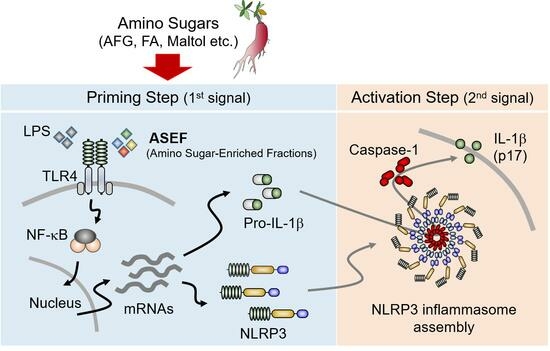
1. Introduction
Inflammasomes are multi-protein assemblies expressed in the cells of the immune system and epithelial cells [
1,
2]. Inflammasomes trigger the activation of inflammatory caspases, such as caspase-1, which promote the secretion of pro-inflammatory cytokines (e.g., interleukin [IL]-1β and IL-18) in response to cytosolic danger signals [
1]. Inflammasome activation is a two-step process of priming and activation. In the priming step, the Toll-like receptor (TLR), tumor necrosis factor α-receptor (TNFR), and IL-1 receptor (IL-1R) signaling pathways upregulate the transcription of the pro-form of the cytokines and sensor proteins (e.g., nucleotide-binding oligomerization domain [NOD], leucine-rich repeat [LRR], and pyrin domain-containing 3 [NLRP3]) through nuclear factor (NF)-κB and others [
1,
3]. On the other hand, the activation step induces the assembly of inflammasome components (e.g., NLRP3, apoptosis-associated speck-like protein containing a caspase recruitment domain [ASC], and caspase-1) in response to triggers such as nigericin and adenosine triphosphate (ATP). The activation of the NLRP3 inflammasome leads to the maturation and secretion of IL-1β and IL-18, and the induction of inflammatory cell death (i.e., pyroptosis) [
2]. NLRP3 inflammasomes have been known to play a role in the pathogenesis of various infectious (e.g., COVID-2019) and non-infectious diseases (e.g., diabetes, arteriosclerosis, and Alzheimer’s diseases) and there has been significant progress in research on the regulators of inflammasome activation [
1,
3,
4].
Ginseng (
Panax ginseng Meyer) is a well-known herbal medicine with several health benefits, including immunity-boosting properties [
5]. Korean Red Ginseng is made by steaming and drying the roots of fresh ginseng, which enhances its efficacy, safety, and preservation [
5]. Korean Red Ginseng extract (RGE) contains various bioactive components, such as saponins, polysaccharides, peptides, fatty acids, mineral oils, and amino sugars, with pharmacological properties. Among these, research on the saponins of ginseng, ginsenosides, has been the most extensive [
3,
5]. Ginsenosides are well known for their anti-inflammatory effects that are exerted through the inhibition of pro-inflammatory cytokine expression. On the other hand, the other substances are known to enhance the phagocytic activity of macrophages against pathogens [
5,
6]. RGE has been reported to selectively inhibit NLRP3, absent in melanoma 2 (AIM2), and non-canonical (i.e., caspase-4/5 in humans and caspase-11 in mice) inflammasomes, and it is known that ginsenosides have this inhibitory effect [
3,
4].
In an earlier study, RGE was split into saponin and non-saponin fractions and their roles in inflammasome activation were tested [
7]. The saponin fractions, containing high levels of ginsenosides, inhibited both the priming and activation steps. However, the non-saponin fractions of RGE, with significantly depleted levels of ginsenosides, induced the priming step for activation of the NLRP3 inflammasome [
7]. Interestingly, the non-saponin fractions inhibited AIM2 inflammasomes, and maltol, a non-saponin substance, attenuated NLRP3 and caspase-11 inflammasomes [
8,
9]. However, the specific substance in the non-saponin fraction that promotes the priming step of inflammasome activation has not yet been determined. Therefore, this study investigated the effects of the amino sugar-enriched fraction (ASEF) of RGE on inflammasome activation to understand which component, saponin or non-saponin components of RGE, plays a more crucial role in regulating the priming step in NLRP3 inflammasomes. The ASEF contains increased amino sugars such as arginine–fructose–glucose (AFG), fructose–arginine (FA), and maltol, without significant changes in the total amount of ginsenosides. The ASEF treatment was carried out during the two distinct steps of NLRP3 inflammasome activation in mouse macrophages and the role of the ASEF on inflammasome activation and its mechanism were studied. The results of this study provide a scientific basis for the immune-boosting effects of RGE.
