Frequent Gene Amplification Predicts Poor Prognosis in Gastric Cancer
Abstract
:1. Introduction
2. Results
2.1. Highly Frequent Amplification of ERBB4, C-MET and CD44 in Gastric Cancer
2.2. Association of Amplification of ERBB4, C-MET and CD44 with Clinicopathological Characteristics in Gastric Cancer
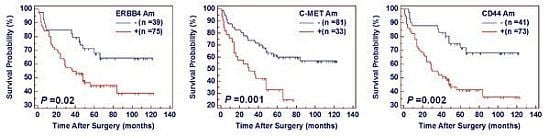
2.3. The Effect of Amplification of ERBB4, C-MET and CD44 on Poor Survival in Gastric Cancer
3. Discussion
4. Experimental Section
4.1. Patients
4.2. Tissues and DNA Preparation
4.3. Copy Number Analysis
4.4. Statistical Analysis
5. Conclusions
Acknowledgments
- Conflicts of InterestThe authors declare no conflict of interest.
References
- Crew, K.D.; Neugut, A.I. Epidemiology of gastric cancer. World J. Gastroenterol 2006, 12, 354–362. [Google Scholar]
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA Cancer J. Clin 2011, 61, 69–90. [Google Scholar]
- Kim, H.J.; Karpeh, M.S. Surgical approaches and outcomes in the treatment of gastric cancer. Semin. Radiat. Oncol 2002, 12, 162–169. [Google Scholar]
- Peng, D.F.; Sugihara, H.; Mukaisho, K.; Tsubosa, Y.; Hattori, T. Alterations of chromosomal copy number during progression of diffuse-type gastric carcinomas: Metaphase- and array-based comparative genomic hybridization analyses of multiple samples from individual tumours. J. Pathol 2003, 201, 439–450. [Google Scholar]
- Buffart, T.E.; van Grieken, N.C.; Tijssen, M.; Coffa, J.; Ylstra, B.; Grabsch, H.I.; van de Velde, C.J.; Carvalho, B.; Meijer, G.A. High resolution analysis of DNA copy-number aberrations of chromosomes 8, 13, and 20 in gastric cancers. Virchows Arch 2009, 455, 213–223. [Google Scholar]
- Zhang, D.; Wang, Z.; Luo, Y.; Xu, Y.; Liu, Y.; Yang, W.; Zhang, X. Analysis of DNA copy number aberrations by multiple ligation-dependent probe amplification on 50 intestinal type gastric cancers. J. Surg. Oncol 2011, 103, 124–132. [Google Scholar]
- Schwab, M. Amplification of oncogenes in human cancer cells. Bioessays 1998, 20, 473–479. [Google Scholar]
- Albertson, D.G. Gene amplification in cancer. Trends Genet 2006, 22, 447–455. [Google Scholar]
- Hirono, Y.; Tsugawa, K.; Fushida, S.; Ninomiya, I.; Yonemura, Y.; Miyazaki, I.; Endou, Y.; Tanaka, M.; Sasaki, T. Amplification of epidermal growth factor receptor gene and its relationship to survival in human gastric cancer. Oncology 1995, 52, 182–188. [Google Scholar]
- Sakakura, C.; Hagiwara, A.; Yasuoka, R.; Fujita, Y.; Nakanishi, M.; Masuda, K.; Kimura, A.; Nakamura, Y.; Inazawa, J.; Abe, T.; et al. Amplification and over-expression of the AIB1 nuclear receptor co-activator gene in primary gastric cancers. Int. J. Cancer 2000, 89, 217–223. [Google Scholar]
- Park, J.H.; Lee, B.L.; Yoon, J.; Kim, J.; Kim, M.A.; Yang, H.K.; Kim, W.H. Focal adhesion kinase (FAK) gene amplification and its clinical implications in gastric cancer. Hum. Pathol 2010, 41, 1664–1673. [Google Scholar]
