Phytochemical Analysis and Antioxidant Activities of Various Extracts from the Aerial Part of Anemone baicalensis Turcz.: In Vitro and In Vivo Studies
Abstract
:1. Introduction
2. Results and Discussion
2.1. Preliminary Identification of Phytochemicals
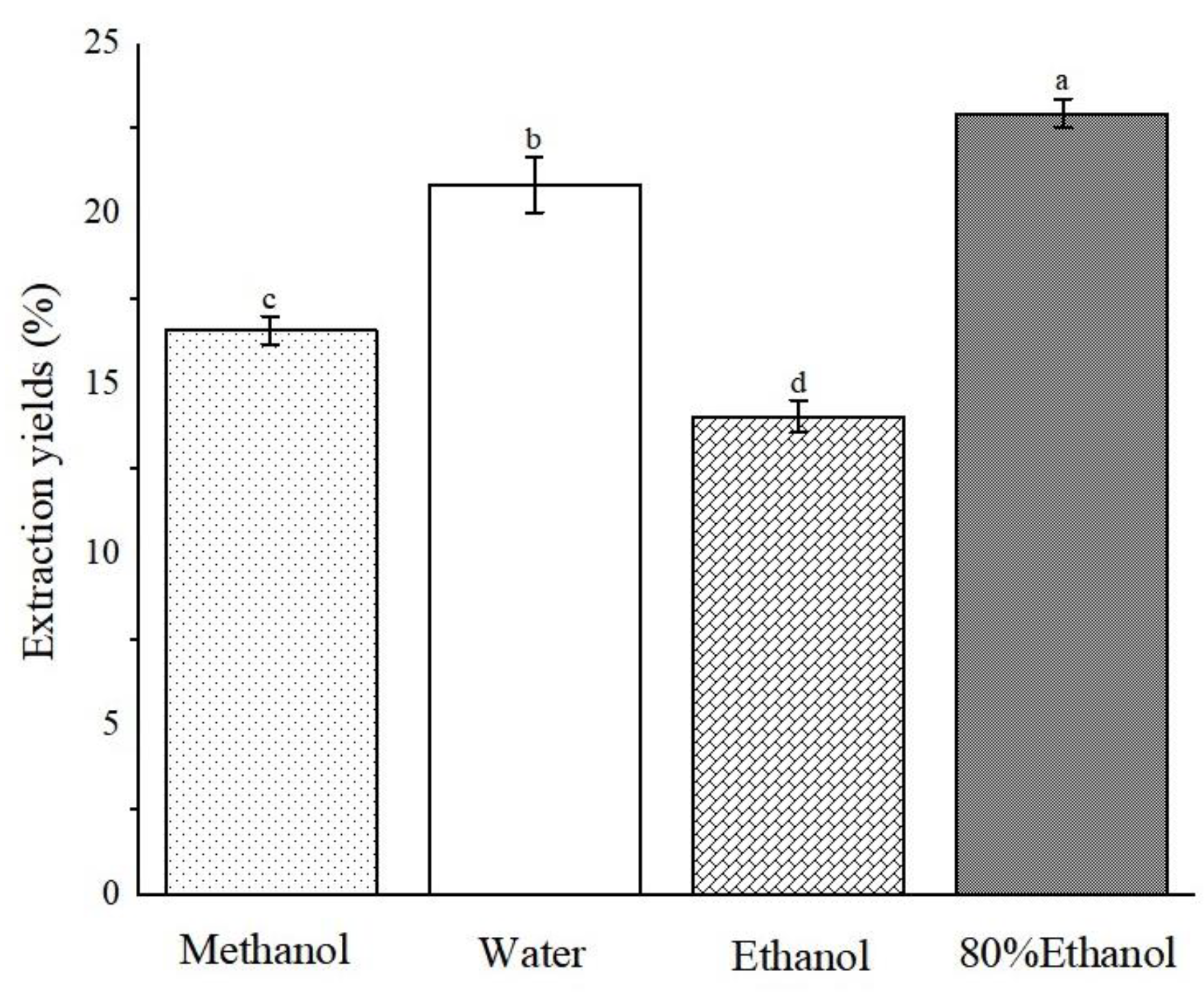
2.2. Extraction Yields of Different Extracts of ABAP
2.3. Content of Active Ingredients
2.3.1. Total Carbohydrate Content (TCC)
2.3.2. Total Protein Content (TProC)
2.3.3. Total Monoterpenoid Content (TMC)
2.3.4. Total Alkaloid Content (TAC)
2.3.5. Total Phenolic Content (TPheC)
2.3.6. Total Phenolic Acid Content (TPAC)
2.3.7. Total Flavonoid Content (TFC)
2.3.8. Total Tannin Content (TTanC), Gallotannin Content (GC), and Condensed Tannin Content (CTC)
2.3.9. Total Triterpenoid Content (TTriC)
2.4. Antioxidant Activity In Vitro
2.4.1. 1,1-Diphenyl-2-Picrylhydrazyl Radical (DPPH) and 2,2′-Azino-Bis(3-Ethylbenzothiazoline-6-Sulfonicacid) Diammonium Salt (ABTS)
2.4.2. Hydroxyl Radicals and Superoxide Radicals
2.4.3. Ferric Reducing Antioxidant Power (FRAP) and Cupric Reducing Antioxidant Capacity (CUPRAC)
2.4.4. Metal Chelation
2.4.5. Hydrogen Peroxide (H2O2) and Singlet Oxygen
2.4.6. β-Carotene Bleaching
2.4.7. Hypochlorous Acid (HClO) and Nitric Oxide (NO)
2.5. UHPLC-MS Analysis
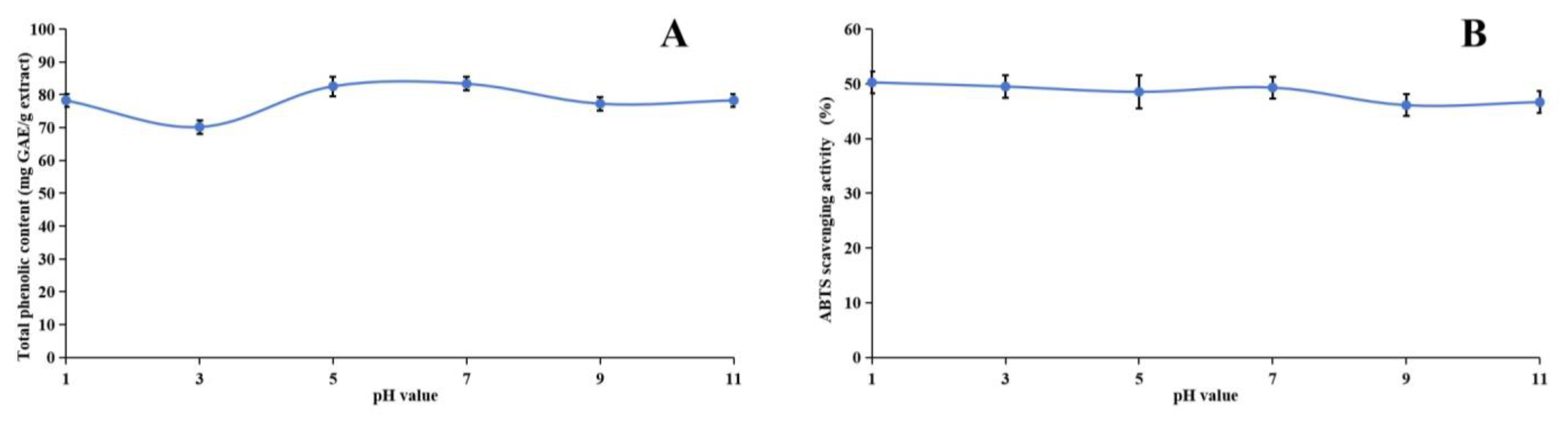
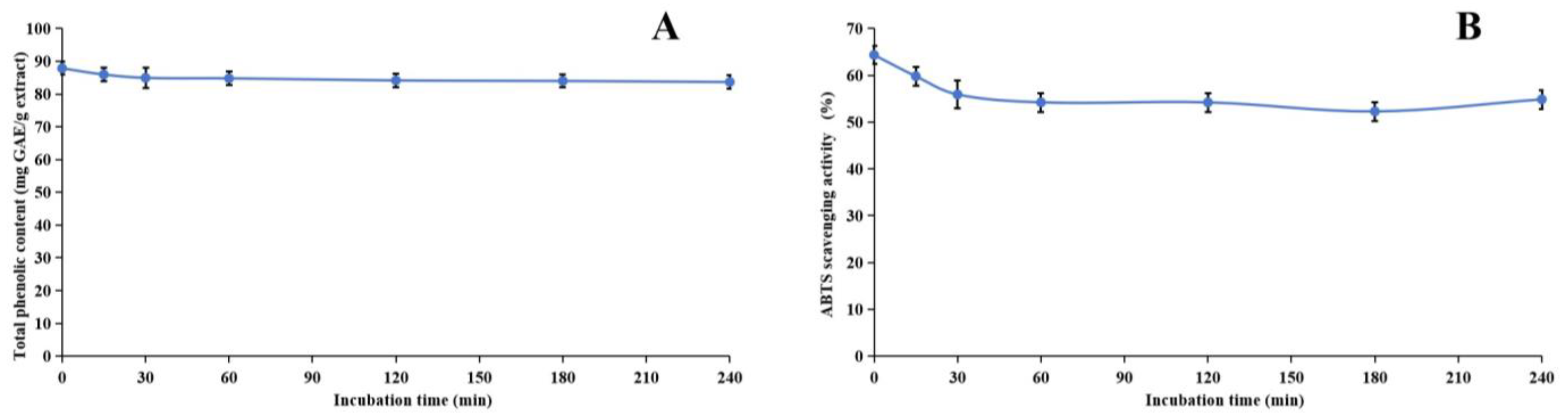
2.6. Stability of Methanol Extract
2.7. Oxidative Stability of Oils
2.8. Oral Acute Toxicity
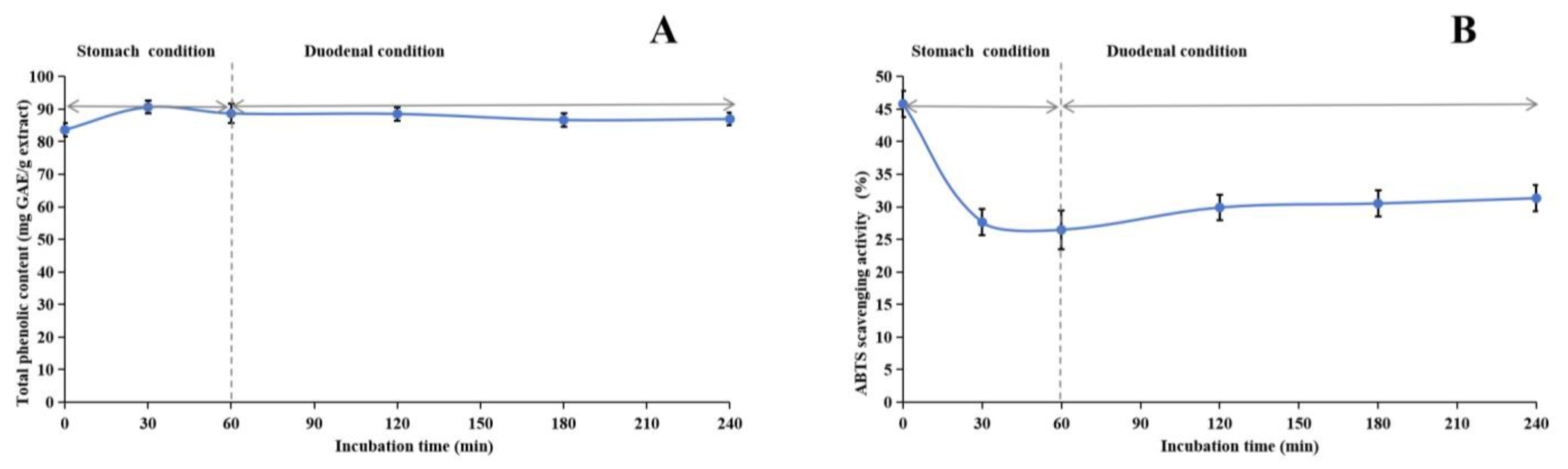
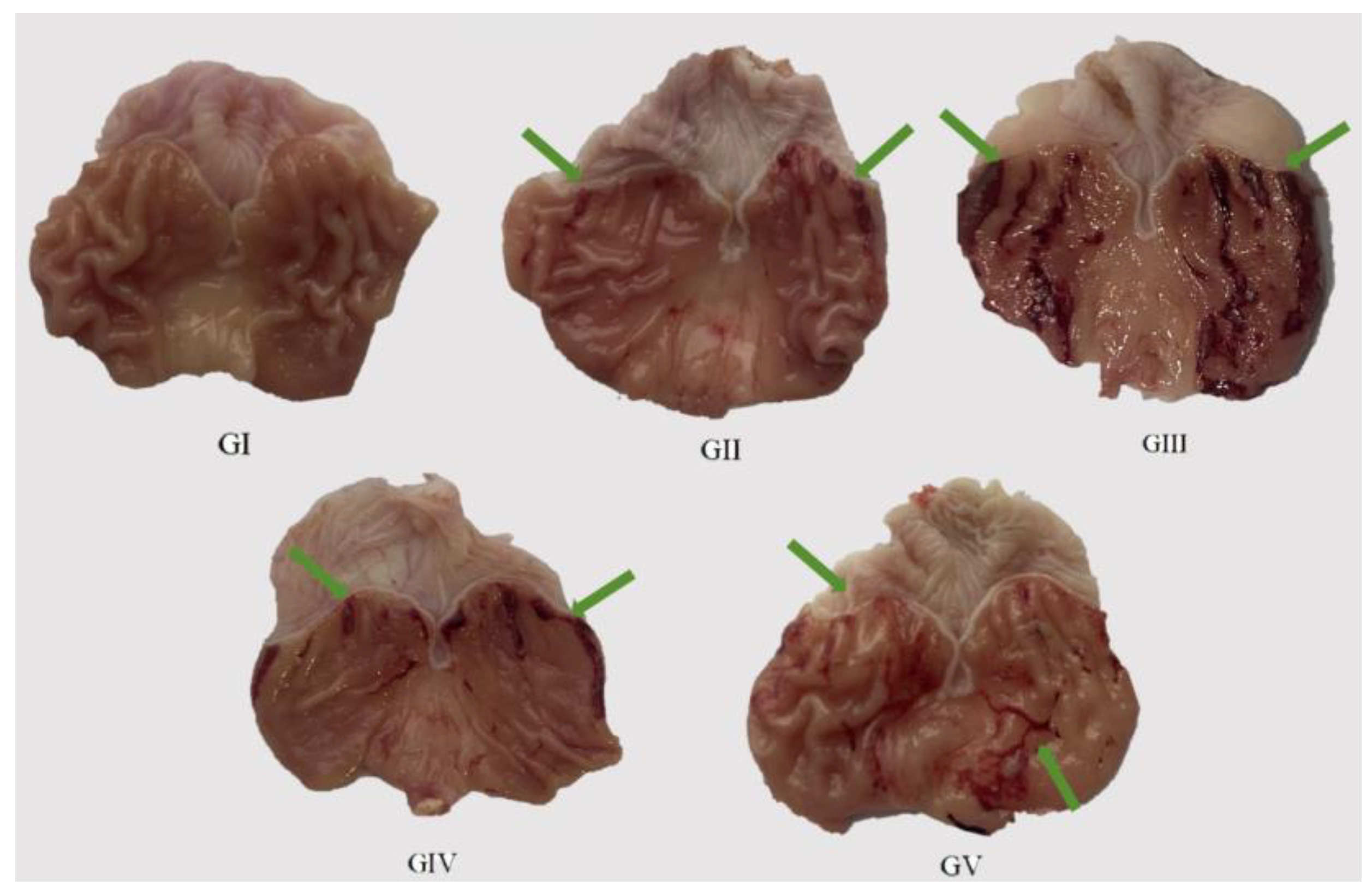
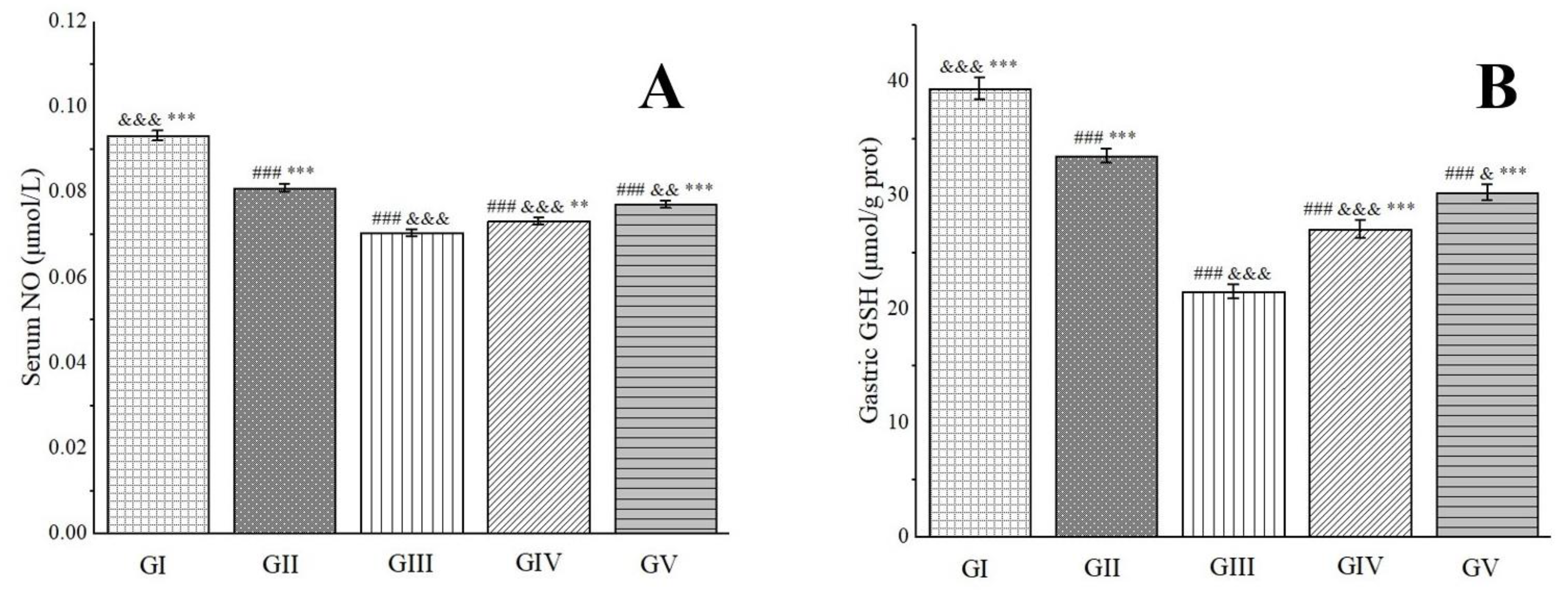
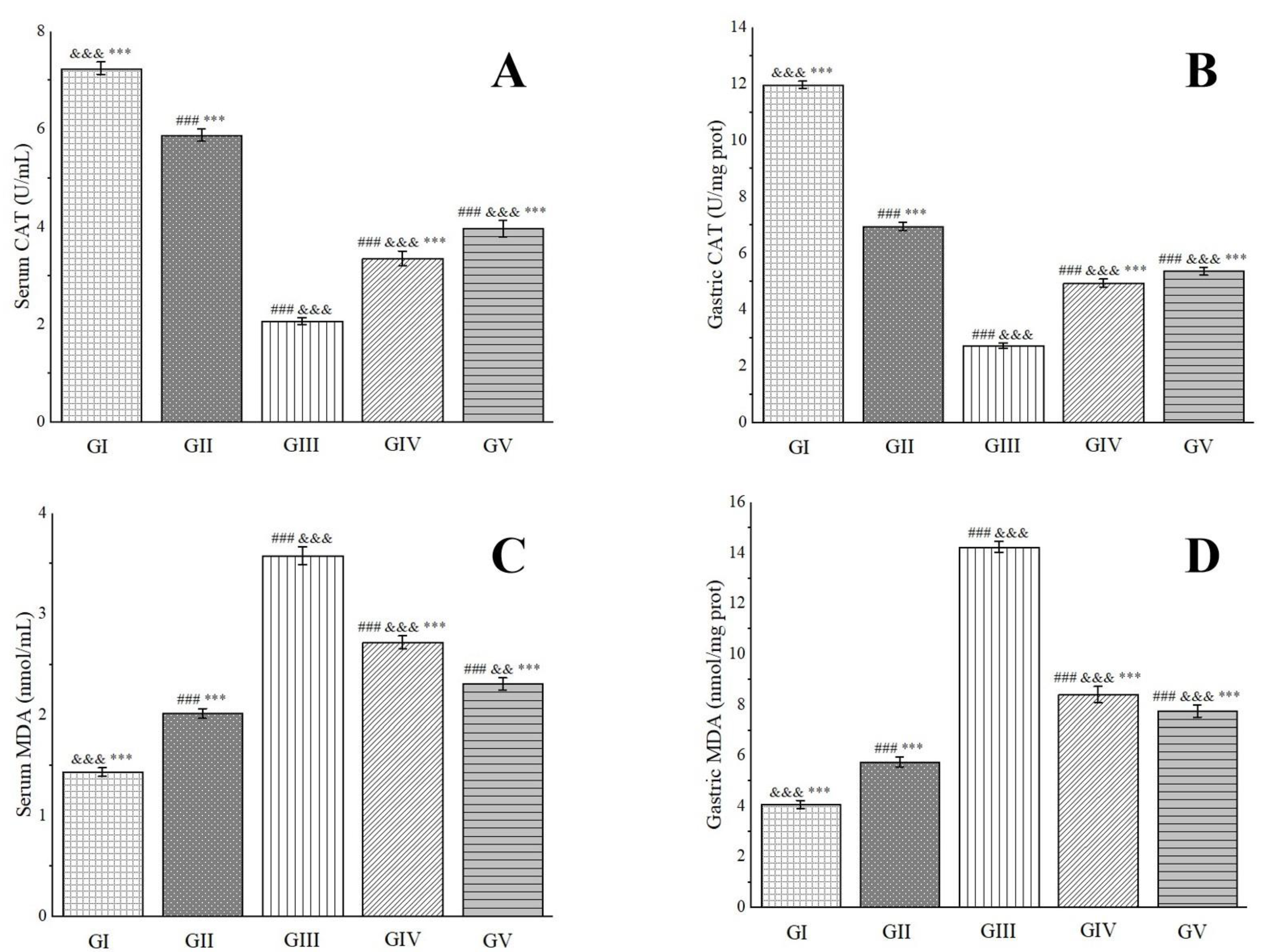
2.9. Gastric Protective Activity
3. Material and Methods
3.1. Materials
3.2. Methods
3.2.1. Qualitative Phytochemical Analysis
3.2.2. Preparation of Different Extracts of ABAP
3.2.3. Quantitative Phytochemical Analysis
3.2.4. Antioxidant Activity Assays
3.2.5. UHPLC-MS Method
3.2.6. Stability Studies of Methanol Extract
3.2.7. Oxidative Stability Studies of Oils
3.2.8. Oral Acute Toxicity Study
3.2.9. Gastric Protective Experiments
3.2.10. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Flora of China Editorial Committee of Chinese Academy of Sciences. The Flora of China; Science Press: Beijing, China, 1980; Volume 28, p. 20. [Google Scholar]
- Liu, Y.; Liu, L.; Tian, C.K.; Zhou, D.Z. Research progress of studies on chemical constituents and biologic activities of Anemone species. China J. Chin. Mater. Med. 2019, 44, 912–919. [Google Scholar] [CrossRef]
- Biswas, T.; Dwivedi, U.N. Plant triterpenoid saponins: Biosynthesis, in vitro production, and pharmacological relevance. Protoplasma 2019, 256, 1463–1486. [Google Scholar] [CrossRef] [PubMed]
- Liao, X.; Li, B.G.; Ding, L.S.; Pan, Y.J.; Chen, Y.Z. New triterpenoid saponins from Anemone begoniifolia. Acta Pharm. Sin. 2000, 35, 821–825. [Google Scholar] [CrossRef]
- Liao, X.; Li, B.G.; Wang, M.K.; Ding, L.S.; Pan, Y.J.; Chen, Y.Z. The chemical constituents from Anemone rivularis. Chem. J. Chin. Univ. 2001, 22, 1338–1341. [Google Scholar]
- Gong, W.Z. Research on the Chemical Composition and Biological Activity of Anemone cathayensis. Master’s Thesis, Minzu University of China, Beijing, China, 2011. [Google Scholar]
- Wang, J.L.; Liu, K.; Gong, W.Z.; Wang, Q.; Xu, D.T.; Liu, M.F.; Bi, K.L.; Song, Y.F. Anticancer, antioxidant, and antimicrobial activities of anemone (Anemone cathayensis). Food Sci. Biotechnol. 2012, 21, 551–557. [Google Scholar] [CrossRef]
- Li, H.; Wang, X.Y.; Wang, X.Y.; Hua, D.; Liu, Y.; Tang, H.F. Two new triterpenoid saponins from the aerial parts of Anemone taipaiensis. J. Asian Nat. Prod. Res. 2015, 17, 576–585. [Google Scholar] [CrossRef]
- Wang, X.Y.; Li, H.; Lu, Y.Y.; Xu, J.; Zhang, Y.H.; Tang, H.F. Saponin constituents from the aerial parts of Anemone taipaiensis. Global Tradit. Chin. Med. 2022, 15, 2001–2009. [Google Scholar]
- Yokosuka, A.; Sano, T.; Hashimoto, K.; Sakagami, H.; Mimaki, Y. Triterpene glycosides from the whole plant of Anemone hupehensis var. japonica and their cytotoxic activity. Chem. Pharm. Bull. 2009, 57, 1425–1430. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.Y.; Li, Y.; Zhao, B.; Zhou, H.L.; Liu, S.H.; Gao, F. Chemical composition and content determination analysis of the aboveground parts of Anemone raddeana rhizoma. Acad. Period. Chang. Coll. Tradit. Chin. Med. 2005, 21, 43–44. [Google Scholar] [CrossRef]
- Sun, J.Y.Ι. Studies on the Chemical Constituents of Ligusticum chuanxiong Hort. and Codonopsis tangshen Oliv. ΙΙ. Evaluation on Pharmacodynamics of Analgesia and Anti-Inflammation of Anemone vitifolia Buch.-Ham. Master’s Thesis, Ningxia Medical University, Yinchuan, China, 2016. [Google Scholar]