3. Discussion
In general, it is well established that the ginsenosides of RGE inhibit the expression and inflammasome-mediated maturation of inflammatory cytokines. However, research on cytokine secretion related to the other components of RGE, such as amino sugars, has been relatively limited. In this study, it was demonstrated that the ASEF not only increased cytokine expression in macrophages but also upregulated the expression of genes associated with inflammasome components, such as NLRP3 and pro-IL-1β. Successful inflammasome activation requires two distinct steps: priming and activation. During the priming step, the gene expression of the inflammasome components is upregulated, followed by the activation of caspase-1 through protein polymerization during the activation step. The effect of the ASEF in increasing gene expression is similar to that of the TLR ligands, which induce the priming step. Through mechanistic studies, it was found that the ASEF mainly induces the priming step of the inflammasome through the TLR4 signaling pathway, with a low level of involvement of the TLR1 signaling pathway. Macrophages primed with the ASEF induced complete NLRP3 inflammasome activation, similar to cells primed with LPS. Furthermore, no inhibitory effect on priming and activation of the NLRP3 inflammasome was observed due to the ginsenosides contained in the ASEF. In this study, maltol was believed to be a component that promoted the priming step, but other ingredients of ASEF might play a role in inducing the priming step due to the distinct characteristics of maltol compared to the ASEF. Taken together, the increase in the cytokine expression and inflammasome priming induced by the ASEF may contribute to the immunomodulatory effects of ginseng.
Among the several pharmacological properties that benefit health, the effects of ginseng, including Korean Red Ginseng, on inflammation have been extensively studied. Notably, ginsenosides have been in focus as anti-inflammatory molecules. Ginsenosides modulate several inflammatory signals and pathways, including glucocorticoid receptors, NF-κB, mitogen-activated protein kinase, and antioxidants, resulting in a reduction in the production of pro-inflammatory cytokines [
12,
13]. Also, the role of ginseng on inflammasomes has been extensively studied. Ginseng has been shown to inhibit the activation of NLRP3, AIM2, and non-canonical (caspase-4/5 in human and caspase-11 in mice) in inflammasomes, but not the NLR family CARD domain-containing protein 4 (NLRC4) inflammasome [
14,
15]. Several ginsenosides (e.g., Rg1, Rg3, Rh3, Rd, and compound K) have been shown to attenuate the activation of NLRP3 and AIM2 inflammasomes [
3,
4]. In addition to the anti-inflammatory effects, there are several reports of the immune-boosting effects of ginseng [
5]. Ginseng has been reported to increase the generation of inflammatory mediators such as nitric oxide (NO) by upregulating inducible nitric oxide synthase in macrophages, thereby enhancing phagocytic activity [
16]. Notably, polysaccharides extracted from ginseng have been shown to increase IL-1β and TNFα secretion and promote NO production in macrophages and mice models, resulting in increased anti-pathogenic activity [
16,
17]. In a clinical study with human subjects, the intake of ginseng extract was found to increase phagocytic activity [
6]. Collectively, while ginsenosides exhibit anti-inflammatory effects, non-ginsenosides exhibit immune-boosting effects. Notably, as revealed in this study, the promotion of inflammasome priming by the ASEF can be regarded as an immunity-boosting effect, as it prepares the host to be ready to fight against pathogens. ASEF-mediated enhancement of the priming step of inflammasomes might induce faster and more robust inflammatory responses (e.g., IL-1β secretion and pyroptosis) against invading pathogens in the host, effectively controlling infectious diseases. Furthermore, the presence of ASEF in RGE could serve as a marker due to its role in leading the immune-boosting effect, while ginsenosides could be considered markers of immunomodulation.