- Begnami, M.D.; Fukuda, E.; Fregnani, J.H.; Nonogaki, S.; Montagnini, A.L.; da Costa, W.L., Jr; Soares, F.A. Prognostic implications of altered human epidermal growth factor receptors (HERs) in gastric carcinomas: HER2 and HER3 are predictors of poor outcome. J. Clin. Oncol 2011, 29, 3030–3036. [Google Scholar]
- Muraoka-Cook, R.S.; Feng, S.M.; Strunk, K.E.; Earp, H.S., III. ErbB4/HER4: Role in mammary gland development, differentiation and growth inhibition. J. Mammary Gland Biol. Neoplasia 2008, 13, 235–246. [Google Scholar]
- Kamath, S.; Buolamwini, J.K. Targeting EGFR and HER-2 receptor tyrosine kinases for cancer drug discovery and development. Med. Res. Rev 2006, 26, 569–594. [Google Scholar]
- Liu, X.; Yao, W.; Newton, R.C.; Scherle, P.A. Targeting the c-MET signaling pathway for cancer therapy. Expert Opin. Investig. Drugs 2008, 17, 997–1011. [Google Scholar]
- Sierra, J.R.; Tsao, M.S. c-MET as a potential therapeutic target and biomarker in cancer. Ther. Adv. Med. Oncol 2011, 3, S21–S35. [Google Scholar]
- Lee, J.; Seo, J.W.; Jun, H.J.; Ki, C.S.; Park, S.H.; Park, Y.S.; Lim, H.Y.; Choi, M.G.; Bae, J.M.; Sohn, T.S.; et al. Impact of MET amplification on gastric cancer: Possible roles as a novel prognostic marker and a potential therapeutic target. Oncol. Rep 2011, 25, 1517–1524. [Google Scholar]
- Toschi, L.; Cappuzzo, F. Clinical implications of MET gene copy number in lung cancer. Future Oncol 2010, 6, 239–247. [Google Scholar]
- Go, H.; Jeon, Y.K.; Park, H.J.; Sung, S.W.; Seo, J.W.; Chung, D.H. High MET gene copy number leads to shorter survival in patients with non-small cell lung cancer. J. Thorac. Oncol 2010, 5, 305–313. [Google Scholar]
- Park, S.; Choi, Y.L.; Sung, C.O.; An, J.; Seo, J.; Ahn, M.J.; Ahn, J.S.; Park, K.; Shin, Y.K.; Erkin, O.C.; et al. High MET copy number and MET overexpression: Poor outcome in non-small cell lung cancer patients. Histol. Histopathol 2012, 27, 197–207. [Google Scholar]
- Zöller, M. CD44: Can a cancer-initiating cell profit from an abundantly expressed molecule? Nat. Rev. Cancer 2011, 11, 254–267. [Google Scholar]
- Takaishi, S.; Okumura, T.; Tu, S.; Wang, S.S.; Shibata, W.; Vigneshwaran, R.; Gordon, S.A.; Shimada, Y.; Wang, T.C. Identification of gastric cancer stem cells using the cell surface marker CD44. Stem Cells 2009, 27, 1006–1020. [Google Scholar]
- Rossi, E.; Klersy, C.; Manca, R.; Zuffardi, O.; Solcia, E. Correlation between genomic alterations assessed by array comparative genomic hybridization, prognostically informative histologic subtype, stage, and patient survival in gastric cancer. Hum. Pathol 2011, 42, 1937–1945. [Google Scholar]
- Ubukata, H.; Nagata, H.; Tabuchi, T.; Konishi, S.; Kasuga, T.; Tabuchi, T. Why is the coexistence of gastric cancer and duodenal ulcer rare? Examination of factors related to both gastric cancer and duodenal ulcer. Gastric Cancer 2011, 14, 4–12. [Google Scholar]
