Design, Synthesis, and Anti-Cancer Evaluation of Novel Water-Soluble Copper(I) Complexes Bearing Terpyridine and PTA Ligands
Abstract
1. Introduction
2. Results and Discussion
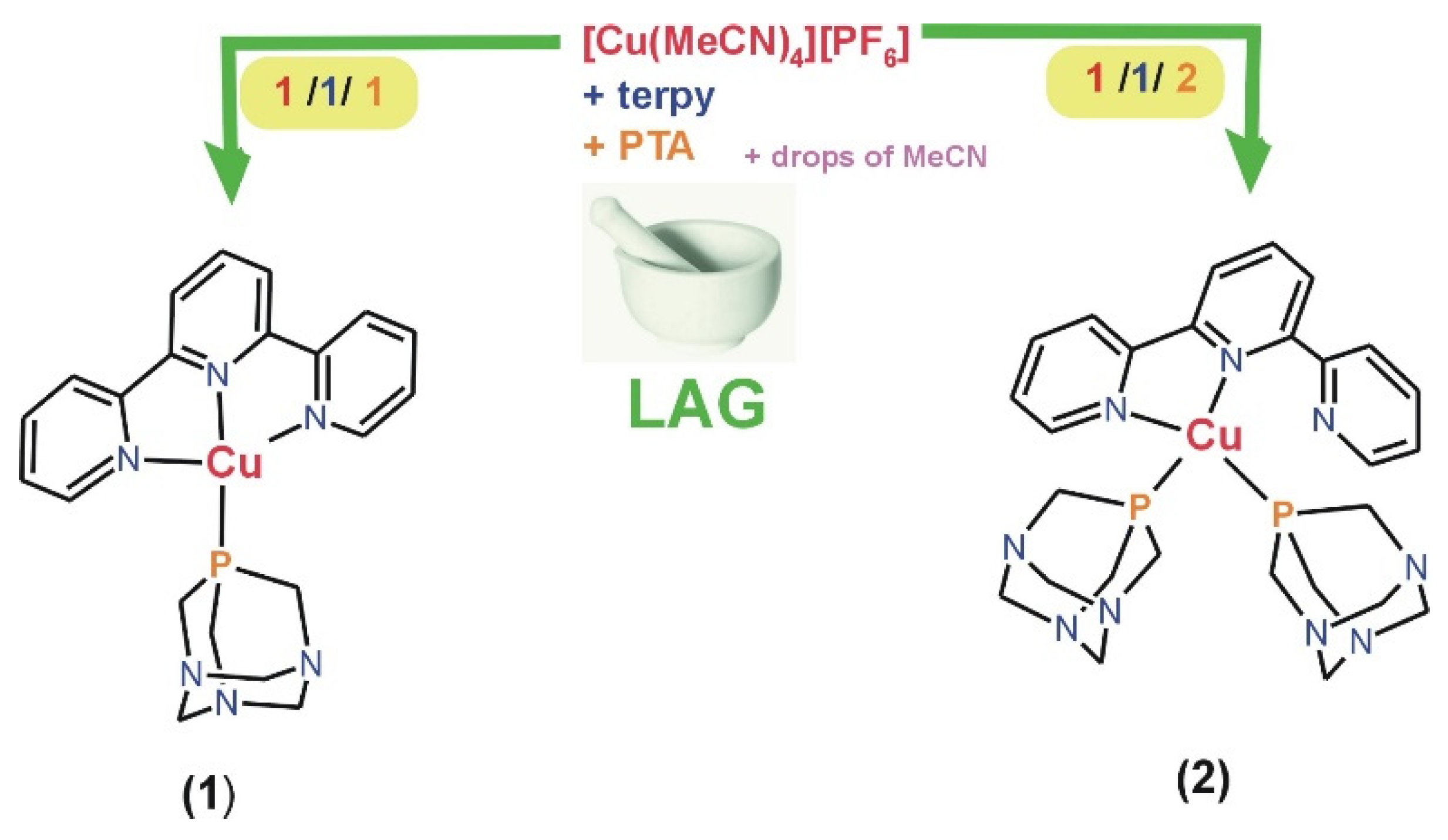
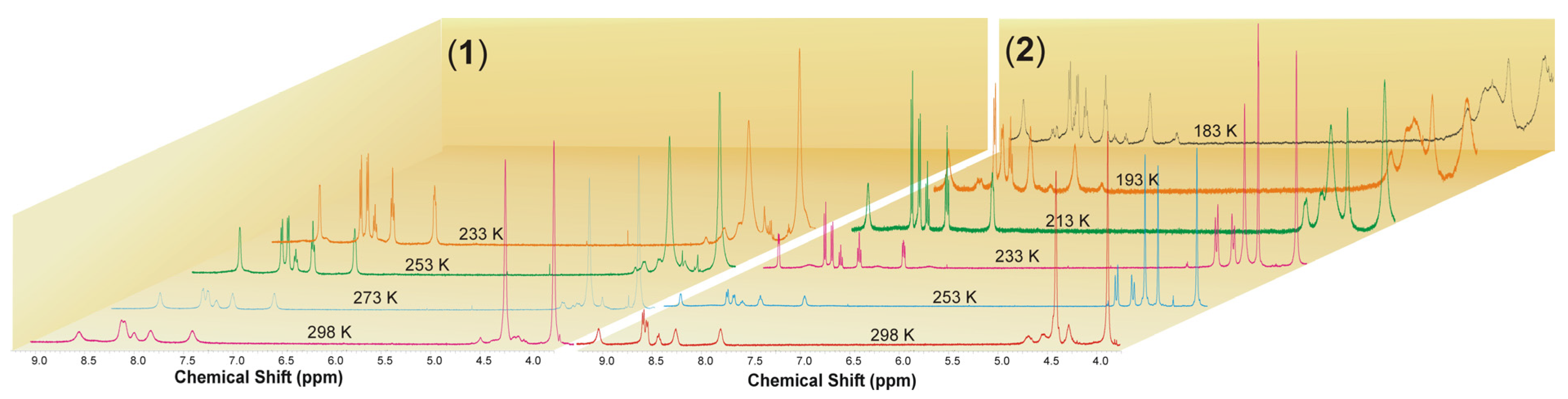
2.1. Synthesis and Characterization
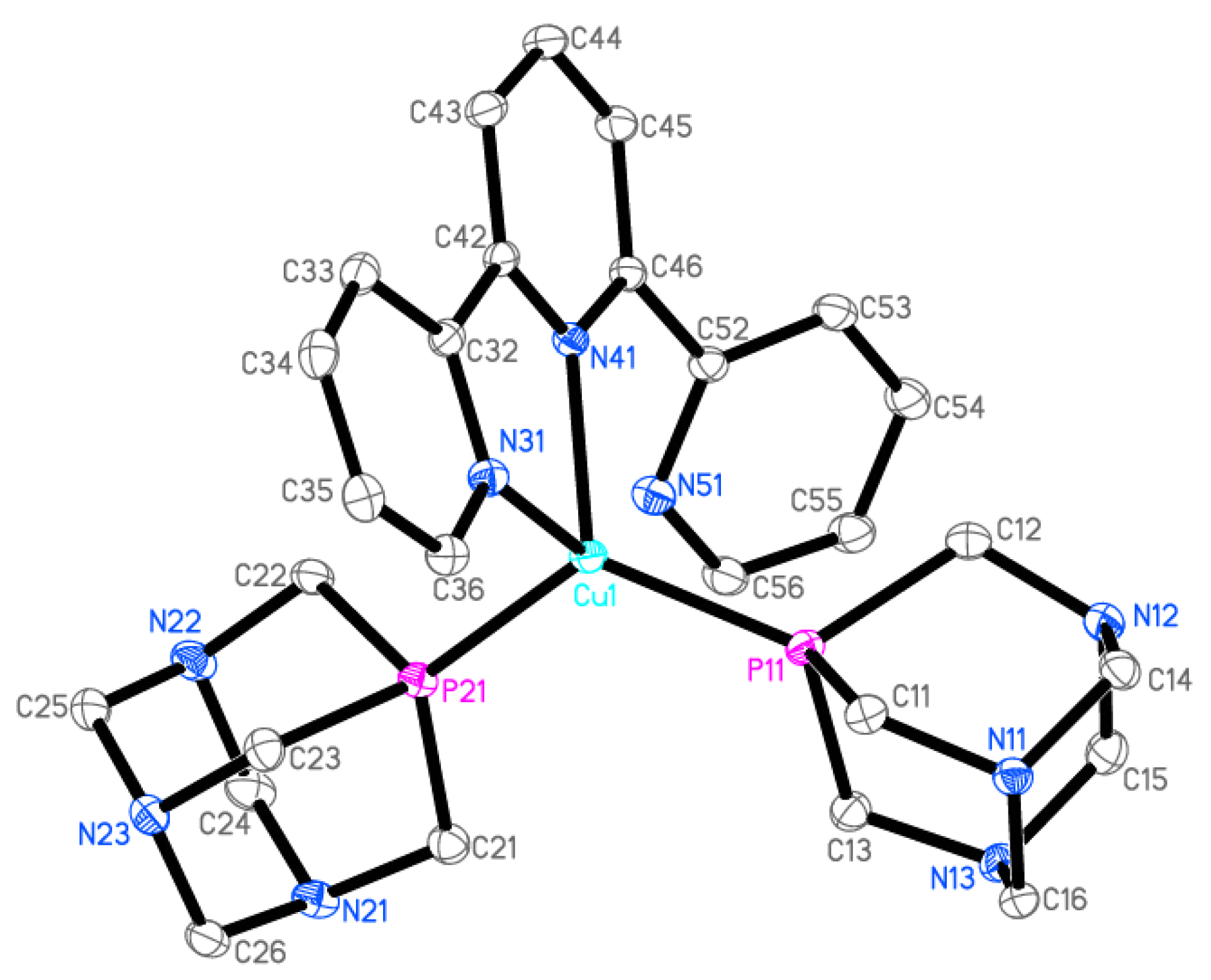
2.2. Crystal Structure of 2
2.3. Cytotoxic Activity
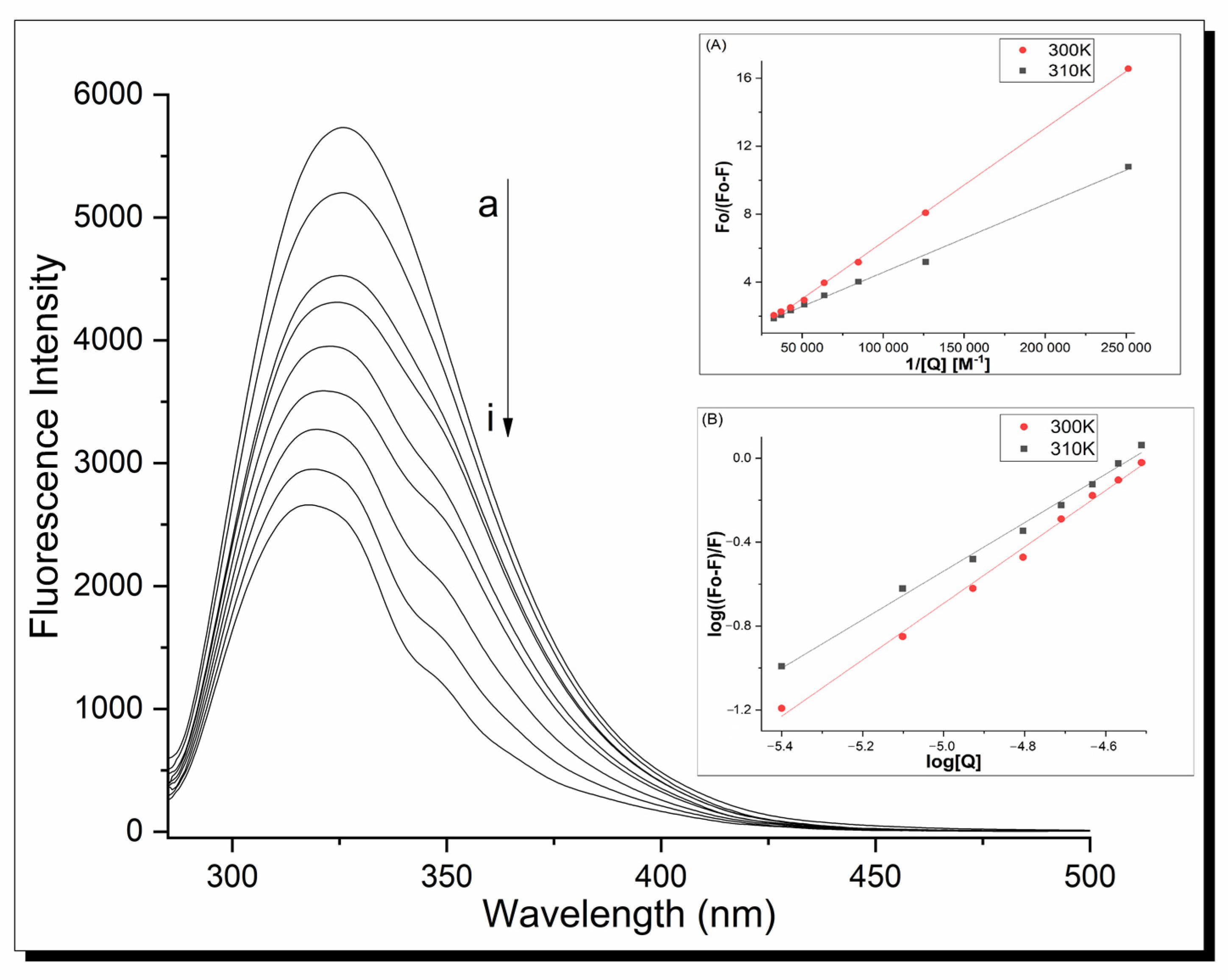
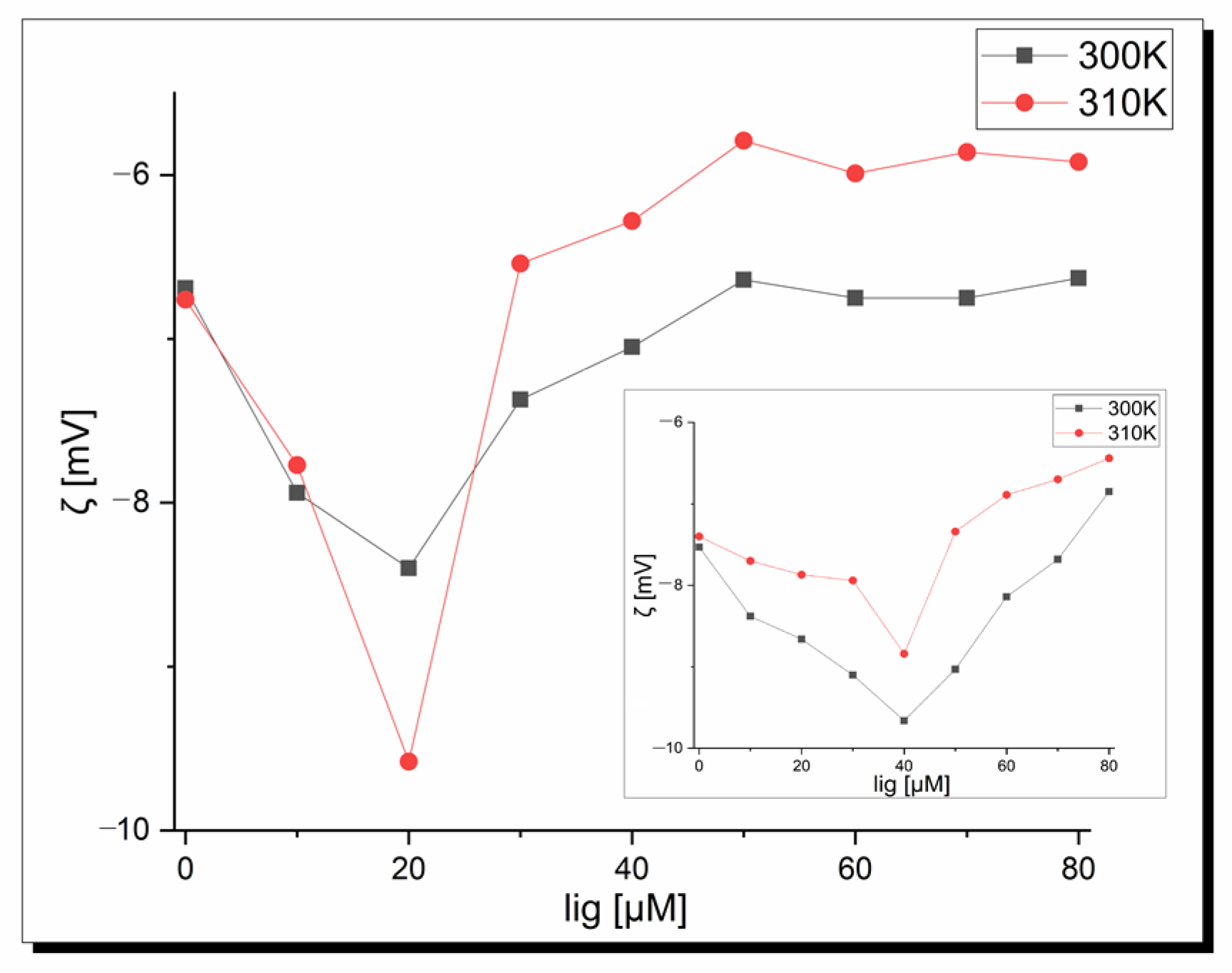
2.4. Apo-Transferrin Interactions
3. Materials and Methods
3.1. Chemicals
3.2. Synthesis of 1 and 2
3.3. X-ray Crystallography
3.4. X-ray Crystal Structure Determination
3.5. Cell Cultures
3.6. Cytotoxicity Assay
3.7. Apo-Transferrin Interactions
3.7.1. Fluorescence Measurements
3.7.2. Circular Dichroism Spectroscopy
3.7.3. Zeta Potential Measurements
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Soerjomataram, I.; Bray, F. Planning for tomorrow: Global cancer incidence and the role of prevention 2020–2070. Nat. Rev. Clin. Oncol. 2021, 18, 663–672. [Google Scholar] [CrossRef]
- Marzano, C.; Pellei, M.; Tisato, F.; Santini, C. Copper complexes as anticancer agents. Anticancer Agents Med. Chem. 2009, 9, 185–211. [Google Scholar] [CrossRef]
- Santini, C.; Pellei, M.; Gandin, V.; Porchia, M.; Tisato, F.; Marzano, C. Advances in copper complexes as anticancer agents. Chem. Rev. 2014, 114, 815–862. [Google Scholar] [CrossRef]
- Denoyer, D.; Clatworthy, S.A.S.; Cater, M.A. Copper Complexes in Cancer Therapy. Met. Ions Life Sci. 2018, 18, 469–506. [Google Scholar]
- Duncan, C.; White, A.R. Copper complexes as therapeutic agents. Metallomics 2012, 4, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Rajalakshmi, S.; Fathima, A.; Rao, J.R.; Nair, B.U. Antibacterial activity of copper (II) complexes against Staphylococcus aureus. RSC Adv. 2014, 4, 32004–32012. [Google Scholar] [CrossRef]
- Teguh, H.S.; Fahimah, M. Synthesis of metal-organic (complexes) compounds copper(II)-imidazole for antiviral HIV candidate. Ind. J. Trop. Infect. Dis. 2016, 6, 5–11. [Google Scholar]
- Wei, C.; He, Y.; Shi, X.; Song, Z. Terpyridine-metal complexes: Applications in catalysis and supramolecular chemistry. Coord. Chem. Rev. 2019, 385, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Murray, N.S.; Keller, S.; Constable, E.C.; Housecroft, C.E.; Neuburger, M.; Prescimone, A. [Cu(N^N)(P^P)]+ complexes with 2,2′:6′,2″-terpyridine ligands as the N^N domain. Dalton Trans. 2015, 44, 7626–7633. [Google Scholar] [CrossRef] [PubMed]
