Recent Advances in Preclinical Studies of the Theranostic Agent [64Cu]CuCl2
Abstract
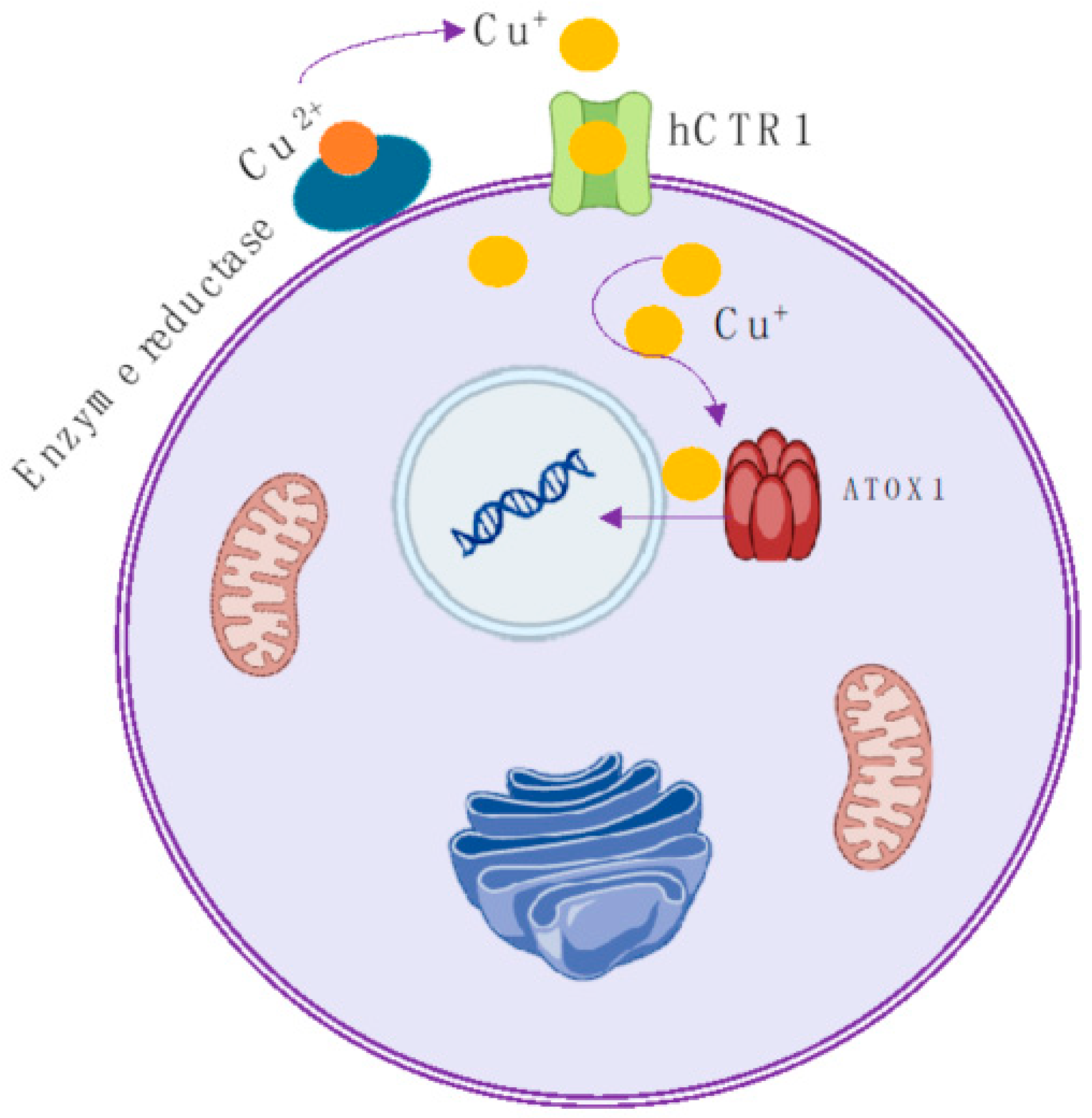
1. Introduction
2. Copper Radiochemistry
2.1. 64Cu Production
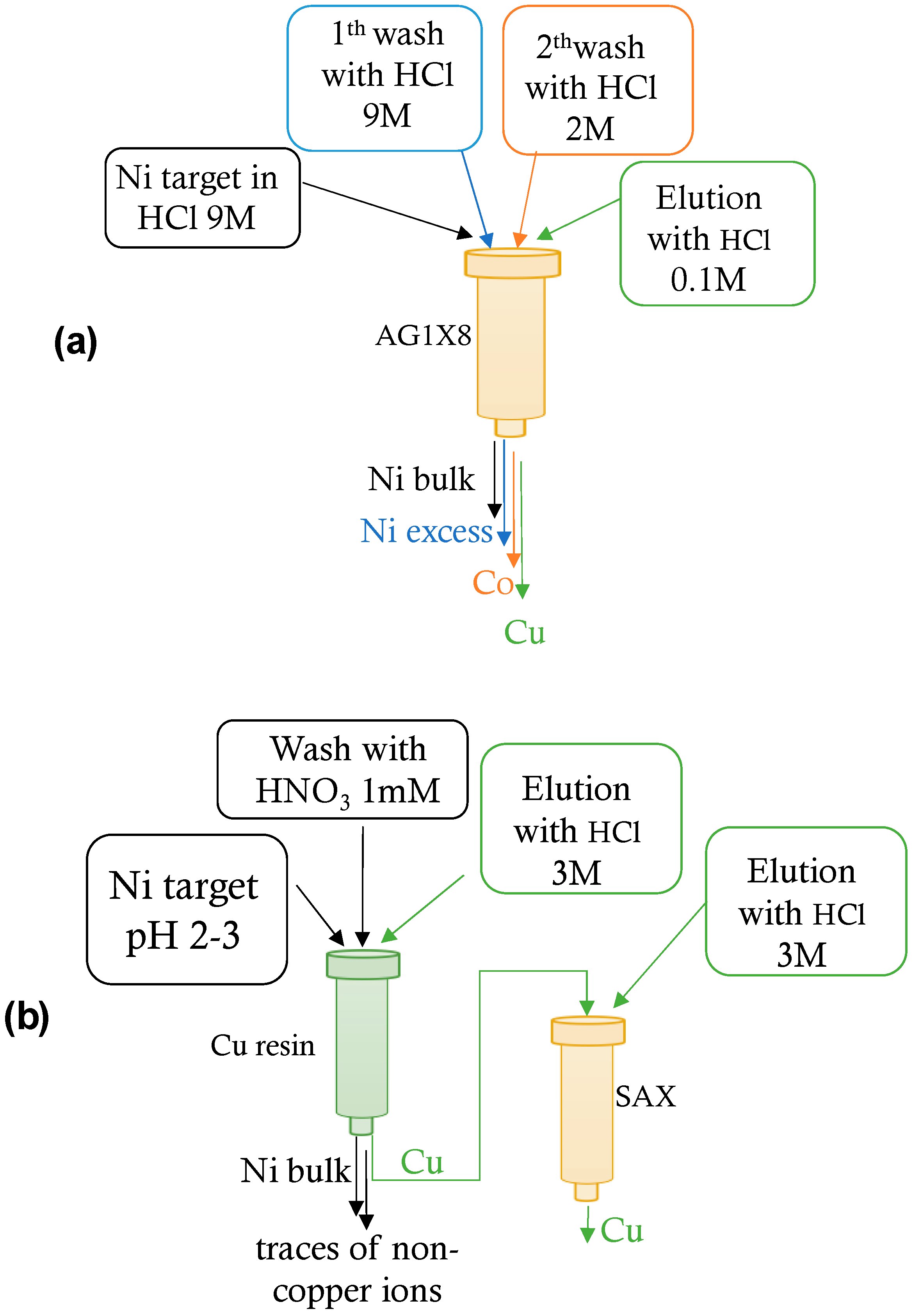
2.2. 64Cu Separation and Purification
3. Preclinical Studies
3.1. Prostate Cancer
3.2. Breast Cancer
3.3. Glioblastoma
3.4. Melanoma
3.5. Wilson’s Disease
3.6. Lung Cancer
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Peng, F. Recent Advances in Cancer Imaging with 64CuCl2 PET/CT. Nucl. Med. Mol. Imaging 2022, 56, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Solomons, N.W. Biochemical, Metabolic, and Clinical Role of Copper in Human Nutrition. J. Am. Coll. Nutr. 1985, 4, 83–105. [Google Scholar] [CrossRef]
- Gutfilen, B.; Souza, S.; Valentini, G. Copper-64: A Real Theranostic Agent. Drug Des. Dev. Ther. 2018, 12, 3235–3245. [Google Scholar] [CrossRef] [PubMed]
- Hatori, Y.; Lutsenko, S. The Role of Copper Chaperone Atox1 in Coupling Redox Homeostasis to Intracellular Copper Distribution. Antioxidants 2016, 5, 25. [Google Scholar] [CrossRef]
- Wang, H.; Chen, X. Visualization of Copper Metabolism by 64CuCl2-PET. Mol. Imaging Biol. 2012, 14, 14–16. [Google Scholar] [CrossRef][Green Version]
- Todde, S.; Boschi, S. Compendio di Radiochimica e Radiofarmacia; Minerva Medica: Torino, Italy, 2020; ISBN 978-88-553-2035-1. [Google Scholar]
- Boschi, A.; Martini, P.; Janevik-Ivanovska, E.; Duatti, A. The Emerging Role of Copper-64 Radiopharmaceuticals as Cancer Theranostics. Drug Discov. Today 2018, 23, 1489–1501. [Google Scholar] [CrossRef]
- Hao, G.; Singh, A.N.; Oz, O.K.; Sun, X. Recent Advances in Copper Radiopharmaceuticals. Curr. Radiopharm. 2011, 4, 109–121. [Google Scholar] [CrossRef] [PubMed]
- Qaim, S.M.; Scholten, B.; Neumaier, B. New Developments in the Production of Theranostic Pairs of Radionuclides. J. Radioanal. Nucl. Chem. 2018, 318, 1493–1509. [Google Scholar] [CrossRef]
