Expression of IL-8, IL-6 and IL-1β in Tears as a Main Characteristic of the Immune Response in Human Microbial Keratitis
Abstract
:1. Introduction
2. Results
2.1. Demographics, and Relevant Medical History of Patients with Microbial Keratitis
| Characteristics of Patients | Patients with Fungal Keratitis (n = 14) | Patients with Gram-Positive Bacterial Keratitis (n = 8) | Patients with Gram-Negative Bacterial Keratitis (n = 6) | Healthy Volunteers (n = 48) | p Value |
|---|---|---|---|---|---|
| Males/Females | 10/4 | 3/5 | 3/3 | 24/24 | – |
| Age (mean ± SD, years) | 49 ± 19 | 65 ± 19 | 51 ± 19 | 52 ± 19 | NS |
| Microbiological diagnosis (number of patients) | Cephalosporium spp. (1) Curvularia spp. (1) Candida albicans (1) Trichoderma spp. (1) Aspergillus spp. (1) Fusarium spp. (9) | S. pneumonie (3) Streptococcus spp. (2) Micrococcus varians (1) S. pneumonie and S. epidermidis (2) | K. pneumonie (1) K. ozanae (1) Pseudomonas sp. (3) S. marcescens (1) | – | – |
| Relevant medical history | Diabetes (4) Eye trauma (3) Diabetes and eye trauma (1) Alcoholism (1) Lens wear (1) None (4) | Diabetes (2) Previous herpes infection (2) Eye trauma (1) Congenital glaucoma (1) Rheumatoid arthritis (2) | Penetrating keratoplasty (1) Cosmetic lens/Lens wear (2) Diabetes (2) Drug user (1) | – | – |
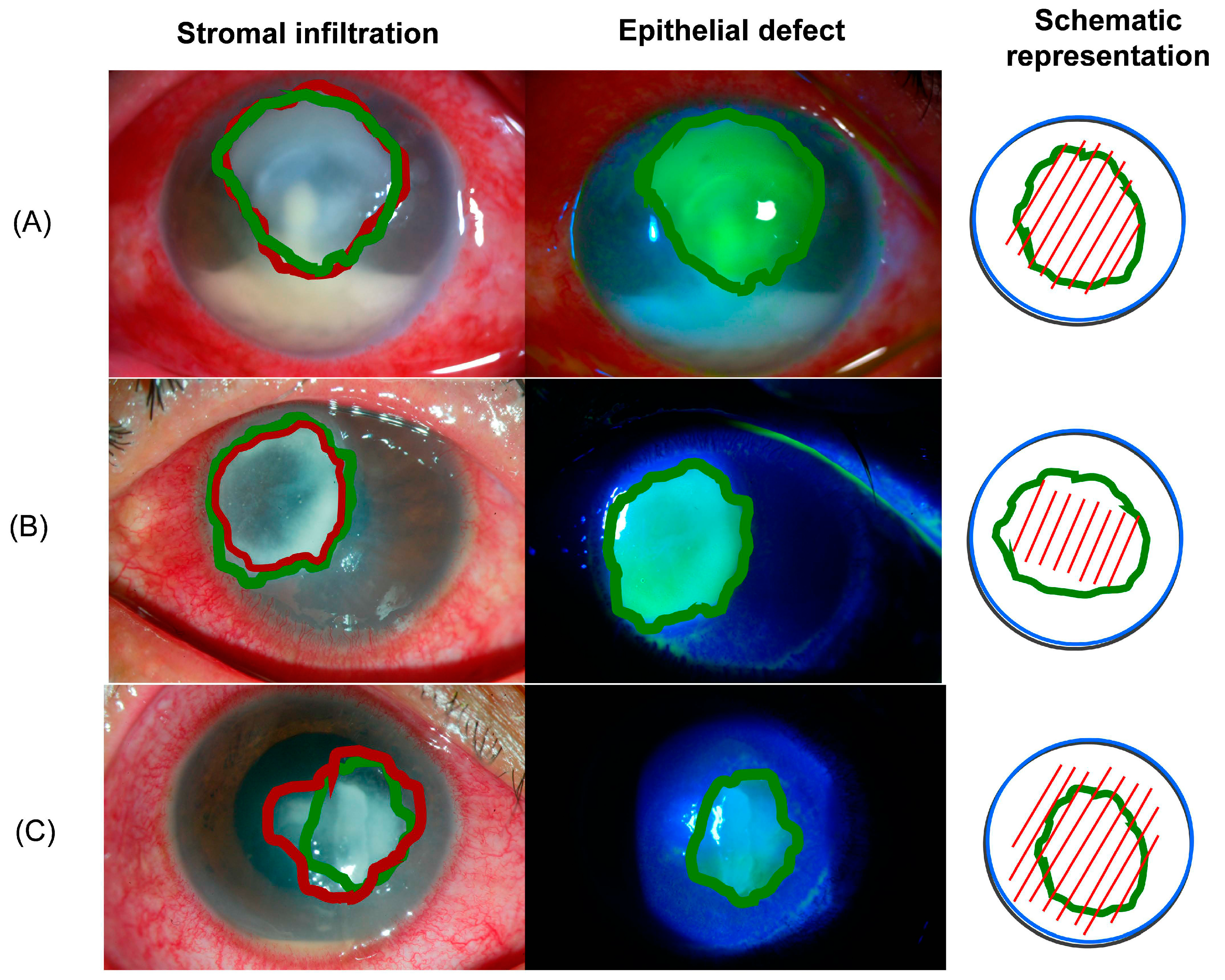
2.2. Ocular Findings of Patients with Microbial Keratitis
| Ocular Findings | Fungal Keratitis (n = 14) | Gram-Positive Bacterial Keratitis (n = 8) | Gram-Negative Bacterial Keratitis (n = 6) | p Value |
|---|---|---|---|---|
| Epithelial defect: Horizontal diameter (mm) | 5.3 ± 1.9 | 4.8 ± 1.9 | 5.6 ± 1.8 | NS between any groups of patients. |