- Ainscough, E.W.; Brodie, A.M.; Ingham, S.L.; Waters, J.M. Synthesis and structural characterisation of five-co-ordinate copper(I) and silver(I) 2,2′:6′,2″-terpyridine complexes. J. Chem. Soc. Dalton Trans. 1994, 2, 215–220. [Google Scholar] [CrossRef]
- Feng, Q.; Li, D.; Yin, Y.; Feng, X.; Cai, J. Synthesis, structure and spectroanalysis studies of a five-coordinated Cu(I) complex [Cu(4′-Phtpy)(PPh3)2](BF4). Acta Chim. Sinica 2002, 60, 2167–2171. [Google Scholar]
- Polson, M.I.J.; Hana, G.S.; Taylor, N.J. [4′-(2-Bromo-5-pyrid-yl)-2,2′:6′,2′′-terpyridine-κ3 N,N′,N′′]bis(triphenyl-phosphine-κP)-copper(I) tetra-fluorido-borate dichloro-methane hemisolvate. Acta Crystallogr. Sect. Struct. Rep. Online 2008, 64, m205. [Google Scholar] [CrossRef]
- Phillips, D.; Gonsalvi, L.; Romerosa, A.; Vizza, F.; Peruzzini, M. Coordination chemistry of 1,3,5-triaza-7-phosphaadamantane (PTA): Transition metal complexes and related catalytic, medicinal and photoluminescent applications. Coord. Chem. Rev. 2004, 248, 955–993. [Google Scholar] [CrossRef]
- Bravo, J.; Bolaño, J.; Gonsalvi, L.; Peruzzini, M. Coordination chemistry of 1,3,5-triaza-7-phosphaadamantane (PTA) and derivatives. Part II. The quest for tailored ligands, complexes and related applications. Coord. Chem. Rev. 2010, 254, 555–607. [Google Scholar] [CrossRef]
- Guerriero, A.; Peruzzini, M.; Gonsalvi, L. Coordination chemistry of 1,3,5-triaza-7-phosphatricyclo[3.3.1.1]decane (PTA) and derivatives. Part III. Variations on a theme: Novel architectures, materials and applications. Coord. Chem. Rev. 2018, 355, 328–361. [Google Scholar] [CrossRef]
- Armstrong, D.S.; Kirk, M.; Murphy, C.; Guerriero, A.; Peruzzini, M.; Gonsalvi, L.; Phillips, A.D. Water-Soluble Silver(I) Complexes Featuring the Hemilabile 3,7-Dimethyl-1,3,5-triaza-7-phosphabicyclo[3.3.1]nonane Ligand: Synthesis, Characterization, and Antimicrobial Activity. Inorg. Chem. 2018, 57, 6309–6323. [Google Scholar]
- Rancan, M.; Tessarolo, J.; Quicic, S.; Armelao, L. Post-assembly guest oxidation in a metallo-supramolecular host and structural rearrangement to a coordination polymer. Chem. Commun. 2014, 50, 13761–13764. [Google Scholar] [CrossRef]
- Shenderovich, I.G. 1,3,5-Triaza-7-Phosphaadamantane (PTA) as a 31P NMR Probe for Organometallic Transition Metal Complexes in Solution. Molecules 2021, 26, 1390. [Google Scholar] [CrossRef]
- Battistin, F.; Balducci, G.; Iengo, E.; Demitri, N.; Alessio, E. Neutral 1,3,5-Triaza-7-phosphaadamantane-Ruthenium(II) Complexes as Precursors for the Preparation of Highly Water-Soluble Derivatives. Eur. J. Inorg. Chem. 2016, 2016, 2850–2860. [Google Scholar] [CrossRef]