- Jalilian, A.R.; Osso, J.A., Jr.; Vera-Araujo, J.; Kumar, V.; Harris, M.J.; Gutfilen, B.; Guérin, B.; Li, H.; Zhuravlev, F.; Chakravarty, R.; et al. IAEA Contribution to the Development of 64Cu Radiopharmaceuticals for Theranostic Applications. Q. J. Nucl. Med. Mol. Imaging 2020, 64, 338–345. [Google Scholar] [CrossRef]
- Alves, F.; Alves, V.H.P.; Do Carmo, S.J.C.; Neves, A.C.B.; Silva, M.; Abrunhosa, A.J. Production of Copper-64 and Gallium-68 with a Medical Cyclotron Using Liquid Targets. Mod. Phys. Lett. A 2017, 32, 1740013. [Google Scholar] [CrossRef]
- Wadas, T.; Wong, E.; Weisman, G.; Anderson, C. Copper Chelation Chemistry and Its Role in Copper Radiopharmaceuticals. Curr. Pharm. Des. 2007, 13, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Fiedler, L.; Kellner, M.; Oos, R.; Böning, G.; Ziegler, S.; Bartenstein, P.; Zeidler, R.; Gildehaus, F.J.; Lindner, S. Fully Automated Production and Characterization of 64Cu and Proof-of-Principle Small-Animal PET Imaging Using 64Cu-Labelled CA XII Targeting 6A10 Fab. ChemMedChem 2018, 13, 1230–1237. [Google Scholar] [CrossRef] [PubMed]
- Jauregui-Osoro, M.; De Robertis, S.; Halsted, P.; Gould, S.-M.; Yu, Z.; Paul, R.L.; Marsden, P.K.; Gee, A.D.; Fenwick, A.; Blower, P.J. Production of Copper-64 Using a Hospital Cyclotron: Targetry, Purification and Quality Analysis. Nucl. Med. Commun. 2021, 42, 1024–1038. [Google Scholar] [CrossRef] [PubMed]
- Moore, G.E.; Kraus, K.A. Anion Exchange Studies. IV. 1,2Cobalt and Nickel in Hydrochloric Acid Solutions. J. Am. Chem. Soc. 1952, 74, 843–844. [Google Scholar] [CrossRef]
- Ohya, T.; Minegishi, K.; Suzuki, H.; Nagatsu, K.; Fukada, M.; Hanyu, M.; Zhang, M.-R. Development of a Remote Purification Apparatus with Disposable Evaporator for the Routine Production of High-Quality 64Cu for Clinical Use. Appl. Radiat. Isot. 2019, 146, 127–132. [Google Scholar] [CrossRef]
- Alves, V.H.; Do Carmo, S.J.C.; Alves, F.; Abrunhosa, A.J. Automated Purification of Radiometals Produced by Liquid Targets. Instruments 2018, 2, 17. [Google Scholar] [CrossRef]
- Hicks, R.J.; Jackson, P.; Kong, G.; Ware, R.E.; Hofman, M.S.; Pattison, D.A.; Akhurst, T.A.; Drummond, E.; Roselt, P.; Callahan, J.; et al. 64Cu-SARTATE PET Imaging of Patients with Neuroendocrine Tumors Demonstrates High Tumor Uptake and Retention, Potentially Allowing Prospective Dosimetry for Peptide Receptor Radionuclide Therapy. J. Nucl. Med. 2019, 60, 777–785. [Google Scholar] [CrossRef]
