Diurnal Profiles of Melatonin Synthesis-Related Indoles, Catecholamines and Their Metabolites in the Duck Pineal Organ
Abstract
:1. Introduction

2. Results and Discussion
2.1. Characteristics of Chromatographic Assays

| Peak No. | Compound | Intra-Day Precision | Inter-Day Precision | ||
|---|---|---|---|---|---|
| Retention Time MIN–MAX | Peak Area RSD (%) | Retention Time MIN–MAX | Peak Area RSD (%) | ||
| 1 | 5-HTRP | 3.453–3.455 | 1.8 | 3.451–3.470 | 2.2 |
| 2 | 5-HT | 4.240–4.244 | 1.9 | 4.240–4.248 | 2.5 |
| 3 | TRP | 6.295–6.297 | 2.2 | 2.293–6.306 | 2.8 |
| 4 | 5-HIAA | 7.654–7.656 | 2.4 | 7.650–7.770 | 3.2 |
| 5 | 5-HTOL | 9.120–9.145 | 2.7 | 9.100–9.157 | 3.7 |
| 6 | NAS | 14.200–14.203 | 2.4 | 14.192–14.205 | 2.6 |
| 7 | 5-MTAM | 15.200–15.208 | 2.1 | 15.197–15.230 | 3.2 |
| 8 | 5-MIAA | 21.270–21.314 | 2.4 | 21.240–21.327 | 3.5 |
| 9 | 5-MTOL | 23.057–23.067 | 2.8 | 23.054–23.087 | 3.4 |
| 10 | MLT | 24.604–24.607 | 2.5 | 24.600–24.630 | 3.7 |
| Peak No. | Compound | Parameters | Indole Added (pg on Column) | |||
|---|---|---|---|---|---|---|
| 25 | 50 | 250 | 500 | |||
| 1 | 5-HTRP | Mean recovery (%) | 102.3 | 99.2 | 100.2 | 101.2 |
| RSD (%) | 3.2 | 2.2 | 1.8 | 2.0 | ||
| 2 | 5-HT | Mean recovery (%) | 99.8 | 101.2 | 102.3 | 99.2 |
| RSD (%) | 2.8 | 3.2 | 2.8 | 2.9 | ||
| 3 | TRP | Mean recovery (%) | 102.2 | 99.8 | 100.2 | 99.8 |
| RSD (%) | 3.4 | 2.5 | 2.2 | 3.2 | ||
| 4 | 5-HIAA | Mean recovery (%) | 98.8 | 100.6 | 100.5 | 99.8 |
| RSD (%) | 2.9 | 3.2 | 1.9 | 2.8 | ||
| 5 | 5-HTOL | Mean recovery (%) | 102.4 | 100.1 | 100.2 | 98.7 |
| RSD (%) | 1.7 | 2.2 | 1.9 | 2.4 | ||
| 6 | NAS | Mean recovery (%) | 99.8 | 101.7 | 98.8 | 102.3 |
| RSD (%) | 2.1 | 1.2 | 1.2 | 2.4 | ||
| 7 | 5-MTAM | Mean recovery (%) | 99.7 | 100.1 | 101.4 | 102.4 |
| RSD (%) | 1.7 | 2.4 | 2.7 | 2.8 | ||
| 8 | 5-MIAA | Mean recovery (%) | 102.1 | 101.2 | 102.3 | 99.8 |
| RSD (%) | 2.7 | 2.7 | 2.6 | 2.4 | ||
| 9 | 5-MTOL | Mean recovery (%) | 101.7 | 98.5 | 99.5 | 97.5 |
| RSD (%) | 1.9 | 2.3 | 2.4 | 3.2 | ||
| 10 | MLT | Mean recovery (%) | 103.2 | 100.1 | 1002 | 99.5 |
| RSD (%) | 2.6 | 2.1 | 1.9 | 2.4 | ||
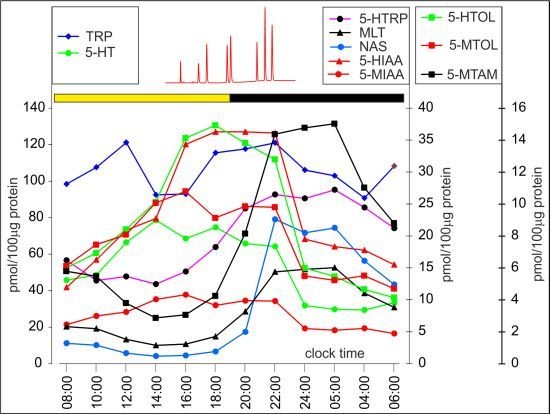
2.2. Pineal Indoles—Diurnal Rhythms and Quantitative Relations
2.2.1. Serotonin and Its Precursors



2.2.2. Direct Derivatives of Serotonin

2.2.3. Melatonin and Other 5-Methoxyindoles


2.3. Catecholamines and Their Metabolites

3. Experimental Section
3.1. Animal Experimental Procedures
3.2. Analytical Procedures
3.2.1. Chemicals
3.2.2. Sample Preparation for Indole and Catecholamine Assays
3.2.3. Indole Assay
3.2.4. Validation of the Indole Assay
3.2.5. Assay of Catecholamines and Their Metabolites
3.2.6. Protein Assay
3.3. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Reiter, R.J. Pineal melatonin: Cell biology of its synthesis and of its physiological interactions. Endocr. Rev. 1991, 12, 151–180. [Google Scholar]
- Simonneaux, V.; Ribelayga, C. Generation of the melatonin endocrine message in mammals: A review of the complex regulations of melatonin synthesis by norepinephrine, peptides and other pineal transmitters. Pharmacol. Rev. 2003, 55, 325–395. [Google Scholar]
