Protective Effect of Tulbaghia violacea Harv. on Aortic Pathology, Tissue Antioxidant Enzymes and Liver Damage in Diet-Induced Atherosclerotic Rats
Abstract
:1. Introduction
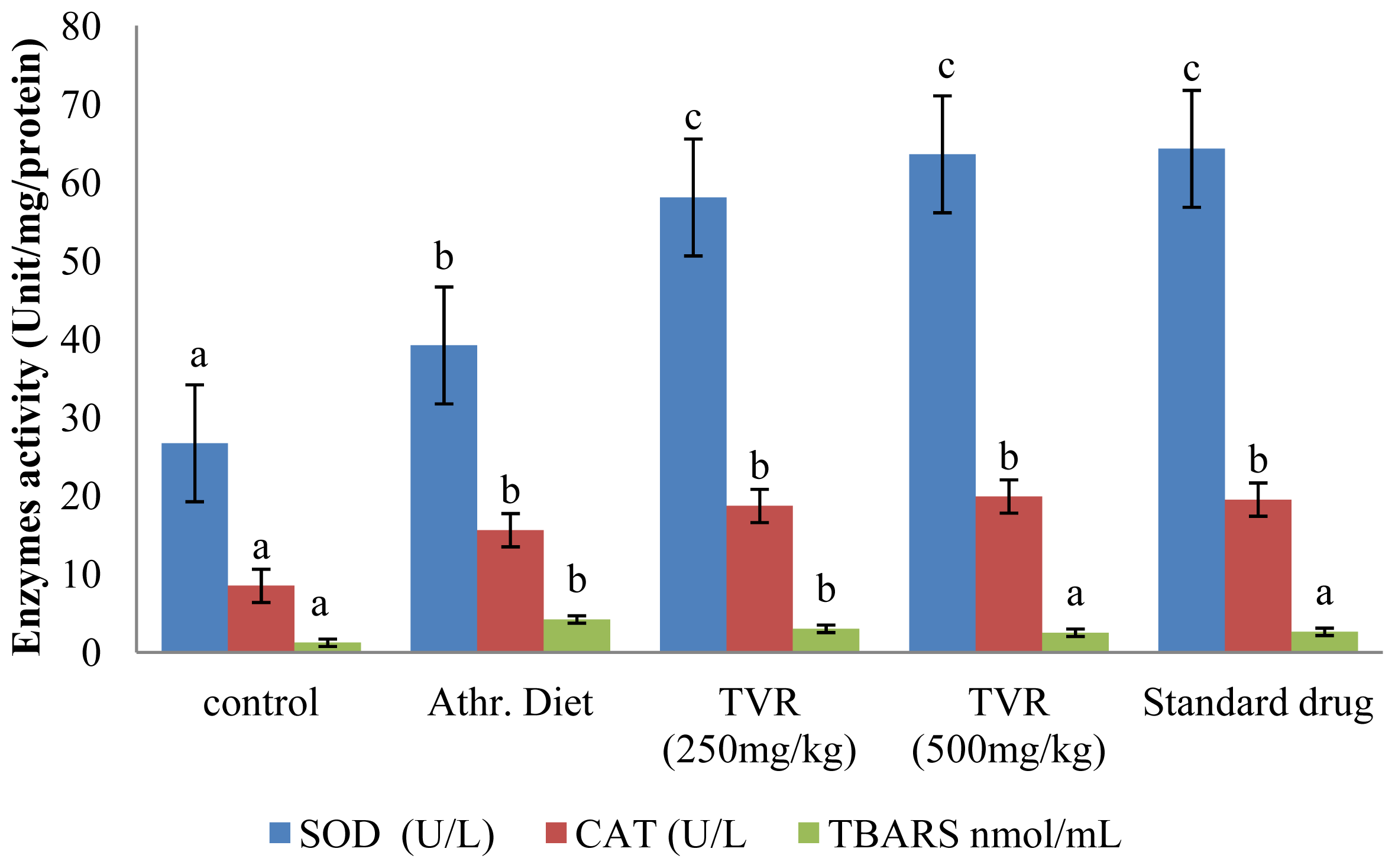
2. Results and Discussion
3. Materials and Methods
3.1. Plant Collection and Extract Preparation
3.2. Animals
3.3. Experiment Design
- Group 1: Experimental animals fed with a standard diet and orally administered 1 mL distilled water served as control.