- Manrique-Arias, J.C.; Carrasco-Hernández, J.; Reyes, P.G.; Ávila-Rodríguez, M.A. Biodistribution in Rats and Estimates of Doses to Humans from 64CuCl2, a Potential Theranostic Tracer. Appl. Radiat. Isot. 2016, 115, 18–22. [Google Scholar] [CrossRef]
- Jørgensen, J.T.; Persson, M.; Madsen, J.; Kjær, A. High Tumor Uptake of 64Cu: Implications for Molecular Imaging of Tumor Characteristics with Copper-Based PET Tracers. Nucl. Med. Biol. 2013, 40, 345–350. [Google Scholar] [CrossRef]
- Capasso, E.; Durzu, S.; Piras, S.; Zandieh, S.; Knoll, P.; Haug, A.; Hacker, M.; Meleddu, C.; Mirzaei, S. Role of 64CuCl2 PET/CT in Staging of Prostate Cancer. Ann. Nucl. Med. 2015, 29, 482–488. [Google Scholar] [CrossRef]
- Paparo, F.; Peirano, A.; Matos, J.; Bacigalupo, L.; Rossi, U.; Mussetto, I.; Bottoni, G.; Ugolini, M.; Introini, C.; Ruggieri, F.G.; et al. Diagnostic Value of Retrospectively Fused 64CuCl2 PET/MRI in Biochemical Relapse of Prostate Cancer: Comparison with Fused 18F-Choline PET/MRI, 64CuCl2 PET/CT, 18F-Choline PET/CT, and mpMRI. Abdom. Radiol. 2020, 45, 3896–3906. [Google Scholar] [CrossRef] [PubMed]
- Peng, F.; Lu, X.; Janisse, J.; Muzik, O.; Shields, A.F. PET of Human Prostate Cancer Xenografts in Mice with Increased Uptake of 64CuCl2. J. Nucl. Med. 2006, 47, 1649–1652. [Google Scholar] [PubMed]
- Peng, F.; Liu, J.; Wu, J.; Lu, X.; Muzik, O. Mouse Extrahepatic Hepatoma Detected on MicroPET Using Copper (II)-64 Chloride Uptake Mediated by Endogenous Mouse Copper Transporter 1. Mol. Imaging Biol. 2005, 7, 325–329. [Google Scholar] [CrossRef] [PubMed]
- Cai, H.; Wu, J.; Muzik, O.; Hsieh, J.-T.; Lee, R.J.; Peng, F. Reduced 64Cu Uptake and Tumor Growth Inhibition by Knockdown of Human Copper Transporter 1 in Xenograft Mouse Model of Prostate Cancer. J. Nucl. Med. 2014, 55, 622–628. [Google Scholar] [CrossRef]