- Battistin, F.; Balducci, G.; Milani, B.; Alessio, E. Water-Soluble Ruthenium(II) Carbonyls with 1,3,5-Triaza-7-phosphaadamantane. Inorg. Chem. 2018, 57, 6991–7005. [Google Scholar] [CrossRef] [PubMed]
- García-Álvarez, J.; Díez, J.; Vidal, C.; Vicent, C. New Ag(I)−Iminophosphorane Coordination Polymers as Efficient Catalysts Precursors for the MW-Assisted Meyer−Schuster Rearrangement of Propargylic Alcohols in Water. Inorg. Chem. 2013, 52, 6533–6542. [Google Scholar] [CrossRef]
- Scalambra, F.; Holzmann, N.; Bernasconi, L.; Imberti, S.; Romerosa, A. The Interaction of Water with cis and trans {Ru(bpy)2(PTA)2}2+ (PTA = 1,3,5-Triaza-7-phosphaadamantane) Studied by Neutron Scattering and Ab Initio Calculations. Eur. J. Inorg. Chem. 2019, 2019, 1162–1169. [Google Scholar] [CrossRef]
- Pettinari, R.; Condello, F.; Marchetti, F.; Pettinari, C.; Smoleński, P.; Riedel, T.; Scopelliti, R.; Dyson, P.J. Dicationic Ruthenium(II)-Arene-Curcumin Complexes Containing Methylated 1,3,5-Triaza-7-phosphaadamantane: Synthesis, Structure, and Cytotoxicity. Eur. J. Inorg. Chem. 2017, 2017, 2905–2910. [Google Scholar] [CrossRef]
- Pettinari, C.; Marchetti, F.; Lupidi, G.; Quassinti, L.; Bramucci, M.; Petrelli, D.; Vitali, L.A.; Guedes da Silva, M.F.C.; Martins, L.M.D.R.S.; Smoleński, P.; et al. Synthesis, Antimicrobial and Antiproliferative Activity of Novel Silver(I) Tris(pyrazolyl)methanesulfonate and 1,3,5-Triaza-7-phosphadamantane Complexes. Inorg. Chem. 2011, 50, 11173–11184. [Google Scholar] [CrossRef] [PubMed]
- Smoleński, P.; Jaros, S.W.; Pettinari, C.; Lupidi, G.; Quassinti, L.; Bramucci, M.; Vitali, L.A.; Petrelli, D.; Kochel, A.; Kirillov, A.M. New water-soluble polypyridine silver(i) derivatives of 1,3,5-triaza-7-phosphaadamantane (PTA) with significant antimicrobial and antiproliferative activities. Dalton Trans. 2013, 42, 6572–6581. [Google Scholar] [CrossRef] [PubMed]
- Smoleński, P.; Pettinari, C.; Marchetti, F.; Guedes da Silva, M.F.C.; Lupidi, G.; Patzmay, G.V.B.; Petrelli, D.; Vitali, L.; Pombeiro, A.J.L. Syntheses, Structures, and Antimicrobial Activity of New Remarkably Light-Stable and Water-Soluble Tris(pyrazolyl)methanesulfonate Silver(I) Derivatives of N-Methyl-1,3,5-triaza-7-phosphaadamantane Salt—[mPTA]BF4. Inorg. Chem. 2015, 54, 434–440. [Google Scholar] [CrossRef] [PubMed]
- Jaros, S.W.; Guedes da Silva, M.F.C.; Król, J.; Oliveira, M.C.; Smoleński, P.; Pombeiro, A.J.L.; Kirillov, A.M. Bioactive Silver-Organic Networks Assembled from 1,3,5-Triaza-7-Phosphaadamantane and Flexible Cyclohexanecarboxylate Blocks. Inorg. Chem. 2016, 55, 1486–1496. [Google Scholar] [CrossRef] [PubMed]
