1. Introduction
Filamentous fungi in the genus
Aspergillus are ubiquitous opportunistic pathogens, which are most notable as causative agents of highly enervating human diseases such as aspergillosis [
1]. They form extremely invasive human infections, particularly in immunocompromised patients or in people suffering chronic granulomatosis [
2,
3,
4].
Aspergillus flavus and
Aspergillus parasiticus also produce highly (hepato)carcinogenic aflatoxins, which contaminate various agricultural/food commodities [
5]. Filamentous fungi in the genus
Penicillium also frequently cause food contamination or postharvest decay, where
P. expansum is the main producer of the mycotoxin patulin that negatively affects human and animal health [
6].
Mycotic diseases/infections are becoming a serious problem since effective antifungal drugs or fungicides, especially agents for treating drug/fungicide-resistant fungi, are very limited. Development of fungal resistance to conventional antimycotic agents not only triggers global public health issues, but also threatens the safety of food supplies, especially for the products susceptible to mycotoxin contamination [
7,
8]. For instance, continuous applications of widely used fungicides, such as strobilurins, fludioxonils,
etc., to agricultural fields resulted in the development of fungal resistance to (and thus, escape from) the toxicities of fungicides. Moreover, applying fungicides at suboptimal concentrations or time-points of fungal growth can potentiate toxin production by mycotoxigenic fungi [
9,
10]. Fungicide-potentiation of mycotoxin production in fungi, especially those resistant to fungicides, has been reported in various aflatoxin-, trichothecene-, citrinin-, and patulin-producing fungal pathogens (See
Table 1). Accordingly, there is an urgent demand to increase the efficacy of conventional antimycotic drugs/fungicides or develop new intervention strategies, which can secure the safe production of crops and food as well as public health.
Table 1.
Fungicide potentiation of mycotoxin production in fungal pathogens.
Table 1.
Fungicide potentiation of mycotoxin production in fungal pathogens.
| Fungi | Fungicide | Key Features (Potentiation of Mycotoxin Production) |
|---|
| Aspergillus parasiticus | Anilinopyrimidine | Correlation between fitness parameters and aflatoxigenicity [11] |
| A. parasiticus | Flusilazole | Highly aflatoxigenic, sterol demethylation inhibition-resistant isolates [12] |
| A. parasiticus | Phenylpyrrole | Highly aflatoxigenic, phenylpyrrole resistant isolates [13] |
| Fusarium graminearum | Carbendazim | Increased trichothecene production with carbendazim resistance [14] |
| Fusarium sp. | Strobilurins | Increased deoxynivalenol production by sub-optimal application of strobilurin [9] |
| F. sporotrichioides | Carbendazim | Higher mycotoxin production (T-2 toxin, 4,15-diacetoxyscirpenol, neosolaniol) with carbendazim resistance [15] |
| Penicillium expansum | Tebuconazole, Fludioxonil, etc. | Adverse effect of fitness penalties on the mycotoxigenicity of resistant isolates [16] |
| P. expansum | Benzimidazole | Highly mycotoxigenic field isolates resistant to the benzimidazoles [17] |
| P. verrucosum | Iprodione | Strong induction of mycotoxin biosynthesis by iprodione [18] |
The cell wall integrity system of fungi is an effective target for control of fungal pathogens [
19]. The cell wall integrity pathway is well elucidated and described in the model fungus
Saccharomyces cerevisiae, where the operation of mitogen-activated protein kinase (MAPK) signaling pathway (
viz., cell wall integrity pathway) is controlled by protein kinase C [
20]. The
BCK1 and
SLT2 genes, which encode MAPK kinase kinase (MAPKKK) and MAPK, respectively (
Table 2), in the pathway play crucial roles for maintaining cell wall integrity in fungi [
20]. Studies have shown that genes in the cell wall integrity system in fungi, such as species in the genus
Aspergillus and
S. cerevisiae, are functionally well conserved [
21,
22]. Therefore, cell wall targeting drugs (e.g., echinocandins) could be administered as broad-spectrum antimycotic agents for control of filamentous or yeast fungal pathogens, including species of
Candida ([
23] and references therein).
Nevertheless, despite their usefulness as cell wall targeting drugs, echinocandins generally do not completely inhibit fungal growth ([
24] and references therein), where the determination of precise endpoints for pathogen intervention is very rigorous [
25]. Echinocandin treatment can also trigger a compensatory stimulation of chitin synthesis, which causes the development of fungal resistance to the drugs ([
24] and references therein). Accordingly, development of new drugs or intervention strategies is continually required for effective control of fungal pathogens, especially the strains exhibiting drug or fungicide resistance.
Table 2.
Microbial strains used in this study.
Table 2.
