Characterization of a κ-Carrageenase from Marine Cellulophaga lytica strain N5-2 and Analysis of Its Degradation Products
Abstract
:1. Introduction
2. Results and Discussion
2.1. Identification of Marine Cellulophaga lytica Strain N5-2
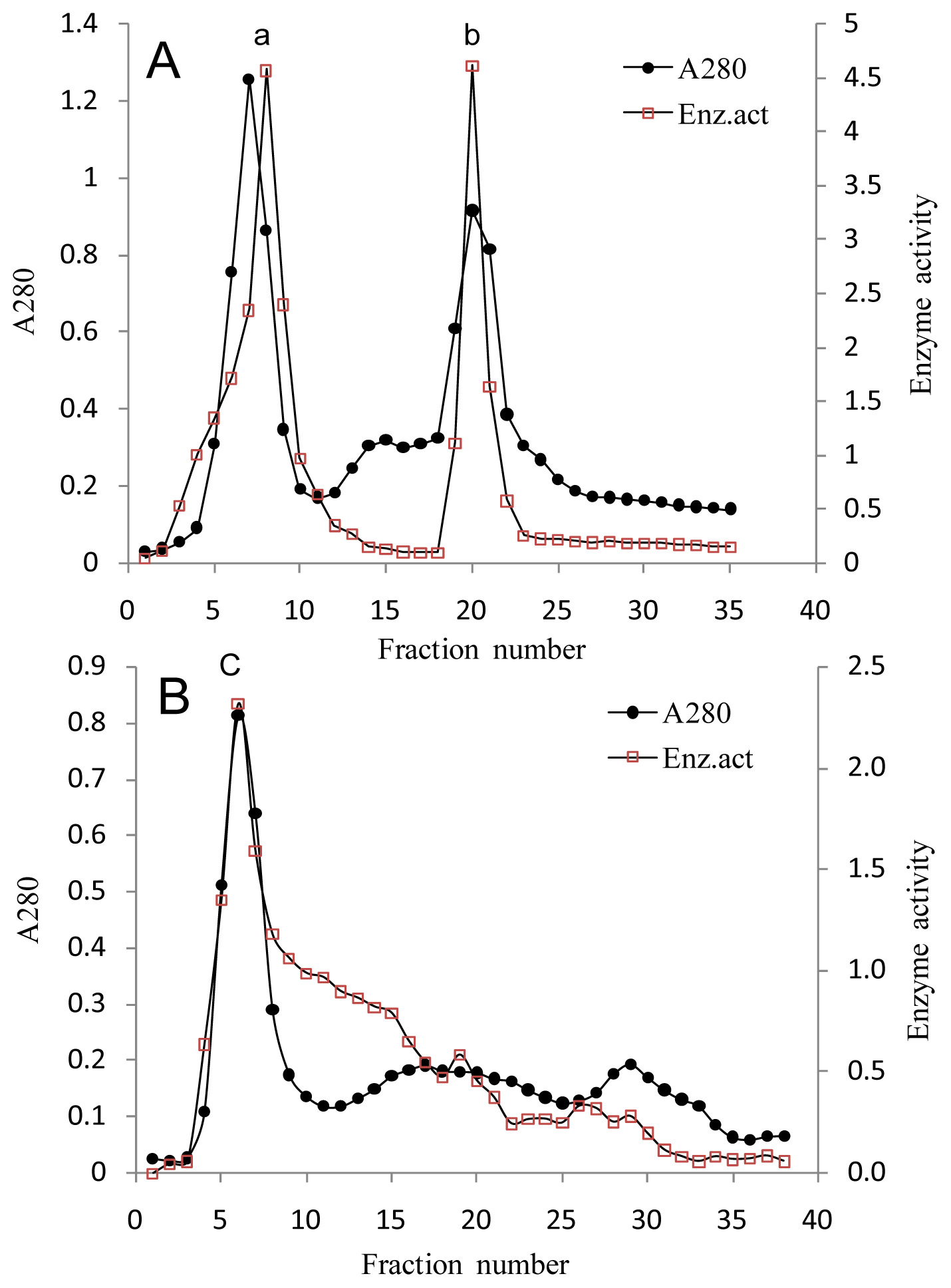
2.2. Purification of κ-Carrageenase
2.3. Properties of the κ-Carragcenase
2.4. Analysis of Degradation Products
3. Materials and Methods
3.1. Microorganism
3.2. Chemicals
3.3. κ-Carrageenase Assay
3.4. Purification of κ-Carrageenase
3.5. Zymography Experiment
3.6. Dependence and Stability of the Enzyme Activity on Temperature and pH
3.7. Kinetic Studies
3.8. Analysis of Degradation Products
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Barbeyron, T.; Gerard, A.; Potin, P. The kappa-carrageenase of the marine bacterium Cytophaga drobachiensis. Structural and phylogenetic relationships within family-16 glycoside hydrolases. Mol. Biol. Evol 1998, 15, 528–537. [Google Scholar]
- Strohmeier, M.; Hrmova, M.; Fischer, M.; Harvey, A.J.; Fincher, G.B.; Pleiss, J. Molecular modeling of family GH16 glycoside hydrolases: Potential roles for xyloglucan transglucosylases/hydrolases in cell wall modification in the poaceae. Protein Sci 2004, 13, 3200–3213. [Google Scholar]
- Zablackis, E.; Vreeland, V.; Kloareg, B. Isolation of protoplasts from kappaphycus alvarezii var. tambalang (Rhodophyta) and secretion of carrageenan fragments by cultured cells. J. Exp. Bot 1993, 44, 1515–1522. [Google Scholar]
- Marion, G.; Patrick, B. Composition and distribution of carrabiose moieties in hybrid κ-/ι-carrageenans using carrageenases. Biomacromolecules 2008, 9, 408–415. [Google Scholar]
- Lemonnier-Le, P.C.; Chatelet, C.; Kloareg, B. Carrageenan oligosaccharides enhance stress-induced microspore embryogenesis in Brassica oleracea var. italica. Plant Sci 2001, 160, 1211–1220. [Google Scholar]
- Nyval, P.; Maud, L.; Richard, D. Enzymatic degradation of κ-carrageenan in aqueous solution. Biomacromolecules 2009, 10, 1757–1767. [Google Scholar]
- Yuan, H.M.; Song, L.M.; Li, X.G. Immunomoduladtion and antitumor activity of κ-carrageenan oligosaccharides. Cancer Lett 2006, 243, 228–234. [Google Scholar]
- Yuan, H.M.; Zhang, W.W.; Li, X.G. Preparation and in vitro antioxidant activity of κ-carrageenan oligosaccharides and their over sulfated, acetylated and phosphorylated derivatives. Carbohydr. Res 2005, 340, 685–692. [Google Scholar]
- Keisuke, I.; Michal, K.W.; Takayuki, T.; Siro, S.; Hiroyuki, O. Novel heparin sulfatemimetic compounds as antitumor agents. Chem. Biol 2004, 11, 367–377. [Google Scholar]
- Gauthier, G.; Gauthier, M.; Christen, R. Phylogenetic analysis of the genera Alteromonas, Shewanella, and Moritella using genes coding for small-subunit rRNA sequences and division of the genus Alteromonas into two genera, Alteromonas (emended) and Pseudoalteromonas gen. nov., and proposal of twelve new species combinations. Int. J. Syst. Bacteriol 1995, 45, 755–761. [Google Scholar]
