Development and Validation of an LC-MS/MS Assay for the Quantitation of MO-OH-Nap Tropolone in Mouse Plasma: Application to In Vitro and In Vivo Pharmacokinetic Studies
Abstract
1. Introduction
2. Results
2.1. Mass Spectrometric and Chromatographic Optimization
2.2. Optimization of Biosamples Extraction
2.3. Method Validation
2.3.1. Method Specificity and Sensitivity
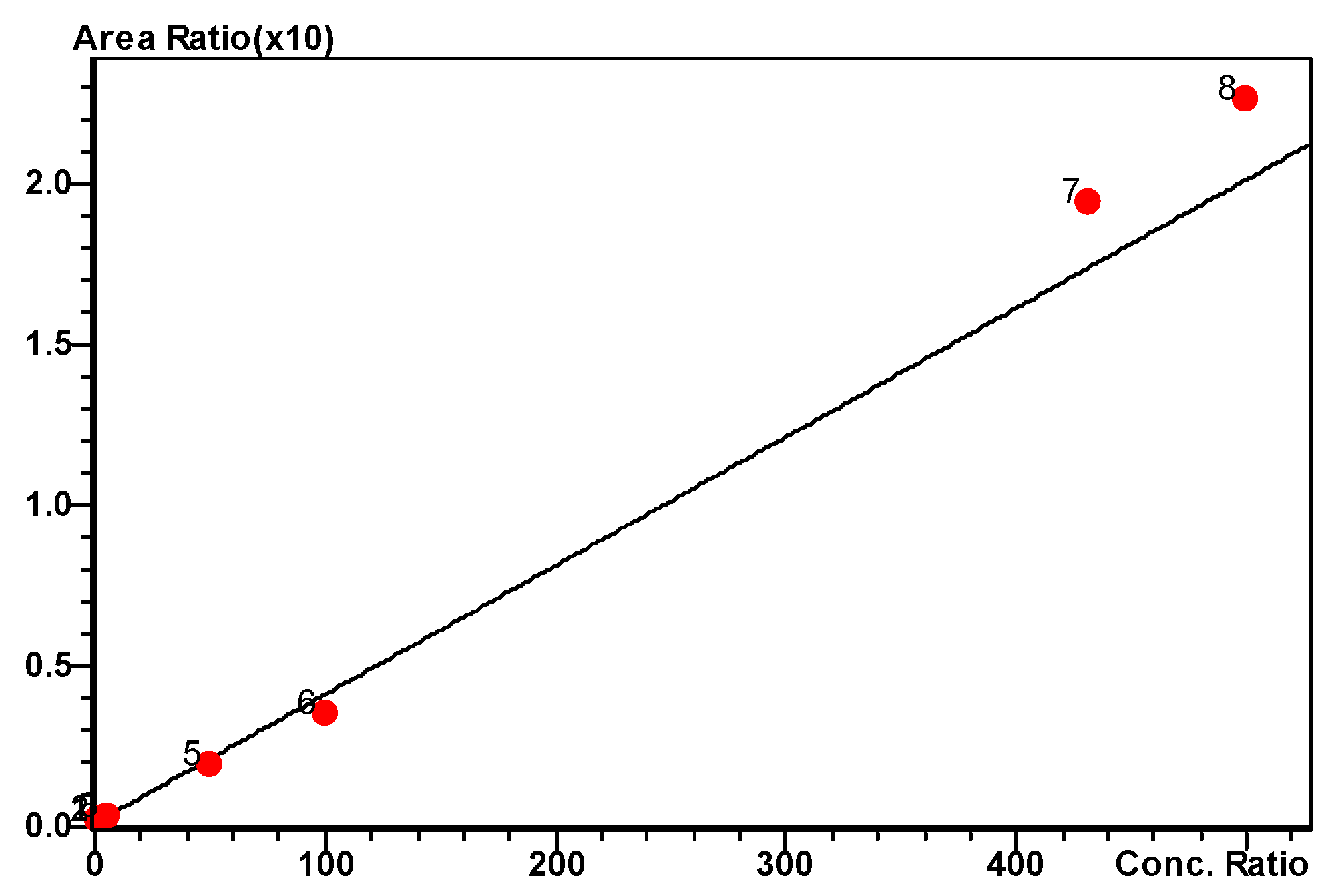
2.3.2. Standard Curve and Linearity
2.3.3. Carry-Over
2.3.4. Accuracy and Precision
2.3.5. Recovery and Matrix Effect
2.3.6. Stability
2.3.7. Dilution Integrity
2.4. In Vitro Studies
2.4.1. Solubility of MO-OH-Nap
2.4.2. Blood to Plasma Ratio (B/P) of MO-OH-Nap
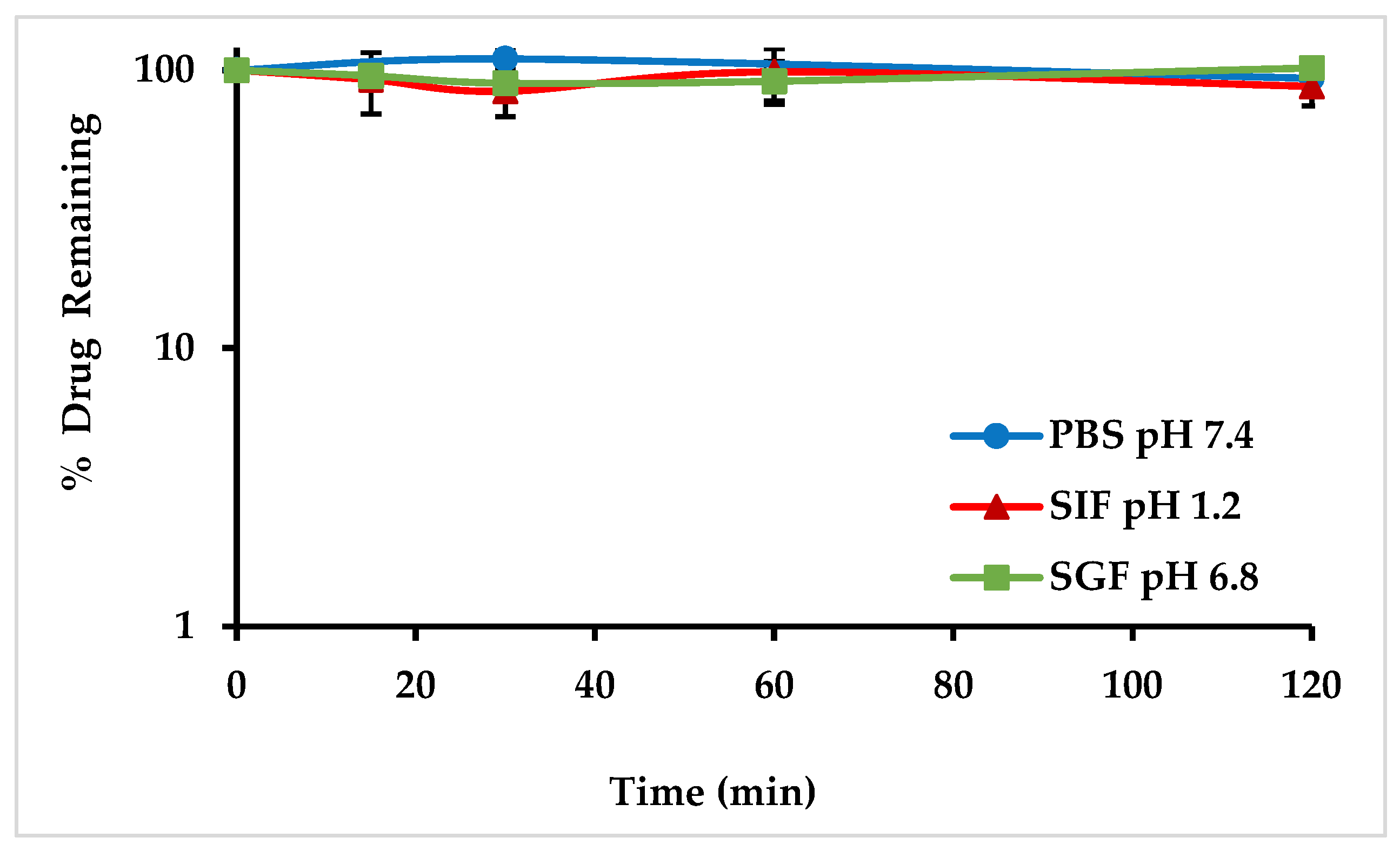
2.4.3. Gastrointestinal Fluids Stability Studies of MO-OH-Nap
2.4.4. Plasma Protein Binding (PPB) Study
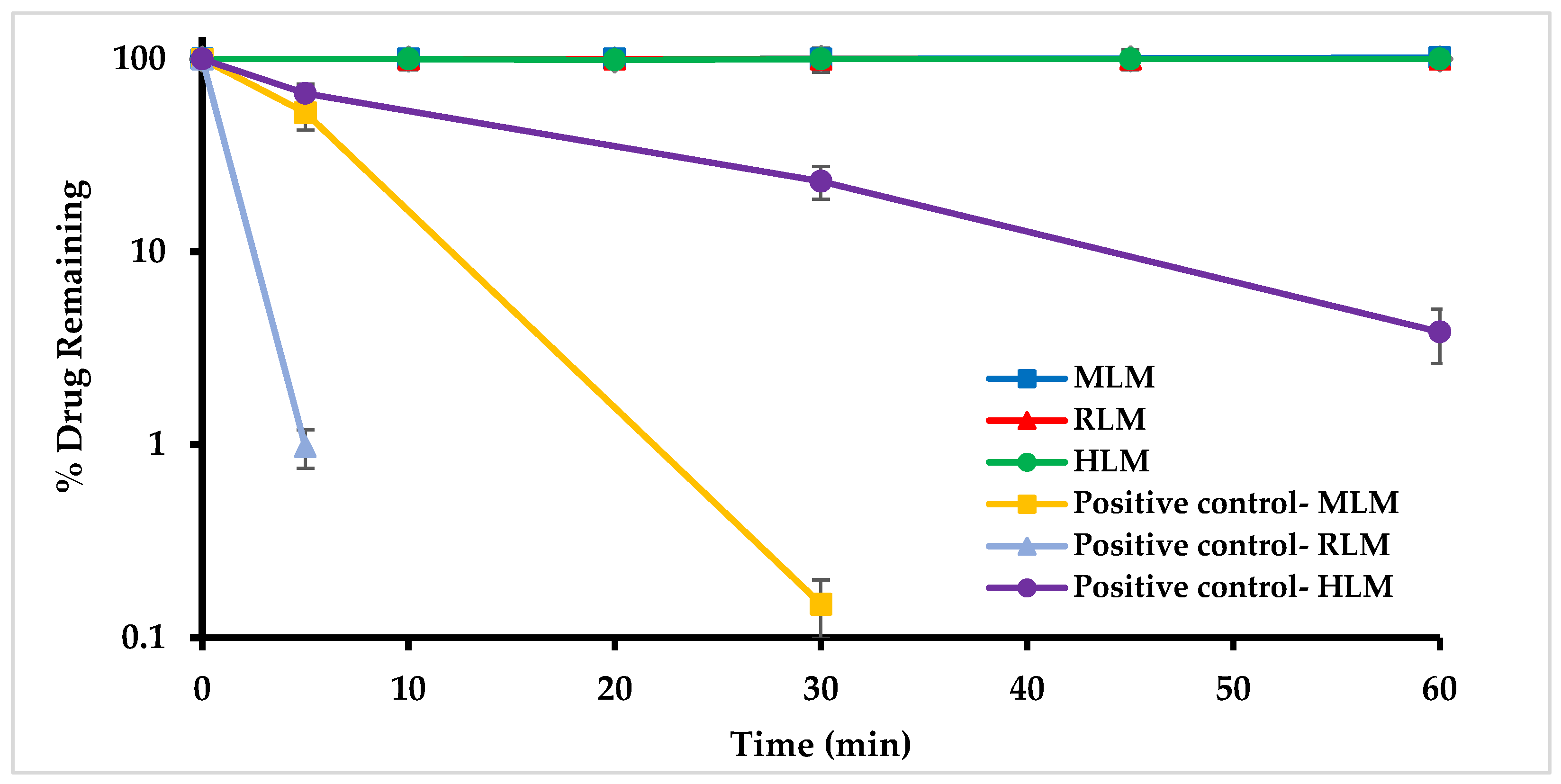
2.4.5. In Vitro Metabolic Stability in Mouse, Rat, and Human Liver Microsomes
2.5. In Vivo Pharmacokinetic Studies
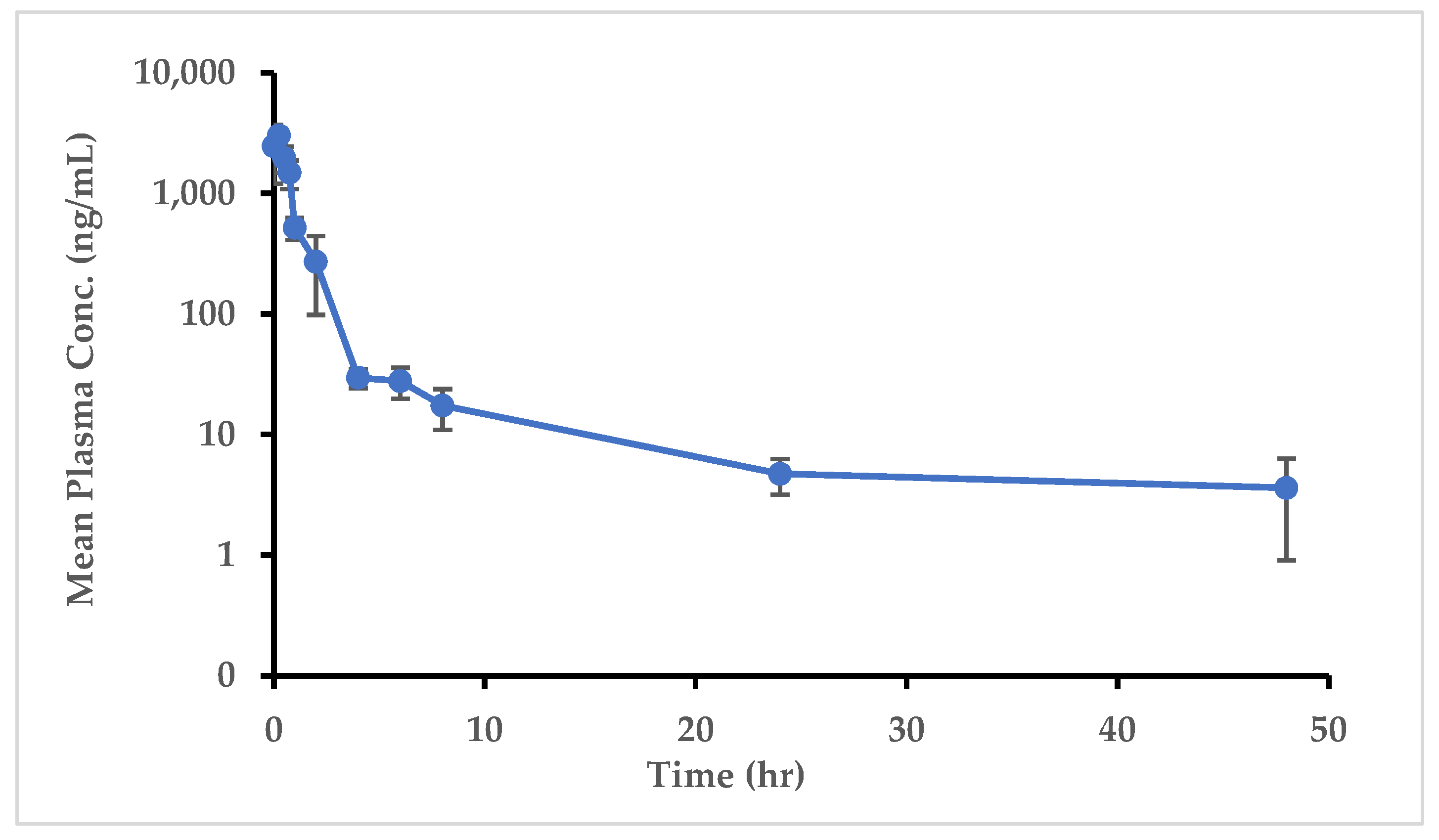
2.5.1. Application of Analytical Method to Pharmacokinetic Animal Study
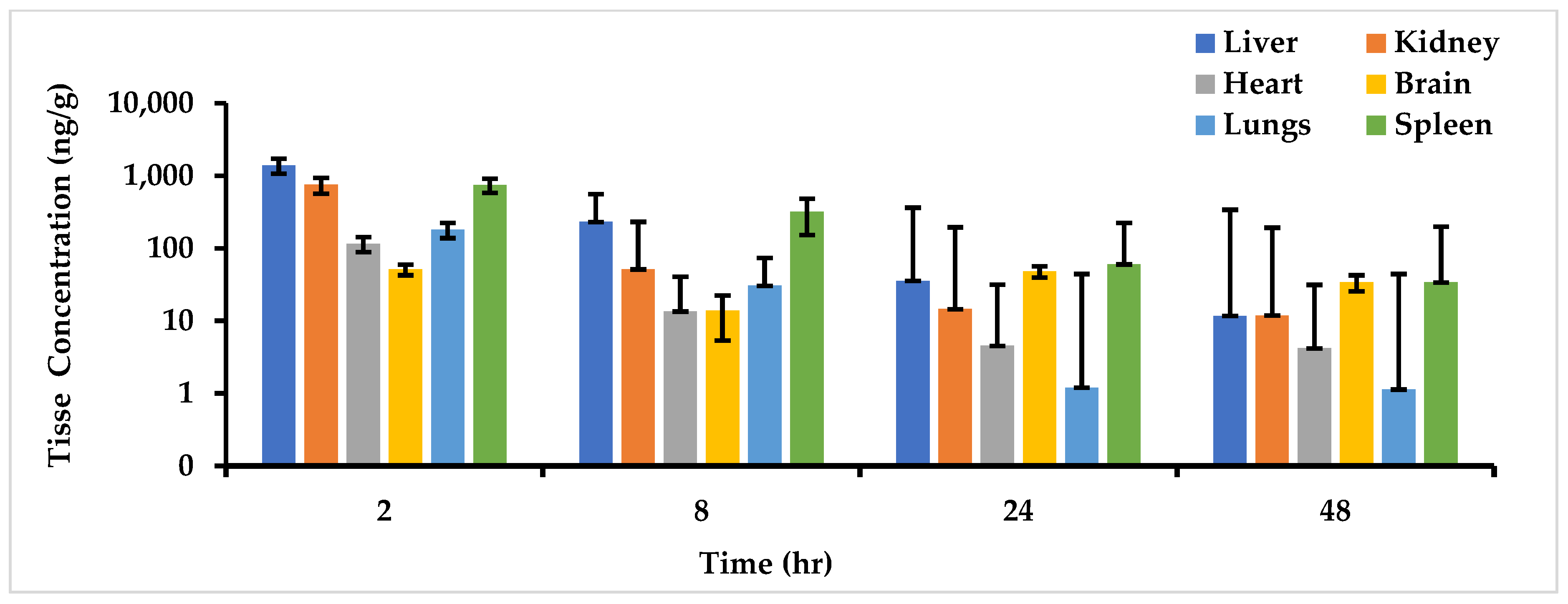
2.5.2. Tissue Distribution Study
3. Discussion
4. Materials and Methods
4.1. Mass Spectrometric and Chromatographic Conditions Optimization
4.2. Liquid Chromatographic and Mass Spectrometric (LC-MS/MS) Conditions for MO-OH-Nap
