The Formation of σ-Hole Bonds: A Physical Interpretation
Abstract
1. σ-Holes
2. Experiment 1: σ-Hole Bonds at Equilibrium
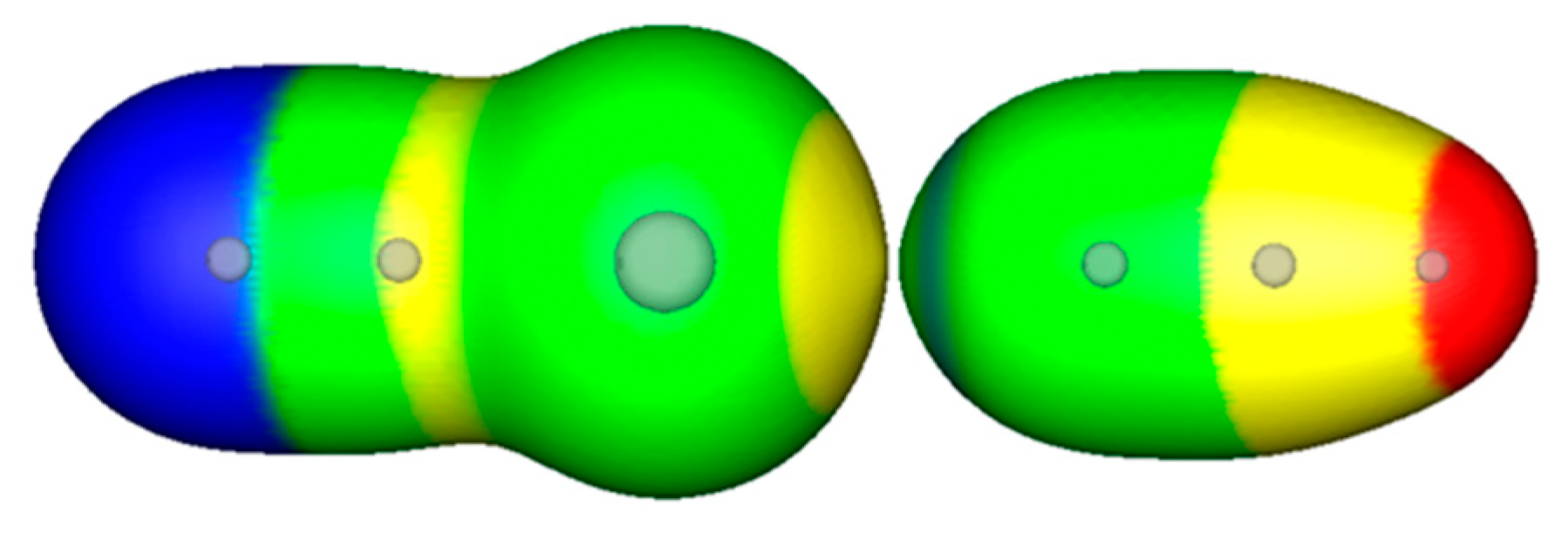
3. Experiment 2: From Infinity until Nearly Merging Electronic Densities in σ-Hole Bonding
4. Discussion and Concluding Remarks
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brinck, T.; Murray, J.S.; Politzer, P. Surface electrostatic potentials of halogenated methanes as indicators of directional intermolecular interactions. Int. J. Quant. Chem. Quant. Biol. Symp. 1992, 44, 57–64. [Google Scholar] [CrossRef]
- Auffinger, P.; Hays, F.A.; Westhof, E.; Shing Ho, P. Halogen bonds in biological molecules. Proc. Nat. Acad. Sci. USA 2004, 101, 16789–16794. [Google Scholar] [CrossRef]
- Metrangolo, P.; Neukirch, H.; Pilati, T.; Resnati, G. Halogen bonding based recognition processes: A world parallel to hydrogen bonding. Acc. Chem. Res. 2005, 38, 386–395. [Google Scholar] [CrossRef]
- Clark, T.; Hennemann, M.; Murray, J.S.; Politzer, P. Halogen bonding: The σ-hole. J. Mol. Model. 2007, 13, 291–296. [Google Scholar] [CrossRef]
- Politzer, P.; Murray, J.S.; Clark, T. Halogen bonding: An electrostatically-driven highly directional noncovalent interaction. Phys. Chem. Chem. Phys. 2010, 12, 7748–7757. [Google Scholar] [CrossRef]
- Clark, T. σ-Holes. WIREs Comput. Mol. Sci. 2013, 3, 13–20. [Google Scholar] [CrossRef]
- Politzer, P.; Murray, J.S.; Clark, T. Halogen bonding and other σ-hole interactions. Phys. Chem. Chem. Phys. 2013, 15, 11178–11189. [Google Scholar] [CrossRef] [PubMed]
- Bauzá, A.; Mooibroek, T.J.; Frontera, A. The bright future of unconventional σ/π-hole interactions. ChemPhysChem 2015, 16, 2496–2517. [Google Scholar] [CrossRef] [PubMed]
- Hobza, P. Non-covalent bonds with σ-hole: Halogen, chalcogen and pnictogen bonds. Chem. Listy 2016, 110, 371–375. [Google Scholar]
- Wang, H.; Wang, W.; Jin, W.H. σ-Hole bond vs π-hole bond: A comparison based on halogen bond. Chem. Rev. 2016, 116, 5072–5104. [Google Scholar] [CrossRef] [PubMed]
- Politzer, P.; Murray, J.S.; Clark, T.; Resnati, G. The σ-hole revisited. Phys. Chem. Chem. Phys. 2017, 19, 32166–32178. [Google Scholar] [CrossRef]
- Lim, J.Y.C.; Beer, P.D. Sigma-hole interactions in anion recognition. Chem 2018, 4, 731–783. [Google Scholar] [CrossRef]
- Panikkattu, V.V.; Tran, A.; Hinha, A.S.; Reinheimer, E.W.; Guidez, E.B.; Aakeröy, C.B. Traversing the tightrope between halogen and chalcogen bonds using structural chemistry and theory. Cryst. Growth Des. 2021, 21, 7168–7178. [Google Scholar] [CrossRef]