- Wang, D.M.; Zhang, Y.J.; Wang, S.S.; Li, D.W. Antioxidant and antifungal activities of extracts and fractions from Anemone taipaiensis, China. Allelopathy J. 2013, 32, 67–77. [Google Scholar]
- Wang, X.Y.; Li, H.; Lu, Y.Y.; Ping, X.D.; Zhang, Y.H.; Tang, H.F. Triterpenoid saponins from aerial parts of Anemone Taipaiensis. Chin. Tradit. Herb. Drugs 2022, 53, 4925–4933. [Google Scholar]
- Wang, J.; Qiu, C.; Zhong, K.; Gao, H.; Huang, Y.N.; Gao, X.L.; Xu, Z.J. Study on anti-yeast activity of active fraction obtained from Urena lobata L. leaves. Sci. Technol. Food Ind. 2015, 36, 107–111. [Google Scholar] [CrossRef]
- Ning, J.L.; Qin, N.; Li, H.F.; Ma, X.Y.; Zhang, F.; Feng, T.; He, J. Anemhupehins D-F, sterols and chromone derivative from Anemone hupehensis and their anti-inflammatory activity. Phytochem. Lett. 2024, 61, 7–10. [Google Scholar] [CrossRef]
- Chepinoga, V.V.; Protopopova, M.V.; Pavlichenko, V.V.; Dudov, S.V. Habitat distribution patterns of nemoral relict plant species on the Khamar-Daban Ridge (the South of Eastern Siberia) according to grid mapping data. Russ. J. Ecol. 2021, 52, 212–222. [Google Scholar] [CrossRef]
- Yu, J.L.; Hu, Y.W.; Zhang, L.Q.; Sun, R.S.; Xiao, J.L.; Jia, A.L. Color Atlas of Medicinal Plants in Jilin Province; Jilin Science and Technology Press: Jilin, China, 2016; Volume 1, p. 212. [Google Scholar]
- Chen, M.H.; He, X.; Sun, H.; Sun, Y.; Li, L.; Zhu, J.Y.; Xia, G.Q.; Guo, X.; Zang, H. Phytochemical analysis, UPLC-ESI-Orbitrap-MS analysis, biological activity, and toxicity of extracts from Tripleurospermum limosum (Maxim.) Pobed. Arabian J. Chem. 2022, 15, 103797. [Google Scholar] [CrossRef]
- He, X.; Chen, M.H.; Sun, H.; Guo, X.; Sun, Y.; Li, L.; Zhu, J.Y.; Xia, G.Q.; Zang, H. Multidirectional insights into the phytochemical, biological, and multivariate analysis of extracts from the aerial part of Swertia perennis Linnaeus. Nat. Prod. Res. 2024, 38, 135–139. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Feng, J.X.; Wei, Z.B.; Sun, H.; Li, L.; Zhu, J.Y.; Xia, G.Q.; Zang, H. Phytochemical analysis, antioxidant activities in vitro and in vivo, and theoretical calculation of different extracts of Euphorbia fischeriana. Molecules 2023, 28, 5172. [Google Scholar] [CrossRef] [PubMed]
- Stipcevic, T.; Knight, C.P.; Kippin, T.E. Stimulation of adult neural stem cells with a novel glycolipid biosurfactant. Acta Neurol. Belg. 2013, 113, 501–506. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Song, M.M.; Wu, S.H.; Zhao, H.B.; Zhang, Y. Plant-based dietary fibers and polysaccharides as modulators of gut microbiota in intestinal and lung inflammation: Current state and challenges. Nutrients 2023, 15, 3321. [Google Scholar] [CrossRef]
- Zhao, H.; Song, A.; Zheng, C.; Wang, M.D.; Song, G.Y. Effects of plant protein and animal protein on lipid profile, body weight and body mass index on patients with hypercholesterolemia: A systematic review and meta-analysis. Acta Diabetol. 2020, 57, 1169–1180. [Google Scholar] [CrossRef]
- Fehér, A.; Gazdecki, M.; Véha, M.; Szakaly, M.; Szakaly, Z. A comprehensive review of the benefits of and the barriers to the switch to a plant-based diet. Sustainability 2020, 12, 4136. [Google Scholar] [CrossRef]
- Juergens, U.R. Anti-inflammatory properties of the monoterpene 1.8-cineole: Current evidence for co-medication in inflammatory airway diseases. Drug Res. 2014, 64, 638–646. [Google Scholar] [CrossRef]
- Kim, S.H.; Lee, S.Y.; Hong, C.Y.; Gwak, K.S.; Park, M.J.; Smith, D.; Choi, I.G. Whitening and antioxidant activities of bornyl acetate and nezukol fractionated from Cryptomeria japonica essential oil. Int. J. Cosmet. Sci. 2013, 35, 484–490. [Google Scholar] [CrossRef] [PubMed]
- Zhong, H.Q.; Xiao, M.Q.; Zarkovic, K.; Zhu, M.J.; Sa, R.; Lu, J.H.; Tao, Y.Z.; Chen, Q.; Xia, L.; Cheng, S.Q.; et al. Mitochondrial control of apoptosis through modulation of cardiolipin oxidation in hepatocellular carcinoma: A novel link between oxidative stress and cancer. Free Radic. Biol. Med. 2017, 102, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.X.; Huang, H.Y.; Zhan, Z.X.; Gao, H.J.; Zhang, C.C.; Lai, J.C.; Cao, J.G.; Li, C.Y.; Chen, Y.; Liu, Z.Q. Berberine inhibits glioma cell migration and invasion by suppressing TGF-β1/COL11A1 pathway. Biochem. Biophys. Res. Commun. 2022, 625, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Bai, R.R.; Yao, C.S.; Zhong, Z.C.; Ge, J.M.; Bai, Z.Q.; Ye, X.Y.; Xie, T.; Xie, Y.Y. Discovery of natural anti-inflammatory alkaloids: Potential leads for the drug discovery for the treatment of inflammation. Eur. J. Med. Chem. 2021, 213, 113165. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.N.; Shi, X.; Hou, J.B.; Gao, S.Y.; Chao, Y.F.; Ding, J.R.; Chen, L.; Qian, Y.; Shao, G.J.; Si, Y.C.; et al. Integrating metabolomics and network pharmacology to explore Rhizoma coptidis extracts against sepsis-associated acute kidney injury. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2021, 1164, 122525. [Google Scholar] [CrossRef] [PubMed]