4.3. Preparation of Stock, Calibration Standards and Quality Control Samples
4.4. Sample Preparation and Extraction Method
4.5. Method Validation
4.5.1. Recovery and Matrix Effect
4.5.2. Stability
4.5.3. Dilution Integrity
4.6. In Vitro Studies
4.6.1. Aqueous Solubility of MO-OH-Nap
4.6.2. Blood to Plasma Ratio (B/P) of MO-OH-Nap
4.6.3. Gastrointestinal Fluid Stability Studies
4.6.4. Plasma Protein Binding (PPB) Study
4.6.5. In Vitro Metabolic Stability
4.7. In Vivo Pharmacokinetic Studies
4.7.1. Biodistribution Study, Animals, Drug Administration and Sampling
4.7.2. Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhao, J. Plant troponoids: Chemistry, biological activity, and biosynthesis. Curr. Med. Chem. 2007, 14, 2597–2621. [Google Scholar] [CrossRef] [PubMed]
- Ononye, S.N.; Vanheyst, M.D.; Giardina, C.; Wright, D.L.; Anderson, A.C. Studies on the antiproliferative effects of tropolone derivatives in Jurkat T-lymphocyte cells. Bioorganic Med. Chem. 2014, 22, 2188–2193. [Google Scholar] [CrossRef]
- Cao, F.; Orth, C.; Donlin, M.J.; Adegboyega, P.; Meyers, M.J.; Murelli, R.P.; Elagawany, M.; Elgendy, B.; Tavis, J.E. Synthesis and Evaluation of Troponoids as a New Class of Antibiotics. ACS Omega 2018, 3, 15125–15133. [Google Scholar] [CrossRef]
