The kin17 Protein in Murine Melanoma Cells
Abstract
:1. Introduction
2. Results
2.1. Isolation of Low and High Metastatic Tumor Cells


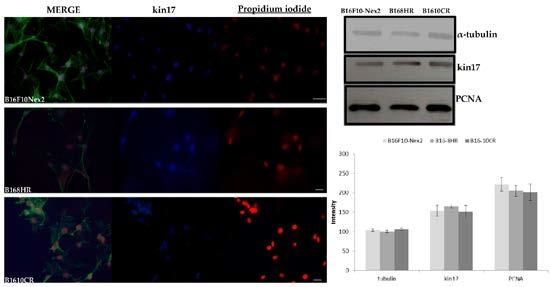
2.2. Immunodetection of the kin17 Protein in Murine Tumor Cell Lines

2.3. Detection of kin17 in Protein Extracts

3. Discussion
4. Experimental Section
4.1. Cell Lines and Culture Conditions
4.2. In Vivo Assays
4.3. Immunodetection
4.4. Protein Extraction
4.5. Western Blotting
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Freitas, Z.F.O.; Rodrigues, E.G.; Oliveira, V.; Carmona, A.K.; Travassos, L.R. Melanoma heterogeneity: Differential, invasive, metastatic properties and profiles of cathepsin B, D and L activities in subclones of the B16F10-NEX2 cell line. Melanoma Res. 2004, 14, 333–344. [Google Scholar] [PubMed]
- Vidwans, S.J.; Flaherty, K.T.; Fisher, D.E.; Tenenbaum, J.M.; Travers, M.D.; Shrager, J. A Melanoma Molecular Disease Model. PLoS ONE 2011, 6, e18257. [Google Scholar] [CrossRef] [PubMed]
- Sarasin, A.; Kauffmann, A. Overexpression of DNA repair genes is associated with metastasis: A new hypothesis. Mutat. Res. 2008, 659, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Zeng, T.; Gao, H.; Yu, P.; He, H.; Ouyang, X.; Deng, L.; Zhang, Y. Up-Regulation of Kin17 Is Essential for Proliferation of Breast Cancer. PLoS ONE 2011, 6, e25343. [Google Scholar] [CrossRef] [PubMed]
- Despras, E.; Miccoli, L.; Créminon, C.; Rouillard, D.; Angulo, J.F.; Biard, D.S.F. Depletion of KIN17, a human DNA replication protein, increases the radio sensitivity of RKO cells. Radiat. Res. 2003, 159, 748–758. [Google Scholar] [CrossRef]
- Biard, D.S.F.; Miccoli, L.; Despras, E.; Frobert, Y.; Creminon, C.; Angulo, J.F. Ionizing radiation triggers chromatin bound Kin17 complex Formation in human cells. J. Biol. Chem. 2003, 277, 19156–19165. [Google Scholar] [CrossRef] [PubMed]
- Miccoli, L.; Frouin, I.; Novac, O.; di Paola, D.; Harper, F.; Zannis-Hadjopoulos, M.; Maga, G.; Biard, D.S.F.; Angulo, J.F. The human stress-activated protein kin17 belongs to the multiprotein DNA replication complex and associates in vivo with mammalian replication origins. Mol. Cell. Biol. 2005, 25, 3814–3830. [Google Scholar] [CrossRef] [PubMed]
- Despras, E. Les proteines kin17, xpc, DNA-PKCS et XRCC4 dans la reponse cellulaire aux dommages de l’ADN. Etude des relations entre la reparation par excision de nucleotides et la recombinaison non homologue dans un modele syngenique humain. In These de Docteur aux Biologie Cellulaire et Moléculaire; Universite Paris V: Paris, France, 2006. [Google Scholar]
- Mazin, A.; Milot, E.; Devoret, R.; Chartrand, P. Kin17, a mouse nuclear protein, binds to bent DNA fragments that are found at illegitimate recombination junctions in mammalian cells. Mol. Gen. Genet. 1994, 244, 435–438. [Google Scholar] [CrossRef] [PubMed]
- Kannouche, P.; Mauffrey, P.; Pinon-Lataillade, G.; Mattei, M.G.; Sarasin, A.; Daya-Grosjean, L.; Angulo, J.F. Molecular cloning and characterization of the human Kin17 cDNA encoding a component of the UVC response that is conserved among metazoans. Carcinogenesis 2000, 21, 1701–1710. [Google Scholar] [CrossRef] [PubMed]
- Kannouche, P.; Pinon-Lataillade, G.; Mauffrey, P.; Faucher, C.; Biard, D.S.F.; Angulo, J.F. Overexpression of kin17 protein forms intranuclear foci in mammalian cells. Biochimie 1997, 79, 599–606. [Google Scholar] [CrossRef]