- Golam, S.; Seiken, M.; Hiroshi, O. Purification of a κ-carrageenase from marine Cytophaga species. Microbiol. Immunol 1987, 31, 869–877. [Google Scholar]
- Khambhty, Y.; Mody, K.; Jha, B. Statistical optimization of medium components for κ-carrageenase production by Pseudomonas elongata. Enzyme Microbial Technol 2007, 40, 813–822. [Google Scholar]
- Michel, G.; Chantalat, L.; Duee, E.; Barbeyron, T.; Henrissat, B.; Kloareg, B.; Dideberg, O. The κ-carrageenase of P carrageenovora features a tunnel-shaped active site: A novel insight in the evolution of Clan-B glycoside hydrolases. Structure 2001, 9, 513–525. [Google Scholar]
- Dun, J.X.; Yao, Z.A.; Wu, H.G.; Wang, F.F.; Zhu, N.N.; Du, Y.G. Screening and characterization of a κ-carrageenase producing strain and the initial analysis of products. Food Sci. Technol 2010, 35, 11–16. (in Chinese). [Google Scholar]
- Kidby, D.K.; Davidson, D.J. A convenient ferricyanide estimation of reducing sugars in the nanomole range. Anal. Biochem 1973, 55, 321–325. [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem 1976, 72, 248–254. [Google Scholar]
- Lineweaver, H.; Bruk, D. The determination of enzyme dissociation constants. Chem. Soc. Rev 1934, 56, 658–666. [Google Scholar]








| Step | Volume (mL) | Total protein a (mg) | Total activity b (units) | Specific activity (units/mg) | Folds | Recovery (%) |
|---|---|---|---|---|---|---|
| Cell-free medium | 1000 | 56.5 | 1648 | 29 | 1 | 100 |
| 40% and 80% (NH4)2SO4 precipitate | 80 | 11 | 1145 | 104 | 4 | 69.5 |
| SephadexG-200 gel-filtration | 5 | 0.86 | 580 | 675 | 23 | 35.2 |
| SephadexG-75 gel-filtration | 2 | 0.235 | 285 | 1170 | 40 | 17.3 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yao, Z.; Wang, F.; Gao, Z.; Jin, L.; Wu, H. Characterization of a κ-Carrageenase from Marine Cellulophaga lytica strain N5-2 and Analysis of Its Degradation Products. Int. J. Mol. Sci. 2013, 14, 24592-24602. https://doi.org/10.3390/ijms141224592
Yao Z, Wang F, Gao Z, Jin L, Wu H. Characterization of a κ-Carrageenase from Marine Cellulophaga lytica strain N5-2 and Analysis of Its Degradation Products. International Journal of Molecular Sciences. 2013; 14(12):24592-24602. https://doi.org/10.3390/ijms141224592
Chicago/Turabian StyleYao, Ziang, Feifei Wang, Zheng Gao, Liming Jin, and Haige Wu. 2013. "Characterization of a κ-Carrageenase from Marine Cellulophaga lytica strain N5-2 and Analysis of Its Degradation Products" International Journal of Molecular Sciences 14, no. 12: 24592-24602. https://doi.org/10.3390/ijms141224592
APA StyleYao, Z., Wang, F., Gao, Z., Jin, L., & Wu, H. (2013). Characterization of a κ-Carrageenase from Marine Cellulophaga lytica strain N5-2 and Analysis of Its Degradation Products. International Journal of Molecular Sciences, 14(12), 24592-24602. https://doi.org/10.3390/ijms141224592