- Jaros, S.W.; Guedes da Silva, M.F.C.; Florek, M.; Smoleński, P.; Pombeiro, A.J.L.; Kirillov, A.M. Silver(I)-1,3,5-Triaza-7-phosphaadamantane Coordination Polymers Driven by Substituted Glutarate and Malonate Building Blocks: Self-assembly Synthesis, Structural Features, and Antimicrobial Properties. Inorg. Chem. 2016, 55, 5886–5894. [Google Scholar] [CrossRef]
- Smoleński, P. Ligands—Synthesis, Characterization and Role in Biotechnology, 1st ed.; Gawryszewska, P., Smoleński, P., Eds.; Nova Science Publishers, Inc.: New York, NY, USA, 2014; pp. 141–176. [Google Scholar]
- Jaros, S.W.; Śliwińska-Hill, U.; Białońska, A.; Nesterov, D.S.; Kuropka, P.; Sokolnicki, J.; Bażanów, B.; Smoleński, P. Light-stable polypyridine silver(i) complexes of 1,3,5-triaza-7-phosphaadamantane (PTA) and 1,3,5-triaza-7-phosphaadamantane-7-sulfide (PTA[double bond, length as m-dash]S): Significant antiproliferative activity of representative examples in aqueous media. Dalton Trans. 2019, 48, 11235–11249. [Google Scholar] [CrossRef] [PubMed]
- Jaros, S.W.; Krogul-Sobczak, A.; Bażanów, B.; Florek, M.; Poradowski, D.; Nesterov, D.S.; Śliwińska-Hill, U.; Kirillov, A.M.; Smoleński, P. Self-Assembly and Multifaceted Bioactivity of a Silver(I) Quinolinate Coordination Polymer. Inorg. Chem. 2021, 60, 15435–15444. [Google Scholar] [CrossRef]
- Jaros, S.W.; Sokolnicki, J.; Wołoszyn, A.; Haukka, M.; Kirillov, A.M.; Smoleński, P. Novel 2D Coordination Network Built from Hexacopper(I)-Iodide Clusters and Cagelike Aminophosphine Blocks for Reversible “Turn-On” Sensing of Aniline. J. Mater. Chem. C 2018, 6, 1670–1678. [Google Scholar] [CrossRef]
- Śliwa, E.I.; Sliwińska-Hill, U.; Bażanów, B.; Siczek, M.; Kłak, J.; Smoleński, P. Synthesis, Structural, and Cytotoxic Properties of New Water-Soluble Copper(II) Complexes Based on 2,9-Dimethyl-1,10-Phenanthroline and Their One Derivative Containing 1,3,5-Triaza-7-Phosphaadamantane-7-Oxide. Molecules 2020, 25, 741. [Google Scholar] [CrossRef]
- Śliwa, E.I.; Nesterov, D.S.; Kłak, J.; Jakimowicz, P.; Kirillov, A.M.; Smoleński, P. Unique Copper−Organic Networks Self-Assembled from 1,3,5-Triaza-7-Phosphaadamantane and Its Oxide: Synthesis, Structural Features, and Magnetic and Catalytic Properties. Cryst. Growth Des. 2018, 18, 2814–2823. [Google Scholar] [CrossRef]
- Saravani, H.; Rezvani, A.R.; Hadadzadeh, H.; Safari, N. An Investigation of Z-in Distortion in Mononuclear Cu(II) Complex with Terpyridine Ligands, [Cu(terpy)2](PF6)2. Iran. J. Chem. Chem. Eng. 2007, 26, 103–110. [Google Scholar]