- Group II: Negative control rats were fed with an atherogenic diet comprised of normal rat chow plus 4% cholesterol, 1% cholic acid and 0.5% thiouracil.
- Group III and IV: These experimental animals were fed with an atherogenic diet comprised of the normal rat chow plus 4% cholesterol, 1% cholic acid and 0.5% thiouracil, but also supplemented orally with extract of T. violacea (0.25 g/kg and 0.50 g/kg body weight, respectively) once daily for two weeks.
- Group V: These rats were fed with an atherogenic diet [normal rat chow plus 4% cholesterol, 1% cholic acid and 0.5% thiouracil) supplemented with standard atorvastatin orally (30 mg/kg body weight) suspended in distilled water once daily for two weeks.
3.4. Body Weight, Food Intake and Water Consumption
3.5. Sample Collection
3.6. Liver and Aorta Homogenate Preparation
3.7. Hematological Analysis
3.8. Biochemical Assay
3.9. Determination of Catalyse Activity (CAT)
3.10. Determination of Superoxide Dismutase Activity
3.11. Estimation of Malonyldialdehyde (MDA)
3.12. Histopathological Analysis of the Liver and Aorta
3.13. Statistical Analysis
4. Conclusion
Acknowledgements
References
- Braunwald, E. Shattuck lecture-cardiovascular medicine at the turn of the millennium: Triumphs, concerns, and opportunities. N. Engl. J. Med 1997, 337, 1360–1369. [Google Scholar]
- Shiao, M.S.; Chiu, J.J.; Chang, B.W.; Wang, J.; Jen, W.P.; Wu, Y.J.; Chen, Y.L. In search of antioxidants and anti-atherosclerotic agents from herbal medicines. Biofactors 2008, 34, 147–157. [Google Scholar]
- Anuradha, S.; Sugumar, V.R. Impact of coconut oil replacement in diet among obese adolescent girls. Indian Coconut J 2009, 52, 12–16. [Google Scholar]
- Bibave, K.H.; Shenoy, P.A.; Mahamuni, S.P.; Bandawane, D.D.; Nipate, S.S.; Chaudhari, P.D. Preclinical evaluation methods for screening of anti-atherosclerotic drugs: An overview. Asian J. Biomed. Pharm. Sci 2011, 1, 1–14. [Google Scholar]
- Lewis, S.J. Prevention and treatment of atherosclerosis: A practitioner’s guide for 2008. Am. J. Med 2009, 122, S38–S50. [Google Scholar]
- Jain, N.C.; Schalm, O.W.; Carroll, E.J. Veterinary Hematology; Lea & Febiger: Philadelphia, PA, USA, 1986; pp. 235–245. [Google Scholar]
- Davidson, M.H.; Tooth, P.P. Comparative effect of lipid lowering therapies. Prog. Cardiovasc. Dis 2004, 47, 173–204. [Google Scholar]
- Olorunnisola, O.S.; Bradley, G.; Afolayan, A.J. Ethnobotanical information on plants used for the management of cardiovascular diseases in Nkonkobe municipality of South Africa. J. Med. Plants Res 2011, 5, 4256–4260. [Google Scholar]
- Bungu, L.; van de Venter, M.; Frost, C. Evidence for an in vitro anticoagulant and antithrombotic activity in Tulbaghia violacea. Afr. J. Biotechnol 2008, 7, 681–688. [Google Scholar]
- Olorunnisola, O.S.; Bradley, G.; Afolayan, A.F. Antioxidant properties and cytotoxicity evaluation of methanolic extract of dried and fresh rhizomes of Tulbaghia violacea. Afr. J. Pharm. Pharmacol 2011, 5, 2490–2497. [Google Scholar]