- Hardeland, R. Melatonin metabolism in the central nervous system. Curr. Neuropharmacol. 2010, 8, 168–181. [Google Scholar]
- Reiter, R.J. The melatonin rhythm: Both a clock and a calendar. Experientia 1993, 49, 654–664. [Google Scholar]
- Hardeland, R. Melatonin, hormone of darkness and more: Occurrence, control mechanisms, actions and bioactive metabolites. Cell Mol. Life Sci. 2008, 65, 2001–2018. [Google Scholar]
- Falcón, J.; Besseau, L.; Fuentès, M.; Sauzet, S.; Magnanou, E.; Boeuf, G. Structural and functional evolution of the pineal melatonin system in vertebrates. Ann. N. Y. Acad. Sci. 2009, 1163, 101–111. [Google Scholar]
- Klein, D.C. Arylalkylamine N-acetyltransferase: “The Timezyme”. J. Biol. Chem. 2007, 282, 4233–4237. [Google Scholar]
- Maronde, E.; Stehle, J.H. The mammalian pineal gland: Known facts, unknown facets. Trends Endocrinol. Metab. 2007, 18, 142–149. [Google Scholar]
- Stehle, J.H.; von Gall, C.; Schomerus, C.; Korf, H.W. Of rodents and ungulates and melatonin: Creating a uniform code for darkness by different signaling mechanisms. J. Biol. Rhythms. 2001, 16, 312–325. [Google Scholar]
- Lewczuk, B.; Zheng, W.; Prusik, M.; Cole, P.A.; Przybylska-Gornowicz, B. N-bromoacetyltryptamine strongly and reversibly inhibits in vitro melatonin secretion from mammalian pinealocytes. Neuro Endocrinol. Lett. 2005, 26, 581–592. [Google Scholar]
- Okano, T.; Fukada, Y. Photoreception and circadian clock system of the chicken pineal gland. Microsc. Res. Tech. 2001, 53, 72–80. [Google Scholar]
- Holthues, H.; Engel, L.; Spessert, R.; Vollrath, L. Circadian gene expression patterns of melanopsin and pinopsin in the chick pineal gland. Biochem. Biophys. Res. Commun. 2005, 326, 160–165. [Google Scholar]
- Fukada, Y.; Okano, T. Circadian clock system in the pineal gland. Mol. Neurobiol. 2002, 25, 19–30. [Google Scholar]
- Zawilska, J.B.; Berezińska, M.; Rosiak, J.; Skene, D.J.; Vivien-Roels, B.; Nowak, J.Z. Suppression of melatonin biosynthesis in the chicken pineal gland by retinally perceived light-involvement of D1-dopamine receptors. J. Pineal Res. 2004, 36, 80–86. [Google Scholar]
- Cantwell, E.L.; Cassone, V.M. Chicken suprachiasmatic nuclei: I. Efferent and afferent connections. J. Comp. Neurol. 2006, 496, 97–120. [Google Scholar]
- Cassone, V.M.; Forsyth, A.M.; Woodlee, G.L. Hypothalamic regulation of circadian noradrenergic input to the chick pineal gland. J. Comp. Physiol. A 1990, 167, 187–192. [Google Scholar]
- Pratt, B.L.; Takahashi, J.S. Alpha-2 adrenergic regulation of melatonin release in chick pineal cell cultures. J. Neurosci. 1987, 7, 3665–3674. [Google Scholar]
- Murakami, N.; Nakamura, H.; Nishi, R.; Marumoto, N.; Nasu, T. Comparison of circadian oscillation of melatonin release in pineal cells of house sparrow, pigeon and Japanese quail, using cell perfusion system. Brain Res. 1994, 651, 209–214. [Google Scholar]
- Natesan, A.; Geetha, L.; Zatz, M. Rhythm and soul in the avian pineal. Cell Tissue Res. 2002, 309, 35–45. [Google Scholar]
