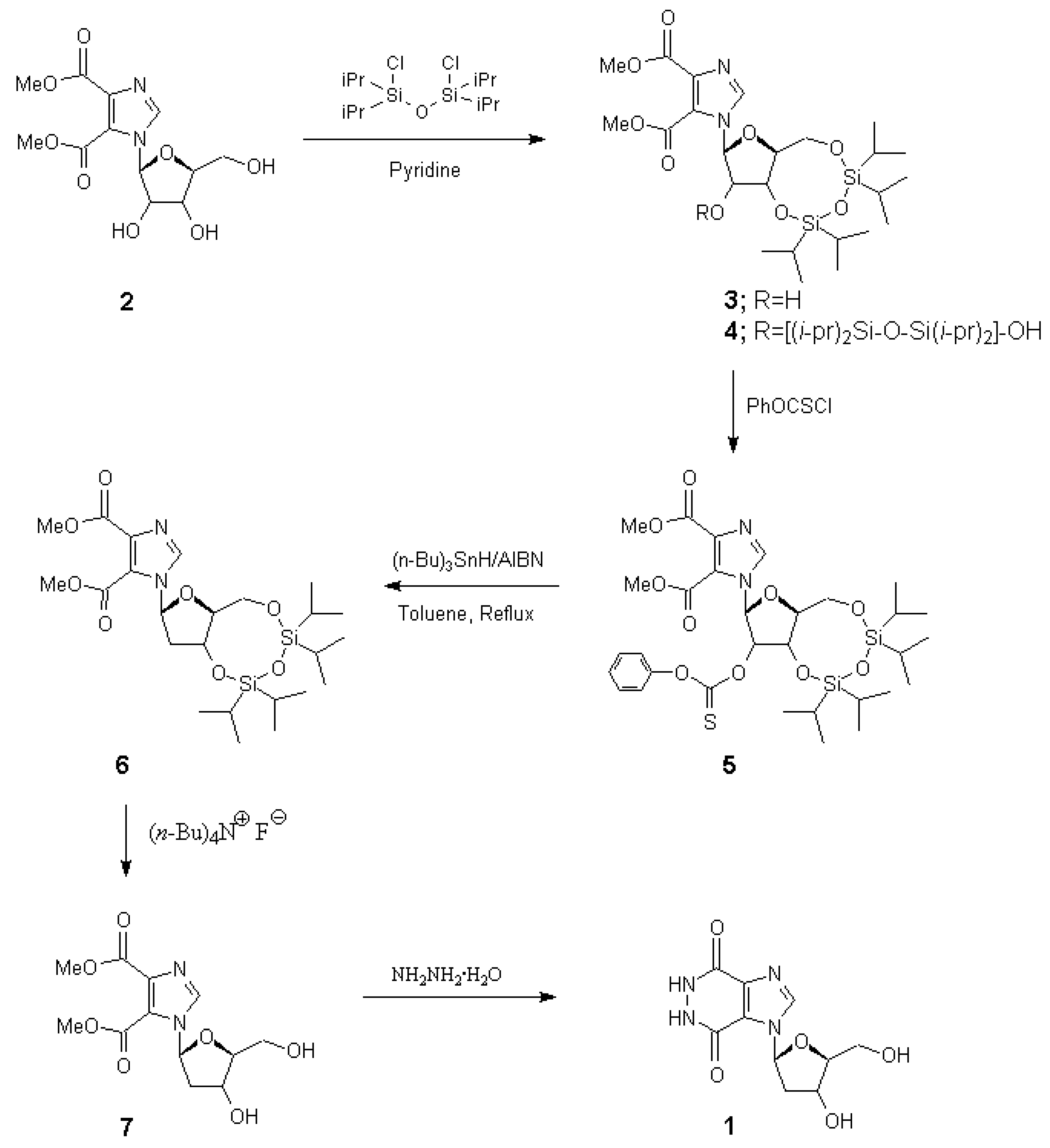
Abstract
Synthesis of the title compound,1-(2'-deoxy-β-d-ribofuranosyl)-1H-imidazo[4,5- d]pyridazine-4,7(5H,6H)-dione (1), is reported. It was synthesized in five steps, commencing with methyl 1-(β-d-ribofuranosyl)imidazo-4,5-dicarboxylate (2). The 3',5'- hydroxyl groups of 2 were protected with a bis-silylating agent to form 3, which was then converted into the corresponding 2'-thionocarbonate derivative 5. The reduction of the latter with tri-n-butyltin hydride (to form 6), followed by silyl deprotection with tetra-n- butylammonium fluoride, afforded 7. Treatment of the latter with hydrazine hydrate yielded the target nucleoside 1.