- Gaidamashivili, M.; van Staden, J. Interaction of lectin-like proteins of South African medicinal plants with Staphylococcus aureus and Bacillus subtilis. J. Ethnopharm 2001, 80, 131–135. [Google Scholar]
- Bungu, L.; Frost, C.L.; Brauns, S.C.; van de Venter, M. Tulbaghia violacea inhibits growth and induces apoptosis in cancer cells in vitro. Afr. J. Biotechnol 2006, 5, 1936–1943. [Google Scholar]
- McGaw, L.J.; Jager, A.K.; van Staden, J. Antibacterial, anthelmintic and anti-amoebic activity in South African medicinal plants. J. Ethnopharmacol 2000, 72, 247–263. [Google Scholar]
- Van Wyk, B.-E.; van Oudtshoorn, B.; Gericke, N. Medicinal Plants of South Africa, 1st ed; Brizza Publications: Pretoria, South Africa, 1997; p. 138. [Google Scholar]
- Leopold, J.A.; Loscalzo, J. Oxidative mechanisms and atherosthrombotic cardiovascular disease. Drug Discov. Today 2008, 5, 5–13. [Google Scholar]
- Kanthlal, S.K.; Sureesh, V.; Arunachalam, G.; Royal Frank, P.; Kameshwaran, S. Anti obesity and hypolipidemic activity of methanolic extracts of Taberenaemontana Divaricata on atherogenic diet induced obesity in rats. Int. Res. J. Pharm. 2012, 3, 157–161. [Google Scholar]
- Azonov, J.A.; Khorram Khorshid, H.R.; Novitsky, Y.A.; Farhadi, M.; Ghorbanoghli, Z.; Shahhosseiny, M.H. Protective effects of setarud (IMODTM) on development of diet-induced hypercholesterolemia in rabbits. DARU J. Pharm. Sci 2008, 16, 218–222. [Google Scholar]
- Shyam Sunder, A.; Rama Narsimha Reddy, A.; Rajeshwar, Y.; Kiran, G.; Krishna Prasad, D.; Baburao, B.; Thirumurugu, S.; Karthik, A. Protective effect of methanolic extract of Trianthema portulacastrum in atherosclerotic diet induced renal and hepatic changes in rats. Der Pharmacia. Lettre 2010, 2, 540–545. [Google Scholar]
- Shankar, V.; Kaur, H.; Dahiya, K.; Gupta, M.S. Comparison of fasting and postprandial lipid profile in patients of coronary heart disease. Bombay Hosp. J 2008, 50, 445–449. [Google Scholar]
- Gokkusu, C.; Mostafazadeh, T. Changes of oxidative stress in various tissues by long-term administration of vitamin E in hypercholesterolemic rats. Clin. Chim. Acta 2003, 328, 155–161. [Google Scholar]
- Maxfield, F.R.; Tabas, I. Role of cholesterol and lipid organization in disease. Nature 2005, 438, 612–621. [Google Scholar]
- Chia, B.L. Cholesterol and coronary artery disease-issues in the 1990s. Singapore Med. J 1991, 32, 291–294. [Google Scholar]
- Yakubu, M.T.; Akanji, M.A.; Oladiji, A.T. Alterations in serum lipid profile of male rats by oral administration of aqueous extract of Fadogia agrestis stem. Res. J. Med. Plant 2008, 2, 66–73. [Google Scholar]
- Saikia, H.; Lama, A. Effect of Bougainvillea spectabilis leaves on serum lipids in albino rats fed with high fat diet. Int. J. Pharm. Sci. Drug Res 2011, 3, 141–145. [Google Scholar]
- Chigozie, J.I.; Chidinma, C.I. Alteration of plasma lipid profiles and atherogenic indices by Stachytarpheta jamaicensis L. (Vahl). Biokemistri 2009, 21, 71–7726. [Google Scholar]
- Raghuveer, C. Beneficial effect of allium sativum and allium tuberosum on experimental hyperlipidemia and atherosclerosis. Pak. J. Physiol 2008, 4, 7–9. [Google Scholar]