Microbial strains used in this study.
| Aspergillus | Characteristics | Source/References |
|---|
| A. flavus 3357 | Plant pathogen (aflatoxin), Human pathogen (aspergillosis), Reference aflatoxigenic strain used for genome sequencing | NRRL a [26] |
| A. flavus 4212 | Plant pathogen (aflatoxin), Human pathogen (aspergillosis) | NRRL |
| A. parasiticus 5862 | Plant pathogen (aflatoxin) | NRRL |
| A. parasiticus 2999 | Plant pathogen (aflatoxin) | NRRL |
| A. fumigatus AF293 | Human pathogen (aspergillosis), Parental strain, Reference clinical strain used for genome sequencing | [26,27] |
| A. fumigatus sakAΔ | Human pathogen (aspergillosis), Mitogen-Activated Protein Kinase (MAPK) gene deletion mutant derived from AF293 | [27] |
| A. fumigatus mpkCΔ | Human pathogen (aspergillosis), Mitogen-Activated Protein Kinase (MAPK) gene deletion mutant derived from AF293 | [28] |
| P. expansum W1 | Plant pathogen (patulin), Parental strain | [29] |
| P. expansum FR2 | Plant pathogen (patulin), Fludioxonil resistant mutant derived from P. expansum W1 | [29] |
| P. expansum W2 | Plant pathogen (patulin), Parental strain | [29] |
| P. expansum FR3 | Plant pathogen (patulin), Fludioxonil resistant mutant derived from P. expansum W2 | [29] |
| Saccharomyces | Characteristics | Source/References |
| S. cerevisiae BY4741 | Model yeast, Parental strain (Mat a his3Δ1 leu2Δ0 met15Δ0 ura3Δ0) | [30] |
| S. cerevisiae slt2Δ | MAPK mutant in cell wall integrity system derived from BY4741 | [30] |
| S. cerevisiae bck1Δ | MAPK kinase kinase (MAPKKK) mutant derived from BY4741 | [30] |
Antifungal chemosensitization is an intervention scheme for effective control of pathogenic fungi, where co-application of a selected natural or synthetic compound (
viz., a chemosensitizer or a chemosensitizing agent) with a conventional antifungal drug can intensify the drug efficacy [
31]. Chemosensitization strategy makes the fungal pathogens highly susceptible to the drug co-administered, where the chemosensitizer significantly impaired fungal defense to the conventional drug. By definition, comparing to the traditional combination therapy (
viz., combined application of two or more commercial drugs), a chemosensitizer itself does not have to exhibit a high extent of antifungal potency. However, chemosensitization not only magnifies the efficacy of the antimycotic drug co-applied, but also overcomes pathogen resistance to conventional drugs [
31]. For example, co-application of piperazinyl quinolone with the azole drug fluconazole (FLC) resulted in overcoming FLC resistance of
C. albicans, while the compound showed no antimycotic activity when applied alone [
32]. Also, co-administration of cyclobutene-dione (squarile) derivatives with FLC elevated the drug activity during the treatment of
C. albicans, where the chemosensitizer(s) modulated the major facilitator superfamily transporter (Mdr1p; responsible for FLC resistance) of the pathogen [
33]. Chemosensitization mediated by a
d-octapeptide derivative further overcame FLC resistance in
S. cerevisiae and pathogenic fungi [
34]. Collectively, fungal intervention via chemosensitization could be an alternative to (or complement) current antifungal practices, for example, combination therapy.
Natural products that present no significant medical or environmental side effects are potential sources of antimycotic or antimycotoxigenic agents, either in their nascent structure or as leads for more potent derivatives [
35]. For instance, benzo derivatives (such as vanillic or caffeic acid) not only inhibited the growth of filamentous fungal pathogens
, but also disrupted the production of mycotoxins [
36]. The redox-active natural products, such as phenolic agents, can be potent redox cyclers that prevent fungal growth by interfering cellular redox homeostasis (thus, triggering fungal oxidative stress) or by disrupting the integrity of cellular components [
37,
38]. For defense, the fungal antioxidant system or cell wall/membrane integrity system play important roles for fungal tolerance to the phenolic agents administered [
37,
38].
