Sulfur Bridged Multidentate Ligands Based on (Bi)pyridyl-(Bi)-1,3,4-Thiadiazolyl Conjugates
Abstract
:Introduction
Results and Discussion

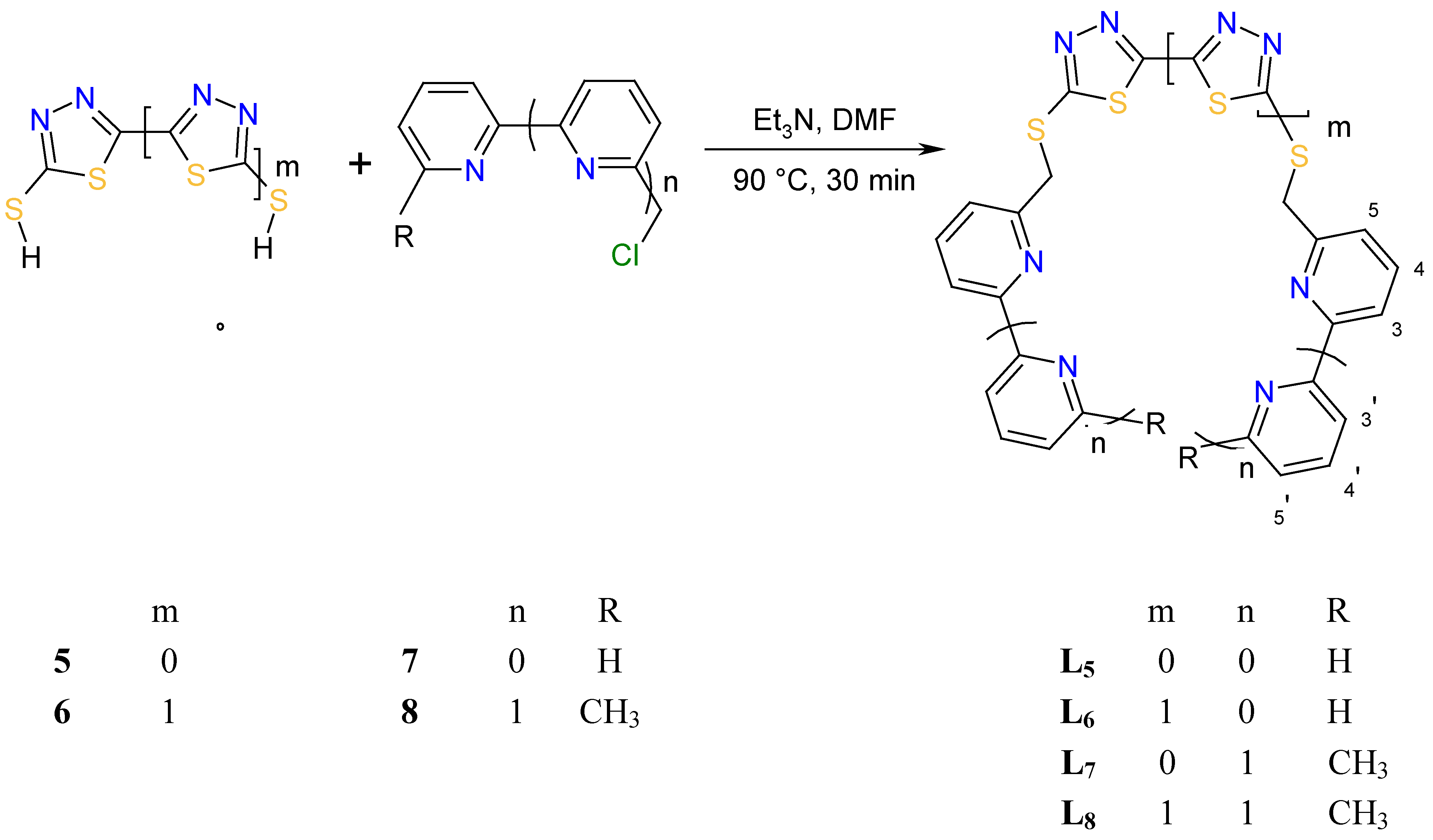
| Proton | L1 | L2 | L3 | L4 | L5 | L6 | L7 | L8 |
|---|---|---|---|---|---|---|---|---|
| 3 | 7.34 d J = 8.0 | 7.41 d J = 8.0 | 8.31 d J = 7.5 | 8.37 d J = 7.5 | 7.41 d J = 8.0 | 7.48 d J = 7.5 | 8.16 d J = 8.0 | 8.20 d J = 8.0 |
| 4 | 7.62 t J = 7.5 | 7.67 t J = 7.8 | 7.79 dt J = 7.5, 1.5 | 7.81 t J = 7.5 | 7.65 dt J = 7.5, 2.0 | 7.67 dt J = 7.5, 1.0 | 7.76 t J = 8.0 | 7.79 t J = 8.0 |
| 5 | 7.34 d J = 8.0 | 7.41 d J = 8.0 | 7.44 d J = 8.0 | 7.49 d J = 7.5 | 7.20 t J = 6.0 | 7.22 t J = 7.5 | 7.39 d J = 7.5 | 7.45 d J = 8.0 |
| 6 | 8.54 dd J = 3.5, 2.0 | 8.57 dd J = 4.0, 1.0 | ||||||
| 3’ | 8.31 d J = 7.5 | 8.37 d J = 7.5 | 8.37 d J = 7.5 | |||||
| 4’ | 7.79 dt J = 7.5, 1.5 | 7.81 t J = 7.5 | 7.68 t J = 7.5 | 7.69 t J = 8.0 | ||||
| 5’ | 7.44 d J = 8.0 | 7.49 d J = 7.5 | 7.16 d J = 7.5 | 7.17 d J = 7.5 | ||||
| CH2 | 4.57 s | 4.72 s | 4.69 s | 4.83 s | 4.58 | 4.72 s | 4.68 s | 4.83 s |
| CH3 | 2.73 s | 2.84 s | 2.73 s | 2.84 s | 2.61 s | 2.61 s |
| Lig. | Yield (%) (Cryst. Solv. ) | Melting Point (°C) | Molecular Formula | %C Found(Calcd) | %H Found(Calcd) | %N Found(Calcd) | 13C NMR |
|---|---|---|---|---|---|---|---|
| L1 | 84 (EtOH) | 94−95 | C13H13N5S6 | 36.13 (36.17) | 3.33 (3.03) | 16.04 (16.22) | 167.0, 164.0, 155.6, 137.7, 122.1, 39.8, 16.7 |
| L2 | 63 (DMF) | 207−212 | C17H13N9S6 | 33.82 (34.04) | 2.22 (2.18) | 21.06 (21.02) | 170.1, 168.2, 158.8, 158.1, 155.2, 137.8, 123.3, 39.7, 16.7 |