- Efenberger-Szmechtyk, M.; Nowak, A.; Czyzowska, A. Plant extracts rich in polyphenols: Antibacterial agents and natural preservatives for meat and meat products. Crit. Rev. Food Sci. Nutr. 2021, 61, 149–178. [Google Scholar] [CrossRef] [PubMed]
- Bonaccio, M.; Pounis, G.; Cerletti, C.; Donati, M.B.; Iacoviello, L.; de Gaetano, G.; MOLI-SANI Study Investigators. Mediterranean diet, dietary polyphenols and low grade inflammation: Results from the MOLI-SANI study. Br. J. Clin. Pharmacol. 2017, 83, 107–113. [Google Scholar] [CrossRef]
- Zhang, L.L.; Wang, W.B.; Yue, X.Y.; Wu, G.S.; Yue, P.X.; Gao, X.L. Gallic acid as a copigment enhance anthocyanin stabilities and color characteristics in blueberry juice. J. Food Sci. Technol. 2020, 57, 1405–1414. [Google Scholar] [CrossRef]
- Fu, L.L.; Xiao, Q.; Ru, Y.; Hong, Q.L.; Weng, H.F.; Zhang, Y.H.; Chen, J.; Xiao, A.F. Bio-based active packaging: Gallic acid modified agarose coatings in grass carp (Ctenopharyngodon idellus) preservation. Int. J. Biol. Macromol. 2024, 255, 128196. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.M.; Tait, A.R.; Kitts, D.D. Flavonoid composition of orange peel and its association with antioxidant and anti-inflammatory activities. Food Chem. 2017, 218, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.F.; Li, X.F.; Wu, K.; Wang, M.K.; Liu, P.; Wang, X.S.; Deng, R.X. Antioxidant activities and chemical constituents of flavonoids from the flower of Paeonia ostii. Molecules 2016, 22, 5. [Google Scholar] [CrossRef]
- Chen, L.; Teng, H.; Xie, Z.L.; Cao, H.; Cheang, W.S.; Skalicka-Woniak, K.; Georgiev, M.I.; Xiao, J.B. Modifications of dietary flavonoids towards improved bioactivity: An update on structure-activity relationship. Crit. Rev. Food Sci. Nutr. 2018, 58, 513–527. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.J.; Shon, M.S.; Kim, G.N.; Lee, S.C. Antioxidant and anti-adipogenic activities of persimmon tannins. Food Sci. Biotechnol. 2014, 23, 1689–1694. [Google Scholar] [CrossRef]
- Villanueva, X.; Zhen, L.L.; Ares, J.N.; Vackier, T.; Lange, H.; Crestini, C.; Steenackers, H.P. Effect of chemical modifications of tannins on their antimicrobial and antibiofilm effect against Gram-negative and Gram-positive bacteria. Front. Microbiol. 2023, 13, 987164. [Google Scholar] [CrossRef] [PubMed]
- Munteanu, I.G.; Apetrei, C. Analytical methods used in determining antioxidant activity: A review. Int. J. Mol. Sci. 2021, 22, 3380. [Google Scholar] [CrossRef] [PubMed]
- Amorati, R.; Valgimigli, L. Methods to measure the antioxidant activity of phytochemicals and plant extracts. J. Agric. Food Chem. 2018, 66, 3324–3329. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Lu, J.C.; He, W.F.; Chi, H.D.; Yamashita, K.; Manabe, M.; Kodama, H. Antiperoxidation activity of triterpenoids from rhizome of Anemone raddeana. Fitoterapia 2009, 80, 105–111. [Google Scholar] [CrossRef]
- Li, C.Q.; Yao, C.; Zhu, R.Y.; Huang, Y.X.; Kang, W.Y.; Wang, J.M. Spectrum-effect relationship in antioxidant activity of Ligustri lucidi Fructus based on DPPH, ABTS and FRAP assays. China J. Chin. Mater. Med. 2016, 41, 1670–1677. [Google Scholar] [CrossRef]
- Apak, R.; Güçlü, K.; Ozyürek, M.; Karademir, S.E. Novel total antioxidant capacity index for dietary polyphenols and vitamins C and E, using their cupric ion reducing capability in the presence of neocuproine: CUPRAC method. J. Agric. Food Chem. 2004, 52, 7970–7981. [Google Scholar] [CrossRef]
- Lomozová, Z.; Hrubša, M.; Conte, P.F.; Papastefanaki, E.; Moravcová, M.; Catapano, M.C.; Proietti Silvestri, I.; Karlíčková, J.; Kučera, R.; Macáková, K.; et al. The effect of flavonoids on the reduction of cupric ions, the copper-driven Fenton reaction and copper-triggered haemolysis. Food Chem. 2022, 394, 133461. [Google Scholar] [CrossRef]
- Lucius, R.; Sievers, J. Postnatal retinal ganglion cells in vitro: Protection against reactive oxygen species (ROS)-induced axonal degeneration by cocultured astrocytes. Brain Res. 1996, 743, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Lodge, D.M.; Neville, D.; Brown, T.; Rupani, H.; Babu, K.S.; Bishop, L.; Heiden, E.; Gates, J.; Longstaff, J.; Winter, J.; et al. Bedside measurement of exhaled breath condensate hydrogen peroxide differentiates lung cancer and interstitial lung disease from healthy controls. Thorax 2019, 74, A133. [Google Scholar] [CrossRef]
- Burton, G.W.; Ingold, K.U. β-Carotene: An unusual type of lipid antioxidant. Science 1984, 224, 569–573. [Google Scholar] [CrossRef]