- Grabowski, S. σ-Hole bonds and the VSEPR Model—From the tetrahedral structure to the trigonal bipyramid. Sci 2022, 4, 17. [Google Scholar] [CrossRef]
- Guidez, E.B. Quasi-atomic orbital analysis of halogen bonding interactions. J. Chem. Phys. 2023, 159, 194307. [Google Scholar] [CrossRef] [PubMed]
- Bauzá, A.; Frontera, A. Aerogen bonding interaction: A new supramolecular force? Angew. Chem. Int. Ed. 2015, 54, 7340–7343. [Google Scholar] [CrossRef] [PubMed]
- Stenlid, J.H.; Brinck, T. Extending the σ-hole concept to metals: An electrostatic interpretation of the effects of nanostructure in gold and platinum catalysis. J. Am. Chem. Soc. 2017, 139, 11012–11015. [Google Scholar] [CrossRef] [PubMed]
- Stenlid, J.H.; Johansson, A.J.; Brinck, T. σ-Holes and σ-lumps direct the Lewis basic and acidic interactions of noble metal nanoparticles: Introducing regium bonds. Phys. Chem. Chem. Phys. 2018, 20, 2676–2692. [Google Scholar] [CrossRef] [PubMed]
- Joy, J.; Jemmis, E.D. Designing M-bond (X-M···Y, M = transition metal): σ-hole and radial density distribution. J. Chem. Sci. 2019, 131, 117. [Google Scholar] [CrossRef]
- Bauzá, A.; Alkorta, I.; Elguero, J.; Mooibroek, T.J.; Frontera, A. Spodium bonds: Noncovalent interactions involving Group 12 elements. Angew. Chem. Int. Ed. 2020, 59, 17482–17487. [Google Scholar] [CrossRef] [PubMed]
- Resnati, G.; Metrangolo, P. Celebrating 150 years from Mendelev: The periodic table of chemical interactions. Coord. Chem. Rev. 2020, 420, 213409. [Google Scholar] [CrossRef]
- Daolio, A.; Pizzi, A.; Calabrese, M.; Terraneo, G.; Bordignon, S.; Frontera, A.; Resnati, G. Anion⋯Anion coinage bonds: The case of tetrachloridoaurate. Angew. Chem. Int. Ed. 2021, 60, 14385–14389. [Google Scholar] [CrossRef] [PubMed]
- Bauzá, A.; Frontera, A. Noncovalent interactions involving group 6 in biological systems: The case of Molybdopterin and Tungstopterin cofactors. Chem. Eur. J. 2022, 28, e202201660. [Google Scholar] [CrossRef] [PubMed]
- Murray, J.S.; Politzer, P. Molecular electrostatic potentials and noncovalent interactions. WIRES Comput. Mol. Sci. 2017, 7, e1326. [Google Scholar] [CrossRef]
- Sen, K.D.; Politzer, P. Characteristic features of the electrostatic potentials of singly-negative monoatomic ions. J. Chem. Phys. 1989, 90, 4370–4372. [Google Scholar] [CrossRef]
- Politzer, P.; Murray, J.S. The neglected nuclei. Molecules 2021, 26, 2982. [Google Scholar] [CrossRef] [PubMed]