- Sugawara, K.; Ohbayashi, M.; Shimizu, K.; Hatori, M.; Kamei, H.; Konishi, M.; Oki, T.; Kawaguchi, H. BMY-28438 (3,7-dihydroxytropolone), a new antitumor antibiotic active against B16 melanoma. I. Production, isolation, structure and biological activity. J. Antibiot. 1988, 41, 862–868. [Google Scholar] [CrossRef] [PubMed]
- Tomita, K.; Hoshino, Y.; Nakakita, Y.; Umezawa, S.; Miyaki, T.; Oki, T.; Kawaguchi, H. BMY-28438 (3,7-dihydroxytropolone), a new antitumor antibiotic active against B16 melanoma. II. Taxonomy of producing organism. J. Antibiot. 1989, 42, 317–321. [Google Scholar] [CrossRef][Green Version]
- Li, J.; Falcone, E.R.; Holstein, S.A.; Anderson, A.C.; Wright, D.L.; Wiemer, A.J. Novel α-substituted tropolones promote potent and selective caspase-dependent leukemia cell apoptosis. Pharmacol. Res. 2016, 113, 438–448. [Google Scholar] [CrossRef][Green Version]
- Liu, S.; Yamauchi, H. Hinokitiol, a metal chelator derived from natural plants, suppresses cell growth and disrupts androgen receptor signaling in prostate carcinoma cell lines. Biochem. Biophys. Res. Commun. 2006, 351, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Zhu, H.; Wu, W.; Shen, Y.; Lin, X.; Wu, Y.; Liu, L.; Tang, J.; Zhou, Y.; Sun, F.; et al. Neoantimycin F, a Streptomyces-Derived Natural Product Induces Mitochondria-Related Apoptotic Death in Human Non-Small Cell Lung Cancer Cells. Front. Pharmacol. 2019, 10, 1042. [Google Scholar] [CrossRef] [PubMed]