- Podrez, E.A.; Abu-Soud, H.M.; Hazen, S.L. Myeloperoxidase-generated oxidants and atherosclerosis. Free Radic. Biol. Med. 2000, 28, 1717–1725. [Google Scholar] [CrossRef]
- Ahmad, R.; Ahsan, H. Singlet oxygen species and systemic lupus erythematosus: A brief review. J. Immunoass. Immunochem. 2019, 40, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Singha, S.; Das Gupta, B.; Sarkar, A.; Jana, S.; Bharadwaj, P.K.; Sharma, N.; Haldar, P.K.; Mukherjee, P.K.; Kar, A. Chemo-profiling and exploring therapeutic potential of Momordica dioica Roxb. ex Willd. for managing metabolic related disorders: In-vitro studies, and docking based approach. J. Ethnopharmacol. 2024, 331, 118351. [Google Scholar] [CrossRef]
- Wu, H.T.; Lin, T.T.; Chen, Y.P.; Chen, F.E.; Zhang, S.D.; Pang, H.Y.; Huang, L.S.; Yu, C.; Wang, G.H.; Wu, C. Ethanol extract of Rosa laevigata Michx. fruit inhibits inflammatory responses through NF-κB/MAPK signaling pathways via AMPK activation in RAW 264.7 macrophages. Molecules 2023, 28, 2813. [Google Scholar] [CrossRef] [PubMed]
- Tsugawa, H.; Nakabayashi, R.; Mori, T.; Yamada, Y.; Takahashi, M.; Rai, A.; Sugiyama, R.; Yamamoto, H.; Nakaya, T.; Yamazaki, M.; et al. A cheminformatics approach to characterize metabolomes in stable-isotope-labeled organisms. Nat. Methods 2019, 16, 295–298. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Yu, Y.C.; Cai, M.R.; Zhang, Z.Q.; Bai, J.; Wu, H.M.; Li, P.; Zhao, T.T.; Ni, J.; Yin, X.B. UPLC-MS/MS method for the determination of the herb composition of Tangshen formula and the in vivo pharmacokinetics of its metabolites in rat plasma. Phytochem. Anal. 2022, 33, 402–426. [Google Scholar] [CrossRef] [PubMed]
- Wan, S.L.; Xie, X.X.; Yang, G.J.; Feng, F. Discovery of the toxicity-related quality markers and mechanisms of Zhi-Zi-Hou-Po decoction based on chinmedomics combined with differentially absorbed components and network pharmacology. J. Ethnopharmacol. 2024, 320, 117408. [Google Scholar] [CrossRef] [PubMed]
- Lela, L.; Ponticelli, M.; Caddeo, C.; Vassallo, A.; Ostuni, A.; Sinisgalli, C.; Faraone, I.; Santoro, V.; De Tommasi, N.; Milella, L. Nanotechnological exploitation of the antioxidant potential of Humulus lupulus L. extract. Food Chem. 2022, 393, 133401. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, R.; Halabi, E.A.; Karar, M.G.E.; Kuhnert, N. Identification and characterisation of the phenolics of Ilex glabra L. Gray (Aquifoliaceae) leaves by liquid chromatography tandem mass spectrometry. Phytochemistry 2014, 106, 141–155. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.Q.; Hu, L.P.; Liu, G.M.; Zhang, D.S.; He, H.J. Evaluation of the nutritional quality of Chinese kale (Brassica alboglabra Bailey) using UHPLC-quadrupole-orbitrap MS/MS-based metabolomics. Molecules 2017, 22, 1262. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.Q.; Zhu, H.L.; Tan, J.; Wang, H.; Wang, Z.Y.; Li, P.Y.; Zhao, C.F.; Liu, J.P. Comparative analysis of chemical constituents of Moringa oleifera leaves from China and India by ultra-performance liquid chromatography coupled with quadrupole-time-of-flight mass spectrometry. Molecules 2019, 24, 942. [Google Scholar] [CrossRef]
- Gao, Y.H.; Ma, K.; Zhu, Z.B.; Zhang, Y.; Zhou, Q.; Wang, J.; Guo, X.W.; Luo, L.T.; Wang, H.T.; Peng, K.; et al. Modified Erchen decoction ameliorates cognitive dysfunction in vascular dementia rats via inhibiting JAK2/STAT3 and JNK/BAX signaling pathways. Phytomedicine 2023, 114, 154797. [Google Scholar] [CrossRef] [PubMed]
- Żurek, N.; Pycia, K.; Pawłowska, A.; Potocki, L.; Kapusta, I.T. Chemical profiling, bioactive properties, and anticancer and antimicrobial potential of Juglans regia L. leaves. Molecules 2023, 28, 1989. [Google Scholar] [CrossRef]
- Mandim, F.; Petropoulos, S.A.; Dias, M.I.; Pinela, J.; Kostic, M.; Soković, M.; Santos-Buelga, C.; Ferreira, I.C.F.R.; Barros, L. Seasonal variation in bioactive properties and phenolic composition of cardoon (Cynara cardunculus var. altilis) bracts. Food Chem. 2021, 336, 127744. [Google Scholar] [CrossRef] [PubMed]
- Fiorentino, A.; Ricci, A.; D’Abrosca, B.; Golino, A.; Izzo, A.; Pascarella, M.T.; Piccolella, S.; Esposito, A. Kaempferol glycosides from Lobularia maritima and their potential role in plant interactions. Chem. Biodivers. 2009, 6, 204–217. [Google Scholar] [CrossRef] [PubMed]
- Świeca, M.; Herok, A.; Piwowarczyk, K.; Sikora, M.; Ostanek, P.; Gawlik-Dziki, U.; Kapusta, I.; Czyż, J. Potentially bioaccessible phenolics from Mung bean and Adzuki bean sprouts enriched with probiotic-antioxidant properties and effect on the motility and survival of AGS human gastric carcinoma cells. Molecules 2020, 25, 2963. [Google Scholar] [CrossRef]