| Epithelial defect: Vertical diameter (mm) | 5.7 ± 2.2 | 5.5 ± 2.3 | 5 ± 2.4 | NS between any groups of patients. |
| Stromal lesion: Horizontal infiltration diameter (mm) | 5.7 ± 2.4 | 4.9 ± 2.1 | 5 ± 2.4 | NS between any groups of patients. |
| Stromal lesion: Vertical infiltration diameter (mm) | 6.0 ± 2.6 | 5.6 ± 2.3 | 4.3 ± 1.5 | NS between any groups of patients. |
| Hypopyon (percentage) | 21 ± 22 | 37.5 ± 31 | 7.6 ± 8.6 | NS between any groups of patients. |
2.3. Frequency of CD3−CD56+ NK Cells Is Increased in Peripheral Blood Mononuclear Cells from Patients with Gram-Negative Bacterial Keratitis
2.4. IL-1β, IL-6, and IL-8 Are Increased in Tears Samples of Patients with Microbial Keratitis

| PBMC Subpopulations | Fungal Keratitis (n = 14) | Gram-Positive Bacterial Keratitis (n = 8) | Gram-Negative Bacterial Keratitis (n = 6) | Healthy Volunteers (n = 48) | p Value |
|---|---|---|---|---|---|
| Percentage of CD3+ cells in PBMC | 57 ± 10 | 55 ± 8 | 63 ± 9 | 62 + 11 | NS between any groups |
| Percentage CD4+ cells in PBMC | 34 ± 10 | 30 ± 3 | 31 ± 10 | 40 ± 13 | NS between any groups |
| Percentage of CD8+ cells in PBMC | 25 ± 11 | 24 ± 10 | 26 ± 10 | 28 ± 12 | NS between any groups |
| Percentage of CD19+ cells in PBMC | 12 ± 8 | 12 ± 9 | 10.1 ± 5 | 12 ± 7 | NS between any groups |
| Percentage of CD3− CD56+ cells in PBMC | 28 ± 20 | 27 ± 13 | 39 ± 21 * | 25 ± 12 * | p < 0.05 |
| CD4+IFN-γ+ cells (After PMA/ionomicyn stimulation) | 12 ± 10 | 16 ± 6 | 11 ± 6 | 10 ± 5 | NS between any groups |
| CD4+IL-4+ (After PMA/ionomicyn stimulation) | 1 ± 1 | 3 ± 3 | 2 ± 2 | 4 ± 2 | NS between any groups |