- Hsiao, C.J.; Hsiao, S.H.; Chen, W.L.; Guh, J.H.; Hsiao, G.; Chan, Y.J.; Lee, T.H.; Chung, C.L. Pycnidione, a fungus-derived agent, induces cell cycle arrest and apoptosis in A549 human lung cancer cells. Chem. Biol. Interact. 2012, 197, 23–30. [Google Scholar] [CrossRef]
- Chen, S.M.; Wang, B.Y.; Lee, C.H.; Lee, H.T.; Li, J.J.; Hong, G.C.; Hung, Y.C.; Chien, P.J.; Chang, C.Y.; Hsu, L.S.; et al. Hinokitiol up-regulates miR-494-3p to suppress BMI1 expression and inhibits self-renewal of breast cancer stem/progenitor cells. Oncotarget 2017, 8, 76057–76068. [Google Scholar] [CrossRef]
- Huang, C.-H.; Lu, S.-H.; Chang, C.-C.; Thomas, P.A.; Jayakumar, T.; Sheu, J.-R. Hinokitiol, a tropolone derivative, inhibits mouse melanoma (B16-F10) cell migration and in vivo tumor formation. Eur. J. Pharmacol. 2015, 746, 148–157. [Google Scholar] [CrossRef]
- Huang, C.H.; Jayakumar, T.; Chang, C.C.; Fong, T.H.; Lu, S.H.; Thomas, P.A.; Choy, C.S.; Sheu, J.R. Hinokitiol Exerts Anticancer Activity through Downregulation of MMPs 9/2 and Enhancement of Catalase and SOD Enzymes: In Vivo Augmentation of Lung Histoarchitecture. Molecules 2015, 20, 17720–17734. [Google Scholar] [CrossRef] [PubMed]
- Nozoe, T. Substitution products of tropolone and allied compounds. Nature 1951, 167, 1055–1057. [Google Scholar] [CrossRef] [PubMed]
- Ononye, S.N.; VanHeyst, M.D.; Oblak, E.Z.; Zhou, W.; Ammar, M.; Anderson, A.C.; Wright, D.L. Tropolones as lead-like natural products: The development of potent and selective histone deacetylase inhibitors. ACS Med. Chem. Lett. 2013, 4, 757–761. [Google Scholar] [CrossRef]
