol derived bio-building block: An alternative to Guerbet chemistry. Green Chem. 2017, 19, 169–174. [Google Scholar] [CrossRef]
- Chen, Z.; Zeng, A.P. Protein engineering approaches to chemical biotechnology. Curr. Opin. Biotechnol. 2016, 42, 198–205. [Google Scholar] [CrossRef]
- Fasan, R.; Jennifer Kan, S.B.; Zhao, H. A Continuing Career in Biocatalysis: Frances H. Arnold. ACS Catal. 2019, 9, 9775–9788. [Google Scholar] [CrossRef] [PubMed]
- Notin, P.; Rollins, N.; Gal, Y.; Sander, C.; Marks, D. Machine learning for functional protein design. Nat. Biotechnol. 2024, 42, 216–228. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wu, Y.Q.; Xu, N.; Zhao, Q.; Yu, H.L.; Xu, J.H. Engineering of Cyclohexanone Monooxygenase for the Enantioselective Synthesis of (S)-Omeprazole. ACS Sustain. Chem. Eng. 2019, 7, 7218–7226. [Google Scholar] [CrossRef]
- Chen, F.F.; Zheng, G.W.; Liu, L.; Li, H.; Chen, Q.; Li, F.L.; Li, C.X.; Xu, J.H. Reshaping the Active Pocket of Amine Dehydrogenases for Asymmetric Synthesis of Bulky Aliphatic Amines. ACS Catal. 2018, 8, 2622–2628. [Google Scholar] [CrossRef]
- Zheng, G.W.; Liu, Y.Y.; Chen, Q.; Huang, L.; Yu, H.L.; Lou, W.Y.; Li, C.X.; Bai, Y.P.; Li, A.T.; Xu, J.H. Preparation of Structurally Diverse Chiral Alcohols by Engineering Ketoreductase CgKR1. ACS Catal. 2017, 7, 7174–7181. [Google Scholar] [CrossRef]
- Brezovsky, J.; Babkova, P.; Degtjarik, O.; Fortova, A.; Gora, A.; Iermak, I.; Rezacova, P.; Dvorak, P.; Smatanova, I.K.; Prokop, Z.; et al. Engineering a de Novo Transport Tunnel. ACS Catal. 2016, 6, 7597–7610. [Google Scholar] [CrossRef]
- Kokkonen, P.; Bednar, D.; Pinto, G.; Prokop, Z.; Damborsky, J. Engineering enzyme access tunnels. Biotechnol. Adv. 2019, 37, 107386. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.K.; Nixon, P.F.; Duggleby, R.G. Effects of deletions at the carboxyl terminus of Zymomonas mobilis pyruvate decarboxylase on the kinetic properties and substrate specificity. Biochemistry 2000, 39, 9430–9437. [Google Scholar] [CrossRef] [PubMed]
- Knoll, M.; Muller, M.; Pleiss, J.; Pohl, M. Factors mediating activity, selectivity, and substrate specificity for the thiamin diphosphate-dependent enzymes benzaldehyde lyase and benzoylformate decarboxylase. Chembiochem 2006, 7, 1928–1934. [Google Scholar] [CrossRef]
- Rother, D.; Kolter, G.; Gerhards, T.; Berthold, C.; Gauchenova, E.; Knoll, M.; Pleiss, J.; Muller, M.; Schneider, G.; Pohl, M. S-Selective mixed carboligation by structure-based design of the pyruvate decarboxylase from Acetobacter pasteurianus. Chemcatchem 2011, 3, 1587–1596. [Google Scholar] [CrossRef]
- Cui, H.; Zhang, L.; Eltoukhy, L.; Jiang, Q.; Korkunc, S.; Jaeger, K.; Schwaneberg, U.; Davari, M. Enzyme hydration determines resistance in organic cosolvents. ACS Catal. 2020, 10, 14847–14856. [Google Scholar] [CrossRef]