| CD8+IFN-γ+ (After PMA/ionomicyn stimulation) | 30 ± 27 | 16 ± 6 * | 31 ± 23 | 34 ± 20 * | p < 0.05 |
| CD8+IL-4+ (After PMA/ionomicyn stimulation) | 1 ± 2 * | 1 ± 1 * | 2 ± 2 * | 10 ± 7 * | p < 0.05, when compared with healthy volunteers |
| Tear Cytokine | Fungal Keratitis | Gram-Positive Bacterial Keratitis | Gram-Negative Bacterial Keratitis | Contralateral Non-Infected Eye | p Value (Compared with Contralateral Non-Infected Eye) |
|---|---|---|---|---|---|
| IL-1β pg/mL | 1135 ± 764 * | 64 ± 40 | 423 ± 240 * | 58 ± 89 * | <0.05 |
| IL-6 pg/mL | 4172 ± 1873 * | 758 ± 1166 | 1596 ± 971 * | 25 ± 17 * | <0.05 |
| IL-8 pg/mL | >2500 * | >2500 * | >2500 * | 420 ± 377 * | <0.05 |
| IL-10 pg/mL | 43 ± 35 | 34 ± 23 | 107 ± 166 | 13 ± 3 | NS |
| IL-12p70 pg/mL | 12 ± 6 | 12 ± 1 | 4.0 ± 1 | 5 ± 5 | NS |
| TNF-α pg/mL | 28 ± 21 | 8 ± 4 | 8 ± 2 | 14 ± 7 | NS |


3. Discussion
4. Material and Methods
4.1. Patients
4.2. Clinical Evaluation
4.3. Monoclonal Antibodies and Reagents
4.4. Peripheral Blood Mononuclear Cells
4.5. Immunofluorescence Staining of Cell Surface Markers
4.6. Cell Cultures
4.7. Immunofluorescence Staining of Intracellular Cytokines
4.8. Flow Cytometric Analysis
4.9. Determination of Soluble Cytokines
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Whitcher, J.P.; Srinivasan, M.; Upadhyay, M.P. Corneal blindness. A global perspective. Bull. World Health Organ. 2001, 79, 214–221. [Google Scholar] [PubMed]
- World Health Organization. Global Initiative for the Elimination of Avoidable Blindness: Action plan 2006–2011, Vision 2020. The right to sight. 2007. Available online: http://apps.who.int/iris/bitstream/10665/43754/1/9789241595889_eng.pdf?ua=1 (accessed on 7 November 2014).