- Aly, S.H.; Elissawy, A.M.; Mahmoud, A.M.A.; El-Tokhy, F.S.; Mageed, S.S.A.; Almahli, H.; Al-Rashood, S.T.; Binjubair, F.A.; Hassab, M.A.E.; Eldehna, W.M.; et al. Synergistic effect of Sophora japonica and Glycyrrhiza glabra flavonoid-rich fractions on wound healing: In vivo and molecular docking studies. Molecules 2023, 28, 2994. [Google Scholar] [CrossRef] [PubMed]
- Feng, Z.L.; Fang, Z.J.; Chen, C.; Vong, C.T.; Chen, J.L.; Lou, R.H.; Hoi, M.P.M.; Gan, L.S.; Lin, L.G. Anti-hyperglycemic effects of refined fractions from Cyclocarya paliurus leaves on streptozotocin-induced diabetic mice. Molecules 2021, 26, 6886. [Google Scholar] [CrossRef]
- Bouymajane, A.; Filali, F.R.; Moujane, S.; Majdoub, Y.O.E.; Otzen, P.; Channaoui, S.; Ed-Dra, A.; Bouddine, T.; Sellam, K.; Boughrous, A.A.; et al. Phenolic compound, antioxidant, antibacterial, and in silico studies of extracts from the aerial parts of Lactuca saligna L. Molecules 2024, 29, 596. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.M.; Li, Z.Y.; Cui, Q.H.; Yan, B.B.; Achi, J.G.; Zhao, Y.G.; Rong, L.J.; Du, R.K. Integrated serum pharmacochemistry and investigation of the anti-influenza a virus pneumonia effect of Qingjin Huatan decoction. J. Ethnopharmacol. 2024, 323, 117701. [Google Scholar] [CrossRef] [PubMed]
- Jayasinghe, U.L.; Jayasooriya, C.P.; Fujimoto, Y. Oleanane glycosides from the leaves of Diploclisia glaucescens. Fitoterapia 2002, 73, 406–410. [Google Scholar] [CrossRef]
- Ling, Y.; Lin, Z.Q.; Zha, W.J.; Lian, T.; You, S.S. Rapid detection and characterisation of triterpene saponins from the root of Pulsatilla chinensis (Bunge) regel by HPLC-ESI-QTOF-MS/MS. Phytochem. Anal. 2016, 27, 174–183. [Google Scholar] [CrossRef] [PubMed]
- Li, X.C.; Wang, D.Z.; Wu, S.G.; Yang, C.R. Triterpenoid saponins from Pulsatilla campanella. Phytochemistry. 1990, 29, 595–599. [Google Scholar] [CrossRef]
- Lv, C.N.; Fan, L.; Wang, J.; Qin, R.L.; Xu, T.Y.; Lei, T.L.; Lu, J.C. Two new triterpenoid saponins from rhizome of Anemone amurensis. J. Asian Nat. Prod. Res. 2015, 17, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.Z.; Zhang, J.W.; Wang, C.Z.; Zhou, L.D.; Zhang, Q.H.; Yuan, C.S. Polydopamine-coated magnetic molecularly imprinted polymers with fragment template for identification of Pulsatilla saponin metabolites in rat feces with UPLC-Q-TOF-MS. J. Agric. Food Chem. 2018, 66, 653–660. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Zhang, X.; Lv, C.N.; Yu, Y.; Zhang, Y.; Lu, J.C. Quantitative and qualitative analyses of cytotoxic triterpenoids in the rhizomes of Anemone raddeana using HPLC and HPLC-ESI-Q/TOF-MS. J. Food Drug Anal. 2018, 26, 1113–1121. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Wang, Y.; Zhang, X.Q.; Zhao, S.X.; Ye, W.C. Chemical constituents of Pulsatilla dahurica. Chem. Nat. Compd. 2009, 45, 764–765. [Google Scholar] [CrossRef]
- Shokry, A.A.; El-Shiekh, R.A.; Kamel, G.; Bakr, A.F.; Sabry, D.; Ramadan, A. Anti-arthritic activity of the flavonoids fraction of ivy leaves (Hedera helix L.) standardized extract in adjuvant induced arthritis model in rats in relation to its metabolite profile using LC/MS. Biomed. Pharmacother. 2022, 145, 112456. [Google Scholar] [CrossRef]
- Ye, W.C.; Zhang, Q.W.; Zhao, S.X.; Che, C.T. Four new oleanane saponins from Anemone anhuiensis. Chem. Pharm. Bull. 2001, 49, 632–634. [Google Scholar] [CrossRef] [PubMed]
- Tabatadze, N.; Tabidze, B.; Mshvildadze, V.; Elias, R.; Dekanosidze, G.; Balansard, G.; Kemertelidze, E. Triterpene glycosides from the tubers of Leontice smirnowii. Chem. Nat. Compd. 2009, 45, 453–454. [Google Scholar] [CrossRef]
- Li, F.; Xu, K.J.; Ding, L.S.; Wang, M.K. Rapid analysis of triterpenoidic saponins in Anemone raddeana using electrospray ionization multi-stage mass spectrometry combined with silica gel column chromatography. Chin. J. Anal. Chem. 2011, 39, 219–224. [Google Scholar]
- Ye, W.; Pan, G.; Zhang, Q.; Che, C.T.; Wu, H.; Zhao, S. Five new triterpene saponins from Pulsatilla patens var. multifida. J. Nat. Prod. 1999, 62, 233–237. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.D.; Zhang, Q.; Zheng, Y.L.; Lu, J.C. Anti-breast cancer and toxicity studies of total secondary saponin from Anemone raddeana rhizome on MCF-7 cells via ROS generation and PI3K/AKT/mTOR inactivation. J. Ethnopharmacol. 2020, 259, 112984. [Google Scholar] [CrossRef]
- Ng, W.L.; Tan, J.K.; Gnanaraj, C.; Shah, M.D.; Nor Rashid, N.; Abdullah, I.; Yong, Y.S. Cytotoxicity of Physalis minima Linn (Solanaceae) fruit against HCT116 and HT29 colorectal cancer cell lines. Nat. Prod. Res. 2024, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Wang, L.; Zhan, X.; Wang, B.; Wu, J.W.; Zhou, A. A UPLC-Q-TOF/MS-based plasma metabolomics approach reveals the mechanism of compound Kushen Injection-based intervention against non-small cell lung cancer in Lewis tumor-bearing mice. Phytomedicine 2020, 76, 153259. [Google Scholar] [CrossRef] [PubMed]