- Ishida, S.; Lee, J.; Thiele, D.J.; Herskowitz, I. Uptake of the Anticancer Drug Cisplatin Mediated by the Copper Transporter Ctr1 in Yeast and Mammals. Proc. Natl. Acad. Sci. USA 2002, 99, 14298–14302. [Google Scholar] [CrossRef]
- Guerreiro, J.; Alves, V.; Abrunhosa, A.; Paulo, A.; Gil, O.; Mendes, F. Radiobiological Characterization of 64CuCl2 as a Simple Tool for Prostate Cancer Theranostics. Molecules 2018, 23, 2944. [Google Scholar] [CrossRef]
- Pinto, C.I.G.; Bucar, S.; Alves, V.; Fonseca, A.; Abrunhosa, A.J.; Da Silva, C.L.; Guerreiro, J.F.; Mendes, F. Copper-64 Chloride Exhibits Therapeutic Potential in Three-Dimensional Cellular Models of Prostate Cancer. Front. Mol. Biosci. 2020, 7, 609172. [Google Scholar] [CrossRef]
- Serban, R.M.; Niculae, D.; Manda, G.; Neagoe, I.; Dobre, M.; Niculae, D.A.; Temelie, M.; Mustăciosu, C.; Leonte, R.A.; Chilug, L.E.; et al. Modifications in Cellular Viability, DNA Damage and Stress Responses Inflicted in Cancer Cells by Copper-64 Ions. Front. Med. 2023, 10, 1197846. [Google Scholar] [CrossRef]
- Kim, K.I.; Jang, S.J.; Park, J.H.; Lee, Y.J.; Lee, T.S.; Woo, K.S.; Park, H.; Choe, J.G.; An, G.I.; Kang, J.H. Detection of Increased 64Cu Uptake by Human Copper Transporter 1 Gene Overexpression Using PET with 64CuCl2 in Human Breast Cancer Xenograft Model. J. Nucl. Med. 2014, 55, 1692–1698. [Google Scholar] [CrossRef]
- Reardon, D.A.; Rich, J.N.; Friedman, H.S.; Bigner, D.D. Recent Advances in the Treatment of Malignant Astrocytoma. J. Clin. Oncol. 2006, 24, 1253–1265. [Google Scholar] [CrossRef]
- Cilliers, K.; Muller, C.J.F.; Page, B.J. Trace Element Concentration Changes in Brain Tumors: A Review. Anat. Rec. 2020, 303, 1293–1299. [Google Scholar] [CrossRef]
- Pinto, C.I.G.; Branco, A.D.M.; Bucar, S.; Fonseca, A.; Abrunhosa, A.J.; Da Silva, C.L.; Guerreiro, J.F.; Mendes, F. Evaluation of the Theranostic Potential of [64Cu]CuCl2 in Glioblastoma Spheroids. EJNMMI Res. 2024, 14, 26. [Google Scholar] [CrossRef] [PubMed]
- Ren, G.; Pan, Y.; Cheng, Z. Molecular Probes for Malignant Melanoma Imaging. Curr. Pharm. Biotechnol. 2010, 11, 590–602. [Google Scholar] [CrossRef]