Terpenoid phenols, such as carvacrol (
5-isopropyl-2-methylphenol) and its structural isomer thymol (
2-isopropyl-5-methylphenol) (
Figure 1), have been demonstrated to be effective natural antimycotic agents by inhibiting the growth or activity of planktonic or biofilms of fungal pathogens ([
39] and references therein). Carvacrol and thymol are generally regarded as safe (GRAS) reagents [
40], and thus, are currently used as food additives. Genome-wide transcription profiling (microarray) study in the model fungus
S. cerevisiae disclosed that genes in metabolic (energy, pyrimidine), biosynthetic, stress responses (oxidative, heat shock),
etc., were highly up- or down-regulated with the treatment of carvacrol, where the ion homeostasis mutant (
vmaΔ) was also hypersensitive to carvacrol treatment [
39]. Similar microarray analysis in
S. cerevisiae treated with thymol also revealed that genes involved in metabolism (sulfur, protein, thiamin, nucleic acid,
etc.), mitochondrial function, organellar ribosome, cell proliferation,
etc., were up- or down-regulated by thymol application [
41]. However, effects of carvacrol or thymol on the function of cell wall integrity system were undetermined in these studies.
Figure 1.
Structures of benzo derivatives, carvacrol, thymol, and aflatoxins used/detected in this study. (1) Benzaldehyde and derivatives: (1a) 2-Hydroxy-4-methoxybenzaldehyde, (1b) 2-Methyl-4-methoxybenzaldehyde, (1c) 3,5-Dimethoxybenzaldehyde, (1d) 2,3-Dimethoxybenzaldehyde, (1e) 2,5-Dimethoxybenzaldehyde, (1f) 2-Methoxybenzaldehyde, (1g) 2,4-Dimethoxybenzaldehyde, (1h) 3-Methoxybenzaldehyde, (1i) 4-Methoxybenzaldehyde, (1j) 2,4,5-Trimethoxybenzaldehyde, (1k) 3,4-Dimethoxybenzaldehyde, (1l) 4-Hydroxy-3-methoxybenzaldehyde, (1m) 4-Hydroxy-2-methoxybenzaldehyde, (1n) 3,5-Dimethoxy-4-hydroxybenzaldehyde; (2) Carvacrol (5-Isopropyl-2-methylphenol); (3) Thymol (2-Isopropyl-5-methylphenol); (4) Aflatoxin B1; (5) Aflatoxin B2; (6) Aflatoxin G1; (7) Aflatoxin G2.
Figure 1.
Structures of benzo derivatives, carvacrol, thymol, and aflatoxins used/detected in this study. (1) Benzaldehyde and derivatives: (1a) 2-Hydroxy-4-methoxybenzaldehyde, (1b) 2-Methyl-4-methoxybenzaldehyde, (1c) 3,5-Dimethoxybenzaldehyde, (1d) 2,3-Dimethoxybenzaldehyde, (1e) 2,5-Dimethoxybenzaldehyde, (1f) 2-Methoxybenzaldehyde, (1g) 2,4-Dimethoxybenzaldehyde, (1h) 3-Methoxybenzaldehyde, (1i) 4-Methoxybenzaldehyde, (1j) 2,4,5-Trimethoxybenzaldehyde, (1k) 3,4-Dimethoxybenzaldehyde, (1l) 4-Hydroxy-3-methoxybenzaldehyde, (1m) 4-Hydroxy-2-methoxybenzaldehyde, (1n) 3,5-Dimethoxy-4-hydroxybenzaldehyde; (2) Carvacrol (5-Isopropyl-2-methylphenol); (3) Thymol (2-Isopropyl-5-methylphenol); (4) Aflatoxin B1; (5) Aflatoxin B2; (6) Aflatoxin G1; (7) Aflatoxin G2.
The benzo derivative 2-hydroxy-4-methoxybenzaldehyde (2H4M) (
Figure 1) is also a GRAS reagent [
40], and hence, is currently used as a food additive. The 2H4M has been isolated from different plants as a natural compound, where it functioned as an insect repellent to protect food sources,
etc. [
42,
43]. In this study, two
S. cerevisiae cell wall integrity mutants (
bck1Δ,
slt2Δ), where genes in cell wall integrity MAPK pathway were deleted (
Table 2), were examined to evaluate the efficacy of targeting cell wall integrity via natural product-based antifungal chemosensitization (namely, monoterpenoid phenols + 2H4M). The same intervention strategy was also investigated in filamentous fungal pathogens, such as
Aspergillus and
Penicillium, according to the Clinical Laboratory Standards Institute (CLSI) M38-A [
44]. Results showed that: (1) 2H4M functioned as an effective antifungal chemosensitizer to augment the potency of monoterpenoid phenols; and (2) 2H4M overcame the tolerance of
A. fumigatus MAPK mutants (
sakAΔ,
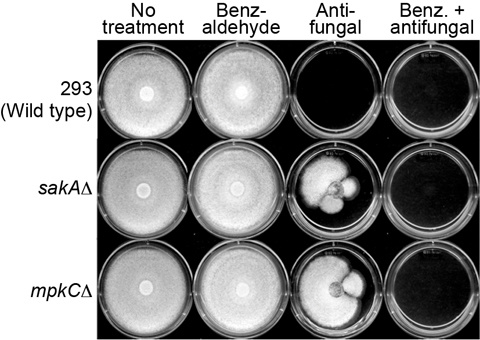
mpkCΔ) to fludioxonil, a phenylpyrrole fungicide.