- Brehm, A.; Pfeiler, G.; Pacini, G.; Vierhapper, H.; Roden, M. Relationship between serum lipoprotein ratios and insulin resistance in obesity. Clin. Chem 2004, 50, 2316–2322. [Google Scholar]
- Law, M.R. Lowering heart disease risk with cholesterol reduction: evidence from observational studies and clinical trials. Eur. Heart J. Suppl 1999, 1, S3–S8. [Google Scholar]
- Botham, K.M.; Mayes, P.A. Harper’s Illustrated Biochemistry, 26th ed; The McGraw-Hill Companies Inc: New York, NY, USA, 2006; pp. 217–245. [Google Scholar]
- Bainton, D.; Miller, N.E.; Bolton, C.H.; Yarnell, J.W.; Sweetnam, P.M.; Baker, A.I.; Lewis, B.; Elwood, P.C. Plasma triglyceride and high density lipoprotein cholesterol as predictors of ischaemic heart disease in British men. Br. Heart J 1992, 68, 60–66. [Google Scholar]
- Yeh, Y.Y.; Liu, L. Cholesterol lowering effect of garlic extracts and organosulfur compounds Human and animal studies. J. Nutr 2001, 131, 389s–393s. [Google Scholar]
- Wu, Y.; Li, J.; Wang, J.; Si, Q.; Zhang, J.; Jiang, Y.; Chu, L. Anti-atherogenic effects of centipede acidic protein in rats fed an atherogenic diet. J. Ethnopharmac 2009, 122, 509–516. [Google Scholar]
- Sharma, S.; Kumar, S.; Wiseman, D.A.; Kallarackal, S.; Ponnala, S.; Elgaish, M.; Tian, J.; Fineman, J.R.; Black, S.M. Perinatal changes in superoxide generation in the ovine lung: Alterations associated with increased pulmonary blood flow. Vascul. Pharmacol 2010, 53, 38–52. [Google Scholar]
- Rantanachamnong, P.; Matsathit, Y.; Sanvarinda, Y.; Piyachaturawat, P.; Phivthong Ngam, L. Effects of Curcurma comosa Roxa. On platelet aggregation and atherosclerosis plaque development in hypercholesterolemic Rabbits. Int. J. Pharmacol 2012, 8, 234–242. [Google Scholar]
- Hoak, J.C. Platelets and atherosclerosis. Thromb. Haemost 1988, 14, 202. [Google Scholar]
- Chandler, A.; Hand, R. Phagocytized platelets: A source of lipids in human thrombi and atherosclerotic plaques. Science 1961, 134, 946. [Google Scholar]
- Mendelsohn, M.E.; Loscalzo, J. Role of platelets in cholesteryl ester formation by U-937 cells. J. Clin. Invest 1988, 81, 62. [Google Scholar]
- Ware, J.A.; Coller, B.S. Platelet Morphology, Biochemistry and Function. In Williams’ Hematology; Beutler, E., Lichtman, M.A., Coller, B.S., Kipps, T.J., Eds.; McGraw Hill: New York, NY, USA, 1994; p. 1161. [Google Scholar]
- Stein, B.; Fuster, V.; Israel, D.H.; Cohen, M.; Badimon, L.; Badimon, J.J.; Chesebro, J.H. Platelet inhibitor agents in cardiovascular disease: An update. J. Am. Coll. Cardiol 1989, 14, 813. [Google Scholar]
- Ryszawa, N.A.; Kawczyñska-dród, J.; Pryjma, M.; Czesnikiewicz-guzik, T.; Adamek-guzik1, M.; Naruszewicz, R.; Korbut, T.; Guzik, J. Effects of novel plant antioxidants on platelet superoxide production and aggregation in atherosclerosis. J. Physiol. Pharmacol 2006, 57, 611–626. [Google Scholar]