- Barrett, R.K.; Underwood, H. The superior cervical ganglia are not necessary for entrainment or persistence of the pineal melatonin rhythm in Japanese quail. Brain Res. 1992, 569, 249–254. [Google Scholar]
- Harumi, T.; Matsushima, S. Separation and assay methods for melatonin and its precursors. J. Chromatogr. B: Biomed. Sci. Appl. 2000, 747, 95–110. [Google Scholar]
- Raynaud, F.; Pévet, P. Determination of 5-methoxyindoles in pineal gland and plasma samples by high-performance liquid chromatography with electrochemical detection. J. Chromatogr. 1991, 564, 103–113. [Google Scholar]
- Moujir, F.; Santana, C.; Hernández, F.J.; Reiter, R.J.; Abreu, P. Daily time course of the contents in monoamines and their metabolites in the pineal gland of Syrian hamster. Neurosci. Lett. 1997, 223, 77–80. [Google Scholar]
- Miguez, J.M.; Recio, J.; Sanchez-Barcelo, E.; Aldegunde, M. Changes with age in daytime and nighttime contents of melatonin, indoleamines, and catecholamines in the pineal gland: A comparative study in rat and Syrian hamster. J. Pineal Res. 1998, 25, 106–115. [Google Scholar]
- Ceinos, R.M.; Rábade, S.; Soengas, J.L.; Míguez, J.M. Indoleamines and 5-methoxyindoles in trout pineal organ in vivo: Daily changes and influence of photoperiod. Gen. Comp. Endocrinol. 2005, 144, 67–77. [Google Scholar]
- López-Patiño, M.A.; Rodríguez-Illamola, A.; Gesto, M.; Soengas, J.L.; Míguez, J.M. Changes in plasma melatonin levels and pineal organ melatonin synthesis following acclimation of rainbow trout (Oncorhynchus mykiss) to different water salinities. J. Exp. Biol. 2011, 214, 928–936. [Google Scholar] [CrossRef]
- Chin, J.R. Determination of six indolic compounds, including melatonin, in rat pineal using high-performance liquid chromatography with serial fluorometric-electrochemical detection. J. Chromatogr. B: Biomed. Sci. Appl. 1990, 528, 111–121. [Google Scholar] [CrossRef]
- Anderson, G.M.; Young, J.G.; Cohen, D.J.; Young, S.N. Determination of indoles in human and rat pineal. J. Chromatogr. 1982, 228, 155–163. [Google Scholar] [CrossRef]
- Muñoz, J.L.; Ceinos, R.M.; Soengas, J.L.; Míguez, J.M. A simple and sensitive method for determination of melatonin in plasma, bile and intestinal tissues by high performance liquid chromatography with fluorescence detection. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2009, 877, 2173–2177. [Google Scholar]
- Hamase, K.; Hirano, J.; Kosai, Y.; Tomita, T.; Zaitsu, K. A sensitive internal standard method for the determination of melatonin in mammals using precolumn oxidation reversed-phase high-performance liquid chromatography. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2004, 811, 237–241. [Google Scholar]
- Todoroki, K.; Ishimaru, K.; Yoshida, H.; Yoshitake, T.; Nohta, H.; Yamaguchi, M. Simultaneous liquid chromatographic measurement of melatonin and related indoles through post-column electrochemical demethylation and fluorescence derivatization. Anal. Sci. 2006, 22, 281–286. [Google Scholar] [CrossRef]
- Zawilska, J.B.; Vivien-Roels, B.; Skene, D.J.; Pévet, P.; Nowak, J.Z. Phase-shifting effects of light on the circadian rhythms of 5-methoxytryptophol and melatonin in the chick pineal gland. J. Pineal Res. 2000, 29, 1–7. [Google Scholar]