- Ji, T.; Wang, J.; Xu, Z.; Cai, H.D.; Su, S.L.; Peng, X.; Ruan, H.S. Combination of mulberry leaf active components possessed synergetic effect on SD rats with diabetic nephropathy by mediating metabolism, Wnt/β-catenin and TGF-β/Smads signaling pathway. J. Ethnopharmacol. 2022, 292, 115026. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.W.; Xu, H.Y.; Zhao, H.; Geng, Y.; Ren, Y.L.; Guo, L.; Shi, J.S.; Xu, Z.H. Edgeworthia gardneri (Wall.) Meisn. water extract improves diabetes and modulates gut microbiota. J. Ethnopharmacol. 2019, 239, 111854. [Google Scholar] [CrossRef] [PubMed]
- Kerboua, M.; Monia, A.A.; Samba, N.; Silva, L.; Raposo, C.; Díez, D.; Rodilla, J.M. Phytochemical composition of Lichen Parmotrema hypoleucinum (J. Steiner) hale from Algeria. Molecules 2022, 27, 5229. [Google Scholar] [CrossRef] [PubMed]
- Narita, Y.; Inouye, K. Degradation kinetics of chlorogenic acid at various pH values and effects of ascorbic acid and epigallocatechin gallate on its stability under alkaline conditions. J. Agric. Food Chem. 2013, 61, 966–972. [Google Scholar] [CrossRef] [PubMed]
- Sultana, B.; Anwar, F.; Przybylski, R. Antioxidant potential of corncob extracts for stabilization of corn oil subjected to microwave heating. Food Chem. 2006, 104, 997–1005. [Google Scholar] [CrossRef]
- Lue, B.M.; Sorensen, A.D.M.; Jacobsen, C.; Guo, Z.; Xu, X.B. Antioxidant efficacies of rutin and rutin esters in bulk oil and oil-in-water emulsion. Eur. J. Lipid Sci. Technol. 2017, 119, 1600049. [Google Scholar] [CrossRef]
- Fossmark, R.; Martinsen, T.C.; Waldum, H.L. Adverse effects of proton pump inhibitors-evidence and plausibility. Int. J. Mol. Sci. 2019, 20, 5203. [Google Scholar] [CrossRef]
- Azlina, M.F.N.; Qodriyah, H.M.S.; Akmal, M.N.; Ibrahim, I.A.A.; Kamisah, Y. In vivo effect of Piper sarmentosum methanolic extract on stress-induced gastric ulcers in rats. Arch. Med. Sci. 2019, 15, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Jabbar, A.A.J.; Mothana, R.A.; Abdulla, M.A.; Abdullah, F.O.; Ahmed, K.A.A.; Hussen, R.R.; Hawwal, M.F.; Fantoukh, O.I.; Hasson, S. Mechanisms of anti-ulcer actions of Prangos pabularia (L.) in ethanol-induced gastric ulcer in rats. Saudi Pharm. J. 2023, 31, 101850. [Google Scholar] [CrossRef] [PubMed]
- Nur Azlina, M.F.; Kamisah, Y.; Chua, K.H.; Ibrahim, I.A.; Qodriyah, H.M. Preventive effects of tocotrienol on stress-induced gastric mucosal lesions and its relation to oxidative and inflammatory biomarkers. PLoS ONE. 2015, 10, e0139348. [Google Scholar] [CrossRef] [PubMed]
- Su, L.J.; Zhang, J.H.; Gomez, H.; Kellum, J.A.; Peng, Z.Y. Mitochondria ROS and mitophagy in acute kidney injury. Autophagy 2023, 19, 401–414. [Google Scholar] [CrossRef] [PubMed]
- Shimoyama, A.T.; Santin, J.R.; Machado, I.D.; de Oliveira e Silva, A.M.; Pereira de Melo, I.L.; Mancini-Filho, J.; Farsky, S.H.P. Antiulcerogenic activity of chlorogenic acid in different models of gastric ulcer. Naunyn Schmiedebergs Arch. Pharmacol. 2013, 386, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Gou, L.S.; Fu, X.B.; Li, S.; Lan, N.; Yin, X.X. Protective effect of rutin against acute gastric mucosal lesions induced by ischemia-reperfusion. Pharm. Biol. 2013, 51, 914–919. [Google Scholar] [CrossRef]
- Jing, Z.T.; Liu, W.; Xue, C.R.; Wu, S.X.; Chen, W.N.; Lin, X.J.; Lin, X. AKT activator SC79 protects hepatocytes from TNF-α-mediated apoptosis and alleviates D-Gal/LPS-induced liver injury. Am. J. Physiol. Gastrointest. Liver Physiol. 2019, 316, G387–G396. [Google Scholar] [CrossRef] [PubMed]
- Wang, O.; Cheng, Q.; Liu, J.; Wang, Y.; Zhao, L.; Zhou, F.; Ji, B.P. Hepatoprotective effect of Schisandra chinensis (Turcz.) Baill. lignans and its formula with Rubus idaeus on chronic alcohol-induced liver injury in mice. Food Funct. 2014, 5, 3018–3025. [Google Scholar] [CrossRef]
- Elizabeth, A.V.; Alison, M.D.; Brian, A.M. Hydrogen peroxide sensing and signaling. Mol. Cell 2007, 26, 1–14. [Google Scholar] [CrossRef]
- Nassir, F.; Ibdah, J.A. Role of mitochondria in alcoholic liver disease. World J. Gastroenterol. 2014, 20, 2136–2142. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.C.; Zhao, X.; Sun, P.; Qian, J.; Shi, Y.H.; Wang, R. Preventive effect of Gardenia jasminoides on HCl/ethanol induced gastric injury in mice. J. Pharmacol. Sci. 2017, 133, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Olaleye, M.T.; Akinmoladun, A.C. Comparative gastroprotective effect of post-treatment with low doses of rutin and cimetidine in rats. Fundam. Clin. Pharmacol. 2013, 27, 138–145. [Google Scholar] [CrossRef]