- Asaoka, Y.; Ikenoue, T.; Koike, K. New targeted therapies for gastric cancer. Expert Opin. Investig. Drugs 2011, 20, 595–604. [Google Scholar]
- Yoong, J.; Michael, M.; Leong, T. Targeted therapies for gastric cancer: Current status. Drugs 2011, 71, 1367–1384. [Google Scholar]
- Smolen, G.A.; Sordella, R.; Muir, B.; Mohapatra, G.; Barmettler, A.; Archibald, H.; Kim, W.J.; Okimoto, R.A.; Bell, D.W.; Sgroi, D.C.; et al. Amplification of MET may identify a subset of cancers with extreme sensitivity to the selective tyrosine kinase inhibitor PHA-665752. Proc. Natl. Acad. Sci. USA 2006, 103, 2316–2321. [Google Scholar]
- Ji, M.; Guan, H.; Gao, C.; Shi, B.; Hou, P. Highly frequent promoter methylation and PIK3CA amplification in non-small cell lung cancer (NSCLC). BMC Cancer 2011, 11. [Google Scholar] [CrossRef]
- Wu, G.; Mambo, E.; Guo, Z.; Hu, S.; Huang, X.; Gollin, S.M.; Trink, B.; Ladenson, P.W.; Sidransky, D.; Xing, M. Uncommon mutation but common amplifications of the PIK3CA gene in thyroid tumors. J. Clin. Endocrinol. Metab 2005, 90, 4688–4693. [Google Scholar]
- Engelman, J.A.; Zejnullahu, K.; Mitsudomi, T.; Song, Y.; Hyland, C.; Park, J.O.; Lindeman, N.; Gale, C.M.; Zhao, X.; Christensen, J.; et al. MET amplification leads to gefitinib resistance in lung cancer by activating ERBB3 signaling. Science 2007, 316, 1039–1043. [Google Scholar]
- Hou, P.; Liu, D.; Shan, Y.; Hu, S.; Studeman, K.; Condouris, S.; Wang, Y.; Trink, A.; El-Naggar, A.K.; Tallini, G.; et al. Genetic alterations and their relationship in the phosphatidylinositol 3-kinase/Akt pathway in thyroid cancer. Clin. Cancer Res 2007, 13, 1161–1170. [Google Scholar]
- Kawano, O.; Sasaki, H.; Okuda, K.; Yukiue, H.; Yokoyama, T.; Yano, M.; Fujii, Y. PIK3CA gene amplification in Japanese non-small cell lung cancer. Lung Cancer 2007, 58, 159–160. [Google Scholar]





| Genes | Male vs. Female | Age 1 | Tumor Localization 2 | Tumor Size 3 | Differentiation 4 |
|---|---|---|---|---|---|
| ERBB4 | 0.97 (0.39–2.39) | 1.12 (0.79–1.54) | 0.82 (0.53–1.29) | 0.88 (0.55–1.40) | 2.62 (1.23–5.59) * |
| C-MET | 0.95 (0.38–2.41) | 1.29 (0.90–1.85) | 0.92 (0.59–1.44) | 1.28 (0.80–2.05) | 1.92 (0.87–4.27) |
| CD44 | 1.06 (0.43–2.61) | 1.15 (0.82–1.63) | 0.80 (0.52–1.25) | 0.88 (0.56–1.39) | 2.28 (1.08–4.79) * |
| Genes | Tumor Invasion 5 | Tumor Stage 6 | Lymph Node Metastasis | No. of LNM 7 | Survival Status 8 |
| ERBB4 | 1.09 (0.65–1.85) | 1.21 (0.81–1.83) | 1.88 (0.89–4.01) | 1.56 (0.99–2.46) | 3.06 (1.41–6.63) ** |
| C-MET | 2.00 (1.03–3.89) * | 1.33 (0.86–2.08) | 1.81 (0.80–4.09) | 1.25 (0.82–1.92) | 3.42 (1.51–7.71) ** |
| CD44 | 1.28 (0.76–2.13) | 1.37 (0.91–2.06) | 2.23 (1.05–4.73) * | 1.70 (1.08–2.69) * | 4.08 (1.86–8.94) ** |
| Genes | Age 1 | Differentiation 2 | Tumor Stage 3 | Lymph Node Metastasis | Survival Status 4 |
|---|---|---|---|---|---|