Introduction
The target nucleoside, 1-(2'-deoxy-β-d-ribofuranosyl)-1H-imidazo[4,5-d]pyridazine-4,7(5H,6H)- dione (1), can be considered an analogue of purine nucleoside in which a pyridazine moiety replaces a pyrimidine in fusion to an imidazole ring. Our molecular modeling studies [1] suggest that nucleoside 1 has some unique structural features that would make it an attractive building block for oligonucleotides with potential antisense and triple-helical applications. These features include, but are not limited to, its potential capability to base-pair with cytidine forming three H-bonds, like guanosine,
 but with a significantly shortened sugar-sugar (C-1' to C-1') distance, which in turn might lead to a decreased interstrand span and consequently somewhat compressed double-helix. The study of such effect on the double-helical or triple-helical conformations, interactions, and stability would be interesting and rewarding.
but with a significantly shortened sugar-sugar (C-1' to C-1') distance, which in turn might lead to a decreased interstrand span and consequently somewhat compressed double-helix. The study of such effect on the double-helical or triple-helical conformations, interactions, and stability would be interesting and rewarding.

Results and Discussion
The synthesis of the ribose analogue of 1, containing a 2'-OH group in place of 2'-H, has long been reported in the literature, both by imidazole ring-closure [2f] as well as by glycosylation of the parent heterocyclic base, imidazo[4,5-d]pyridazine-4,7(5H,6H)-dione [2b]. Surprisingly, however, the target 2'-deoxy analogue 1, which is a necessary building block for the preparation of DNA analogues, is as yet unknown. We report herein the synthesis of 1 in five steps starting from methyl 1-(β-d- ribofuranosyl)imidazo-4,5-dicarboxylate (2) [2].
Nucleoside 2 (Scheme I) was selectively protected at the 3',5'-position using the Markiewicz reagent [3] 1,3-dichloro-1,1,3,3-tetraisopropyldisiloxane to form the corresponding 3',5'-O-(1,1,3,3- tetraisopropyldisiloxan-1,3-diyl) (TIPDS) derivative 3 as the major product, along with a small amount of the completely silyl-protected derivative 4. Functionalization of 3 at O-2' with phenyl chlorothionocarbonate (phenoxythiocarbonyl chloride), employing 4-(dimethylamino)pyridine (DMAP) as a catalyst, afforded the 3',5'-O-TIPDS-protected 2'-O-(phenoxythiocarbonyl) ester (5). Free radical-mediated Barton deoxygenation [4] with tributylstannane (tributyltin hydride), employing α,α- azobis(isobutyronitrile) (AIBN) as a radical initiator in toluene at reflux, gave satisfactory conversion into the 3',5'-TIPDS- protected-2'-deoxynucleoside (6). The use of excess tributylstannane makes the conversion almost quantitative, and extended reaction times do not give any by-products. The silyl deprotection of 6 was achieved by treatment with tetra-n-butylammonium fluoride, which gave the required imidazole precursor, methyl 1-(2'-deoxy-ß-d-erythropentofuranosyl)-4,5- imidazoledicarboxylate (7).
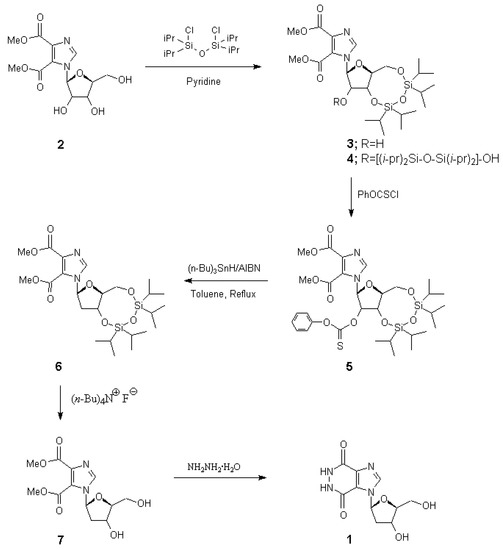
SCHEME I.