- Qin, C.; Liu, H.; Chen, K.; Hu, X.; Ma, X.; Lan, X.; Zhang, Y.; Cheng, Z. Theranostics of Malignant Melanoma with 64CuCl2. J. Nucl. Med. 2014, 55, 812–817. [Google Scholar] [CrossRef]
- Jiang, L.; Tu, Y.; Hu, X.; Bao, A.; Chen, H.; Ma, X.; Doyle, T.; Shi, H.; Cheng, Z. Pilot Study of 64Cu(I) for PET Imaging of Melanoma. Sci. Rep. 2017, 7, 2574. [Google Scholar] [CrossRef] [PubMed]
- Prodan, C.I.; Holland, N.R.; Wisdom, P.J.; Burstein, S.A.; Bottomley, S.S. CNS Demyelination Associated with Copper Deficiency and Hyperzincemia. Neurology 2002, 59, 1453–1456. [Google Scholar] [CrossRef]
- Tan, J.C.; Burns, D.L.; Jones, H.R. Severe Ataxia, Myelopathy, and Peripheral Neuropathy Due to Acquired Copper Deficiency in a Patient With History of Gastrectomy. J. Parenter. Enter. Nutr. 2006, 30, 446–450. [Google Scholar] [CrossRef] [PubMed]
- Goez, H.R.; Jacob, F.D.; Fealey, R.D.; Patterson, M.C.; Ramaswamy, V.; Persad, R.; Johnson, E.S.; Yager, J.Y. An Unusual Presentation of Copper Metabolism Disorder and a Possible Connection With Niemann-Pick Type C. J. Child. Neurol. 2011, 26, 518–521. [Google Scholar] [CrossRef]
- Khaleeli, Z.; Healy, D.G.; Briddon, A.; Lunn, M.P.; Reilly, M.M.; Land, J.; Giovannoni, G. Copper Deficiency as a Treatable Cause of Poor Balance. BMJ 2010, 340, c508. [Google Scholar] [CrossRef]
- Juhasz-Pocsine, K.; Rudnicki, S.A.; Archer, R.L.; Harik, S.I. Neurologic Complications of Gastric Bypass Surgery for Morbid Obesity. Neurology 2007, 68, 1843–1850. [Google Scholar] [CrossRef]
- Cartwright, G.E.; Wintrobe, M.M. Copper Metabolism in Normal Subjects. Am. J. Clin. Nutr. 1964, 14, 224–232. [Google Scholar] [CrossRef]
- Turnlund, J. Human Whole-Body Copper Metabolism. Am. J. Clin. Nutr. 1998, 67, 960S–964S. [Google Scholar] [CrossRef]
- Peng, F.; Lutsenko, S.; Sun, X.; Muzik, O. Imaging Copper Metabolism Imbalance in Atp7b−/− Knockout Mouse Model of Wilson’s Disease with PET-CT and Orally Administered 64CuCl2. Mol. Imaging Biol. 2012, 14, 600–607. [Google Scholar] [CrossRef] [PubMed]