- Ceresa, C.; Nicolini, G.; Semperboni, S.; Gandin, V.; Monfrini, M.; Avezza, F.; Alberti, P.; Bravin, A.; Pellei, M.; Santini, C.; et al. Evaluation of the Profile and Mechanism of Neurotoxicity of Water-Soluble [Cu(P)4]PF6 and [Au(P)4]PF6 (P = thp or PTA) Anticancer Complexes. Neurotox. Res. 2018, 34, 93–108. [Google Scholar] [CrossRef] [PubMed]
- Kirillov, A.M.; Smoleński, P.; Guedes da Silva, M.F.C.; Pombeiro, A.J.L. The First Copper Complexes Bearing the 1,3,5-Triaza-7-phosphaadamantane (PTA) Ligand. Eur. J. Inorg. Chem. 2007, 2007, 2686. [Google Scholar] [CrossRef]
- Frost, B.J.; Mebi, C.A.; Gingrich, P.W. Boron–Nitrogen Adducts of 1,3,5-Triaza-7-phosphaadamantane (PTA): Synthesis, Reactivity, and Molecular Structure. Eur. J. Inorg. Chem. 2006, 2006, 1182–1189. [Google Scholar] [CrossRef]
- Nakamoto, K. Infrared and Raman Spectra of Inorganic and Coordination Compounds, 5th ed.; Wiley: New York, NY, USA, 1997. [Google Scholar]
- Robinson, K.; Gibbs, G.; Ribbe, P. Quadratic elongation: A quantitative measure of distortion in coordination polyhedral. Science 1971, 172, 567–570. [Google Scholar] [CrossRef]
- Fleet, M.E. Distortion Parameters for Coordination Polyhedra. Mineral. Mag. 1976, 40, 531–533. [Google Scholar] [CrossRef]
- Toyota, S.; Oki, M. Structure of Intramolecular Boron–Amine Complexes and Proposal of Tetrahedral Character for Correlation between Molecular Structure and Barrier to Dissociation of the N–B Bonds. Bull. Chem. Soc. Jpn. 1992, 65, 1832–1840. [Google Scholar] [CrossRef]
- Höpfl, H. The tetrahedral character of the boron atom newly defined—A useful tool to evaluate the N→B bond. J. Organomet. Chem. 1999, 581, 129–149. [Google Scholar] [CrossRef]
- Hutchison, A.R.; Mitra, A.; Atwood, D.A. The four coordinate geometric parameter: A new quantification of geometry for four coordinate aluminum and gallium. Main Group Chem. 2005, 4, 187–200. [Google Scholar] [CrossRef]
- Yang, L.; Powell, D.R.; Houser, R.P. Structural variation in copper(i) complexes with pyridylmethylamide ligands: Structural analysis with a new four-coordinate geometry index, τ4. Dalton Trans. 2007, 9, 955–964. [Google Scholar] [CrossRef]
- Qi, Y.; Pradipta, A.R.; Li, M.; Zhao, X.; Lu, L.; Fu, X.; Wei, J.; Hsung, R.P.; Tanaka, K.; Zhou, L. Cinchonine induces apoptosis of HeLa and A549 cells through targeting TRAF6. J. Exp. Clin. Cancer Res. 2017, 36, 35. [Google Scholar] [CrossRef]
- Mateo, D.; Morales, P.; Ávalos, A.; Haza, A.I. Comparative cytotoxicity evaluation of differentsize gold nanoparticles in human dermal fibroblasts. J. Exp. Nanosci. 2015, 10, 1401–1417. [Google Scholar] [CrossRef]