The inflammasome substrate, pro-IL-1β, is synthesized as an inactive precursor and must be cleaved or processed into a mature form (17 kDa) that interacts with the IL-1 receptors [
18]. Two signals are needed to induce the two steps of inflammasome activation, namely priming, and activation for the production of pro-IL-1β and the release of mature IL-1β, respectively [
10,
19]. Inflammasome activation occurs in two phases due to the need for strict control over the powerful inflammatory response and cellular damage caused by NLRP3 inflammasome activation [
11]. The priming step is a key component of the control measures needed to ensure a timely and appropriate inflammatory response [
11]. Pro-IL-1β synthesis is generally induced by many stimuli, including TLR ligands and cytokines such as TNF-alpha [
18]. However, in general, LPS stimulation through TLR4 induces pro-IL-1β production [
10]. Transcriptional increases in inflammasome substrates (i.e., pro-IL-1β) occur during priming. Although pro-IL-18 is constitutively expressed, it is also increased by TLR signals [
11]. Therefore, increasing the concentration of substrates through priming is essential for inflammasome activation [
11]. The inflammasome sensor protein, NLRP3, itself, is also increased through NF-κB signaling in the priming step, and high levels of NLRP3 expression are essential for NLRP3 inflammasome activation [
11]. To summarize, the increased expression of substrates and the sensor protein NLRP3 during priming appears to be correlated with inflammasome activation. However, the post-transcriptional modifications (e.g., ubiquitination and phosphorylation) of inflammasome components during the priming step may be additional signals as NLRP3 activation can occur even when protein synthesis is inhibited during the priming step [
11,
20]. Although this study did not investigate the probable post-transcriptional modifications caused by the ASEF, the study confirmed that the ASEF clearly increases transcriptional priming and induces inflammasome activation.
5. Materials and Methods
Unless otherwise indicated, all materials were purchased from Daejung Chemicals & Metals Co., Ltd. (Siheung-si, Republic of Korea), Welgene (Gyeongsan-si, Republic of Korea), or Life Sciences (Pocheon-si, Republic of Korea).
5.1. Preparation of ASEF, AFG, and FA
The ASEF was obtained from the Korea Ginseng Corporation (KGC, Daejeon, Republic of Korea). To prepare the ASEF, the powdered RGE (KGC, Daejeon, Republic of Korea) was dissolved in absolute ethanol (EtOH), and then the EtOH was removed. The residue was re-dissolved in water, and the eluted water fraction was evaporated to dryness in vacuo. Through this process, the concentration of ginsenosides did not change significantly, but the concentration of amino sugars, namely AFG and maltol, was increased to about three-fold in the ASEF when compared to that in RGE (
Supplementary Table S1) [
22,
23].
5.2. Analysis of Contents of Contents of ASEF
The ASEF was analyzed for arginine, sugars, amino sugars, and ginsenosides by chromatographic analysis, as shown in
Figure 6 and
Table 1, in accordance with previous studies [
7,
22,
23,
24]. Briefly, the standard materials were purchased from Chromadex (Irvine, CA, USA) for ginsenosides, from Ambo Institute (Seoul, Republic of Korea) for ginsenosides and amino sugars, and from Sigma-Aldrich (Supelco Inc., Burlington, MA, USA) for arginine, sugar, and amino sugars. In total, 0.3 g of ASEF dissolved in 20 mL of methanol (70%) for ginsenosides and 0.1 to 0.5 g of ASEF dissolved in 10 mL of distilled water for arginine, sugars, and amino sugars were mixed using an ultrasonicator (SD-200H, Sungdong Ultrasonics, Seoul, Republic of Korea). For the analysis of amino acids, 1 mL of the ASEF solution in distilled water was added to 10 mL of 0.02 N HCl and mixed. After centrifugation, the supernatant was filtered through a PVDF membrane filter (0.22 µm, φ47 mm, Nylon, Whatman, Maidstone, Kent, UK). The filtrates were subjected to the following chromatographic analyses: Ginsenosides were analyzed using a UPLC/PDA system (Column: ACQUITY UPLC BEH Phenyl [2.1 × 100 mm, 1.7 µm], Detector: UV 203 nm, Acquity UPLC System, Waters, Milford, MA, USA); Maltol was analyzed using an HPLC/UVD system (Column: Discovery C18 [4.6 × 250 mm, 5 µm], Detector: UV 275 nm, Alliance 2695 HPLC system, Waters); Sugars, FA, and AFG were analyzed using a Bio-LC/PAD system (CarboPac PA-10 column [4 × 250 mm], Au working electrode and Ag/AgCl reference electrode, Dionex, Sunnyvale, CA, USA); and Arginine was analyzed using an LA8080 amino acid analyzer (Hitachi, Tokyo, Japan). Arg, arginine; AFG, arginine–fructose–glucose; FA, fructose–arginine. AFG and FA were produced using the methods described in
Supplementary Materials [
25].