4. Conclusions
In this study, levels of compound interactions between monoterpenoid phenols (carvacrol, thymol) and a chemosensitizer (2H4M) were determined for the enhancement of antifungal efficacy. Key features identified for the antifungal or antimycotoxigenic potential of compounds are as follows: (1) Among fifteen benzaldehyde analogs examined, nine compounds inhibited the growth of bck1Δ and slt2Δ, the cell wall integrity mutants of S. cerevisiae, with structure-activity relationship. 2H4M exhibited the highest antifungal activity among test compounds; (2) 2H4M possessed a chemosensitizing capability to carvacrol or thymol in yeasts, where chemosensitization enhanced the antimycotic potency of test compounds. The 2H4M, a cell wall perturbing chemosensitizer, and monoterpenoid phenols could affect common cellular targets, i.e., cell wall integrity system of fungi, which results in synergistic inhibition of fungal growth; (3) In yeast chemosensitization, thymol required much lower concentration to achieve complete inhibition of yeast growth compared to carvacrol, thus reflecting structure-activity relationship; (4) In filamentous fungal tests, co-administration of 2H4M with carvacrol or thymol resulted in the achievement of synergism in Aspergillus and Penicillium strains; (5) Carvacrol or thymol further exacerbated the vulnerability (namely, defects in countering oxidative stress) of the oxidative MAPK mutants (sakAΔ, mpkCΔ) of A. fumigatus; (6) A. fumigatus or A. flavus were more susceptible to thymol compared to carvacrol, while the level of antimycotic activity of carvacrol or thymol was vastly similar in A. parasiticus or P. expansum (compound-strain relationship); (7) Co-application of 2H4M with fludioxonil overcame fungal tolerance to fludioxonil, where the cell wall interfering capability of 2H4M might enhance the susceptibility of the oxidative MAPK mutants (A. fumigatus sakAΔ, mpkCΔ) to fludioxonil, possibly by increased penetration of fludioxonil into the fungal cell through perturbed cell wall; (8) Thymol and carvacrol enhanced aflatoxin production in A. flavus and A. parasiticus. Although 2H4M also potentiated the aflatoxin production in A. flavus, this compound reduced the aflatoxin production in A. parasiticus at all concentrations examined (i.e., strain specificity for the antimycotoxigenic activity of 2H4M); (9) Thymol or carvacrol also enhanced the sclerotial differentiation in A. flavus or A. parasiticus, depending on dosages and types of test compounds. A. flavus 3357 produced higher number of sclerotia compared to A. flavus 4212 (w/carvacrol or thymol), while A. parasiticus developed more sclerotia with thymol than with carvacrol. However, sclerotia development was almost not detected with the treatment of 2H4M.
In previous studies in yeasts, another signaling pathway, namely the “intact” oxidative MAPK pathway, was also shown to be important for fungal susceptibility to cell wall perturbing agents [
50,
51,
52,
53]. Results showed that mutations in the antioxidant system could develop fungal resistance to cell wall disrupting agents. For example, the
S. cerevisiae oxidative MAPK pathway mutants (e.g., MAPK or MAPK kinase mutants, the upstream transmembrane osmosensor or histidine kinase osmosensor mutants in the same signaling cascade,
etc.) exhibited tolerance to the cell wall-perturbing agents, such as calcofluor white [
51,
52,
53]. Therefore, existence of the “intact” oxidative MAPK pathway of fungi is necessary for effective control of pathogens. Nonetheless, the antioxidant mutants of
A. fumigatus (
sakAΔ,
mpkCΔ) tested in this study did not develop tolerance to 2H4M or monoterpenoid phenols, while 2H4M further overcame fungal tolerance to the phenylpyrrole fungicide fludioxonil.
In conclusion, 2H4M, a natural phenolic compound, possesses a potential to serve as an antimycotic chemosensitizer in combination with monoterpenoid phenols. 2H4M-mediated chemosensitization, as described in this study, can modulate/debilitate the cell wall integrity system of fungal strains, which can lower effective doses of antimycotic agent co-administered. Future studies are needed for comprehensive determination of optimum chemosensitization in various fungal pathogens by including additional cell wall disrupting agents. It would also be interesting to know whether antifungal activity correlates with chemosensitizing activity, or whether a circumstance similar to that for piperazinyl quinolone with fluconazole [
32], exists for benzaldehyde analogs with no antifungal activity.