- Zawilska, J.B.; Rosiak, J.; Vivien-Roels, B.; Skene, D.J.; Pevet, P.; Nowak, J.Z. Daily variation in the concentration of 5-methoxytryptophol and melatonin in the duck pineal gland and plasma. J. Pineal Res. 2002, 32, 214–218. [Google Scholar] [CrossRef]
- Zawilska, J.B.; Berezinska, M.; Rosiak, J.; Vivien-Roels, B.; Skene, D.J.; Pevet, P.; Nowak, J.Z. Daily variation in the concentration of melatonin and 5-methoxytryptophol in the goose pineal gland, retina, and plasma. Gen. Comp. Endocrinol. 2003, 134, 296–302. [Google Scholar] [CrossRef]
- Grady, R.K., Jr.; Caliguri, A. Day/night differences in pineal indoles in the adult pigeon (Columba livia). Comp. Biochem. Physiol. C 1984, 78, 141–143. [Google Scholar] [CrossRef]
- Cassone, V.M.; Takahashi, J.S.; Blaha, C.D.; Lane, R.F.; Menaker, M. Dynamics of noradrenergic circadian input to the chicken pineal gland. Brain Res. 1986, 384, 334–341. [Google Scholar] [CrossRef]
- Siuciak, J.A.; Gamache, P.H.; Dubocovich, M.L. Monoamines and their precursors and metabolites in the chicken brain, pineal, and retina: Regional distribution and day/night variation. J. Neurochem. 1992, 58, 722–729. [Google Scholar]
- Vaarmann, A.; Kask, A.; Mäeorg, U. Novel and sensitive high-performance liquid chromatographic method based on electrochemical coulometric array detection for simultaneous determination of catecholamines, kynurenine and indole derivatives of tryptophan. J. Chromatogr.B Anal. Technol. Biomed. Life Sci. 2002, 769, 145–153. [Google Scholar] [CrossRef]
- Piesiewicz, A.; Podobas, E.; Kędzierska, U.; Joachimiak, E.; Markowska, M.; Majewski, P.; Skwarlo-Sonta, K. Season-related differences in the biosynthetic activity of the neonatal chicken pineal gland. Open Ornithol. J. 2010, 3, 134–140. [Google Scholar] [CrossRef]
- Guchhait, P.; Haldar, C. A reproductive phase-dependent effect of dietary l-tryptophan on pineal gland and gonad of a nocturnal bird, Indian spotted owlet Athene brama. Acta Biol. Hung. 2001, 52, 1–7. [Google Scholar] [CrossRef]
- Sugden, D.; Namboodiri, M.A.; Klein, D.C.; Grady, R.K., Jr; Mefford, I.N. Ovine pineal indoles: Effects of l-tryptophan or l-5-hydroxytryptophan administration. J. Neurochem. 1985, 44, 769–772. [Google Scholar] [CrossRef]
- Reiter, R.J.; King, T.S.; Steinlechner, S.; Steger, R.W.; Richardson, B.A. Tryptophan administration inhibits nocturnal N-acetyltransferase activity and melatonin content in the rat pineal gland. Evidence that serotonin modulates melatonin production via a receptor-mediated mechanism. Neuroendocrinology 1990, 52, 291–296. [Google Scholar] [CrossRef]
- Mateos, S.S.; Sánchez, C.L.; Paredes, S.D.; Barriga, C.; Rodríguez, A.B. Circadian levels of serotonin in plasma and brain after oral administration of tryptophan in rats. Basic Clin. Pharmacol.Toxicol. 2009, 104, 52–59. [Google Scholar] [CrossRef]
- Miguez, J.M.; Recio, J.; Vivien-Roels, B.; Pévet, P. Daily variation in the content of indoleamines, catecholamines and related compounds in the pineal gland of Syrian hamsters kept under long and short photoperiods. J. Pineal Res. 1995, 19, 139–148. [Google Scholar] [CrossRef]