- Haney, S.L.; Allen, C.; Varney, M.L.; Dykstra, K.M.; Falcone, E.R.; Colligan, S.H.; Hu, Q.; Aldridge, A.M.; Wright, D.L.; Wiemer, A.J.; et al. Novel tropolones induce the unfolded protein response pathway and apoptosis in multiple myeloma cells. Oncotarget 2017, 8, 76085–76098. [Google Scholar] [CrossRef] [PubMed]
- Haney, S.L.; Feng, D.; Kollala, S.S.; Chhonker, Y.S.; Varney, M.L.; Williams, J.T.; Ford, J.B.; Murry, D.J.; Holstein, S.A. Investigation of the activity of a novel tropolone in osteosarcoma. Drug Dev. Res. 2024, 85, e22129. [Google Scholar] [CrossRef]
- Bryant, B.E.; Fernelius, W.C.; Douglas, B.E. Formation Constants of Metal Complexes of Tropolone and Its Derivatives. I. Tropolone1. J. Am. Chem. Soc. 1953, 75, 3784–3786. [Google Scholar] [CrossRef]
- Haney, S.L.; Varney, M.L.; Safranek, H.R.; Chhonker, Y.S.; G-Dayanandan, N.; Talmon, G.; Murry, D.J.; Wiemer, A.J.; Wright, D.L.; Holstein, S.A. Tropolone-induced effects on the unfolded protein response pathway and apoptosis in multiple myeloma cells are dependent on iron. Leuk. Res. 2019, 77, 17–27. [Google Scholar] [CrossRef]
- Food and Drug Administration. Bioanalytical Method Validation Guidance for Industry; Food and Drug Administration: Silver Spring, MD, USA, 2022.