- Politzer, P.; Laurence, P.R.; Jayasuriya, K. Molecular electrostatic potentials: An effective tool for elucidation of biochemical phenomena. Environ. Health Perspect. 1985, 61, 191. [Google Scholar] [CrossRef]
- Murray, J.S.; Politzer, P. Can counter-intuitive halogen bonding be Coulombic? ChemPhysChem 2021, 22, 1201–1207. [Google Scholar] [CrossRef]
- Scheiner, S. Does a halogen bond require positive potential on the acid and negative potential on the base? Phys. Chem. Chem. Phys. 2023, 25, 7184–7194. [Google Scholar] [CrossRef]
- Murray, J.S.; Politzer, P. Interaction and polarization energy relationships in σ-hole and π-hole bonding. Crystals 2020, 10, 76. [Google Scholar] [CrossRef]
- Bartlett, R.J.; Weinstein, H. Theoretical treatment of multiple site reactivity in large molecules. Chem. Phys. Lett. 1975, 30, 441–447. [Google Scholar] [CrossRef]
- Bonaccorsi, R.; Scrocco, E.; Tomasi, J. A representation of the polarization term in the interaction energy between a molecule and a point-like charge. Theoret. Chim. Acta 1976, 43, 63–73. [Google Scholar] [CrossRef]
- Francl, M.M. Polarization correction to electrostatic potentials. J. Phys. Chem. 1985, 89, 428–433. [Google Scholar] [CrossRef]
- Reed, A.E.; Curtiss, L.A.; Weinhold, F. Intermolecular interactions form a natural bond orbital, donor-acceptor viewpoint. Chem. Rev. 1988, 88, 899–926. [Google Scholar] [CrossRef]
- Solimannejad, M.; Malekani, M.; Alkorta, I. Cooperative and diminutive unusual weak bonding in F3CX⋯HMgH⋯Y and F3CX⋯Y⋯HMgH trimers (X = Cl, Br; Y=HCN, and HNC). J. Phys. Chem. A 2010, 114, 12106–12111. [Google Scholar] [CrossRef] [PubMed]
- Geronimo, I.; Singh, N.J.; Kim, K.S. Can electron-rich π systems bind atoms? J. Chem. Theor. Comput. 2011, 7, 825–829. [Google Scholar] [CrossRef] [PubMed]
- Politzer, P.; Riley, K.E.; Bulat, F.A.; Murray, J.S. Perspectives on halogen bonding and other σ-hole interactions: Lex parsimoniae (Occam’s Razor). Comput. Theor. Chem. 2012, 998, 2–8. [Google Scholar] [CrossRef]
- Politzer, P.; Murray, J.S.; Clark, T. Mathematical modeling and physical reality in noncovalent interactions. J. Mol. Model. 2015, 21, 52. [Google Scholar] [CrossRef] [PubMed]
- Clark, T. Polarization, donor-acceptor interactions and covalent contributions in weak interactions: A clarification. J. Mol. Model. 2017, 23, 297. [Google Scholar] [CrossRef]
- Edwards, A.J.; Mackenzie, C.F.; Spackman, P.R.; Jayatilaka, D.; Spackman, M.A. Intermolecular interactions in molecular crystals: What’s in a name? Faraday Discuss. 2017, 203, 93–112. [Google Scholar] [CrossRef]