- Míguez, J.M.; Recio, J.; Vivien-Roels, B.; Pévet, P. Diurnal changes in the content of indoleamines, catecholamines, and methoxyindoles in the pineal gland of the Djungarian hamster (Phodopus sungorus): Effect of photoperiod. J. Pineal Res. 1996, 21, 7–14. [Google Scholar] [CrossRef]
- Steinlechner, S.; Baumgartner, I.; Klante, G.; Reiter, R.J. Melatonin synthesis in the retina and pineal gland of Djungarian hamsters at different times of the year. Neurochem. Int. 1995, 27, 245–251. [Google Scholar] [CrossRef]
- Green, C.B.; Besharse, J.C.; Zatz, M. Tryptophan hydroxylase mRNA levels are regulated by the circadian clock, temperature, and cAMP in chick pineal cells. Brain Res. 1996, 738, 1–7. [Google Scholar] [CrossRef]
- Florez, J.C.; Takahashi, J.S. Quantitative two-dimensional gel electrophoretic analysis of clock-controlled proteins in cultured chick pineal cells: Circadian regulation of tryptophan hydroxylase. J. Biol. Rhythm. 1996, 11, 241–257. [Google Scholar] [CrossRef]
- Florez, J.C.; Takahashi, J.S. Regulation of tryptophan hydroxylase by cyclic AMP, calcium, norepinephrine, and light in cultured chick pineal cells. J. Neurochem. 1996, 67, 242–250. [Google Scholar]
- Hedlund, L.; Ralph, C.L.; Chepko, J.; Lynch, H.J. A diurnal serotonin cycle in the pineal body of Japanese quail: Photoperiod phasing and the effect of superior cervical ganglionectomy. Gen. Comp. Endocrinol. 1971, 16, 52–58. [Google Scholar] [CrossRef]
- Majewski, P.M.; Adamska, I.; Pawlak, J.; Barańska, A.; Skwarło-Sońta, K. Seasonality of pineal gland activity and immune functions in chickens. J. Pineal Res. 2005, 39, 66–72. [Google Scholar] [CrossRef]
- Piesiewicz, A.; Kędzierska, U.; Podobas, E.; Adamska, I.; Zuzewicz, K.; Majewski, P.M. Season-dependent postembryonic maturation of the diurnal rhythm of melatonin biosynthesis in the chicken pineal gland. Chronobiol. Int. 2012, 29, 1227–1238. [Google Scholar] [CrossRef]
- Mefford, I.N.; Chang, P.; Klein, D.C.; Namboodiri, M.A.; Sugden, D.; Barchas, J. Reciprocal day/night relationship between serotonin oxidation and N-acetylation products in the rat pineal gland. Endocrinology 1983, 113, 1582–1586. [Google Scholar] [CrossRef]
- Chattoraj, A.; Liu, T.; Zhang, L.S.; Huang, Z.; Borjigin, J. Melatonin formation in mammals: In vivo perspectives. Rev. Endocr. Metab. Disord. 2009, 10, 237–243. [Google Scholar]
- Liu, T.; Borjigin, J. Relationship between nocturnal serotonin surge and melatonin onset in rodent pineal gland. J. Circadian Rhythm. 2006, 4, 12. [Google Scholar] [CrossRef]
- Harumi, T.; Matsushima, S. Indoleamine metabolism in the pineal gland of the Chinese hamster, Cricetulus griseus. Gen. Comp. Endocrinol. 1998, 109, 133–139. [Google Scholar] [CrossRef]
- Johnson, R.D.; Lewis, R.J.; Canfield, D.V.; Dubowski, K.M.; Blank, C.L. Utilizing the urinary 5-HTOL/5-HIAA ratio to determine ethanol origin in civil aviation accident victims. J. Forensic Sci. 2005, 50, 670–675. [Google Scholar]
- Ceinos, R.M.; Polakof, S.; Illamola, A.R.; Soengas, J.L.; Míguez, J.M. Food deprivation and refeeding effects on pineal indoles metabolism and melatonin synthesis in the rainbow trout Oncorhynchus mykiss. Gen. Comp. Endocrinol. 2008, 156, 410–417. [Google Scholar] [CrossRef]