- Alsenz, J.; Kansy, M. High throughput solubility measurement in drug discovery and development. Adv. Drug Deliv. Rev. 2007, 59, 546–567. [Google Scholar] [CrossRef]
- Censi, R.; Di Martino, P. Polymorph Impact on the Bioavailability and Stability of Poorly Soluble Drugs. Molecules 2015, 20, 18759–18776. [Google Scholar] [CrossRef]
- Moldoveanu, S.C.; David, V. Chapter 5—Retention Mechanisms in Different HPLC Types. In Essentials in Modern HPLC Separations; Moldoveanu, S.C., David, V., Eds.; Elsevier: Amsterdam, The Netherlands, 2013; pp. 145–190. [Google Scholar]
- Sonawane, L.V.; Poul, B.N.; Usnale, S.V.; Waghmare, P.V.; Surwase, L.H. Bioanalytical method validation and its pharmaceutical application-a review. Pharm. Anal. Acta 2014, 5, 2. [Google Scholar] [CrossRef]
- Simeoli, R.; Cairoli, S.; Greco, M.; Bellomo, F.; Mancini, A.; Rossi, C.; Dionisi Vici, C.; Emma, F.; Goffredo, B.M. A New and Rapid LC-MS/MS Method for the Determination of Cysteamine Plasma Levels in Cystinosis Patients. Pharmaceuticals 2024, 17, 649. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-H.; Mondal, G.; Butawan, M.; Bloomer, R.J.; Yates, C.R. Development of a liquid chromatography-tandem mass spectrometry (LC–MS/MS) method for characterizing caffeine, methylliberine, and theacrine pharmacokinetics in humans. J. Chromatogr. B 2020, 1155, 122278. [Google Scholar] [CrossRef] [PubMed]
- Côté, C.; Bergeron, A.; Mess, J.-N.; Furtado, M.; Garofolo, F. Matrix Effect Elimination During LC–MS/MS Bioanalytical Method Development. Bioanalysis 2009, 1, 1243–1257. [Google Scholar] [CrossRef] [PubMed]
- Aldhafiri, W.N.; Chhonker, Y.S.; Zhang, Y.; Coulter, D.W.; McGuire, T.R.; Li, R.; Murry, D.J. Assessment of tissue distribution and metabolism of MP1, a novel pyrrolomycin, in mice using a validated LC-MS/MS method. Molecules 2020, 25, 5898. [Google Scholar] [CrossRef] [PubMed]
- Thakare, R.; Chhonker, Y.S.; Gautam, N.; Alamoudi, J.A.; Alnouti, Y. Quantitative analysis of endogenous compounds. J. Pharm. Biomed. Anal. 2016, 128, 426–437. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.K.; Valicherla, G.R.; Joshi, P.; Shahi, S.; Syed, A.A.; Gupta, A.P.; Hossain, Z.; Italiya, K.; Makadia, V.; Singh, S.K. Determination of permeability, plasma protein binding, blood partitioning, pharmacokinetics and tissue distribution of Withanolide A in rats: A neuroprotective steroidal lactone. Drug Dev. Res. 2018, 79, 339–351. [Google Scholar] [CrossRef] [PubMed]
- Valicherla, G.R.; Riyazuddin, M.; Shahi, S.; Gupta, A.P.; Syed, A.A.; Husain, A.; Gayen, J.R. LC-ESI-MS/MS assay development and validation of a novel antidiabetic peptide PSTi8 in mice plasma using SPE: An application to pharmacokinetics. J. Pharm. Biomed. Anal. 2020, 180, 113074. [Google Scholar] [CrossRef] [PubMed]
- Davies, B.; Morris, T. Physiological parameters in laboratory animals and humans. Pharm. Res. 1993, 10, 1093–1095. [Google Scholar] [CrossRef] [PubMed]