- Clark, T.; Murray, J.S.; Politzer, P. A perspective on quantum mechanics and chemical concepts in describing noncovalent interactions. Phys. Chem. Chem. Phys. 2018, 20, 30076–30082. [Google Scholar] [CrossRef]
- Murray, J.S.; Politzer, P. In search of the ‘impenetrable volume’ of a molecule in a noncovalent complex. Mol. Phys. 2018, 116, 570–577. [Google Scholar] [CrossRef]
- Murray, J.S.; Zadeh, D.H.; Lane, P.; Politzer, P. The role of ‘excluded’ electronic charge in noncovalent interactions. Mol. Phys. 2019, 117, 2260–2266. [Google Scholar] [CrossRef]
- Bader, R.F.W.; Carroll, M.T.; Cheeseman, J.R.; Chang, C. Properties of atoms in molecules: Atomic volumes. J. Am. Chem. Soc. 1987, 109, 7968–7979. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 09, Revision A.1; Gaussian, Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 16W, Version 1.1; Gaussian, Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Bulat, F.A.; Toro-Labbé, A.; Brinck, T.; Murray, J.S.; Politzer, P. Quantitative analysis of molecular surfaces: Areas, volumes, electrostatic potentials and average local ionization energies. J. Mol. Model. 2010, 16, 1679–1691. [Google Scholar] [CrossRef]
- Zhao, Y.; Truhlar, D.G. Density functionals with broad applicability in chemistry. Acc. Chem. Res. 2008, 41, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Truhlar, D.G. The M06 suite of density functional for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: Two new functionals and systematic testing of four M06-class functionals and 12 other functionals. Theor. Chem. Acc. 2008, 120, 215–241. [Google Scholar]
- Roos, G.; Murray, J.S. Intramolecular repulsion visible through the electrostatic potential in disulfides: Analysis via varying iso-density contours. J. Phys. Chem. A 2023, 127, 8354–8364. [Google Scholar] [CrossRef]
- Roos, G.; Murray, J.S. Probing intramolecular interactions using molecular electrostatic potentials: Changing electron density contours to unveil both attractive and repulsive interactions. Phys. Chem. Chem. Phys. 2024; submitted. [Google Scholar]
- Bondi, A. van der Waals volumes and radii. J. Phys. Chem. 1964, 68, 441–451. [Google Scholar] [CrossRef]
- Rowland, R.; Taylor, R. Intermolecular nonbonded contact distances in organic crystal structures: Comparison with distances to van der Waals radii. Phys. Chem. 1996, 100, 7384–7391. [Google Scholar] [CrossRef]
- Dance, I. Distance criteria for crystal packing analysis of supramolecular motifs. New J. Chem. 2003, 27, 22–27. [Google Scholar] [CrossRef]