- Mazin, A.; Timchenko, T.; Mènissier-De Murcia, J.; Schreiber, V.; Angulo, J.F.; Gilbert de, M.; Devoret, R. Kin17, a mouse nuclear zinc finger protein that binds preferentially to curved DNA. Nucleic Acids Res. 1994, 22, 4335–4341. [Google Scholar] [CrossRef] [PubMed]
- Angulo, J.F.; Rouer, E.; Mazin, A.; Mattei, M.-G.; Tissier, A.; Horellou, P.; Benarous, R.; Devoret, R. Identification and expression of the cDNA of Kin17, a zinc finger gene located on mouse chromosome 2, encoding a new DNA-binding protein. Nucleic Acids Res. 1991, 19, 5117–5123. [Google Scholar] [CrossRef] [PubMed]
- Le Maire, A.; Schiltz, M.; Braud, S.; Gondry, M.; Charbonnier, J.-B.; Zinn-Justin, S.; Stura, E. Crystallization and halide phasing of the C-terminal domain of human KIN17. Acta Crystallogr. F 2006, 62, 245–248. [Google Scholar] [CrossRef] [PubMed]
- Kyrpides, N.C.; Woese, C.R.; Ouzounis, C.A. KOW: A novel motif linking a bacterial transcription factor with ribosomal proteins. Trends Biochem. Sci. 1996, 21, 425–426. [Google Scholar] [CrossRef]
- Steiner, T.; Kaiser, J.T.; Marinkovic, S.; Huber, R.; Wahl, M.C. Crystal structures of transcription factor NusG in light of its nucleic acid- and protein-binding activities. EMBO J. 2002, 21, 4641–4651. [Google Scholar] [CrossRef] [PubMed]
- Cloutier, P.; Lavallée-Adam, M.; Faubert, D.; Blanchette, M.; Coulombe, B. A Newly Uncovered Group of Distantly Related Lysine Methyltransferases Preferentially Interact with Molecular Chaperones to Regulate Their Activity. PLoS Genet. 2013, 9, e1003210. [Google Scholar] [CrossRef] [PubMed]
- Cloutier, P.; Lavallée-Adam, M.; Faubert, D.; Blanchette, M.; Coulombe, B. Methylation of the DNA/RNA-binding protein Kin17 by METTL22 affects its association with chromatin. J. Proteom. 2014, 100, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Gasser, S.M.; Laemmli, U.K. Improved methods for the isolation of individual and clustered mitotic chromosomes. Exp. Cell Res. 1987, 173, 85–98. [Google Scholar] [CrossRef]
- Phi-Van, L.; von Kries, J.P.; Ostertag, W.; Strätling, W.H. The chicken lysozyme 5′ matrix attachment region increases transcription from a heterologous promoter in heterologous cells and dampens position effects on the expression of transfected genes. Mol. Cell. Biol. 1990, 10, 2302–2307. [Google Scholar] [CrossRef] [PubMed]
- Laemmli, U.K.; Käs, E.; Poljak, L.; Adachi, Y. Scaffold-associated regions: cis-Acting determinants of chromatin structural loops and functional domains. Curr. Opin. Genet. Dev. 1992, 2, 275–285. [Google Scholar] [CrossRef]
- Ottaviani, D.; Lever, E.; Takousis, P.; Sheer, D. Anchoring the genome. Genome Biol. 2008, 9, 201. [Google Scholar] [CrossRef] [PubMed]
- Miccoli, L.; Biard, D.S.F.; Frouin, I.; Harper, F.; Maga, G.; Angulo, J.F. Selective interactions of human kin17 and RPA proteins with chromatin and the nuclear matrix in a DNA damage- and cell cycle-regulated manner. Nucleic Acids Res. 2003, 31, 4162–4175. [Google Scholar] [CrossRef] [PubMed]
- Kelman, Z. PCNA: Structure, functions and interactions. Oncogene 1997, 14, 629–640. [Google Scholar] [CrossRef] [PubMed]