| L3 | 90 (EtOH-CHCl3) | 157−158 | C18H16N6S6 | 42.83 (42.49) | 3.06 (3.17) | 16.74 (16.52) | 166.9, 164.4, 155.2, 154.9, 137.7, 123.1, 120.0, 40.3, 16.7 |
| L4 | 71 (DMF) | 257−258 | C22H16N10S8 | 39.17 (39.03) | 2.03 (2.38) | 20.92 (20.69) | 170.1, 168.6, 158.6, 158.2, 155.2, 154.4, 137.9, 123.2, 120.2, 40.2, 16.7 |
| L5 | 95 (MeOH-H2O) | 80−81 | C14H12N4S3 | 50.91 (50.58) | 3.57 (3.64) | 16.94 (16.85) | 164.9, 155.4, 149.5, 136.9, 123.3, 122.7, 39.9 |
| L6 | 80 (AcOEt) | 163−165 | C26H22N6S3 | 61.01 (60.67) | 3.90 (4.31) | 16.41 (16.33) | 165.4, 157.9, 156.0, 154.8, 154.7, 137.6, 137.0, 123.4, 122.7, 119.9, 118.1, 40.4, 24.6 |
| L7 | 73 (Dioxane) | 159−160 | C16H12N6S4 | 46.52 (46.13) | 2.49 (2.90) | 19.74 (20.17) | 168.3, 158.6, 155.0, 149.6, 136.9, 123.3, 122.8, 39.8 |
| L8 | 80 Dioxane/DMF | 248−250 | C28H22N8S4 | 55.98 (56.16) | 3.85 (3.70) | 18.92 (18.71) | 168.8, 158.6, 157.9, 156.2, 154.7, 154.2, 137.7, 137.0, 123.5, 122.7, 120.0, 118.1, 40.3, 26.6 |
Conclusions
Experimental
General
5-Mercapto-5'-methylthio-2,2'-bi-1,3,4-thiadiazole (2).
General Procedure for the Nucleophilic Displacement Reactions: Synthesis of 2,6-Bis[5-methylthio-1,3,4-thiadiazol-2-ylthio)methyl]pyridine (L1)
Acknowledgements
References
- Balzani, V.; Scandola, F. Supramolecular Photochemistry; Ellis Horwood: Chichester, 1991. [Google Scholar] Lehn, J. M. Supramolecular Chemistry - Concepts and Perspectives; VCH: Weinheim, 1995. [Google Scholar] Balzani, V.; Juris, A.; Venturi, M.; Campagna, S.; Serroni, S. Chem. Rev. 1996, 96, 759, and refs. therein.