- Tiago, M.; de Oliveira, E.M.; Brohem, C.A.; Pennacchi, P.C.; Paes, R.D.; Haga, R.B.; Campa, A.; de Moraes Barros, S.B.; Smalley, K.S.; Maria-Engler, S.S. Fibroblasts Protect Melanoma Cells from the Cytotoxic Effects of Doxorubicin. Tissue Eng. Part A 2014, 20, 2412–2421. [Google Scholar] [CrossRef]
- Zaremba-Czogalla, M.; Jaromin, A.; Sidoryk, K.; Zagórska, A.; Cybulski, M.; Gubernator, J. Evaluation of the In Vitro Cytotoxic Activity of Caffeic Acid Derivatives and Liposomal Formulation against Pancreatic Cancer Cell Lines. Materials 2020, 13, 5813. [Google Scholar] [CrossRef]
- Galandáková, A.; Franková, J.; Ambrožová, N.; Habartová, K.; Pivodová, V.; Zálešák, B.; Šafářová, K.; Smékalová, M.; Ulrichová, J. Effects of silver nanoparticles on human dermal fibroblasts and epidermal keratinocytes. Hum. Exp. Toxicol. 2016, 35, 946–957. [Google Scholar] [CrossRef]
- Musiał, M.; Zorębski, E.; Malarz, K.; Kuczak, M.; Mrozek-Wilczkiewicz, A.; Jacquemin, J.; Dzida, M. Cytotoxicity of Ionic Liquids on Normal Human Dermal Fibroblasts in the Context of Their Present and Future Applications. ACS Sustain. Chem. Eng. 2021, 9, 7649–7657. [Google Scholar] [CrossRef]
- Cheng, Y.; Xia, Q.; Lu, Z.; Luan, X.; Fan, L.; Wang, Z.; Luo, D. Maslinic acid attenuates UVB-induced oxidative damage in HFF-1 cells. J. Cosmet. Dermatol. 2023, 22, 2352–2360. [Google Scholar] [CrossRef]
- De Mello, G.H.; Medianeira da Silva D’Ávila, C.; Viana, A.R.; Krause, L.M.F.; Cadoná, F.C. Cocoa presents cytotoxicity against melanoma cancer cell lines (A-375 e B16-F10) and improves chemotherapy activity by increasing oxidative stress. J. Food Biochem. 2022, 46, e14512. [Google Scholar] [CrossRef] [PubMed]
- Aghajanshakeri, S.; Ataee, R.; Karami, M.; Aghajanshakeri, S.; Shokrzadeh, M. Cytomodulatory characteristics of Granulocyte-Macrophage Colony-Stimulating Factor (GM-CSF) against cypermethrin on skin fibroblast cells (HFF-1). Toxicology 2023, 499, 153655. [Google Scholar] [CrossRef]
- Lakowicz, J.R. Principles of Fluorescence Spectroscopy, 3rd ed.; Springer: New York, NY, USA, 2006. [Google Scholar]
- Kragh-Hansen, U. Molecular aspects of ligand binding to serum albumin. Pharmacol. Rev. 1981, 33, 17–53. [Google Scholar] [PubMed]
- Du, H.; Xiang, J.; Zhang, Y.; Tang, Y.; Xu, G. Binding of V(IV) to human transferrin: Potential relevance to anticancer activity of vanadocene dichloride. J. Inorg. Biochem. 2008, 102, 146–149. [Google Scholar] [CrossRef] [PubMed]
- Michon, J.; Frelon, S.; Garnier, C.; Coppin, F. Determinations of Uranium(VI) Binding Properties with some Metalloproteins (Transferrin, Albumin, Metallothionein and Ferritin) by Fluorescence Quenching. J. Fluoresc. 2010, 20, 581–590. [Google Scholar] [CrossRef]
