2.2. Optimization of Polymerization Conditions
The catalytic properties of water-soluble salen complexes were investigated by changing the metal ions in the active centre and varying the reaction conditions in order to optimize the polymerization degree of isoeugenol (2-methoxy-4-(1-propenyl) phenol) (
Figure 1). The polymerization degree was evaluated by GPC analyses.
The first reaction followed was the oxidation of isoeugenol in a 1:1 water-ethanol solution with different catalysts. Ethanol is required as a cosolvent as isoeugenol is not water-soluble. Two types of bulky salen complexes were used: the SulphoSalen (SS,
1) and the PhosphoSalen (PS,
2). The effectiveness of four different metal ions acting as the active center were tested: Manganese (Mn), Iron (Fe), Cobalt (Co) and Copper (Cu) for Sulphosalen complexes (
1); Copper (Cu) and Cobalt (Co) for Phosphosalen complexes (
2). During the reaction, isoeugenol consumption was monitored by TLC. The reaction was carried out for 48 hours at room temperature under stirring. In these conditions the main products are isoeugenol oligomers generated by coupling reactions of phenoxy radicals. The concentration of isoeugenol in the solution was equal to 1% w/v and the concentration of catalyst 0.1% w/v. At the end of reaction, the molecular weight of isoeugenol oligomers were determined on extracted products by GPC analysis (
Table 1).
Mn- and Fe-Salen catalyzed oxidations are known to occur through metal-oxo complexes in the presence of hydrogen peroxide, while Co- and Cu- complexes activate dioxygen forming a 1:1 metal to oxygen superoxo or a 2:1 metal to oxygen peroxo complexes as the active oxidizing species. As reported in
Table 1, Iron, Manganese and Copper complexes were activated by the presence of H
2O
2, while Cobalt and Copper complexes were exposed to a slight overpressure of molecular oxygen (O
2). According to literature data [
7] Copper complexes (CuSS and CuPS) were found to be more active in the presence of H
2O
2 and led to DHPs with higher molecular weights. The obtained products were mainly oligomeric compounds with a low amount (maximum 5% w/w) of monomeric side-chain oxidation by-products. These oxidation products were characterized by GC-MS analysis, and consist mainly in vanillin (3-methoxy-4-hydroxybenzaldehyde,
5), acetovanillone (1-(3-methoxy-4-hydroxyphenyl)-ethanone,
6) and vanillylmandelic acid (hydroxy-(3-hydroxy-4-methoxyphenyl) acetic acid,
7) (
Figure 2).
These preliminary GPC data showed that Cu(SS) catalyzes the formation of polymers with the highest M
n and M
w in the presence of H
2O
2 as the oxidant. Afterwards, the effect of pH on the polymerization reaction catalyzed by Cu(SS) was taken into account monitoring the M
n and M
w values of DHPs synthesized into different buffer solution. As shown in
Table 2, the molecular weight of the isoeugenol DHP was lowest at pH 4.5 and greatest at pH 9. An increased activity of Sulphosalen at higher pH value was also reported by Sippola [
18].
Another parameter influencing DHPs structure is the monomer addition rate to the reaction mixture. The reagent could be added either slowly and continuatively (Zutropf method ZT) or by means of an unique, batch-mode addition (Zuluaf method ZL). Literature data report a lower molecular weight for Zuluaf DHPs, which are assumed to precipitate faster than Zutropf polymers [
19]. Differences in the products composition are presumably related to the concentration of radicals. The aim of the present work is to point out changes in DHPs molecular weight distribution as the monomer addition rate is varied. Therefore, potential differences in intermonomeric linkages type and distribution have not been taking into account. In these experiments a slow rate addition was achieved by means of a step-wise approach.
According to literature data, GPC analyses showed an higher molecular weight for Zutropf DHPs (
Table 3) [
19].