- McNulty, J.A.; Prechel, M.M.; Simmons, W.H. Correlations of serotonin and its metabolites in individual rat pineal glands over light: Dark cycles and after acute light exposure. Life Sci. 1986, 39, 1–6. [Google Scholar] [CrossRef]
- Young, S.N.; Anderson, G.M. Factors influencing melatonin, 5-hydroxytryptophol, 5-hydroxyindoleacetic acid, 5-hydroxytryptamine and tryptophan in rat pineal glands. Neuroendocrinology 1982, 35, 464–468. [Google Scholar] [CrossRef]
- Liu, T.; Borjigin, J. N-acetyltransferase is not the rate limiting enzyme of melatonin synthesis at night. J. Pineal Res. 2005, 39, 91–96. [Google Scholar] [CrossRef]
- Hofman, M.A.; Skene, D.J.; Swaab, D.F. Effect of photoperiod on the diurnal melatonin and 5-methoxytryptophol rhythms in the human pineal gland. Brain Res. 1995, 671, 254–260. [Google Scholar] [CrossRef]
- Lakhdar-Ghazal, N.; Vivien-Roels, B.; Pevet, P. Seasonal variations in pineal 5-methoxytryptophol (5-ML) concentrations and in the daily pattern of pineal 5-ML and melatonin in the desert rodent Jaculus orientalis: Effect of prolonged illumination during the night. J. Pineal Res. 1992, 13, 28–35. [Google Scholar] [CrossRef]
- Tsang, C.W.; Leung, O.K.; Lee, P.P.; Pang, S.F. Mass spectrometric identification and quantification of endogenous 5-methoxyindole-3-acetic acid in quail tissues. Biochem. Biophys. Res. Commun. 1995, 209, 1132–1139. [Google Scholar] [CrossRef]
- Balemans, M.G.; Ebels, I.; Hendriks, H.G.; van Berlo, M.F. Changes in the circadian rhythmicity of hydroxyindole-O-methyl transferase (HIOMT) activity in the synthesis of 5-methoxyindoles in the pineal gland of 28 day old male Wistar rats exposed to white, red and green light. J. Neural Transm. 1982, 53, 293–303. [Google Scholar] [CrossRef]
- Beck, O.; Pévet, P. Analysis of melatonin, 5-methoxytryptophol and 5-methoxyindoleacetic acid in the pineal gland and retina of hamster by capillary column gas chromatography-mass spectrometry. J. Chromatogr. 1984, 311, 1–8. [Google Scholar] [CrossRef]
- Grace, M.S.; Cahill, G.M.; Besharse, J.C. Melatonin deacetylation: Retinal vertebrate class distribution and Xenopus laevis tissue distribution. Brain Res. 1991, 559, 56–63. [Google Scholar] [CrossRef]
- Beck, O.; Jonsson, G. In vivo formation of 5-methoxytryptamine from melatonin in rat. J. Neurochem. 1981, 36, 2013–2018. [Google Scholar] [CrossRef]
- Galzin, A.M.; Eon, M.T.; Esnaud, H.; Lee, C.R.; Pévet, P.; Langer, S.Z. Day-night rhythm of 5-methoxytryptamine biosynthesis in the pineal gland of the golden hamster (Mesocricetus auratus). J. Endocrinol. 1988, 118, 389–397. [Google Scholar] [CrossRef]
- Li, P.; Pang, S.F.; Chan, C.L.; Tsang, C.W. Identification and diurnal studies of pineal and serum 5-methoxytryptamine in the rat and quail. Neurosci. Lett. 1997, 228, 63–65. [Google Scholar] [CrossRef]
- Yáñez, J.; Meissl, H. Secretion of the methoxyindoles melatonin, 5-methoxytryptophol, 5-methoxyindoleacetic acid, and 5-methoxytryptamine from trout pineal organs in superfusion culture: Effects of light intensity. Gen. Comp. Endocrinol. 1996, 101, 165–172. [Google Scholar] [CrossRef]