- Mazuryk, O.; Kurpiewska, K.; Lewiński, K.; Stochel, G.; Brindell, M. Interaction of apo-transferrin with anticancer ruthenium complexes NAMI-A and its reduced form. J. Inorg. Biochem. 2012, 116, 11–18. [Google Scholar] [CrossRef]
- Ross, P.D.; Subramanian, S. Thermodynamics of Protein Association Reactions: Forces Contributing to Stability. Biochemistry 1981, 20, 3096–3102. [Google Scholar] [CrossRef]
- Kelly, S.M.; Jess, T.J.; Price, N.C. How to Study Proteins by Circular Dichroism. Biochim. Biophys. Acta 2005, 1751, 119–139. [Google Scholar] [CrossRef] [PubMed]
- Prieto, G.; Sabın, J.; Ruso, J.M.; Gonzalez-Perez, A.; Sarmiento, F. A study of the interaction between proteins and fully-fluorinated and fully-hydrogenated surfactants by zeta potential measurements. Colloids Surf. A 2004, 249, 51–55. [Google Scholar] [CrossRef]
- Rahnama, E.; Mahmoodian-Moghaddam, M.; Khorsand-Ahmadi, S.; Saberi, M.R.; Chamani, J. Binding site identification of metformin to human serum albumin and glycated human serum albumin by spectroscopic and molecular modeling techniques: A comparison study. J. Biomol. Struct. Dyn. 2015, 33, 513–533. [Google Scholar] [CrossRef]
- Müller, R.H. Zetapotential und Partikelladung in der Laborprax Is—Einführung in Die Theorie, Praktische Meßdurchführung, Dateninterpretation; Wissen Schaftliche Verlagsgesellschaft: Stuttgart, Germany, 1996. [Google Scholar]
- Daigle, D.J.; Pepperman, A.B.; Vail, S.L.J., Jr. Synthesis of a monophosphorus analog of hexamethylenetetramine. Heterocycl. Chem. 1974, 11, 407–408. [Google Scholar] [CrossRef]
- Daigle, D.J.; Decuir, T.J.; Robertson, J.B.; Darensbourg, D.J. 1,3,5-Triaza-7-Phosphatricyclo-[3.3.1.13,7]Decane and Derivatives. Inorg. Synth. 1998, 32, 40–42. [Google Scholar]
- Kubas, G.J.; Monzyk, B.; Crumbliss, A.L. Tetrakis(acetonitrile)copper(1+) Hexafluorophosphate(1−): [Cu(NCMe)4]+. Inorg. Synth. 1990, 28, 68–90. [Google Scholar]
- Groessl, M.; Reisner, E.; Hartinger, C.G.; Eichinger, R.; Semenova, O.; Timerbaev, A.R.; Jakupec, M.A.; Arion, V.B.; Keppler, B.K. Structure-activity relationships for NAMI-A-type complexes (HL)[trans-RuCl4L(S-dmso)ruthenate(III)] (L = imidazole, indazole, 1,2,4-triazole, 4-amino-1,2,4-triazole, and 1-methyl-1,2,4-triazole): Aquation, redox properties, protein binding, and antiproliferative activity. J. Med. Chem. 2007, 50, 2185–2193. [Google Scholar] [PubMed]
- Agilent: CrysAlis PRO; Agilent Technologies Ltd.: Yarnton, UK, 2014.
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. 2015, 71, 3–8. [Google Scholar]
- XP—Interactive Molecular Graphics, v. 5.1 in SHELXTL; Bruker Analytical X-ray System: Madison, WI, USA, 1998.