5.3. Cell Culturing
To generate BMDMs, the bone marrow cells were isolated from the femur and tibia of mice (C57BL/6, Nara Biotech, Seoul, Republic of Korea) and incubated in differentiation media for 7 d. The differentiation media contained fetal bovine serum (FBS, 10%), antibiotics, and L929 cell-conditioned media (50%), which included the macrophage colony-stimulating factor. The above steps to generate BMDMs were based on previously published studies [
8,
9,
23]. All animal experiments were carried out in accordance with the Guide for the Care and Use of Laboratory Animals, National Institutes of Health, USA, and approved by the Institutional Animal Care and Use Committee of Kangwon National University (IACUC; approval no. KW-230328-2).
5.4. Cell Treatment
For mRNA and protein expression assays, BMDMs were seeded in culturing plates, and then the cells were treated with the ASEF (100 µg/mL), heat-killed Listeria monocytogenes (HKLM, 1%), or LPS (10 ng/mL; Sigma-Aldrich Co., St. Louis, MO, USA) with/without Anti-mTLR2-IgG (100 ng/mL; mabg-mtlr2, InvivoGen, San Diego, CA, USA), CU-CPT22 (5 mM; 4884, Tocris Bioscience, Minneapolis, MN, USA), TAK-242 (5 µM; CLI-095, tlrl-cli95, InvivoGen), or polymyxin B (100 µg/mL; tlrl-pmb, InvivoGen) for 3 h. The BMDMs were treated with 1 mg/mL of arginine, fructose, glucose, maltose, AFG, and FA for 3 h.
For the assay of inflammasome activation, BMDMs were primed with the ASEF (100 µg/mL) or LPS (1 µg/mL) for 3 h and treated with adenosine triphosphate (ATP, 5 mM; InvivoGen) with/without ASEF (100 µg/mL) or maltol for 1 h. The cellular supernatant (Sup) was harvested in a tube, and the cells were lysed with a lysis buffer containing a proteinase inhibitor cocktail. The lysate (Lys) was collected after centrifugation, and the remaining cell pellets were treated with suberic acid bis (2 mM, Sigma-Aldrich Co.) for 1 h. The cross-linked pellets (Pellet) were collected by centrifugation and then re-suspended in a loading dye buffer [
26].
5.5. Reverse Transcription Polymerase Chain Reaction (RT-PCR) and Quantitative Real-Time PCR (qPCR)
The total RNA was prepared with NucleoZOL (Macherey-Nagel Co., KG, Düren, Germany) and synthesized to complementary DNA (cDNA) by a synthesis kit (Enzynomics Inc., Daejeon, Republic of Korea). For RT-PCR, the transcripts were amplified using nTaq polymerase (Enzynomics) and a cycler (Thermo Fisher Scientific Inc. Grand Island, NY, USA). The band of PCR products was separated by agarose gel electrophoresis and visualized by staining with ethidium bromide. For qPCR, the transcripts were quantified using a PCR system (Eco Real-Time PCR, Illumina, San Diego, CA, USA) and an enzyme mixture containing SYBR Green (Enzynomics). The quantitation was normalized with a housekeeping gene (glyceraldehyde 3-phosphate dehydrogenase [GAPDH]). The gene-specific primers are provided in the
Supplementary Materials.
5.6. Western Blot Analysis
Samples of the cellular supernatant, lysate, or pellets were separated using a sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) using a gel running system (Bio-Rad Laboratories, Hercules, CA, USA). The gels were further transferred onto a polyvinylidene difluoride membrane (PVDF; GE Healthcare, Chicago, IL, USA). The membranes were soaked in skim milk (5%) and then probed with an anti-mouse IL-1β antibody (AF-401-NA, R&D Systems, Minneapolis, MN, USA), anti-NLRP3 antibody (AG-20B-0014-C100, AdipoGen Life Sciences, San Diego, CA, USA), anti-caspase-1 antibody (AG-20B-0042-C100, AdipoGen), or anti-actin antibody (sc-1615, Santa Cruz Biotechnology, Dallas, TX, USA) overnight at 4 °C. The membrane was further probed with secondary antibodies conjugated with horseradish peroxidase (HRP; sc-2020, sc-2005 or sc-2004, Santa Cruz Biotechnology) for 2 h and then analyzed under a chemiluminescence solution (AbFrontier, Seoul, Republic of Korea) and a chemiluminescence imaging system (EZ-Capture II, ATTO Technology, Tokyo, Japan).
5.7. Statistical Analysis
Statistical software (GraphPad Prism 6, San Diego, CA, CA) was used to perform analyses using the Mann–Whitney test or one-way ANOVA. The p-value is shown in the figures.