- Zawilska, J.B.; Berezińska, M.; Stasikowska, O.; Lorenc, A.; Skene, D.J.; Nowak, J.Z. Posthatching developmental changes in noradrenaline content in the chicken pineal gland. J. Pineal Res. 2005, 38, 123–129. [Google Scholar] [CrossRef]
- Sato, T. Sensory and endocrine characteristics of the avian pineal organ. Microsc. Res. Tech. 2001, 53, 2–11. [Google Scholar] [CrossRef]
- Drijfhout, W.J.; van der Linde, A.G.; Kooi, S.E.; Grol, C.J.; Westerink, B.H. Norepinephrine release in the rat pineal gland: The input from the biological clock measured by in vivo microdialysis. J. Neurochem. 1996, 66, 748–755. [Google Scholar]
- Kaleczyc, J.; Przybylska, B.; Majewski, M.; Lewczuk, B. Immunohistochemical studies on the coexistence of catecholamine-synthesizing enzymes and neuropeptide Y in nerve fibers of the porcine pineal gland. J. Pineal Res. 1994, 17, 20–24. [Google Scholar] [CrossRef]
- Møller, M.; Baeres, F.M. The anatomy and innervation of the mammalian pineal gland. Cell Tissue Res. 2002, 309, 139–150. [Google Scholar] [CrossRef]
- Nowicki, M.; Wojtkiewicz, J.; Seremak, B.; Sulik, M.; Ostaszewski, J.; Lewczuk, B.; Majewski, M.; Przybylska-Gornowicz, B. Specific distribution pattern of nerve fibers containing catecholamine-synthesizing enzymes, neuropeptide Y (NPY) and C-terminal flanking peptide of NPY (CPON) in the pineal gland of the chinchilla (Chinchilla laniger)—An immunohistochemical study. Folia Histochem. Cytobiol. 2003, 41, 193–200. [Google Scholar]
- Acuña-Castroviejo, D.; Escames, G.; Venegas, C.; Díaz-Casado, M.E.; Lima-Cabello, E.; López, L.C.; Rosales-Corral, S.; Tan, D.X.; Reiter, R.J. Extrapineal melatonin: Sources, regulation, and potential functions. Cell. Mol. Life Sci. 2014, 2014. [Google Scholar] [CrossRef]
- Slominski, A.; Pisarchik, A.; Semak, I.; Sweatman, T.; Wortsman, J. Characterization of the serotoninergic system in the C57BL/6 mouse skin. Eur. J. Biochem. 2003, 270, 3335–3344. [Google Scholar] [CrossRef]
- Slominski, A.; Wortsman, J.; Tobin, D.J. The cutaneous serotoninergic/melatoninergic system: Securing a place under the sun. FASEB J. 2005, 19, 176–194. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lewczuk, B.; Ziółkowska, N.; Prusik, M.; Przybylska-Gornowicz, B. Diurnal Profiles of Melatonin Synthesis-Related Indoles, Catecholamines and Their Metabolites in the Duck Pineal Organ. Int. J. Mol. Sci. 2014, 15, 12604-12630. https://doi.org/10.3390/ijms150712604
Lewczuk B, Ziółkowska N, Prusik M, Przybylska-Gornowicz B. Diurnal Profiles of Melatonin Synthesis-Related Indoles, Catecholamines and Their Metabolites in the Duck Pineal Organ. International Journal of Molecular Sciences. 2014; 15(7):12604-12630. https://doi.org/10.3390/ijms150712604
Chicago/Turabian StyleLewczuk, Bogdan, Natalia Ziółkowska, Magdalena Prusik, and Barbara Przybylska-Gornowicz. 2014. "Diurnal Profiles of Melatonin Synthesis-Related Indoles, Catecholamines and Their Metabolites in the Duck Pineal Organ" International Journal of Molecular Sciences 15, no. 7: 12604-12630. https://doi.org/10.3390/ijms150712604