The polymerization degree was then optimized by changing the catalyst (CuSS) concentration and the amount of isoeugenol, expressed as w/v% respect to water-ethanol solution. GPC results are reported in
Table 4. A correlation between the catalyst concentration and the average molecular weight of the DHPs was observed: a rise from 0.2% up to 2% in the amount of catalyst allowed for a doubling of M
w. Otherwise, when the same amount of catalyst was used, a straightforward relationship between isoeugenol concentration and M
w were not identified as the polymerization degree did not seem to be greatly affected by a monomer percentage above 1%.
Two additional parameters play a crucial role in the dehydrogenative polymerization: amount and addition rate of the oxidant into the reaction medium. For this reason, differences in the DHPs molecular weight distribution at different concentrations and at different addition rates of H
2O
2 were investigated. The concentration of H
2O
2 was expressed as an overall molar ratio between H
2O
2 and isoeugenol while the step-wise addition of the oxidant was carried out at regular intervals (four aliquots: one addition in the morning and one addition in the afternoon covering a reaction period of about 48 hours. Eight aliquots: four additions per day, every two hours, over two eight-hour working day. Sixteen aliquots: eight additions per day, every hour, over two eight-hour working day). Results are reported in
Table 5.
Statistical analyses performed by means of the factorial ANOVA point out which parameters may affect the DHP molecular weight. It seems that both of the parameters influence the molecular weight distribution (P-value 0.01 and 0.11 for oxidant concentration and velocity of addition respectively), with the contribution of the oxidant concentration as the most important. Moreover, it is possible to observe an important interaction effect between the parameters (P-value 0.76). The best result in terms of Mn and Mw was accomplished for a H2O2/isoeugenol molar ratio equal to 10 and a relatively fast addition rate (4 different aliquots). Differences in molecular weights are presumably related to different concentrations of the monomeric phenoxy radicals generated during the reaction.
2.3. Polymer Characterization
The DHP synthesized by adopting the best reaction conditions (isoeugenol 1% w/v, CuSS as catalyst 0.2% w/v, pH 9, H
2O
2 as oxidant, molar ratio H
2O
2:isoeugenol 10:1 added in four aliquots, 48 h reaction time) was characterized by means of a range of analytical techniques: GPC,
13C-NMR,
31P-NMR and LC-MS. Gel permeation chromatography provided information about the molecular weight distribution of the product, expressed in terms of M
n and M
w. Results showed that the DHP had a number-average molecular weight (M
n) of about 2,500 g/mol and a weight-average molecular weight (M
w) of about 8,000 g/mol. Therefore, the number of monomer units constituting the polymer was about 15, assuming 164 g/mol the molecular weight of the repeating unit.
13C-NMR was performed on the acetylated isoeugenol polymer [
20]. The spectrum (
Figure 3) showed the characteristic signals of intermonomeric bonds correlated to the β-carbon and the α-carbon of β-
O-4 and β-5 moieties respectively [
12,
21]. In the range between 110–150 ppm fall many broad and partially unresolved peaks related to aromatic and olefinic carbons, as expected on the basis of the complex structure of the dehydrogenated polymer. The area comprised between 167 and 170 ppm enclose two peaks, assigned to the acetylic carbon atom connected to hydroxyl groups (phenols 167–169 ppm and secondary alcohol 169–170 ppm). Moreover, an intense signal originated by methoxylic carbon atoms is found at about 55 ppm.
31P-NMR analysis was performed on the phosphytylated isoeugenol polymer in order to quantify labile -OH groups (different phenols, aliphatic hydroxyls and carboxylic acid) on the polymer [
22]. The acquired spectra showed a signal related to aliphatic -OH (0.60 mmol/g, 147–150 pm), due to the presence of secondary alcohol originated from β-
O-4 linkages. This result is in agreement with the result observed after the
13C-NMR analysis. The absence of primary aliphatic alcohol is associated to the structure of isoeugenol, which does not posses any hydroxyl functionality at the γ position. A slight side-chain oxidation is confirmed by the presence of a limited amount of carboxylic acid (0.03 mmol/g, 134–135 ppm). By means of
31P-NMR spectroscopy it is also possible to detect a moderate amount of condensed phenols, related to 5-5’ and 4-
O-5’ phenolic structures (0.11 mmol/g, 143–145 ppm), and a large content of non-condensed guaiacyl units (0.99 mmol/g, 140–141 pm) (
Figure 4).