- Pignatelli, P.; di Santo, S.; Buchetti, B.; Sanguigni, V.; Brunelli, A.; Violi, F. Polyphenols enhance platelet nitric oxide by inhibiting protein kinase C-dependent NADPH oxidase activation: Effect on platelet recruitment. FASEB J 2006, 20, 1082–1089. [Google Scholar]
- Ada, M.C.; Alfredo, M.G. Diet and Endothelial Function. Biol. Res 2004, 37, 225–230. [Google Scholar]
- Lokhande, P.D.; Dhawaren, B.S.; Jagdale, S.C.; Chabukswar, A.R.; Mukawar, S.A. Cardiac activity of isolated constituents of Inula racemosa. J. Herb. Pharmcother 2006, 6, 81–88. [Google Scholar]
- Lauf, U.; Fata, V.L.; Liao, J.K. Inhibition of 3-hydroxy-3-methylglutary (HMG)-CoA reductase blocks hypoxia-mediated down-regulation of endothelial nitric oxide synthase. J. Biol. Chem 1997, 272, 31725–31729. [Google Scholar]
- Matsuzawa, N.; Takamura, T.; Kurita, S.; Misu, H.; Ota, T.; Ando, H.; Yokoyama, M.; Honda, M.; Zen, Y.; Nakanuma, Y.; et al. Lipid-induced oxidative stress causes steatohepatitis in mice fed an atherogenic diet. Hepatology 2007, 46, 1392–1403. [Google Scholar]
- Panigrahi, B.B.; Panda, P.K.; Patro, V.J. Comparative hepatoprotective activity of different extracts of spirulina against CCl4 induced liver damage in rats. Int. J. Pharm. Sci. Rev. Res 2010, 4, 200–202. [Google Scholar]
- Khatri, A.; Garg, A.; Agrawal, S.S. Evaluation of hepatoprotective activity of aerial part of Tephrosia purpuria L. and stem bark of Tecomella undulata. J. Ethnopharmacol 2009, 122, 1–5. [Google Scholar]
- Mohammad, S.F.; Woodward, S.C. Characterisation of a potent inhibitor of platelet aggregation and release reaction isolated from Allium sativum (Garlic). Thromb. Res 1986, 44, 793–806. [Google Scholar]
- Noori, S.A.; Mahboob, M. Study on the antioxidant effects of cinnamon and garlic extract in liver, kidney and heart tissue of rat. Biosci. Res 2012, 9, 17–22. [Google Scholar]
- Green, L.C.; Wagner, D.A.; Glogowski, J.; Skippe, P.; Wishnok, J.S.; Tannenbaum, S.R. Analysis of nitrate, nitrite and [15N] nitrate in biological fluids. Anal. Biochem 1982, 126, 131–138. [Google Scholar]
- Pari, L.; Latha, M. Protective role of Scoparia dulcis plant extract on brain antioxidant status and lipid peroxidation in STZ diabetic male Wistar rats. BMC Complement Altern. Med 2004, 4. [Google Scholar] [CrossRef] [Green Version]
- Misra, H.P.; Fridovich, I. The role of superoxide anion in the autooxidation of epinephrine and simple assay for superoxide dismutase. J. Biol. Chem 1972, 247, 3170–3175. [Google Scholar]
- Okrainec, K.; Banarjee, D.K.; Eisenberg, M.J. Coronary artery disease in the developing world. Am. Heart J 2004, 148, 7–15. [Google Scholar]


| Treatment | AIW (g) | AFW | % weight gain | FI (g/2 weeks) |
|---|---|---|---|---|
| Control | 151.0 ± 0.11 | 208.0 ± 0.14 | 38.0 | 341.11 |
| Athero-diet | 152.1 ± 0.12 | 237.1 ± 0.12 * | 55.9 | 353.62 |
| Athero-diet + TVR (250 mg/kg) | 150.2 ± 0.10 | 215.2 ± 0.11 | 36.7 | 346.24 |
| Athero-diet + TVR (500 mg/kg) | 153.0 ± 0.13 | 210.3 ± 0.13 | 34.6 | 343.40 |
| Athero-diet + TVR (30 mg ator) | 152.4 ± 0.12 | 209.1 ± 0.11 | 37.2 | 342.21 |
| Treatment (mg/kg) | TG (mg/dL) | TC (mg/dL) | LDL (mg/dL) | VLDL (mg/dL) | HDL-c (mg/dL) | Atherogenic index (AI) |