- Saha, S.; Banerjee, D.; Khetan, A.; Sengupta, J. Epidemiological profile of fungal keratitis in urban population of West Bengal, India. Oman J. Ophthalmol. 2009, 2, 114–118. [Google Scholar] [CrossRef] [PubMed]
- Amer Awan, M.; Reeks, G.; Rahman, M.Q.; Butcher, I.; Ramaesh, K. The patterns of in vitro antimicrobial susceptibility and resistance of bacterial keratitis isolates in Glasgow, United Kingdom. Clin. Exp. Optom. 2010, 93, 354–359. [Google Scholar]
- Deorukhkar, S.; Katiyar, R.; Saini, S. Epidemiological features and laboratory results of bacterial and fungal keratitis: A five-year study at a rural tertiary-care hospital in western Maharashtra, India. Singap. Med. J. 2012, 53, 264–267. [Google Scholar]
- Gopinathan, U.; Sharma, S.; Garg, P.; Rao, G.N. Review of epidemiological features, microbiological diagnosis and treatment outcome of microbial keratitis: Experience of over a decade Indian. J. Ophthalmol. 2009, 57, 273–279. [Google Scholar]
- Bourcier, T.; Thomas, F.; Borderie, V.; Chaumeil, C.; Laroche, L. Bacterial keratitis: Predisposing factors, clinical and microbiological review of 300 cases. Br. J. Ophthalmol. 2003, 87, 834–838. [Google Scholar] [CrossRef] [PubMed]
- Whitcher, J.P.; Srinivasan, M. Corneal ulceration in developing world: A silent epidemic. Br. J. Ophthalmol. 1997, 81, 622–623. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, C.A.; Srinivasan, M.; Whitcher, J.P.; Smolin, G. Incidence of corneal ulceration in Madurai District, South India. Ophthalmic Epidemiol. 1996, 3, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.C.; Lalitha, P.; Srinivasan, M.; Prajna, N.V.; McLeod, S.D.; Acharya, N.R.; Lietman, T.M.; Porco, T.C. Seasonal trends of microbial keratitis in South India. Cornea 2012, 31, 1123–1127. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Camarena, J.C.; Graue-Hernández, E.O.; Chirinos-Saldaña, P.; Navas, A.; Ramírez-Miranda, A.; Vizuet-García, L.; Ortiz-Casas, M.; López-Espinosa, N.L.; Gaona-Juárez, C.; Bautista-Hernández, L.A.; et al. Infectious keratitis: microbiological and antibiotic sensitivity trends. First annual report of the Ocular Microbiology Study Group from the Instituto de Oftalmología “Conde de Valenciana”. Rev. Mex. Oftalmol. 2012, 86, 213–212. [Google Scholar]
- Hernández Camarena, J.C.; Bautista de Lucio, V.M.; Chirinos-Saldaña, P.; Navas, A.; Ramírez Miranda, A.; Climent-Flores, A.; Ortiz Casas, M.; López-Espinosa, N.L.; Gaona-Juárez, C.; Bautista-Hernández, L.A.; et al. Infectious keratitis: Microbiological and antibiotic sensitivity trends. Second Annual Report of the Ocular Microbiology Study Group from the Instituto de Oftalmologia “Conde de Valenciana”. Rev. Mex. Oftalmol. 2013, 87, 100–109. (In Spanish) [Google Scholar]
- Sun, Y.; Karmakar, M.; Roy, S.; Ramadan, R.T.; Williams, S.R.; Howell, S.; Shive, C.L.; Han, Y.; Stopford, C.M.; Rietsch, A.; et al. TLR4 and TLR5 on corneal macrophages regulate Pseudomonas aeruginosa keratitis by signaling through MyD88-dependent and -independent pathways. J. Immunol. 2010, 185, 4272–4283. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Chandra, J.; Mukherjee, P.; Szczotka-Flynn, L.; Ghannoum, M.A.; Pearlman, E. A murine model of contact lens-associated fusarium keratitis. Investig. Ophthalmol. Vis. Sci. 2010, 51, 1511–1516. [Google Scholar] [CrossRef]