- Diamond-Crystal and Molecular Structure Visualization; Crystal Impact-Dr. H. Putz & Dr. K.; Brandenburg GbR: Bonn, Germany; Available online: https://www.crystalimpact.de/diamond (accessed on 14 February 2024).
- Spek, L. Single-crystal structure validation. J. Appl. Cryst. 2003, 36, 7–13. [Google Scholar] [CrossRef]
- ISO 10993-5:2009; Biological Evaluation of Medical Device—Part 5: Tests for In Vitro Cytotoxicity. ISO: Geneva, Switzerland, 2009.




| Bond Distances [Å] | Valence Angles [°] | ||
|---|---|---|---|
| Cu1-P11 | 2.1854(5) | P11-Cu1-P21 | 119.48(2) |
| Cu1-P21 | 2.2126(5) | P11-Cu1-N31 | 118.43(5) |
| Cu1-N31 | 2.0785(16) | P11-Cu1-N41 | 117.71(5) |
| Cu1-N41 | 2.0674(15) | P21-Cu1-N31 | 104.86(5) |
| P21-Cu1-N41 | 109.43(5) | ||
| N31-Cu1-N41 | 79.87(6) | ||
| Formula | C27H35CuF6N9P3 |
|---|---|
| Moiety formula | [Cu(C15H11N3){P(CH2)6N3}2][PF6] |
| FW [g·mol−1] | 756.09 |
| T [K] | 100(2) |
| Wavelength [Å] | 1.54184 |
| Cryst. Syst. | Monoclinic |
| Space group | P21/n |
| a [Å] | 10.4364(3) |
| b [Å] | 13.7629(4) |
| c [Å] | 21.5413(5) |
| β [°] | 98.343(3) |
| V [Å3] | 3061.34(15) |
| Z | 4 |
| Calc d [g·cm−3] | 1.640 |
| µ [mm−1] | 3.150 |
| Tmin/Tmax | 0.706/0.860 |
| θ range [°] | 3.823–75.785 |
| Reflns collected | 30003 |
| Indep refunds (Rint) | 6287 (0.0195) |
| GOF on F2 | 1.057 |
| Final R1/wR2 indices | 0.0331/0.0877 |
| CCDC No. | 2291627 |
| THCDA/100 [43] | FCGP/100 [44] | τ4 [45] | |
|---|---|---|---|
| Tetrahedron | 1.00 | 0.00 | 1.00 |
| Trigonal pyramid | 0.00 | 1.00 | 0.85 |
| [Cu(terpy)(PTA)2]+ | 0.32 | 0.45 | 0.87 |
| Cell Line | 1 | 2 | [Cu(MeCN)4][PF6] | terpy | PTA | [Ag(terpy)(PTA)][NO3] [30] | Cisplatin [30] |
|---|---|---|---|---|---|---|---|
| NHDF | 152.36 ± 2.82 | 171.72 ± 4.77 | 24.75 ± 3.64 | 1.71 ± 0.95 | 127.39 ± 7.84 | 50.07 ± 4.74 | 16.65 ± 2.17 |
| A549 | 0.64 ± 0.35 | 49.87 ± 1.14 | 0.89 ± 1.14 | 0.75 ± 0.23 | 141.84 ± 3.62 | 1.67 ± 2.53 | 33.30 ± 4.23 |
| HeLa | 0.31 ± 0.61 | 5.09 ± 0.79 | 5.02 ± 1.72 | 0.33 ± 0.41 | 110.73 ± 4.77 | 1.67 ± 3.21 | 16.65 ± 3.19 |
| MCF-7 | 1.29 ± 2.47 | 0.23 ± 1.39 | 22.02 ± 2.49 | 0.38 ± 0.17 | 148.19 ± 7.43 | 26.75 ± 2.81 | 33.30 ± 4.28 |
| Compound | T [K] | Ksv [M−1] | Kq [M−1s−1] | Ka [M−1] | n | ∆H [kJ/mol] | ∆S [J/mol] | ∆G [kJ/mol] |
|---|---|---|---|---|---|---|---|---|
| 1 | 300 | 6.26 × 103 | 2.50 × 1012 | 5.39 × 105 | 1.25 | −69.30 | −121.28 | −32.92 |
| 310 | 2.26 × 104 | 9.02 × 1012 | 2.20 × 105 | 1.16 | −31.70 | |||
| 2 | 300 | 4.86 × 103 | 1.94 × 1012 | 1.08 × 106 | 1.34 | −141.20 | −355.17 | −34.65 |
| 310 | 1.40 × 104 | 5.60 × 1012 | 1.74 × 105 | 1.16 | −31.10 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smoleński, P.; Śliwińska-Hill, U.; Kwiecień, A.; Wolińska, J.; Poradowski, D. Design, Synthesis, and Anti-Cancer Evaluation of Novel Water-Soluble Copper(I) Complexes Bearing Terpyridine and PTA Ligands. Molecules 2024, 29, 945. https://doi.org/10.3390/molecules29050945
Smoleński P, Śliwińska-Hill U, Kwiecień A, Wolińska J, Poradowski D. Design, Synthesis, and Anti-Cancer Evaluation of Novel Water-Soluble Copper(I) Complexes Bearing Terpyridine and PTA Ligands. Molecules. 2024; 29(5):945. https://doi.org/10.3390/molecules29050945
Chicago/Turabian StyleSmoleński, Piotr, Urszula Śliwińska-Hill, Anna Kwiecień, Joanna Wolińska, and Dominik Poradowski. 2024. "Design, Synthesis, and Anti-Cancer Evaluation of Novel Water-Soluble Copper(I) Complexes Bearing Terpyridine and PTA Ligands" Molecules 29, no. 5: 945. https://doi.org/10.3390/molecules29050945
APA StyleSmoleński, P., Śliwińska-Hill, U., Kwiecień, A., Wolińska, J., & Poradowski, D. (2024). Design, Synthesis, and Anti-Cancer Evaluation of Novel Water-Soluble Copper(I) Complexes Bearing Terpyridine and PTA Ligands. Molecules, 29(5), 945. https://doi.org/10.3390/molecules29050945