- Alvarez, S. A cartography of the van der Waals territories. Dalton Trans. 2013, 42, 8617–8636. [Google Scholar] [CrossRef] [PubMed]
- Politzer, P.; Murray, J.S. The use and misuse of van der Waals radii. Struct. Chem. 2021, 32, 623–629. [Google Scholar] [CrossRef]



| Complex | ΔE (kcal/mol) | Equilibrium Distance A···B (Å) | Eq. Dist. A···B/Sum of vdW Radii a | Contour X (a.u.) of Nearly-Touching Point b |
|---|---|---|---|---|
| Ar···Ar | −0.2 | 3.76 | 1.00 | 0.0015 |
| H3C-Cl···OCO | −0.5 | 3.26 | 1.00 | 0.0046 |
| NCF···NCH | −1.5 | 2.98 | 0.99 | 0.006 |
| H3C-Cl···NH3 | −1.9 | 3.10 | 0.94 | 0.011 |
| NCF···NH3 | −1.9 | 2.97 | 0.98 | 0.0081 |
| NCH···OCO | −2.2 | 2.24 | 0.82 | 0.011 |
| NCCl···NCH | −3.7 | 2.91 | 0.88 | 0.013 |
| NCBr···NCH | −4.6 | 2.97 | 0.87 | 0.014 |
| NCH···NCH | −4.9 | 2.22 | 0.84 | 0.015 |
| NC(F)Se···NCH c | −5.3 | 2.94 | 0.85 | 0.015 |
| FBr···NCH | −7.1 | 2.64 | 0.78 | 0.026 |
| ClCl···NH3 | −7.9 | 2.60 | 0.79 | 0.0277 |
| NCH···NH3 | −8.5 | 2.10 | 0.76 | 0.0222 |
| NCH···N(CH3)3 | −13.1 | 2.01 | 0.73 | 0.029 |
| FBr···NH3 | −15.6 | 2.38 | 0.70 | 0.056 |
| Complex | ΔE (kcal/mol) | Eq. Dist. A···B/Sum of vdW Radii a | Dist. of A···B at Merging Point of 0.001 a.u. Envelopes/Sum of vdW Radii a |
|---|---|---|---|
| Ar···Ar | −0.2 | 1.00 | 1.14 |
| H3C-Cl···OCO | −0.5 | 1.00 | 1.20 |
| NCF···NCH | −1.5 | 0.99 | 1.22 |
| H3C-Cl···NH3 | −1.9 | 0.94 | 1.29 |
| NCF···NH3 | −1.9 | 0.98 | 1.26 |
| NCH···OCO | −2.2 | 0.82 | 1.20 |
| NCCl···NCH | −3.7 | 0.88 | 1.23 |
| NCBr···NCH | −4.6 | 0.87 | 1.23 |
| NCH···NCH | −4.9 | 0.84 | 1.24 |
| NC(F)Se···NCH b | −5.3 | 0.85 | 1.23 |
| FBr···NCH | −7.1 | 0.78 | 1.23 |
| ClCl···NH3 | −7.9 | 0.79 | 1.28 |
| NCH···NH3 | −8.5 | 0.76 | 1.31 |
| NCH···N(CH3)3 | −13.1 | 0.73 | 1.33 |
| FBr···NH3 | −15.6 | 0.70 | 1.27 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Murray, J.S. The Formation of σ-Hole Bonds: A Physical Interpretation. Molecules 2024, 29, 600. https://doi.org/10.3390/molecules29030600
Murray JS. The Formation of σ-Hole Bonds: A Physical Interpretation. Molecules. 2024; 29(3):600. https://doi.org/10.3390/molecules29030600
Chicago/Turabian StyleMurray, Jane S. 2024. "The Formation of σ-Hole Bonds: A Physical Interpretation" Molecules 29, no. 3: 600. https://doi.org/10.3390/molecules29030600
APA StyleMurray, J. S. (2024). The Formation of σ-Hole Bonds: A Physical Interpretation. Molecules, 29(3), 600. https://doi.org/10.3390/molecules29030600