- Feng, J.X.; Sun, Y.; Wei, Z.B.; Sun, H.; Li, L.; Zhu, J.Y.; Xia, G.Q.; Zang, H. Screening the extract of Laportea bulbifera (Sieb. et Zucc.) Wedd. based on active component content, its antioxidant capacity and exploration of hepatoprotective activity in rats. Molecules 2023, 28, 6256. [Google Scholar] [CrossRef]
- Li, L.; Zang, H.; Jiang, Y.; Zhang, Y.; Mu, S.; Cao, J.; Qu, Y.; Wang, Z.; Qi, W. Acupuncture at Back-Shu and Front-Mu acupoints prevents gastric ulcer by regulating the TLR4/MyD88/NF-κB signaling pathway. Evid. Based Complement. Alternat. Med. 2021, 2021, 8214052. [Google Scholar] [CrossRef] [PubMed]



| Extracting Solvents | TCC (mg GE/g Extract) | TProC (mg BSAE/g Extract) | TMC (mg LE/g Extract) | TAC (mg BHE/g Extract) | TPheC (mg GAE/g Extract) | TPAC (mg CAE/g Extract) | TFC (mg QE/g Extract) | TTanC (mg TAE/g Extract) | GC (mg GAE/g Extract) | CTC (mg GAE/g Extract) | TTriC (mg GRE/g Extract) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Methanol | 424.65 ± 3.08 a | N.T. | 1152.54 ± 19.05 d | 4.36 ± 0.03 b | 30.92 ± 0.15 c | 15.38 ± 0.86 a | 13.73 ± 0.47 b | 22.72 ± 0.11 c | None | None | 86.85 ± 2.43 a |
| Water | 355.38 ± 2.86 c | 493.08 ± 3.15 a | 1430.19 ± 14.12 b | 4.12 ± 0.03 c | 34.53 ± 0.24 a | 11.62 ± 0.79 b | 4.04 ± 0.21 d | 27.25 ± 0.38 a | 4.33 ± 0.11 b | None | 12.30 ± 1.05 d |
| Ethanol | 433.95 ± 5.17 a | N.T. | 1260.91 ± 21.39 c | 5.71 ± 0.01 a | 19.06 ± 0.43 d | 8.97 ± 0.53 c | 6.14 ± 0.12 c | 16.31 ± 0.20 d | None | None | 73.20 ± 1.62 b |
| 80% Ethanol | 389.32 ± 3.24 b | N.T. | 1622.28 ± 15.72 a | 3.00 ± 0.00 d | 31.57 ± 0.58 b | 16.45 ± 0.20 a | 15.44 ± 0.31 a | 25.93 ± 0.18 b | 6.29 ± 0.15 a | None | 42.97 ± 0.75 c |
| Extracting Solvents | DPPH (IC50, μg/mL) | ABTS (IC50, μg/mL) | Hydroxyl Radicals (%, 2500 μg/mL) | Superoxide Radicals (IC50, μg/mL) |
|---|---|---|---|---|
| Methanol | 37.21 ± 1.66 c | 71.21 ± 2.05 d | 47.28 ± 0.43 c | 920.36 ± 3.18 b |
| Water | 93.53 ± 2.35 e | 52.36 ± 0.54 b | 41.12 ± 1.21 d | >2143 c |
| Ethanol | 99.76 ± 2.45 f | 125.03 ± 2.40 e | 40.74 ± 1.62 d | >2143 c |
| 80% Ethanol | 45.14 ± 0.88 d | 58.10 ± 1.36 c | 42.15 ± 1.08 d | >2143 c |
| Trolox * | 2.11 ± 0.10 a | 3.43 ± 0.11 a | 89.18 ± 0.87 a | N.T. |
| BHT * | 9.46 ± 0.22 b | 4.48 ± 0.04 a | 70.14 ± 1.13 b | N.T. |
| Curcumin * | N.T. | N.T. | N.T. | 78.68 ± 0.82 a |
| Extracting Solvents | TEACFRAP | TEACCUPRAC | Iron Chelation (IC50, μg/mL) | Copper Chelation (IC50, μg/mL) |
|---|---|---|---|---|
| Methanol | 0.22 ± 0.00 b | 0.14 ± 0.00 c | 921.30 ± 4.04 b | 473.20 ± 4.85 d |
| Water | 0.20 ± 0.00 b | 0.13 ± 0.00 c | 2019.96 ± 13.63 e | 347.42 ± 3.84 c |
| Ethanol | 0.18 ± 0.00 c | 0.06 ± 0.00 d | 1481.40 ± 11.92 d | 1243.17 ± 5.87 e |
| 80% Ethanol | 0.21 ± 0.00 b | 0.17 ± 0.00 b | 1065.22 ±10.94 c | 317.04 ± 3.12 b |
| Trolox * | 0.92 ± 0.01 a | 0.89 ± 0.02 a | N.T. | N.T. |
| EDTANa2 * | N.T. | N.T. | 2.28 ± 0.11 a | 31.51 ± 0.59 a |
| Extracting Solvents | H2O2 (IC50, μg/mL) | Singlet Oxygen (%, 2000 μg/mL) | β-Carotene Bleaching AAC | HClO (IC50, μg/mL) |
|---|---|---|---|---|
| Methanol | 1143.52 ± 6.17 c | 15.30 ± 1.29 d | 496.45 ± 2.59 c | None |
| Water | 970.74 ± 8.13 b | 5.91 ± 0.39 e | 443.68 ± 2.12 d | None |
| Ethanol | 1522.10 ± 10.39 e | 25.40 ± 1.46 c | 482.21 ± 2.29 c | None |
| 80% Ethanol | 1393.38 ± 8.05 d | 40.20 ± 1.18 b | 483.12 ± 2.37 c | None |
| Trolox * | N.T. | N.T. | N.T. | 11.22 ± 0.10 a |
| Lipoic acid * | N.T. | N.T. | N.T. | 24.75 ± 0.78 a |
| Gallic acid * | 30.74 ± 1.07 a | N.T. | N.T. | N.T. |
| Ferulic acid * | N.T. | 89.37 ± 1.02 a | N.T. | N.T. |
| BHT * | N.T. | N.T. | 873.67 ± 3.56 a | N.T. |
| TBHQ * | N.T. | N.T. | 802.70 ± 4.28 b | N.T. |
| Peak No. | RT (min) | Identification | Molecular Formula | Selective Ion | Full Scan MS (m/z) | MS/MS Fragments (m/z) | |
|---|---|---|---|---|---|---|---|
| Theory | Measured | ||||||
| 1 | 0.93 | Biotin | C10H16N2O3S | [M+NH4]+ | 262.1226 | 262.1223 | 216.1185 |
| 2 | 1.28 | Unknown | 280.1326 | 262.1230 | |||
| 3 | 1.35 | l-Saccharopine | C11H20N2O6 | [M]+ | 276.1321 | 276.1373 | 294.1489, 132.0983 |
| 4 | 2.43 | N-Fructosyl phenylalanine | C15H21NO7 | [M+H]+ | 328.1396 | 328.1316 | 310.1224, 282.1275, 250.1597 |
| 5 | 4.51 | l-Phenylalanine | C9H11NO2 | [M+Na]+ | 188.0688 | 188.0658 | 100.0729 |
| 6 | 6.79 | Thymine | C5H6N2O2 | [M+NH4]+ | 144.0773 | 144.0767 | 127.034, 112.9525 |
| 7 | 7.10 | Chlorogenic acid | C16H18O9 | [M+H]+ | 355.1029 | 355.0952 | 377.0768, 163.0350, 135.0406 |
| 8 | 7.62 | 4-O-Caffeoylquinic acid | C16H18O9 | [M+H]+ | 355.1029 | 355.0953 | 192.1344, 163.0350, 135.0406 |
| 9 | 8.95 | 1-O-Feruloyl-β-d-glucose | C16H20O9 | [M+NH4]+ | 374.1451 | 374.1368 | 194.1137, 163.0347 |
| 10 | 9.08 | Benzyl β-primeveroside | C18H26O10 | [M+NH4]+ | 420.1870 | 420.1781 | 295.0930, 149.0566 |