| ERBB4 | 1.22 (0.81–1.83) | 2.95 (1.27–6.86) * | 0.73 (0.39–1.39) | 1.24 (0.38–3.98) | 3.33 (1.28–8.67) * |
| C-MET | 1.32 (0.88–1.97) | 2.17 (0.92–5.14) | 0.88 (0.45–1.70) | 0.80 (0.23–2.84) | 3.81 (1.35–10.8) * |
| CD44 | 1.19 (0.79–1.78) | 2.49 (1.08–5.79) * | 0.80 (0.43–1.51) | 1.20 (0.38–3.84) | 4.23 (1.62–11.0) ** |
| Covariate | Gene Amplification | P Value | HR * | 95% CI |
|---|---|---|---|---|
| Gender | ERBB4 | 0.04 | 2.00 | 1.02–3.86 |
| Age | ||||
| Differentiation | C-MET | 0.01 | 2.10 | 1.20–3.69 |
| Lymph node metastasis | CD44 | 0.005 | 2.59 | 1.34–5.01 |
| Tumor stage |
| Characteristics | No. of Patients (%) |
|---|---|
| Gender | |
| Male | 101 (78.9) |
| Female | 27 (21.1) |
| Age, years | |
| Mean | 59.42 |
| SD | 13.062 |
| Tumor localization | |
| gastric cardia | 35 (27.3) |
| gastric body | 33 (25.8) |
| gastric antrum | 60 (46.9) |
| Tumor size (cm3) | |
| ≤3 | 43 (33.6) |
| 3–5 | 46 (35.9) |
| >5 | 39 (30.5) |
| Differentiation | |
| well/moderate | 53 (41.4) |
| poor/undifferentiation | 75 (58.6) |
| Tumor invasion | |
| T1 | 14 (10.9) |
| T2 | 22 (17.2) |
| T3 | 90 (70.3) |
| T4 | 2 (1.6) |
| TNM stage | |
| I | 29 (22.7) |
| II | 20 (15.6) |
| III | 73 (57.0) |
| IV | 6 (4.7) |
| Residual tumor | |
| Yes | 14 (10.9) |
| No | 114 (89.1) |
| Lymph node metastasis (LNM) | |
| Yes | 80 (62.5) |
| No | 48 (37.5) |
| No. of LNM | |
| N0 | 48(37.5) |
| N1 (1–6) | 47 (36.7) |
| N2 (7–15) | 27 (21.1) |
| N3 (≥16) | 6 (4.7) |
| Survival status | |
| Dead | 66 (51.6) |
| Alive | 62 (48.4) |
| Genes | Forward Primer Sequence (5′→3′) | Probe Sequence (5′→3′) | Reverse Primer Sequence (5′→3′) | Amplification Efficiency (%) |
|---|---|---|---|---|
| ERBB4 | CCCTGAAGCCAGGCACTGT | 6FAM-CTGCCGCCTCCACCTTACAGACACC-TAMRA | CCTAAAAAACCACAACTGAGCTTACA | 84.2 |
| C-MET | ACCTGCCAGCGACATGTCTT | 6FAM-CCACAATCATACTGCTGACA-TAMRA | GACACTGGCTGGGCTCTTCTATC | 84.1 |
| CD44 | GCTCTGAGCATCGGATTTGAG | 6FAM-CCTGCAGGTAAGAGACCAGCACCCG-TAMRA | AGGCCGCCAGCTTTCC | 85.0 |
| β-Actin | TCACCCACACTGTGCCCATCTACGA | 6FAM-ATGCCCTCCCCCATGCCATCC-TAMRA | TCGGTGAGGATCTTCATGAGGTA | 95.0 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Shi, J.; Yao, D.; Liu, W.; Wang, N.; Lv, H.; He, N.; Shi, B.; Hou, P.; Ji, M. Frequent Gene Amplification Predicts Poor Prognosis in Gastric Cancer. Int. J. Mol. Sci. 2012, 13, 4714-4726. https://doi.org/10.3390/ijms13044714
Shi J, Yao D, Liu W, Wang N, Lv H, He N, Shi B, Hou P, Ji M. Frequent Gene Amplification Predicts Poor Prognosis in Gastric Cancer. International Journal of Molecular Sciences. 2012; 13(4):4714-4726. https://doi.org/10.3390/ijms13044714
Chicago/Turabian StyleShi, Jing, Demao Yao, Wei Liu, Na Wang, Hongjun Lv, Nongyue He, Bingyin Shi, Peng Hou, and Meiju Ji. 2012. "Frequent Gene Amplification Predicts Poor Prognosis in Gastric Cancer" International Journal of Molecular Sciences 13, no. 4: 4714-4726. https://doi.org/10.3390/ijms13044714