- Cui, H.; Eltoukhy, L.; Zhang, L.; Markel, U.; Jaeger, K.; Davari, M.; Schwaneberg, U. Less unfavorable salt bridegs on the enzyme surface result in more organic cosolvent resistance. Angew. Chem. Int. Edit. 2021, 60, 11448–11456. [Google Scholar] [CrossRef] [PubMed]
- Yi, T.; Lim, H.; Lee, S.; Lee, K.; Kim, D. Synthesis of (R,R)-2,3-butanediol from starch in a hybrid cell-free reaction system. J. Ind. Eng. Chem. 2018, 67, 231–235. [Google Scholar] [CrossRef]
- Zheng, L.; Baumann, U.; Reymond, J.L. An efficient one-step-directed and site-saturation mutagenesis protocol. Nucleic Acids Res. 2004, 32, e115. [Google Scholar] [CrossRef] [PubMed]




| Enzyme | Km (mM) a | kcat (s−1) a | kcat/Km(s−1 M−1) | Ref. |
|---|---|---|---|---|
| FLS | 58.46 ± 2.32 | 0.45 ± 0.04 | 7.69 | This study |
| FLS:I28V | 81.95 ± 3.61 | 3.33 ± 0.21 | 40.59 | This study |
| FLS:L482E | 50.95 ± 3.22 | 0.49 ± 0.06 | 9.66 | This study |
| FLS:F484A | 53.28 ± 2.52 | 1.74 ± 0.12 | 32.56 | This study |
| FLS:I28V/L482E | 375.55 ± 6.84 | 8.76 ± 0.13 | 23.34 | This study |
| FLS:L482S | 47.45 ± 1.20 | 0.63 ± 0.01 | 13.30 | [27] |
| Enzyme | Method | Substrate | 2,3-BD (g/L) | Ref. |
|---|---|---|---|---|
| The lysate from E. coli expressing ALS/ALDC/BDH | Cell-free | Glucose | 82.00 | [10] |
| BDH, NOX and Vitreoscilla hemoglobin | Whole-cell | Acetoin | 38.41 | [11] |
| E. coli expressing BDH | Whole-cell | Diacetyl | 26.80 | [13] |
| Bacillus subtilis expressing acetoin reductase and NOX | Whole-cell | Acetoin | 74.50 | [14] |
| The lysates from E. coli expressing ALS/ALDC/BDH and cyanobacteria | Cell-free | Starch | 13.52 | [42] |
| EtDH:D46G, FLS:I28V/L482E, NOX and BDH:S199A | Cell-free | Ethanol | 124.83 | This study |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Lin, H.; Zheng, C.; Yang, B.; Liang, M.; Lin, Y.; Zhang, L. Efficient 2,3-Butanediol Production from Ethanol by a Modified Four-Enzyme Synthetic Biosystem. Molecules 2024, 29, 3934. https://doi.org/10.3390/molecules29163934
Zhang J, Lin H, Zheng C, Yang B, Liang M, Lin Y, Zhang L. Efficient 2,3-Butanediol Production from Ethanol by a Modified Four-Enzyme Synthetic Biosystem. Molecules. 2024; 29(16):3934. https://doi.org/10.3390/molecules29163934
Chicago/Turabian StyleZhang, Jiming, Hui Lin, Chaosong Zheng, Bin Yang, Miao Liang, Yi Lin, and Liaoyuan Zhang. 2024. "Efficient 2,3-Butanediol Production from Ethanol by a Modified Four-Enzyme Synthetic Biosystem" Molecules 29, no. 16: 3934. https://doi.org/10.3390/molecules29163934
APA StyleZhang, J., Lin, H., Zheng, C., Yang, B., Liang, M., Lin, Y., & Zhang, L. (2024). Efficient 2,3-Butanediol Production from Ethanol by a Modified Four-Enzyme Synthetic Biosystem. Molecules, 29(16), 3934. https://doi.org/10.3390/molecules29163934