| 11 | 9.39 | 1-O-Caffeoylquinic acid | C16H18O9 | [M+H]+ | 355.1029 | 355.0959 | 192.1345, 163.0348 |
| 12 | 9.80 | 4-p-Coumaroylquinic acid/3-p-Coumaroylquinic acid | C16H18O8 | [M+H]+ | 339.1080 | 339.1008 | 192.1346, 163.0346 |
| 13 | 11.55 | 3-O-Feruloylquinic acid | C17H20O9 | [M+H]+ | 369.1185 | 369.1107 | 194.1132, 177.0506 |
| 14 | 13.05 | Kaempferol 3-O-sophoroside 7-O-rhamnoside | C33H40O20 | [M+H]+ | 757.2191 | 757.2062 | 779.1869, 611.1505, 432.2707 |
| 15 | 14.85 | Rutin | C27H30O16 | [M+H]+ | 611.1612 | 611.1481 | 633.1341, 465.0949, 303.0447 |
| 16 | 15.30 | Kaempferol 3-O-sophoroside | C27H30O16 | [M+H]+ | 611.1612 | 611.1504 | 633.1323, 465.0953, 303.0447 |
| 17 | 15.97 | Kaempferol 3-glucuronide | C21H18O12 | [M+H]+ | 463.0876 | 463.0786 | 287.0492, 194.1130 |
| 18 | 17.68 | Unknown | 183.0736 | 155.0432, 127.0124 | |||
| 19 | 18.88 | Apigenin 7-glucuronide | C21H18O11 | [M+H]+ | 447.0927 | 447.0824 | 271.0544, 194.1133 |
| 20 | 19.86 | Diosmetin 7-glucuronide | C22H20O12 | [M+H]+ | 477.1033 | 477.0926 | 301.0651, 194.1131 |
| 21 | 22.61 | Unknown | 679.4984 | 359.2275, 340.2541 | |||
| 22 | 25.84 | 1-O-{3-[(3-O-Hexopyranosylhexopyranosyl)oxy]-28-oxoolean-12-en-28-yl}hexopyranose | C48H78O18 | [M+H]+ | 943.5266 | 943.5076 | 960.5358, 781.4589 |
| 23 | 26.05 | Unknown | 905.6643 | 453.3372 | |||
| 24 | 26.37 | Hederagenin 28-O-β-d-glucopyranosyl-(1-3)-α-l-rhamnopyranosyl-(1-4)-β-d-glucopyranosyl-(1-6)-β-d-glucopyranosyl ester | C54H88O23 | [M+H]+ | 1105.5794 | 1105.5566 | 309.2008 |
| 25 | 26.75 | Pulsatiloside A | C47H76O18 | [M+H]+ | 929.5110 | 929.4904 | 768.4514 |
| 26 | 26.95 | Oleanolic acid 28-O-β-d-glucopyranosyl-(1-3)-α-l-rhamnopyranosyl-(1-4)-β-d-glucopyranosyl-(1-6)-β-d-glucopyranosyl ester | C54H88O22 | [M+H]+ | 1089.5845 | 1089.5630 | 619.4108, 455.3442 |
| 27 | 27.61 | Leonloside D | C59H96O27 | [M+H]+ | 1237.6217 | 1237.5969 | 1075.5502, 929.4957, 767.4457 |
| 28 | 28.41 | Raddeanoside R18 | C65H106O31 | [M+H]+ | 1383.6796 | 1383.6523 | 1059.5586, 913.5021 |
| 29 | 29.98 | Cussonoside B | C48H78O17 | [M+NH4]+ | 944.5583 | 944.5383 | 340.2525, 309.1122 |
| 30 | 31.67 | Hederacolchiside F | C65H106O31 | [M+NH4]+ | 1400.7062 | 1400.6834 | 340.2531, 181.1181 |
| 31 | 32.37 | Anhuienoside E | C60H98O26 | [M+NH4]+ | 1252.6690 | 1252.6458 | 944.5346, 340.2530 |
| 32 | 32.52 | Hederacolchiside E | C65H106O30 | [M+NH4]+ | 1384.7113 | 1384.6890 | 1076.5832, 516.2719, 309.1126 |
| 33 | 32.94 | Hederacoside C | C59H96O26 | [M+NH4]+ | 1238.6534 | 1238.6314 | 340.2538, 295.2222 |
| 34 | 33.17 | Raddeanoside R14 | C59H96O26 | [M+NH4]+ | 1238.6534 | 1238.6362 | 369.1878, 348.2680 |
| 35 | 36.34 | 3-O-β-d-glucopyranosyl-(1-2)-β-d-glucopyranosyl-(1-6)-β-d-galactopyranosyl-hederagenin | C48H78O19 | [M+H]+ | 959.5215 | 959.5013 | 981.4881, 797.4541, 455.3442 |
| 36 | 37.75 | Unknown | 274.2677 | 127.0123 | |||
| 37 | 38.18 | Unknown | 318.2931 | 274.2691 | |||
| 38 | 39.79 | Raddeanoside R13 | C47H76O17 | [M+NH4]+ | 930.5427 | 930.5239 | 478.3288, 460.3174 |
| 39 | 41.53 | Gingerglycolipid A | C33H56O14 | [M+H]+ | 677.3748 | 677.3609 | 694.3916, 497.3011, 353.2626 |
| 40 | 41.89 | 1-Palmitoylglycerophosphoinositol | C25H49O12P | [M+H]+ | 573.3040 | 573.2913 | 555.2849, 537.2768 |
| 41 | 42.42 | Sphinganine | C18H39NO2 | [M+H]+ | 302.3059 | 302.2983 | 230.8841, 127.0124, 100.0728 |
| 42 | 45.95 | Linolenic acid | C18H30O2 | [M+H]+ | 279.2324 | 279.2252 | 236.1960, 183.0740 |
| 43 | 47.21 | Stearidonic acid | C18H28O2 | [M+H]+ | 277.2167 | 277.2089 | 155.0428 |
| 44 | 47.60 | Palmitoleic acid | C16H30O2 | [M+Na]+ | 277.2144 | 277.2090 | 196.9607, 140.1148, 130.1559 |
| Group | Ulcer Index (mm) | Ulcer Inhibition Rate (%) |
|---|---|---|
| GI | N.T. | N.T. |
| GII | 13.33 ± 0.83 ### | 81.55 |
| GIII | 72.25 ± 3.43 *** | N.T. |
| GIV | 24.75 ± 1.26 ### *** | 65.74 |
| GV | 17.25 ± 1.68 ### *** | 76.12 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, S.; Xia, G.; Pang, H.; Zhu, J.; Li, L.; Zang, H. Phytochemical Analysis and Antioxidant Activities of Various Extracts from the Aerial Part of Anemone baicalensis Turcz.: In Vitro and In Vivo Studies. Molecules 2024, 29, 4602. https://doi.org/10.3390/molecules29194602
Sun S, Xia G, Pang H, Zhu J, Li L, Zang H. Phytochemical Analysis and Antioxidant Activities of Various Extracts from the Aerial Part of Anemone baicalensis Turcz.: In Vitro and In Vivo Studies. Molecules. 2024; 29(19):4602. https://doi.org/10.3390/molecules29194602
Chicago/Turabian StyleSun, Shuang, Guangqing Xia, Hao Pang, Junyi Zhu, Li Li, and Hao Zang. 2024. "Phytochemical Analysis and Antioxidant Activities of Various Extracts from the Aerial Part of Anemone baicalensis Turcz.: In Vitro and In Vivo Studies" Molecules 29, no. 19: 4602. https://doi.org/10.3390/molecules29194602