It was observed that 7 was unstable in solution, and was completely decomposed in 2-3 days to give the corresponding aglycon methyl imidazole-4,5-dicarboxylate. The ring-closure of 7 with hydrazine hydrate provided the target nucleoside 1-(2'-deoxy-β-d-ribofuranosyl)-1H-imidazo[4,5-d] pyridazine- 4,7(5H,6H)-dione (1). The structure of the latter was consistent with its spectroscopic and microanalytical data.
Experimental
1H-NMR spectra were recorded on a General Electric QE-300 (300 MHz) instrument. The spectral data are reported in the following format: chemical shift (all relative to Me4Si as an internal reference standard unless otherwise indicated), multiplicity (s = singlet, d = doublet, dd = doublet of doublets, t = triplet, q = quartet, m = multiplet, b = broad ), integration, coupling constants, exchangeability after D2O addition, and assignment of resonances. Elemental Microanalyses were performed by Atlantic Microlab, Inc., Norcross, Georgia. The mass spectra were recorded at the Mass Spectral Facility, Department of Biochemistry, Michigan State University. Thin layer chromatography was performed on Merck Kieselgel 60 GF254 plates (0.2 mm thickness). Melting points were determined on a Thomas- Hoover capillary melting point apparatus, and are uncorrected.
Methyl 1-[(3',5'-O-(1,1,3,3-tetraisopropyldisiloxan-1,3-diyl))-ß-d-ribofuranosyl]-4,5-imidazole- dicarboxylate (3) and Methyl 1-[((2'-O-(3-hydroxy-1,1,3,3-tetraisopropyldisiloxyl)-3',5'-O- (1,1,3,3-tetraisopropyldisiloxan-1,3-diyl))-ß-d-ribofuranosyl]-4,5-imidazoledicarboxylate (4)
To a solution of dry methyl 1-β-d-ribofuranosyl-4,5-imidazoledicarboxylate [2] (2) (500 mg, 1.58 mmol) in dry pyridine (10 mL) was added 1,3-dichloro-1,1,3,3-tetraisopropyldisiloxane [3] (550 mg, 1.75 mmol), and the mixture was stirred for 4 h at ambient temperature with protection from moisture. Volatile materials were evaporated in vacuo, and the residue was dissolved in chloroform. The chloroform solution was washed twice with cold water and dried over anhydrous sodium sulfate. The residue after evaporation was purified by silica gel flash chromatography, eluting with chloroform to give 3 and 4 as a colorless liquids, whose yield, spectral and analytical data are given below.
Compound 3: Yield 800 mg (91%), Rf 0.38 (chloroform/methanol, 30:1); 1H-NMR (CDCl3) δ 8.10 (s, 1H, imidazole), 6.11 (s, 1H, 1'-H), 4.44 (dd, 1H, J=4.2 and 9.0 Hz, 3'-H), 4.26 (d, 1H, Jgem=13.5 Hz, 5'-H), 4.18 (d, 1H, J=4.2 Hz, 2'-H), 4.16 (dd, 1H, J=9.0 and 2.4 Hz, 4'-H), 4.03 (dd, 1H, Jgem=13.5 Hz, J5',4'=2.4 Hz, 5'-H), 3.95 (s, 3H, OCH3), 3.94 (s, 3H, OCH3), 2.89 (brs, 1H, 2'-OH, exchangeable with D2O), 1.05 (m, 28H, isopropyl groups); 13C-NMR (CDCl3, 75.48 MHz) δ 12.45 (CHSi), 12.82 (CHSi), 12.90 (CHSi), 13.35 (CHSi), 16.81 (CCH3), 16.87 (CCH3), 16.97 (CCH3), 16.97 (CCH3), 17.26 (CCH3), 17.26 (CCH3), 17.32 (CCH3), 17.42 (CCH3), 52.50 (OCH3), 52.78 (OCH3), 59.91 (C-5'), 68.43 (C-3'), 76.82 (C-2'), 81.87 (C-4'), 91.54 (C-1'), 122.83 (C-4 or 5), 137.26 (C-2), 138.57 (C-5 or 4), 160.26 (C=O), 162.91 (C=O); Anal. Calcd. for C24H42N2O9Si2 (MW 558.78): C, 51.59; H, 7.58; N, 5.01. Found: C, 51.50; H, 7.57; N, 5.04.