- Barigelletti, F.; Flamigni, L.; Guardigli, M.; Juris, A.; Beley, M.; Chodorowski-Kimmes, S.; Collin, J.-P.; Sauvage, J.-P. Inorg. Chem. 1996, 35, 136, and refs. Therein. [CrossRef] [PubMed]Harriman, A.; Ziessel, R. Chem. Commun. 1996, 1707. Shaw, J. R.; Sadler, G. S.; Wacholtz, W. F.; Ryu, C. K.; Schmehl, R. H. New J. Chem. 1996, 20, 749.
- Biagini Cingi, M; Lanfranchi, M; Pellinghelli, M. A.; Tegoni, M. Eur. J. Inorg. Chem. 2000, 703. Mahong, Y.; Song, J.; Xujie, Y.; Lude, L.; Xin, W. Wuji Huaxue Xuebac. 1988, 14, 276. Fennema, B. D. J. R.; De Graaff, R. A. G.; Hage, R.; Haasnoot, J. G.; Reedijk, J.; Vos, J. G. J. Chem. Soc., Dalton Trans. 1991, 1043. Fabretti, A. C.; Giusti, A. Polyedron 1986, 5, 1927. Gajendragad, M. R.; Mohiyuddin, R.; Agarwala, U. Aust. J. Chem. 1975, 37, 2429.
- Orellana, G.; Quiroga, M. L.; Braun, A. M. Helv. Chim. Acta 1987, 70, 2073.
- Mamo, A. J. Heterocyclic Chem. 2000, 37, 1225. [CrossRef] Mamo, A.; Stefio, I.; Parisi, M.F.; Credi, A.; Venturi, M.; Di Pietro, C.; Campagna, S. Inorg. Chem. 1997, 36, 5947. Mamo, A.; Stefio, I.; Poggi, A.; Tringali, C.; Di Pietro, C.; Campagna, S. New. J. Chem. 1997, 21, 1173.
- Pappalardo, S.; Bottino, F.; Di Grazia, M. J. Heterocyclic Chem. 1984, 21, 1113.
- Aue, W. P.; Bartholdi, E.; Ernst, R. R. J. Chem. Phys. 1964, 64, 2229.
- Finocchiaro, P.; Mamo, A.; Tringali, C. Magnetic Resonance in Chemistry 1991, 29, 1165.
- Still, I. W. J.; Plavac, N.; McKinnon, D. M.; Chauhan, M. S. Can. J. Chem. 1976, 54, 1660. [CrossRef] Bartels-Keith, J. R.; Burgess, M. T.; Stevenson, J. M. J. Org. Chem. 1977, 42, 3725. Fujita, E.; Nagao, Y.; Seno, K.; Takao, S.; Miyasada, T.; Rimura, M.; Watson, W. H. J. Chem. Soc., Perkin Trans. 1. 1981, 914.
- Pappalardo, S.; Bottino, F.; Tringali, C. J. Org. Chem. 1987, 52, 405.
- Bottino, F.; Pappalardo, S. Org. Magn. Res. 1981, 16, 1.
- Baker, W.; Buggle, K. M.; McOmie, J. F. W.; Watkins, D. A. M. J. Chem. Soc. 1958, 3594.
- Newkome, G. R.; Puckett, W. E.; Kiefer, G. E.; Gupta, V. K.; Xia, Y.; Coreil, M.; Hackney, M. A. J. Org. Chem. 1982, 47, 4116.
- Kato, H.; Ohta, M. Nippon Kagaku Zasshi 1957, 78, 1588.
- Sample availability: Available from the authors.
© 2003 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Mamo, A.; Pappalardo, A.; Clasadonte, M.T. Sulfur Bridged Multidentate Ligands Based on (Bi)pyridyl-(Bi)-1,3,4-Thiadiazolyl Conjugates. Molecules 2003, 8, 310-317. https://doi.org/10.3390/80300310
Mamo A, Pappalardo A, Clasadonte MT. Sulfur Bridged Multidentate Ligands Based on (Bi)pyridyl-(Bi)-1,3,4-Thiadiazolyl Conjugates. Molecules. 2003; 8(3):310-317. https://doi.org/10.3390/80300310
Chicago/Turabian StyleMamo, Antonino, Andrea Pappalardo, and M. Teresa Clasadonte. 2003. "Sulfur Bridged Multidentate Ligands Based on (Bi)pyridyl-(Bi)-1,3,4-Thiadiazolyl Conjugates" Molecules 8, no. 3: 310-317. https://doi.org/10.3390/80300310