Further confirmation on the DHP structure were obtained by LC-MS analysis. This spectroscopic technique, when soft ionization procedures are applied, act as a powerful tool in lignin investigation [
23,
24]. The LC-MS spectrum of isoeugenol oxidative coupling product is reported in
Figure 5.
Polymerization clusters are distinctly recognizable and regularly occur every 163 amu along with patterns located at +18 amu. Regular raising mass of 163 amu could be regarded as progressive radical couplings resulting in the formation of β-5 or condensed (5-5’, 4-
O-5’) units [
21]. Patterns occurring at +18 amu after every principal peak account for the formation of β-
O-4 bonds, which is subjected to water insertion on the intermediate quinone methide.
Altogether, these results are in agreement with a radical coupling mechanism. The reaction starts with the formation of phenoxy radicals by metal catalyst-mediated H abstraction. Then, the mesomeric delocalization on the phenylpropenoid structure lead to various type of radical coupling reactions. The resulting oligomers are characterized by different intermonomeric linkages, mainly represented by β-
O-4 and β-5 bonds [
25,
15]. On the basis of NMR, GPC and LC-MS qualitative and quantitative data it was possible to attribute a tentative formula, accounting for a ‘lignin-like’ structure, to the isoeugenol DHP (
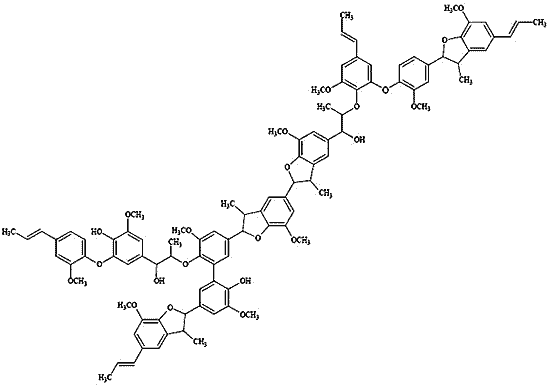
Figure 6). A representative DHP oligomer was assumed to contain about 10 repeating units, connected by β-5, β-
O-4 bonds and condensed units (5-5’, 4-
O-5’) as the principal intermonomeric linkages.
2.4. Radical Scavenging Activity
The obtained DHP is proved to possess valuable antioxidant and antibacterial activity. Elegir and coworkers studied the antibacterial properties of lignocellulosic fibers treated with isoeugenol and laccase as grafting initiator [
17]. The antibacterial activity
versus Staphylococcus aureus was strongly enhanced by the polymerization with respect to the isoeugenol monomer. This important property could be applied in the development of antimicrobial packaging based on lignocellulosic materials. The main mechanism involved in the antioxidant activity of phenolic compounds is supposed to be the scavenging of free radicals. Therefore, the reactivity towards the stable radical DPPH• of both isoeugenol and DHP was tested (
Table 6).
Isoeugenol showed a higher radical scavenging activity, with an effective concentration of DPPH• radical (IC
50) equal to 0.195 mg[AH]/mg[DPPH•]. Alternatively, this result could be expressed in mole (0.48 mol per mol of DPPH•) and it is in agreement with literature data [
21,
26]. The data evidenced that the molar ratio isoeugenol/DPPH• needed for a complete reaction is 1.04, indicating that one mole of phenolic groups reacts with one mole of DPPH•. When the DHP is concerned, the observed radical scavenging activity show a higher IC
50 value, 1.41 mg per mg of DPPH. However, the data is in agreement with the total phenolic content of the DHP (1.1 mmol/g), demonstrating that the radical scavenging activity of the isoeugenol DHP is also related to the amount of phenolic groups.