Compound 4: Yield 120 mg (9%), Rf 0.45 (chloroform/methanol, 30:1); 1H-NMR (CDCl3) δ 8.37 (s, 1H, imidazole), 6.06 (s, 1H, 1'-H), 4.48 (d, 1H, J=3.3 Hz, 2'-H), 4.35 (dd, 1H, J=3.3 and 9.6 Hz, 3'-H), 4.31 (d, 1H, Jgem=13.8 Hz, 5'-H), 4.27 (dd, 1H, J=9.6 and 2.4 Hz, 4'-H), 4.02 (dd, 1H, Jgem=13.8 Hz, J5',4'=2.4 Hz, 5'-H), 3.93 (s, 3H, OCH3), 3.93 (s, 3H, OCH3), 1.03 (m, 56H, isopropyl groups); 13C- NMR (CDCl3, 75.48 MHz) δ 12.51 (CHSi), 12.91 (CHSi), 13.05 (CHSi), 13.05 (CHSi), 13.24 (CHSi), 13.35 (CHSi), 13.39 (CHSi), 13.48 (CHSi), 16.59 (CCH3), 16.78 (CCH3), 16.81 (CCH3), 16.81 (CCH3), 17.06 (CCH3), 17.06 (CCH3), 17.06 (CCH3), 17.23 (CCH3), 17.26 (CCH3), 17.26 (CCH3), 17.30 (CCH3), 17.37 (CCH3), 17.42 (CCH3), 17.51 (CCH3), 17.55 (CCH3), 17.65 (CCH3), 52.56 (OCH3), 53.17 (OCH3), 59.56 (C-5'), 68.47 (C-3'), 77.95 (C-2'), 81.44 (C-4'), 92.15 (C-1'), 121.16 (C-4 or 5), 138.64 (C-2), 139.48 (C-5 or 4), 161.49 (C=O), 163.11 (C=O); Anal. Calcd. for C36H70N2O11Si4 (MW 819.30): C, 52.78; H, 8.61; N, 3.42. Found: C, 52.45; H, 8.89; N, 2.93.
Methyl 1-[(2'-O-phenoxythiocarbonyl)-3',5'-O-(1,1,3,3-tetraisopropyldisiloxan-1,3-diyl)-ß-d-erythro- pentofuranosyl)]-4,5-imidazoledicarboxylate (5)
To a solution of methyl 1-(3',5'-O-(1,1,3,3,-tetraisopropyldisiloxan-1,3-diyl)-β-d-erythropento- furanosyl)-4,5-imidazoledicarboxylate (3) (560 mg, 1 mmol) and DMAP (250 mg, 2.05 mmol) in 15 mL of dried acetonitrile was added phenoxythiocarbonyl chloride (200 μL, 250 mg, 1.45 mmol). The solution was stirred for 16 h at ambient temperature, and evaporated to dryness in vacuo. The residue was applied to column chromatography, eluted with chloroform to give a colorless oily product (660 mg, 95%). Rf 0.60, chloroform/ methanol (30:1); 1H-NMR (CDCl3) δ 8.16 (s, 1H, imidazole), 7.43 (t, 2H, J=7.8 Hz, Phmeta-H), 7.31 (t, 1H, J=7.8 Hz, Phpara-H), 7.13 (d, 2H, J=7.8 Hz, Phortho-H), 6.38(s, 1H, 1'-H), 6.01 (d, 1H, J=4.5 Hz, 2'-H), 4.61 (dd, 1H, J=4.5 and 9.3 Hz, 3'-H), 4.31 (d, 1H, Jgem=13.5 Hz, 5'-H), 4.16 (dd, 1H, J=9.3 and 2.4 Hz, 4'-H), 4.05 (dd, 1H, Jgem=13.5 Hz, J5',4'=2.4 Hz, 5'-H), 3.93 (s, 3H, OCH3), 3.91 (s, 3H, OCH3), 1.06 (m, 28H, isopropyl groups); 13C-NMR (CDCl3, 75.48 MHz) δ 12.80 (CHSi), 12.91 (CHSi), 13.37 (CHSi), 13.49 (CHSi), 16.87 (CCH3), 16.94 (CCH3), 16.96 (CCH3), 17.08 (CCH3), 17.24 (CCH3), 17.27 (CCH3), 17.34 (CCH3), 17.44 (CCH3), 52.49 (OCH3), 52.74 (OCH3), 59.29 (C-5'), 67.69 (C-3'), 82.39 (C-4'), 85.10 (C-2'), 89.40 (C-1'), 121.68 (Ph-C2,6), 126.69 (Ph-C4), 129.57 (Ph-C3,5), 137.13 (C-2), 138.93 (C-5 or 4), 139.24 (C-4 or 5), 153.47 (Ph-C1), 159.79 (C=O), 162.76 (C=O), 193.70 (C=S); Anal. Calcd. for C31H46N2O10SSi2 (MW 694.94): C, 53.58; H, 6.67; N, 4.03; S, 4.61. Found: C, 53.88; H, 7.47; N, 3.53; S, 3.27.