- Osthoff, M.; Brown, K.D.; Kong, D.C.; Daniell, M.; Eisen, D.P. Activation of the lectin pathway of complement in experimental human keratitis with Pseudomonas aeruginosa. Mol. Vis. 2014, 20, 38–45. [Google Scholar] [PubMed]
- Vasanthi, M.; Prajna, N.V.; Lalitha, P.; Mahadevan, K.; Muthukkaruppan, V. A pilot study on the infiltrating cells and cytokine levels in the tear of fungal keratitis patients. Indian J. Ophthalmol. 2007, 55, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, T.; Calvacanti, B.M.; Cruzat, A.; Qazi, Y.; Ishikawa, S.; Osuka, A.; Lederer, J.; Hamrah, P. Correlation between human tear cytokine levels and cellular corneal changes in patients with bacterial keratitis by in vivo confocal microscopy. Investig. Ophthalmol. Vis. Sci. 2014, 55, 7457–7466. [Google Scholar] [CrossRef]
- Dalmon, C.; Porco, T.C.; Lietman, T.M.; Prajna, N.V.; Prajna, L.; Das, M.R.; Kumar, J.A.; Mascarenhas, J.; Margolis, T.P.; Whitcher, J.P.; et al. The clinical differentiation of bacterial and fungal keratitis: A photographic survey. Investig. Ophthalmol. Vis. Sci. 2012, 53, 1787–1791. [Google Scholar] [CrossRef]
- Matthew, A.; Dahlgren, M.D.; Ahila Lingappan, M.D.; Wilhelmus, K.R. The clinical diagnosis of microbial keratitis. Am. J. Ophthalmol. 2007, 143, 940–944. [Google Scholar] [CrossRef] [PubMed]
- Ahn, M.; Yoon, K.C.; Ryu, S.K.; Cho, N.C.; You, I.C. Clinical aspects and prognosis of mixed microbial (bacterial and fungal) keratitis. Cornea 2011, 30, 409–413. [Google Scholar] [CrossRef] [PubMed]
- Benson, W.H.; Lanier, J.D. Current diagnosis and treatment of corneal ulcers. Curr. Opin. Ophthalmol. 1998, 9, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Green, M.; Apel, A.; Stapleton, F. Risk factors and causative organisms in microbial keratitis. Cornea 2008, 27, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Keay, L.; Edwards, K.; Naduvilath, T.; Taylor, H.R.; Snibson, G.R.; Forde, K.; Stapleton, F. Microbial keratitis predisposing factors and morbidity. Ophthalmology 2006, 113, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Passos, R.M.L.; Cariello, A.J.; Yu, M.C.; Höfling-Lima, A.L. Microbial keratitis in the elderly: A 32-year review. Arq. Bras. Oftalmol. 2010, 73, 315–319. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, G.; Hart, O.; Gardiner, C. Putting the natural killer cell in its place. Immunology 2006, 117, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Rizzello, V.; Bonaccorsi, I.; Dongarrà, M.L.; Fink, L.N.; Ferlazzo, G. Role of natural killer and dendritic cell crosstalk in immunomodulation by commensal bacteria probiotics. J. Biomed. Biotechnol. 2011, 2011, 473097. [Google Scholar] [CrossRef] [PubMed]
- Lighvani, S.; Huang, X.; Trivedi, P.P.; Swanborg, R.H.; Hazlett, L.D. Substance p regulates natural killer cell interferon-γ production and resistance to pseudomonas aeruginosa infection. Eur. J. Immunol. 2005, 35, 1567–1575. [Google Scholar] [CrossRef] [PubMed]
- Hazlett, L.D.; Qianqian, L.; Liu, J.; McClellan, S.; Du, W.; Barrett, R.P. NKT cells are critical to initiate an inflammatory response after Pseudomonas aeruginosa ocular infection in susceptible mice. J. Immunol. 2007, 179, 1138–1146. [Google Scholar] [CrossRef] [PubMed]
- Hazlett, L.D. Corneal response to Pseudomonas aeruginosa infection. Prog. Retin. Eye Res. 2004, 23, 1–30. [Google Scholar] [CrossRef] [PubMed]