- Singh, H.; Satish, N.; Babu, T.R.; Singh, A.; Yadav, B.; Singh, S.K.; Wahajuddin, M.; Siddiqui, M.I.; Jagavelu, K.; Sudhakar, G. Functionalized Azirine Based Scaffolds as Endothelin Inhibitors for the Selective Anti-Angiogenic activity. Eur. J. Med. Chem. 2024, 274, 116510. [Google Scholar] [CrossRef]
- Klein, S. The use of biorelevant dissolution media to forecast the in vivo performance of a drug. Aaps J. 2010, 12, 397–406. [Google Scholar] [CrossRef] [PubMed]
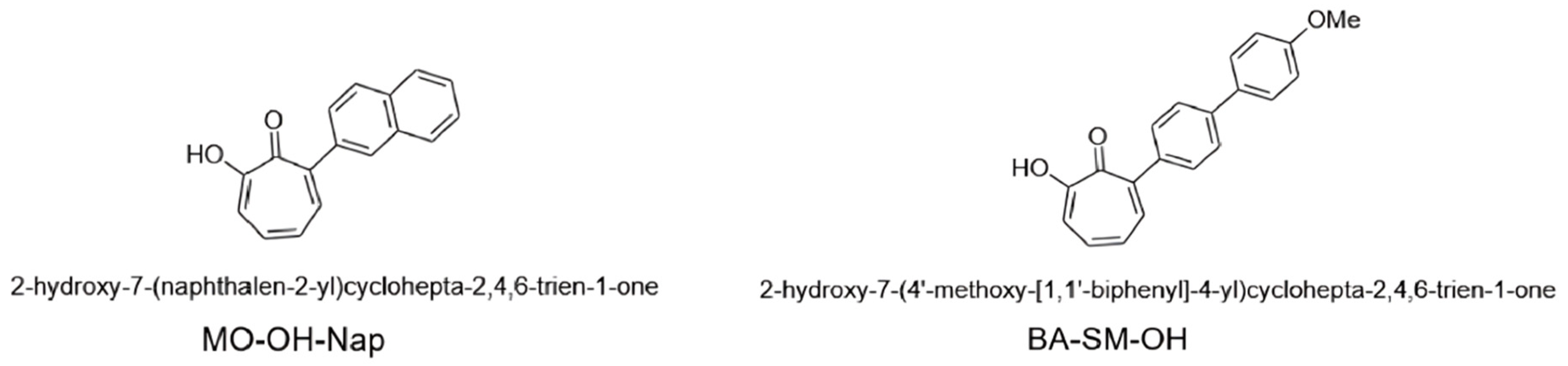
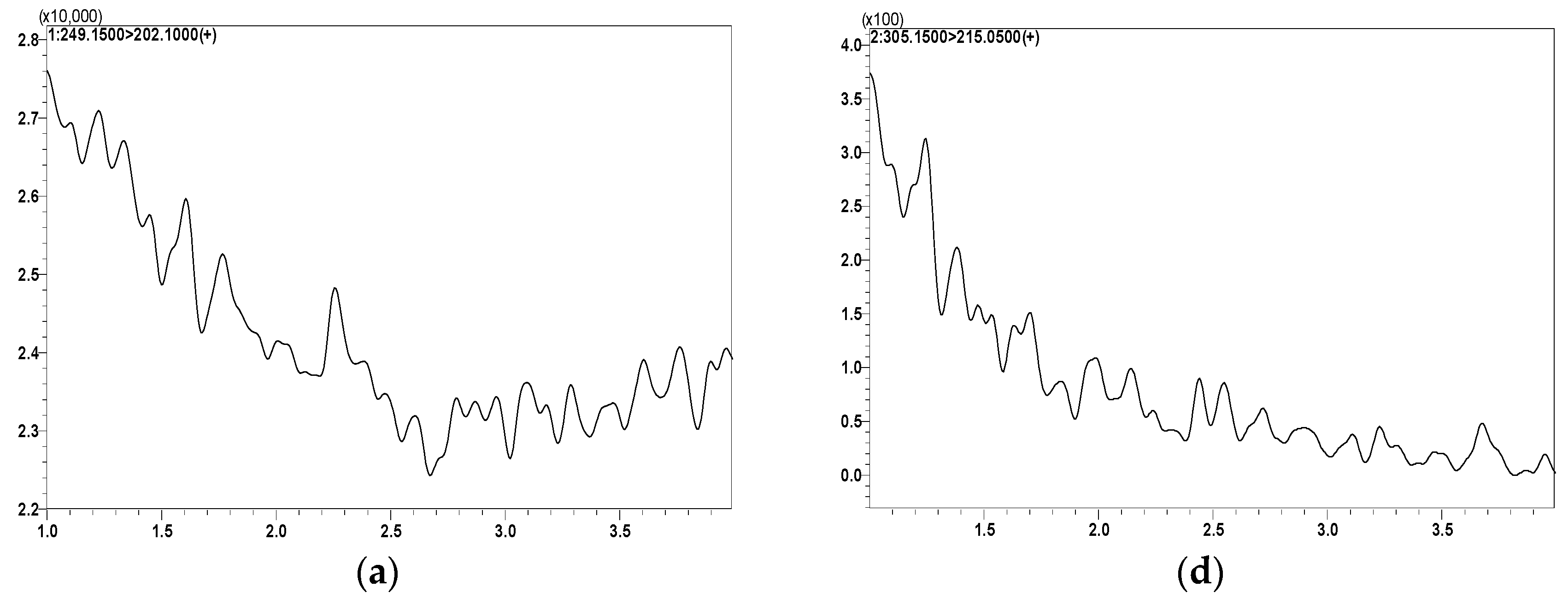

| Analytes | MRM Transition m/z (Q1→Q3) | Q1 (V) | Q3 (V) | CE (V) | Retention Time (min) |
|---|---|---|---|---|---|
| MO-OH-Nap (Target) | 249.10→202.15 | −100 | −23 | −35 | 1.70 |
| 249.10→231.10 | −11 | −23 | −24 | ||
| BA-SM-OH (IS) | 305.10→215.05 | −23 | −22 | −25 | 1.85 |
| 305.10→195.05 | −13 | −25 | −21 |
| Extraction Techniques | Nominal Conc. (ng/mL) | Accuracy | Precision | ||
|---|---|---|---|---|---|
| %Bias Intra–Assay | %Bias Inter–Assay | %RSD Intra–Assay | %RSD Inter–Assay | ||
| PPT | LLOQ (1 ng/mL) | −5.03 | 11.23 | 9.45 | 14.76 |
| LQC (3 ng/mL) | −1.90 | −5.09 | 5.39 | 6.10 | |
| MQC (200 ng/mL) | −6.11 | 0.62 | 7.15 | 9.51 | |
| HQC (375 ng/mL) | 1.64 | 2.12 | 11.73 | 14.67 | |
| SPE | LLOQ (1 ng/mL) | 5.40 | 12.50 | 1.80 | 17.30 |
| LQC (3 ng/mL) | −4.00 | 1.80 | 1.50 | 13.40 | |
| MQC (200 ng/mL) | −6.10 | −3.70 | 13.40 | 10.80 | |
| HQC (375 ng/mL) | −9.20 | −11.20 | 11.60 | 11.20 | |
| Extraction Techniques | Nominal Conc. (ng/mL) | MO-OH-Nap Tropolone | BA-SM-OH (IS) | ||
|---|---|---|---|---|---|
| Mean Extraction Recovery (%) | Mean ME (%) | Mean Extraction Recovery (%) | Mean ME (%) | ||
| PPT | LQC (3 ng/mL) | 79.5 ± 3.0 | 93.12 ± 10.86 | 90.8 ± 10.8 | 89.64 ± 21.52 |
| MQC (200 ng/mL) | 92.7 ± 11.9 | 90.29 ± 20.55 | |||
| HQC (375 ng/mL) | 89.7 ± 7.7 | 86.02 ± 25.83 | |||
| SPE | LQC (3 ng/mL) | 80.6 ± 8.3 | 97.28 ± 6.75 | 55.9 ± 9.2 | 90.59 ± 6.82 |
| MQC (200 ng/mL) | 54.9 ± 0.8 | 94.26 ± 1.74 | |||
| HQC (375 ng/mL) | 53.8 ± 6.0 | 109.49 ± 10.99 | |||
| Nominal Conc. (ng/mL) | Measured Mean Conc. (ng/mL) | % Accuracy |
|---|---|---|
| Bench-top stability at 21 °C, up to 6 h, Mean ± S.D, n = 3. | ||
| LQC (3 ng/mL) | 3.2 ± 0.5 | 107.8 ± 14.8 |
| MQC (200 ng/mL) | 191.6 ± 18.1 | 95.8 ± 9.1 |
| HQC (375 ng/mL) | 380.5 ± 20.9 | 101.5 ± 5.6 |
| Freeze-thaw stability at −80 °C, up to 3 Cycle, Mean ± S.D, n = 3. | ||
| LQC (3 ng/mL) | 3.0 ± 0.4 | 100.5 ± 13.3 |
| MQC (200 ng/mL) | 167.9 ± 1.8 | 84.0 ± 0.9 |
| HQC (375 ng/mL) | 332.2 ± 17.6 | 88.6 ± 4.7 |
| Autosampler stability at 4 °C, up to 24 h, Mean ± S.D, n = 3. | ||
| LQC (3 ng/mL) | 3.1 ± 0.2 | 101.9 ± 5.8 |
| MQC (200 ng/mL) | 180.2 ± 1.10 | 90.1 ± 0.6 |
| HQC (375 ng/mL) | 340.2 ± 20.1 | 90.7 ± 5.3 |
| Long-term stability at −80 °C, 12 months, Mean ± S.D, n = 3. | ||
| LQC (3 ng/mL) | 2.9 ± 0.1 | 95.6 ± 2.4 |
| MQC (200 ng/mL) | 199.5 ± 1.3 | 99.8 ± 0.6 |
| HQC (375 ng/mL) | 347.4 ± 1.5 | 92.7 ± 0.4 |
| Time (min) | MO-OH-Nap B/P Ratio |
|---|---|
| 0 | 1.1 ± 0.1 |
| 30 | 0.9 ± 0.2 |
| 60 | 1.0 ± 0.12 |