- Xie, F.; Xi, Y.; Pascual, J.M.; Muzik, O.; Peng, F. Age-Dependent Changes of Cerebral Copper Metabolism in Atp7b−/− Knockout Mouse Model of Wilson’s Disease by [64Cu]CuCl2-PET/CT. Metab. Brain Dis. 2017, 32, 717–726. [Google Scholar] [CrossRef] [PubMed]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global Cancer Statistics 2018: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA A Cancer J Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Song, D.; Ma, X.; Wu, X.; Jiang, L. Preclinical PET Imaging Study of Lung Cancer with 64CuCl2. Ann. Nucl. Med. 2020, 34, 653–662. [Google Scholar] [CrossRef]
- García-Pérez, F.O.; Medina-Ornelas, S.S.; Barron-Barron, F.; Arrieta-Rodriguez, O. Evaluation of Non-Small Cell Lung Cancer by PET/CT with 64CuCl2: Initial Experience in Humans. Am. J. Nucl. Med. Mol. Imaging 2020, 10, 143. [Google Scholar]
| Radionuclide | T1/2 | Eβmean (keV, int.%) | Eγ (keV, int.%) | E Auger (keV, int.%) |
|---|---|---|---|---|
| 61Cu | 3.33 h | 500 (61) | 283 (12) | 0.84 (52) |
| 511 (123) | 6.54 (20) | |||
| 656 (11) | ||||
| 1185 (4) | ||||
| 64Cu | 12.7 h | β+, 278 (17.6) β−, 19 (38.5) | 511 (32) | 0.84 (58) |
| 1346 (0.5) | 6.54 (22) | |||
| 67Cu | 61.8 h | β−, 141 (11) | 91 (7) | 0.99 (19) |
| 93 (16) | 7.53 (7) | |||
| 185 (49) | 83.65 (12) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Speltri, G.; Porto, F.; Boschi, A.; Uccelli, L.; Martini, P. Recent Advances in Preclinical Studies of the Theranostic Agent [64Cu]CuCl2. Molecules 2024, 29, 4085. https://doi.org/10.3390/molecules29174085
Speltri G, Porto F, Boschi A, Uccelli L, Martini P. Recent Advances in Preclinical Studies of the Theranostic Agent [64Cu]CuCl2. Molecules. 2024; 29(17):4085. https://doi.org/10.3390/molecules29174085
Chicago/Turabian StyleSpeltri, Giorgia, Francesca Porto, Alessandra Boschi, Licia Uccelli, and Petra Martini. 2024. "Recent Advances in Preclinical Studies of the Theranostic Agent [64Cu]CuCl2" Molecules 29, no. 17: 4085. https://doi.org/10.3390/molecules29174085
APA StyleSpeltri, G., Porto, F., Boschi, A., Uccelli, L., & Martini, P. (2024). Recent Advances in Preclinical Studies of the Theranostic Agent [64Cu]CuCl2. Molecules, 29(17), 4085. https://doi.org/10.3390/molecules29174085