- Maga, G.; Hübscher, U. Proliferating cell nuclear antigen (PCNA): A dancer with many partners. J. Cell Sci. 2003, 116, 3051–3060. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Chiara, A.; Pederzoli-Ribeil, M.; Mocek, J.; Mayeux, P.; Millet, A.; Witko-Sarsat, V. Characterization of cytosolic proliferating cell nuclear antigen (PCNA) in neutrophils: Antiapoptotic role of the monomer. J. Leukoc. Biol. 2013, 94, 723–731. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shivji, K.K.; Kenny, M.K.; Wood, R.D. Proliferating cell nuclear antigen is required for DNA excision repair. Cell 1992, 69, 367–374. [Google Scholar] [CrossRef]
- Moldovan, G.L.; Pfander, B.; Jentsch, S. PCNA, the maestro of the replication fork. Cell 2007, 129, 665–679. [Google Scholar] [CrossRef] [PubMed]
- Naryzhny, S.N.; Lee, H. Proliferating cell nuclear antigen in the cytoplasm interacts with components of glycolysis and cancer. FEBS Lett. 2010, 584, 4292–4298. [Google Scholar] [CrossRef] [PubMed]
- Blow, J.J.; Dutta, A. Preventing re-replication of chromosomal DNA. Nat. Rev. Mol. Cell Biol. 2005, 6, 476–486. [Google Scholar] [CrossRef] [PubMed]
- Bell, S.P.; Dutta, A. DNA replication in eukaryoticcells. Annu. Rev. Biochem. 2002, 71, 333–374. [Google Scholar] [CrossRef] [PubMed]
- Prasanth, S.G.; Prasanth, K.V.; Stillman, B. ORC6 involved in DNA replication, chromosome segregation, and cytokinesis. Science 2002, 297, 1026–1031. [Google Scholar] [CrossRef] [PubMed]
- Prasanth, S.G.; Prasanth, K.V.; Siddiqui, K.; Spector, D.L.; Stillman, B. Human ORC2 localizes to centrosomes, centromeresand heterochromatin during chromosome inheritance. EMBO J. 2004, 23, 2651–2663. [Google Scholar] [CrossRef] [PubMed]
- DePamphilis, M.L. The “ORC cycle”: A novel pathway for regulating eukaryotic DNA replication. Gene 2003, 310, 1–15. [Google Scholar] [CrossRef]
- Mobley, A.K.; Braeuer, R.R.; Kamiya, T.; Shoshan, E.; Bar-Eli, M. Driving transcriptional regulators in melanoma metastasis. Cancer Metastasis Rev. 2012, 31, 621–632. [Google Scholar] [CrossRef] [PubMed]
- Paliwal, S.; Hwang, B.H.; Tsai, K.Y.; Mitragotri, S. Diagnostic opportunities based on skin biomarkers. Eur. J. Pharm. Sci. 2013, 50, 546–556. [Google Scholar] [CrossRef] [PubMed]
- Margue, C.; Philippidou, D.; Reinsbach, S.E.; Schmitt, M.; Behrmann, I.; Kreis, S. New Target Genes of MITF-Induced microRNA-211 Contribute to Melanoma Cell Invasion. PLoS ONE 2013, 8, e73473. [Google Scholar] [CrossRef] [PubMed]
- Méndez, J.; Stillman, B. Chromatin Association of Human Origin Recognition Complex, Cdc6, and Minichromosome Maintenance Proteins during the Cell Cycle: Assembly of Prereplication Complexes in Late Mitosis. Mol. Cell. Biol. 2000, 20, 8602–8612. [Google Scholar] [CrossRef] [PubMed]
- Laemmlli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramos, A.C.; Gaspar, V.P.; Kelmer, S.M.G.; Sellani, T.A.; Batista, A.G.U.; De Lima Neto, Q.A.; Rodrigues, E.G.; Fernandez, M.A. The kin17 Protein in Murine Melanoma Cells. Int. J. Mol. Sci. 2015, 16, 27912-27920. https://doi.org/10.3390/ijms161126072
Ramos AC, Gaspar VP, Kelmer SMG, Sellani TA, Batista AGU, De Lima Neto QA, Rodrigues EG, Fernandez MA. The kin17 Protein in Murine Melanoma Cells. International Journal of Molecular Sciences. 2015; 16(11):27912-27920. https://doi.org/10.3390/ijms161126072
Chicago/Turabian StyleRamos, Anelise C., Vanessa P. Gaspar, Sabrina M. G. Kelmer, Tarciso A. Sellani, Ana G. U. Batista, Quirino A. De Lima Neto, Elaine G. Rodrigues, and Maria A. Fernandez. 2015. "The kin17 Protein in Murine Melanoma Cells" International Journal of Molecular Sciences 16, no. 11: 27912-27920. https://doi.org/10.3390/ijms161126072