|---|---|---|---|---|---|---|
| Control | 63.32 ± 0.25 a | 75.11 ± 0.21 a | 7.45 ± 0.23 a | 15.94 ± 0.50 a | 39.10 ± 0.41 a | 0.92 ± 0.12 |
| Ath diet | 99.3 ± 1.23 b | 148.3 ± 0.23 b | 25.24 ± 0.17 b | 38.12 ± 0.19 b | 11.59 ± 0.35 b | 11.80 ± 0.15 |
| TVR (250) | 79.35 ± 0.43 c | 95.21 ± 0.22 c | 19.18 ± 0.15 c | 25.43 ± 0.23 c | 21.61 ± 0.27 c | 3.41 ± 0.14 a |
| TVR (500) | 71. 78 ± 0.21 c | 83.11 ± 0.25 d | 10.44 ± 0.21 d | 19.25 ± 0.14 d | 29.41 ± 0.19 d | 1.83 ± 0.11 b |
| Ator- (30) | 66.16 ± 0.24 a | 80.34 ± 0.31 d | 8.54 ± 0.32 d | 16.19 ± 0.11 d | 31.29 ± 0.16 d | 1.56 ± 0.13 c |
| Group | PLT (g/L) | Fibrinogen (g/L) | NO (μmol/L) |
|---|---|---|---|
| Normal | 405.2 ± 0.25 a | 1.95 ± 0.43 a | 57.33 ± 0.11 a |
| Ath diet | 847.4 ± 0.21 b | 3.21 ± 0.28 b | 46.38 ± 0.18 b |
| TVR (250 mg/kg) | 490.6 ± 0.23 a | 2.63 ± 0.25 c | 51.36 ± 0.25 a |
| TVR (500 mg/kg) | 420.4 ± 0.16 a | 2.01 ± 0.39 d | 53.28 ± 0.35 a |
| Ato- (30mg/kg) | 415.9 ± 0.51 a | 1.99 ± 0.16 d | 55.19 ± 0.22 a |
| Group | AST (IU/L) | ALT (IU/L) | ALP (IU/L) | LDH (IU/L) | T-Bilirubin (mg/dL) | Creatinine (μmol/L) |
|---|---|---|---|---|---|---|
| Normal | 23.12 ± 0.15 a | 26.03 ± 0.13 a | 42.33 ± 0.17 a | 69.18 ± 0.11 a | 0.21 ± 0.21 a | 61.12 ± 0.50 a |
| Ath diet | 47.32 ± 0.21 b | 51.21 ± 0.18 b | 92.38 ± 0.14 b | 138.3 ± 0.23 b | 5.43 ± 0.25 b | 67.11 ± 0.32 b |
| TVR (250 mg/kg) | 38.61 ± 0.33 c | 41.13 ± 0.15 c | 58.22 ± 0.45 c | 98.12 ± 0.20 c | 3.98 ± 0.19 c | 65.01 ± 0.21 c |
| TVR (500 mg/kg) | 31.55 ± 0.16 c | 35.43 ± 0.19 d | 51.14 ± 0.31 c | 79.33 ± 0.35 d | 2.14 ± 0.25 d | 62.23 ± 0.38 a |
| Ato- (30mg/kg) | 28.89 ± 0.51 a | 34.63 ± 0.16 d | 50.69 ± 0.42 c | 73.41 ± 0.31 d | 2.11 ± 0.24 d | 61.99 ± 0.26 a |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Olorunnisola, O.S.; Bradley, G.; Afolayan, A.J. Protective Effect of Tulbaghia violacea Harv. on Aortic Pathology, Tissue Antioxidant Enzymes and Liver Damage in Diet-Induced Atherosclerotic Rats. Int. J. Mol. Sci. 2012, 13, 12747-12760. https://doi.org/10.3390/ijms131012747
Olorunnisola OS, Bradley G, Afolayan AJ. Protective Effect of Tulbaghia violacea Harv. on Aortic Pathology, Tissue Antioxidant Enzymes and Liver Damage in Diet-Induced Atherosclerotic Rats. International Journal of Molecular Sciences. 2012; 13(10):12747-12760. https://doi.org/10.3390/ijms131012747
Chicago/Turabian StyleOlorunnisola, Olubukola S., Graeme Bradley, and Anthony J. Afolayan. 2012. "Protective Effect of Tulbaghia violacea Harv. on Aortic Pathology, Tissue Antioxidant Enzymes and Liver Damage in Diet-Induced Atherosclerotic Rats" International Journal of Molecular Sciences 13, no. 10: 12747-12760. https://doi.org/10.3390/ijms131012747
APA StyleOlorunnisola, O. S., Bradley, G., & Afolayan, A. J. (2012). Protective Effect of Tulbaghia violacea Harv. on Aortic Pathology, Tissue Antioxidant Enzymes and Liver Damage in Diet-Induced Atherosclerotic Rats. International Journal of Molecular Sciences, 13(10), 12747-12760. https://doi.org/10.3390/ijms131012747