| Nominal Conc. of MO-OH-Nap (µg/mL) | % Plasma Protein Bound ± SD | % Device Recovery ± SD | % Drug Remaining at 5 h ± SD | Equilibrium at 5 h ± SD |
|---|---|---|---|---|
| 1 µg/mL | 99.6 ± 0.1 | 103.5 ± 18.7 | 105.6 ± 0.8 | 0.3 ± 0.5 |
| 10 µg/mL | 99.7 ± 0.1 | 119.4 ± 6.6 | 91.1 ± 2.5 | 0.1 ± 0.00 |
| Pharmacokinetic Parameters | Mean | S.D | CV% |
|---|---|---|---|
| Cmax (ng/mL) | 3040.9 | 1088.4 | 35.8 |
| Tmax (h) | 0.3 | 0.2 | 56.4 |
| t1/2 (h) | 17.7 | 7.2 | 40.6 |
| AUC0_∞ (h × ng/mL) | 2859.8 | 499.3 | 17.5 |
| AUC0_last (h × ng/mL) | 2747.9 | 403.1 | 14.7 |
| Vd/F (mL/kg) | 43,727.1 | 10,870.1 | 24.9 |
| CL/F (mL/h/kg) | 1790.6 | 305.8 | 17.1 |
| Parameter | Condition |
|---|---|
| LC-MS/MS model | LC-MS/MS 8060 system (Shimadzu Scientific Instruments, Columbia, MD, USA) equipped with a dual ion source (DUIS) |
| LC-MS/MS Software | LabSolutions LCMS software Version 5.9 (Shimadzu Scientific, Inc., Columbia, MD, USA) |
| LC-MS/MS Ionization mode | ESI+ mode |
| MS parameters | Nebulizing Gas: 2.0 L/min Drying gas flow: 10.0 L/min Heating gas flow: 10.0 L/min Interface Temp: 300 °C DLTemp: 250 °C Block Heater Temp: 400 °C |
| Column | ACE Excel C18 (1.7 µm, 100 × 2.1 mm, Advanced Chromatography Technologies, Ltd., Aberdeen, UK) protected with a C18 guard column (Phenomenex, Torrance, CA, USA) |
| Mobile phase A | Water with 0.05% v/v TFA |
| Mobile phase B | ACN with 0.05% v/v TFA |
| Flow | 0.25 mL/min |
| Isocratic elution | (15% A: 85% B) |
| Total run time | 4 min |
| Injection volume | 10 µL |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aldhafiri, W.N.; Chhonker, Y.S.; Ahmed, N.; Singh, S.K.; Haney, S.L.; Ford, J.B.; Holstein, S.A.; Murry, D.J. Development and Validation of an LC-MS/MS Assay for the Quantitation of MO-OH-Nap Tropolone in Mouse Plasma: Application to In Vitro and In Vivo Pharmacokinetic Studies. Molecules 2024, 29, 4424. https://doi.org/10.3390/molecules29184424
Aldhafiri WN, Chhonker YS, Ahmed N, Singh SK, Haney SL, Ford JB, Holstein SA, Murry DJ. Development and Validation of an LC-MS/MS Assay for the Quantitation of MO-OH-Nap Tropolone in Mouse Plasma: Application to In Vitro and In Vivo Pharmacokinetic Studies. Molecules. 2024; 29(18):4424. https://doi.org/10.3390/molecules29184424
Chicago/Turabian StyleAldhafiri, Wafaa N., Yashpal S. Chhonker, Nusrat Ahmed, Sandeep K. Singh, Staci L. Haney, James B. Ford, Sarah A. Holstein, and Daryl J. Murry. 2024. "Development and Validation of an LC-MS/MS Assay for the Quantitation of MO-OH-Nap Tropolone in Mouse Plasma: Application to In Vitro and In Vivo Pharmacokinetic Studies" Molecules 29, no. 18: 4424. https://doi.org/10.3390/molecules29184424
APA StyleAldhafiri, W. N., Chhonker, Y. S., Ahmed, N., Singh, S. K., Haney, S. L., Ford, J. B., Holstein, S. A., & Murry, D. J. (2024). Development and Validation of an LC-MS/MS Assay for the Quantitation of MO-OH-Nap Tropolone in Mouse Plasma: Application to In Vitro and In Vivo Pharmacokinetic Studies. Molecules, 29(18), 4424. https://doi.org/10.3390/molecules29184424