- De Maria, A.L.; Bozzano, F.; Cantoni, C.; Moretta, L. Revisiting human natural killer cell subset function revealed cytolytic CD56(dim)CD16+ NK cells as rapid producers of abundant IFN-γ on activation. Proc. Natl. Acad. Sci. USA 2011, 108, 728–732. [Google Scholar] [CrossRef] [PubMed]
- Boomer, J.S.; To, K.; Chang, K.C.; Takasu, O.; Osborne, D.F.; Walton, A.; Bricker, T.L.; Jarman, S.D.; Kreisel, D.; Krupnick, A.S.; et al. Immunosuppression in patients who die of sepsis and multiple organ failure. JAMA 2011, 306, 2594–2605. [Google Scholar] [CrossRef] [PubMed]
- Niederkorn, J.Y.L.; Peeler, J.S.; Mellon, J. Phagocytosis of particulate antigens by corneal epithelial cells stimulates interleukin-1 secretion and migration of Langerhans cells into the central cornea. Reg. Immunol. 1989, 2, 83–90. [Google Scholar] [PubMed]
- McGettrick, H.M.; Butler, L.M.; Buckley, C.D.; Rainger, G.E.; Nash, G.B. Tissue stroma as a regulator of leukocyte recruitment in inflammation. J. Leukoc. Biol. 2012, 91, 385–400. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, H.; Ghazanfari, T.; Yaraee, R.; Faghihzadeh, S.; Hassan, Z.M. Roles of IL-8 in ocular inflammations: A review. Ocul. Immunol. Inflamm. 2011, 19, 401–412. [Google Scholar] [CrossRef] [PubMed]
- Karthikeyan, R.S.; Leal, S.M., Jr.; Prajna, N.V.; Dharmalingam, K.; Geiser, D.M.; Pearlman, E.; Lalitha, P. Expression of innate and adaptive immune mediators in human corneal tissue infected with Aspergillus or Fusarium. J. Infect Dis. 2011, 204, 942–950. [Google Scholar] [CrossRef] [PubMed]
- Mesquida, M.; Leszczynska, A.; Llorenç, V.; Adán, A. Interleukin-6 blockade in ocular inflammatory diseases. Clin. Exp. Immunol. 2014, 176, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, T.; Sonoda, K.H.; Ohguro, N.; Ohsugi, Y.; Ishibashi, T.; Cua, D.J.; Kobayashi, T.; Yoshida, H.; Yoshimura, A. Involvement of Th17 cells and the effect of anti-IL-6 therapy in autoimmune uveitis. Rheumatology (Oxford) 2009, 48, 347–354. [Google Scholar] [CrossRef]
- Sakimoto, T.L.; Sugaya, S.; Ishimori, A.; Sawa, M. Anti-inflammatory effect of IL-6 receptor blockade in corneal alkali burn. Exp. Eye Res. 2012, 97, 98–104. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santacruz, C.; Linares, M.; Garfias, Y.; Loustaunau, L.M.; Pavon, L.; Perez-Tapia, S.M.; Jimenez-Martinez, M.C. Expression of IL-8, IL-6 and IL-1β in Tears as a Main Characteristic of the Immune Response in Human Microbial Keratitis. Int. J. Mol. Sci. 2015, 16, 4850-4864. https://doi.org/10.3390/ijms16034850
Santacruz C, Linares M, Garfias Y, Loustaunau LM, Pavon L, Perez-Tapia SM, Jimenez-Martinez MC. Expression of IL-8, IL-6 and IL-1β in Tears as a Main Characteristic of the Immune Response in Human Microbial Keratitis. International Journal of Molecular Sciences. 2015; 16(3):4850-4864. https://doi.org/10.3390/ijms16034850
Chicago/Turabian StyleSantacruz, Concepcion, Marisela Linares, Yonathan Garfias, Luisa M. Loustaunau, Lenin Pavon, Sonia Mayra Perez-Tapia, and Maria C. Jimenez-Martinez. 2015. "Expression of IL-8, IL-6 and IL-1β in Tears as a Main Characteristic of the Immune Response in Human Microbial Keratitis" International Journal of Molecular Sciences 16, no. 3: 4850-4864. https://doi.org/10.3390/ijms16034850