Methyl 1-(2'-Deoxy-3',5'-O-(1,1,3,3-tetraisopropyldisiloxan-1,3-diyl)-ß-d-erythropentofuranosyl)-4,5- imidazoledicarboxylate (6)
A solution of methyl1-(2'-O-phenoxythiocarbonyl-3',5'-O-(1,1,3,3-tetraisopropyldisiloxan-1,3-diyl)- β-d-erythropentofuranosyl)-4,5-imidazoledicarboxylate (5) (695 mg, 1 mmol) and AIBN (32 mg, 0.2 mmol) in 30 mL of dried toluene was purged with oxygen-free nitrogen for 30 min. Tributylstannane (400 μL, 433 mg, 1.49 mmol) was added and the solution was refluxed for 3 h. Tlc showed a single spot with very slightly lower Rf value than that of starting material. The solvent was evaporated and the pure product was obtained as a colorless liquid by column chromatography (hexane/ethyl acetate, 3:1) in almost quantitative yield. Rf 0.12, hexane/ethyl acetate (3:1); 1H-NMR (CDCl3) δ 8.09 (s, 1H, imidazole), 6.33 (dd, 1H, J=7.4 and 1.2 Hz, 1'-H), 4.54 (dt, 1H, J=10.2 and 7.4 Hz, 3'-H), 4.11 (dd, 1H, Jgem=13.2 Hz, J5',4'=2.4 Hz, 5'-H), 4.06 (dd, 1H, Jgem=13.2 Hz, J5',4'=3.0 Hz, 5'-H), 3.93 (s, 3H, OCH3), 3.91 (s, 3H, OCH3), 3.82 (dt, 1H, J=8.2 and 2.6 Hz, 4'-H), 2.64 (ddd, 1H, J=13.2, 10.2 and 7.4 Hz, 2'- H), 2.35 (ddd, 1H, J=13.2, 7.4 and 1.2 Hz, 2'-H), 1.06 (m, 28H, isopropyl groups); 13C-NMR (CDCl3, 75.48 MHz) δ 12.54 (CHSi), 12.94 (CHSi), 13.04 (CHSi), 13.43 (CHSi), 16.86 (CCH3), 16.97 (CCH3), 16.97 (CCH3), 17.09 (CCH3), 17.31 (CCH3), 17.31 (CCH3), 17.39 (CCH3), 17.49 (CCH3), 41.70 (C-2'), 52.34 (OCH3), 52.50 (OCH3), 60.48 (C-5'), 67.52 (C-3'), 85.25 (C-4'), 86.02 (C-1'), 136.41 (C-4 or 5), 137.12 (C-2), 138.41 (C-5 or 4), 160.37 (C=O), 163.04 (C=O); Anal. Calcd. for C24H42N2O8Si2 (MW 542.78): C, 53.11; H, 7.80; N, 5.16. Found: C, 53.39; H, 7.83; N, 4.91.
Methyl 1-(2'-Deoxy-ß-d-erythropentofuranosyl)-4,5-imidazoledicarboxylate (7)
A solution of 1M tetrabutylammonium fluoride in THF (2 mL, 2 mmol) was added to an ice-cooled solution of methyl 1-(2'-deoxy-3',5'-O-(1,1,3,3,-tetraisopropyldisiloxan-1,3-diyl)-ß-d-erythropento- furanosyl)-4,5-imidazoledicarboxylate (6) (543 mg, 1 mmol) in 10 mL of dried THF. The reaction was stopped after 45 min stirring at 0 °C. The solvent was evaporated in vacuo and the pure product was obtained as a foam by column chromatography (chloroform/methanol, 10:1) in 75 % yield. Rf 0.20, chloroform/methanol (10:1); 1H-NMR (CDCl3) δ 8.51 (s, 1H, imidazole), 6.38 (dd, 1H, J=6.2 and 3.4 Hz, 1'-H), 4.82 (brs, 1H, OH, exchangeable with D2O), 4.54 (dd 1H, J=12.8 and 6.6 Hz, 3'-H), 4.15 (brs, 1H, OH, exchangeable with D2O), 3.98 (m, 3H, 4',5'-H), 3.91 (s, 3H, OCH3), 3.89 (s, 3H, OCH3), 2.56 (ddd, 1H, J=13.6, 12.8 and 6.2 Hz, 2'-H), 2.35 (ddd, 1H, J=13.6, 6.6 and 3.4 Hz, 2'-H); 13C-NMR (CDCl3, 75.48 MHz) δ 42.49 (C-2'), 52.42 (OCH3), 52.73 (OCH3), 60.62 (C-5'), 68.91 (C-3'), 87.03 (C-4'), 87.21 (C-1'), 136.50 (C-4 or 5), 136.86 (C-5 or 4), 137.68 (C-2), 160.40 (C=O), 163.05 (C=O); Anal. Calcd. for C12H16N2O7 (MW 300.27): C, 48.00; H, 5.37; N, 9.33. Found: C, 48.21; H, 5.68; N, 9.45.
1-(2'-Deoxy-ß-d-ribofuranosyl)-1H-imidazo[4,5-d]pyridazine-4,7(5H,6H)-dione (1)
A solution of methyl 1-(2'-deoxy-β-d-ribofuranosyl)imidazole-4,5-dicarboxylate (7) (0.15 g, 0.5 mmol) and 99% hydrazine (10 mL) was refluxed for 6 hours. The excess hydrazine was removed by distillation in vacuo and the residue coevaporated several times with water. The crystalline residue was recrystallized from methanol to give white crystals (0.1g, 75%). mp. >250 °C; Rf, 0.34 (chloroform/methanol/30% ammonium hydroxide, 2:2:1); 1H NMR (DMSO-d6) δ 8.62 (s, 1H, imidazole), 6.76 (t, 1H, J=6.2 Hz, 1'-H), 5.31 (brs, 1H, OH, exchangeable with D2O), 5.06 (brs, 1H, OH, exchangeable with D2O), 4.33 (m, 1H, 4'-H), 3.87 (m, 1H, 3’-H), 3.57 (m, 2H, 5'-H), 2.34 (dt, 1H, J=13.6 and 6.3 Hz, 2'-H), 2.08 (d, 1H, J=13.6 Hz, 2'-H). ms: (FAB) m/z 310 (MH+).; Anal. Calcd. For C10H12N4O5.H2O (286.24): C, 41.96; H, 4.93; N, 19.57. Found: C, 41.98; H, 4.92; N, 19.56.
Acknowledgments
This research was supported by a grant from the National Institutes of Health (#RO1 CA 71079). The Michigan State University Mass Spectrometry Facility was supported in part from a grant (# P41RR00480-0053) from the National Institutes of Health.
References and Notes
- Molecular modeling studies were performed on a Silicon Graphics R4000 workstation using InsightII/Discover software from Molecular Simulations, Inc. (MSI), San Diego, California
- (a) Wyss, P. C.; Fischer, U. Helv. Chim. Acta 1978, 61, 3149. (b) Cook, P. D.; Dea, P.; Robins, R.K. J. Heterocyclic Chem. 1978, 15, 1. (c) Cook, P. D.; Robins, R. K. Nucleic Acid Chemistry, Part 1; Townsend, L. B., Tipson, R. S., Eds.; John Wiley, Inc.: New York, 1978; p. 211. [Google Scholar] (d) Fischer, U.; Wyss, P. C. Ger. Offen. 2735458 1978. Chem. Abstr. 89:24726e, 1978. (e) Fischer, U.; Wyss, P. C. Belg. 857512 1978. Chem. Abstr. 89:44128q, 1978. (f) Tapiero, C.; Imbach, J. L.; Panzicka, R. P.; Townsend, L. B. J. Carbohyd. Nucleosides Nucleotides 1976, 3, 191.
- Markiewicz; Wojciech, T. J. Chem. Res. (S) 1979, 1, 24.
- (a) Barton, D. H. R.; Jang, D. O; Jaszberenyi, J. C. Tetrahedron 1993, 49, 2793. (b) Robins, M. J.; Wilson, J. S. Regiospecific and Stereoselective 2'-Deoxygenation of a Ribonucleoside. In Nucleic Acid Chemistry Part 2; Townsend, L. B., Tipson, R. S., Eds.; Wiley: New York, 1991; pp. 194–200. [Google Scholar]
- Sample Availability: Available from the authors
© 2000 by MDPI (http://www.mdpi.org).